Abstract
Anticoagulation therapy is administered to patients to prevent or stop thrombin generation in vivo. Although plasma tests of in vivo thrombin generation have been available for more than 2 decades, they are not routinely used in clinical trials or practice to monitor anticoagulation therapy. We observed a fall in one such marker, the
Introduction
Anticoagulant drugs are used to prevent thrombosis or to treat patients with established thrombosis. Successful treatment halts the ongoing generation of thrombin, fibrin clot propagation, and generation of plasmin, thereby decreasing the risk of thromboembolism.
Heparin and vitamin K antagonists have been used to prevent and treat thrombotic diseases for decades. Because a given dose of these agents can have different effects upon laboratory tests of anticoagulation, health professionals have been taught that anticoagulant monitoring with laboratory tests (typically the prothrombin time and the activated partial thromboplastin time [aPTT]) is required when using these classic agents. The emphasis on maintaining test results “within the therapeutic range” has led many to assume that if the target blood clotting time is achieved, the patient’s blood coagulation system has been adequately inhibited. Although this approach leads to successful prevention or treatment for most patients, it is not based on direct demonstration of anticoagulant efficacy in vivo. Each of these ex vivo laboratory tests has limitations, 1 and in both clinical trials and practice, there are a small percentage of patients who experience progressive disease even when standard guidelines are followed.
Many newer anticoagulants have been developed that can be administered without monitoring. The lack of required routine laboratory monitoring simplifies the management for many patients and physicians. Coincident with the development of these drugs has been a drop-off in investigation of more sophisticated laboratory techniques for determining the efficacy of anticoagulant therapy. End points for determination of efficacy (or therapeutic resistance) are therefore confined to diagnostic imaging tests. Expert opinion, rather than clinical trial data, is the main source of guidance for the management of complex patients. 2
A number of tests have been developed over the years to measure in vivo generation of thrombin. The first generation utilized functional assays to detect circulating fibrin monomers, using end points such as staphylococcal clumping, 3 protamine-induced paracoagulation, 4 ethanol-induced gelation, 5 or augmented plasmin generation by tissue plasminogen activator. 6 The second generation utilized radiolabeled fibrinogen and examined the effects of anticoagulation treatment upon the circulation time of fibrinogen in selected patients. 7 The third generation utilized immunoassays to quantify markers of coagulation system activation. Some target the products of thrombin cleavage of fibrinogen (ie, circulating fibrin monomers or fibrinopeptides), 8 –10 while others measure peptides released when coagulation factor zymogens (such as Factors II, IX, or X) are converted to active enzymes. 11 –13 Measuring thrombin–antithrombin (T-AT) complexes, which are formed following the in vivo generation of thrombin, 11 has also been informative regarding in vivo thrombin generation.
