Abstract
Background
Direct oral anticoagulants (DOACs) have been widely applied in adults for thrombosis prophylaxis. However, the effect of DOACs in pediatric patients with congenital or acquired heart diseases who need anticoagulation therapy remains unclear.
Methods
We systematically searched the databases of PubMed, Embase, and the Cochrane Library, as well as the ClinicalTrials.gov registry and the World Health Organization's International Clinical Trials Registry Platform until June 2024 to identify relevant randomized clinical trials (RCTs). If the number of included studies was less than 5, we performed a narrative review to assess the effect of DOACs in pediatric patients.
Results
Four studies were included. In the UNIVERSE study, thrombotic events occurred in 2% of the rivaroxaban group and 9% of the aspirin group, with bleeding events in 36% and 41%, respectively. The ENNOBLE-ATE study showed no thromboembolic events in the edoxaban group and 1.7% in the SOC group (rate difference: −0.07%, 95% CI: −0.22 to 0.07%). Major bleeding rates were similar (rate difference: −0.03%, 95% CI: −0.18 to 0.12%). The SAXOPHONE trial showed no thromboembolic events in either group and similar major bleeding rates (−0.8%, 95% CI: −8.1 to 3.3%). In the DIVERSITY trial, 81% of dabigatran patients achieved the primary outcome versus 59.3% in the SOC group (Odds ratio: 0.342, 95% CI: 0.081–1.229). No major bleeding occurred in either group.
Conclusion
Existing studies suggest that the use of DOACs hold promise as an effective and safe alternative for preventing and treating thromboembolism in pediatric patients with heart conditions.
Introduction
Pediatric heart disease encompasses a wide spectrum from congenital malformations to acquired conditions, predispose this vulnerable population to an increased risk of thromboembolism,1,2 due to a number of mechanisms including altered hemodynamics, abnormality in vessel wall integrity, the need for surgical intervention and medical device, polycythemia and hyperviscosity, and hypercoagulable states. Notable examples include Fontan procedure/shunt-dependent single ventricle defect, Kawasaki disease with giant coronary aneurysms and intracardiac/intravascular devices. 3 In order to prevent and treat thromboembolic events, anticoagulation therapy is recommended. The current Standard of care (SOC) including vitamin K antagonists (VKAs) and low-molecular-weight heparins (LMWHs) are however associated with certain limitations. The need for frequent dose monitoring due to narrow therapeutic window, possible drug and food interactions, and the lack of suitable pediatric formulation collectively impose significant burden and risk on the patients and healthcare providers. An effective, safe and easy-to-use alternative is therefore highly heralded.
Direct oral anticoagulants (DOACs) represent a newer class of anticoagulants that have transformed clinical practice since the approval of its first drug dabigatran by FDA for adult use in 2010. 4 The paediatric population witnessed the advent of the first approved DOAC dabigatran in 2021 closely followed by rivaroxaban. 5 The other two DOACs apixaban and edoxaban are less explored in children with relatively limited clinical data available, and therefore not yet approved for this population. DOACs offer several potential advantages compared to conventional treatments, including predictable pharmacokinetics, fewer drug interactions, and elimination of frequent monitoring requirements. However, their application in the paediatric setting, especially among patients with heart diseases, remain in the nascent stage, with limited evidence to guide clinical decision making.
This systematic review aims to synthesize existing evidence on the use of DOACs in children with cardiac conditions requiring antithrombotic treatment or thromboprophylaxis by critically analyzing data from recent clinical trials. We endeavor to provide clinicians and caregivers with the latest state of knowledge and hopefully illuminate future research directions for this particularly vulnerable population.
Methods
Inclusion and Exclusion Criteria
According to the PICOS (Population, Intervention, Comparison, Outcomes, Study design) framework, our eligibility criteria included: i) Population (P): pediatric patients diagnosed with cardiac diseases, including congenital or acquired conditions, who are at an elevated risk of thromboembolic events and necessitate anticoagulant prophylaxis; ii) Intervention (I): administration of DOACs, specifically dabigatran, rivaroxaban, apixaban, or edoxaban; iii) Comparison (C): standard of care treatments such as VKAs and LMWHs; iv) outcomes (O): the efficacy and safety outcomes related to the use of DOACs in this patient population, which include but are not limited to rates of thromboembolic events, bleeding events, and other adverse outcomes as defined by the original studies; and v) Study design (S): RCTs or observational studies that assessed the impact of DOACs in the specified pediatric population. We excluded those studies focusing on pharmacokinetics, and studies with no efficacy or safety outcomes were also excluded.
Literature Search Strategies
We systematically searched the databases of PubMed, Embase, and the Cochrane Library (CENTRAL), as well as the ClinicalTrials.gov registry and the World Health Organization's International Clinical Trials Registry Platform (ICTRP) until June 2024 to identify relevant RCTs. Three kinds of search terms were applied as follows: (i) “non-vitamin K antagonist oral anticoagulants” OR “direct oral anticoagulants” OR “novel oral anticoagulants” OR “dabigatran” OR “rivaroxaban” OR “apixaban” OR “edoxaban”; AND (ii) “pediatric” OR “children” OR “childhood”; AND (iii) “heart” OR “cardiac”. The detailed search strategies are presented in Supplemental Table 1. There were no restrictions on the languages in which the literature could be published.
Study Selection
After searching the PubMed and Embase databases, two reviewers screened the titles and abstracts of the retrieved studies. According to the predefined exclusion criteria, we initially deleted some studies if they were animal experiments, case reports, reviews, meta-analyses, comments, and conference abstracts, or other irrelevant articles. After that, we selected eligible studies during the full-text screening process. Any disagreement would be resolved by discussion with each other, or consultation with the senior author.
Data Extraction
The following baseline characteristics of included studies of this systematic review were extracted, mainly including authors and publication years, registry number of RCT, study population (sample size, age, and sex), study design, data source, DOAC type, comparators, the studied outcomes and their definitions, and treatment time.
Risk of Bias Assessment
The Cochrane's Risk of Bias tool 6 was used to assess the bias risk of RCTs. For each of these domains, the risk of bias was categorized as ‘low’, ‘unclear’, or ‘high’.
Narrative Review
Our systematic review followed the guidelines outlined in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). In instances where the number of studies meeting our inclusion criteria was insufficient (less than 5), we opted for a narrative review. A narrative review allows for a qualitative analysis of the available literature, providing a descriptive and contextual understanding of the topic at hand.
Results
Study Selection Process
Figure 1 shows the flow chart of the systematic process in the study selection of this review. In total, 637 studies were identified that were searched in the databases. A total of 616 studies were excluded after the title-/abstract- screenings, and the remaining 21 studies were included in the full-text screenings. Among them, we excluded 17 studies due to the following reasons: (i) RCTs on the treatment of DOACs in pediatric patients with acute venous thromboembolism or acute lymphoblastic leukaemia/lymphoma (n = 8),7–14 and (ii) 9 single-arm observational studies.15–23 Finally, a total of 4 studies (3 RCTs24–26 and 1 post hoc analysis of RCT 27