Another assay quantitates the cross-linked fibrin degradation product known as the
We observed a fall in
Methods
Systematic searches of the PubMed and Embase databases were performed by one of the investigators (C.P.M.) on August 26, 2016. All available articles from inception through the search date were included. The search strategy involved searching for combinations of keywords including “venous thromboembolism,” “
After screening titles and abstracts of studies identified by the database search, candidate studies identified by this initial screening were reviewed in full. Inclusion criteria were as follows: (1) the study population consisted of adult patients with newly diagnosed acute pulmonary embolism or deep vein thrombosis; (2) treatment of VTE in all patients was initiated using therapeutic dosing of anticoagulant drugs (including heparin products with or without the simultaneous initiation of warfarin, direct oral anticoagulants, parenteral direct thrombin inhibitors, and fondaparinux); (3)
Data were extracted from studies by one of the investigators. A standardized form was used to extract first author name, publication date, method of deep vein thrombosis or pulmonary embolism diagnosis, anticoagulation treatment used, population size, and comparator groups, if any. An additional form was used to abstract values of
Results
The PubMed literature search identified 2247 candidate papers, and the Embase search identified 2557 candidate papers. Duplicates from these 2 searches were identified and discarded, leaving 4059 unique articles. Screening and study selection as described above resulted in the identification of 20 articles
20
–39
meeting the inclusion criteria (Figure 1). Two articles
29,32
reported data found in other included reports
31,34
and were not included in the final analysis. Of the 18 included studies that tracked serial changes in one or more markers of coagulation system activation after the initiation of treatment for VTE, 16 reported serial
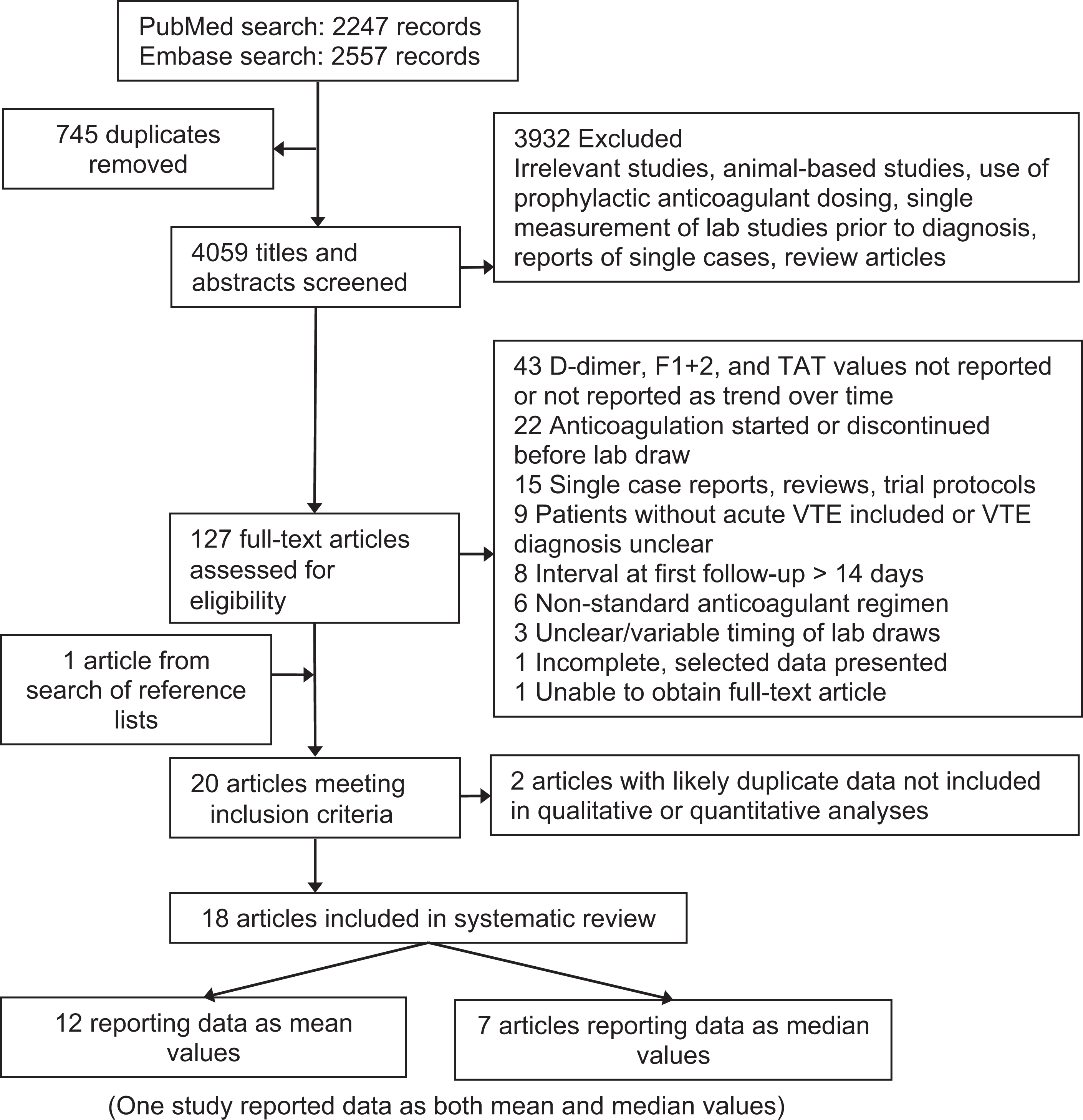
Flow diagram of systematic review. Figure adapted from PRISMA. 66 F1+2 indicates prothrombin fragment F1+2; T-AT, thrombin–antithrombin complex; VTE, venous thromboembolism.
Studies With Serial Measurements of

Abbreviations: aPTT, activated partial thromboplastin time; BID, twice daily; CT, computed tomography; DVT, deep vein thrombosis; F1+2, prothrombin fragment F1+2; IV, intravenous; LMWH, low-molecular-weight heparin; PE, pulmonary embolism; T-AT, thrombin–antithrombin complex; UFH, unfractionated heparin; VTE, venous thromboembolism; VQ scan, ventilation-perfusion scintigraphy.
aNapsagatran-treated groups not included.
bGroup treated with delayed warfarin dosing not included.
cT-AT data could not be accurately abstracted from the figures in this source and was thus not included.
Of the included 18 studies, 12 reported the mean values of laboratory markers for the participants studied and 7 reported median values. (One study reported its
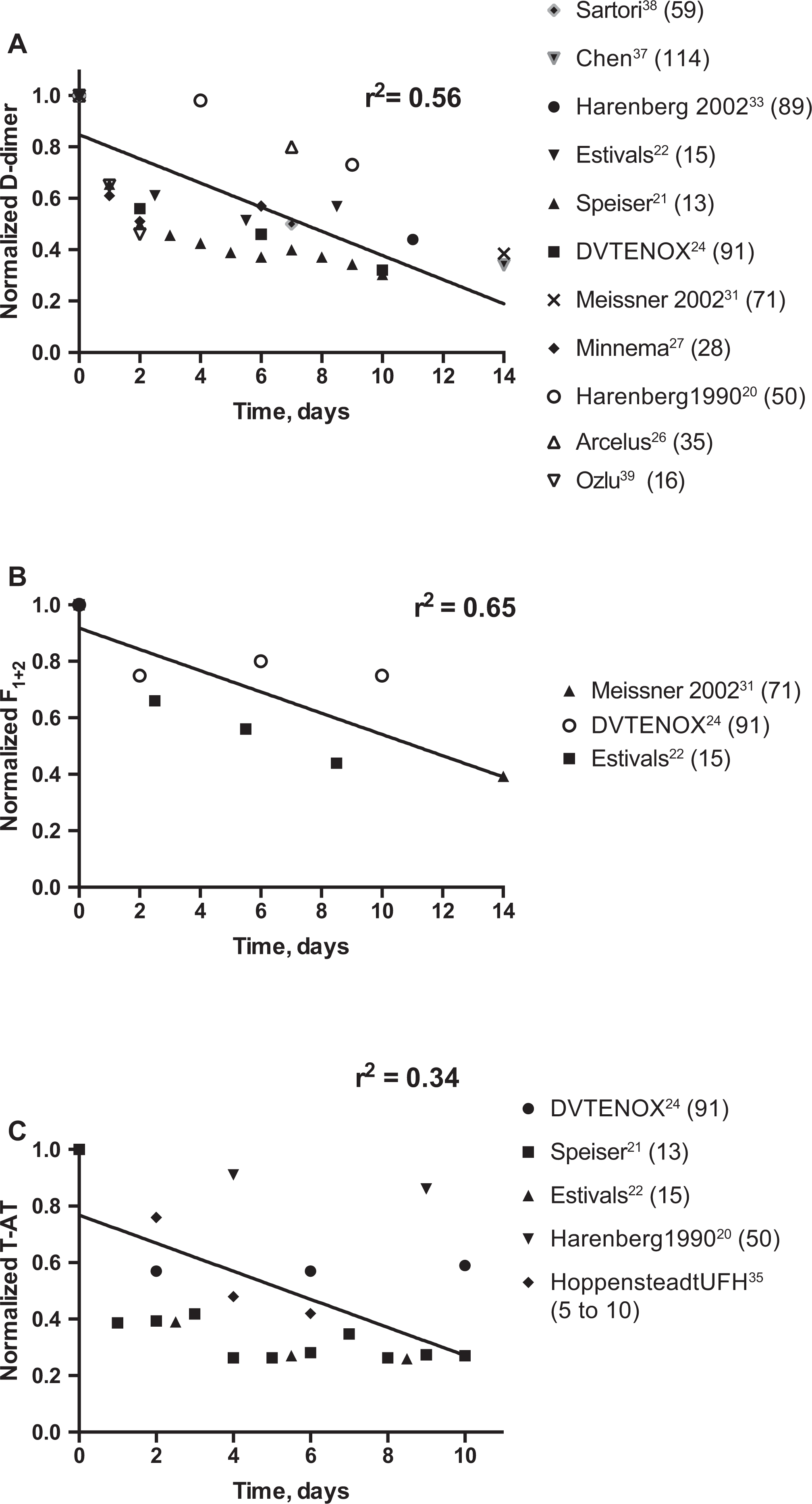
Serial measurements of markers of coagulation system activation in patients receiving UFH or LMWH for venous thromboembolism. A, Normalized
The data of the 7 studies that reported median results were normalized, as shown in Figure 3. Some of the studies reported data for subsets of the study population only and did not report median values of the entire study population; we therefore show the data as reported in the paper. Patients were often grouped based on treatments given (typically low-molecular-weight heparin [LMWH] vs unfractionated heparin) or based on response to treatment. “Responder” in these studies typically indicates stabilization or shrinkage in measured thrombus or the absence of VTE recurrence, though definitions, terminology, and methods of measurement varied by study. Although there were a few exceptions, most markers of thrombin generation in most patient groups declined in these studies during the course of anticoagulation treatment, as was observed in the studies that reported data as mean values.
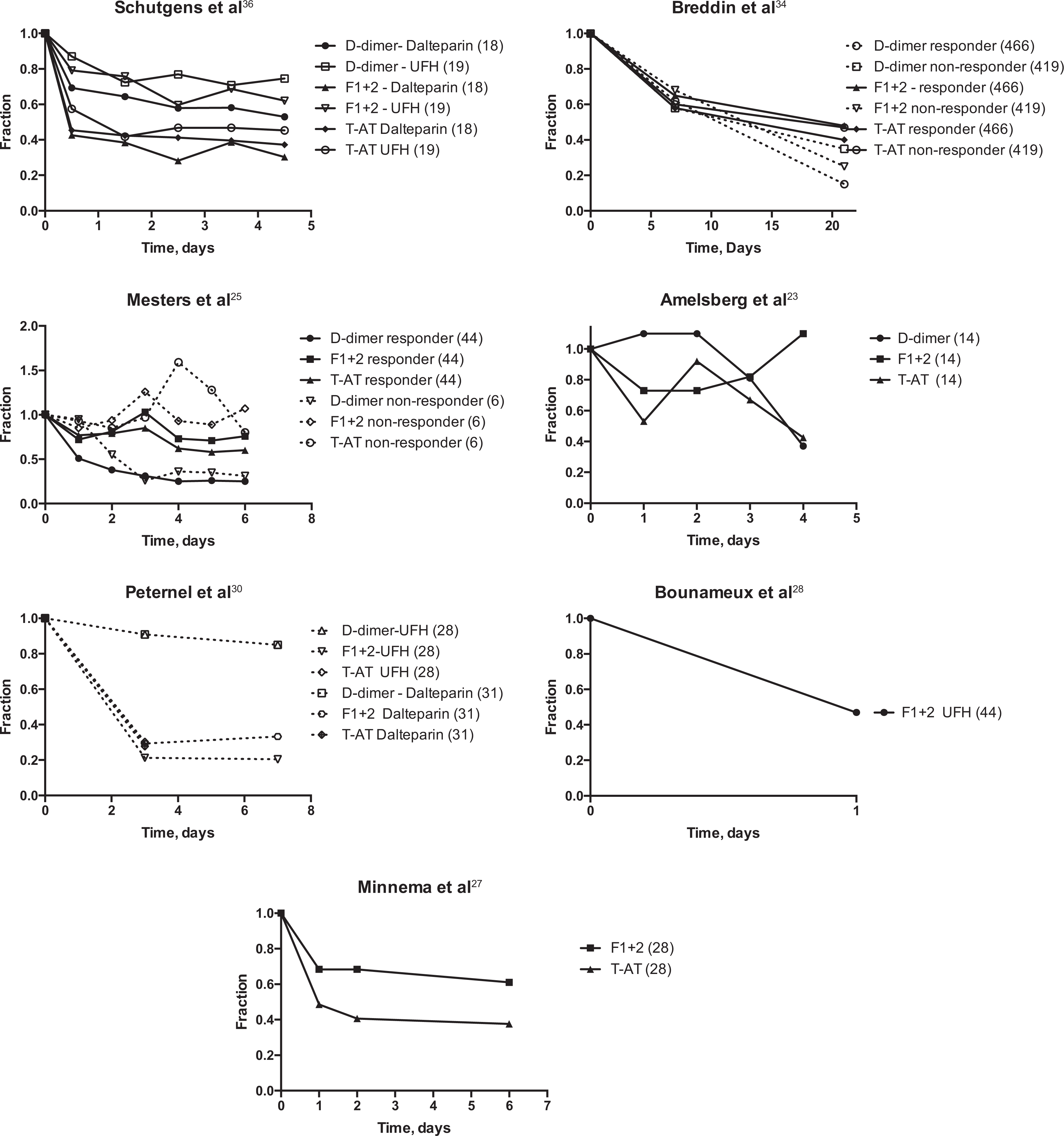
Serial measurements of markers of coagulation system activation in studies reporting median values for patients receiving anticoagulation for VTE. Number of patients in each treatment group are in parentheses. Figure has been adapted by authors from figures in sources indicated. F1+2 indicates prothrombin fragment F1+2; T-AT, thrombin–antithrombin complex; UFH, unfractionated heparin; VTE, venous thromboembolism.
Discussion
In our practice, we observed a serial fall in
The
The decline in all 3 markers over the first 14 days of anticoagulation suggests that one or more might be used as biomarkers for the effectiveness of anticoagulation. Since F1+2 is metabolized by the kidney, fluctuations in renal function may render it a less useful test in medically complex patients. Both the F1+2 and T-AT levels are very sensitive to activation of blood coagulation, and it is possible that routine hospital phlebotomy (rather than the careful techniques used in the published studies) may result in spuriously elevated results, particularly in those with poor venous access or who require blood draws through in-dwelling intravenous catheters.
Using biomarkers to monitor anticoagulation efficacy will not be needed in all patients but may be useful in specific subgroups of patients. We discuss briefly 5 groups of patients whose care might be improved by the use of thrombin generation biomarkers to optimize anticoagulation therapy.
Atrial Fibrillation
A meta-analysis
41
of published trials comparing the new oral anticoagulant agents (NOACs) with warfarin in patients with atrial fibrillation showed that the pooled risk of stroke or embolic events was 3% in patients treated with NOACs and 3.8% in patients treated with warfarin. It has long been recognized that atrial fibrillation is associated with an increase in markers of coagulation system activation and that persistent elevation may be associated with therapeutic failure.
42,43
Adjustment of drug therapy to optimize a marker of in vivo anticoagulation, such as the
Left Ventricular Assist Devices
Left ventricular assist devices are an increasingly common intervention utilized for patients with heart failure. Although the Edmonton Anticoagulation and Platelet Inhibition Protocol, which is based upon thromboelastography, is used for pediatric monitoring,
44
there is little (if any) consensus on the management of adults.
45
There is considerable discordance between anti-Xa levels and aPTT levels in these patients,
46
leaving open the question (as with general treatment of VTE) of which laboratory “target” should be used. Personal observations suggest that in some patients, pump thrombosis may be preceded by a rise in
Obstetric Patients
Although it has been recognized for a number of years that the dose of LMWH required by pregnant patients to maintain “therapeutic” plasma levels increases during pregnancy, there are no data to indicate that maintaining such levels improves outcomes.
Antiphospholipid Antibody Syndrome
Some patients with antiphospholipid antibody syndrome experience recurrent thrombosis despite seemingly adequate anticoagulation treatment. There is little data to indicate how they are best managed. 53 The only studies that attempt to provide guidance to managing clinicians were based upon the use of higher international normalized ratio target ranges, 54,55 and uncertainty remains about the benefits of this approach for the majority of patients. 56,57 A recent report of 151 lupus anticoagulant-positive patients indicates that 20% had recurrent events, half of which were arterial. 58 A variety of “antithrombotic” and “anticoagulant” therapies are used in these patients. Monitoring the degree of on-going thrombin generation in such cases might provide insights into the roles of both anticoagulant and antiplatelet drugs in fibrin generation.
Patients With Cancer
The management of patients with cancer having recurrent thrombosis remains a difficult problem, 59 with few data to drive decision-making. Changing treatment regimens, an evolving inflammatory response to tumor burden, and coexisting clinical morbidities (such as infectious episodes) characterize their course. An individualized (or “personalized”) approach to care, utilizing coagulation system activation markers to decide upon doses of anticoagulant drugs, may result in fewer recurrences in those who experience thrombosis while receiving standard doses of anticoagulants.
Our systematic literature review includes patients with acute VTE for whom only unfractionated heparin or LMWH was used for initial treatment. We anticipate that a decrease in markers of coagulation system activation would also be found if large numbers of patients are studied prior to and after the initiation of therapy using other types of anticoagulants, such as parenteral or oral direct thrombin and Xa inhibitors. Indeed, reports documenting a decline in
Although large controlled studies of clinical outcomes are always desired before adopting new clinical management tools, the financial and logistical requirements of performing studies of even a single thrombin generation biomarker with the ever-increasing number of anticoagulant drugs on the market would be formidable. Establishing that one or more biomarkers of coagulation system activation provide a suitable surrogate for relevant clinical end points would allow investigators to conduct studies in patient subsets during the routine administration of medical care, without pharmaceutical industry backing. Without such studies, clinicians will continue to be dependent upon expert opinion and observations from large drug company-sponsored registration studies that (necessarily) do not include smaller patient populations of interest. Using each patient’s baseline values may be a way of overcoming the objection that tests for thrombin generation biomarkers are not standardized. Although following markers of coagulation system activation will not address the problem of bleeding in patients treated with these drugs, many specialties of medicine may find it helpful to utilize physiologically based measures of anticoagulation efficacy.
Supplemental Material
Supplemental_Appendix - Thrombin Generation Biomarkers Decline With Parenteral Anticoagulation—An Overlooked Means of Anticoagulation Monitoring?
Supplemental_Appendix for Thrombin Generation Biomarkers Decline With Parenteral Anticoagulation—An Overlooked Means of Anticoagulation Monitoring? by Craig P. McFarland, and Stuart E. Lind in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Note
C.P.M. designed and carried out the research, analyzed the data, and wrote the manuscript. S.E.L. designed the research, analyzed the data, and wrote the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
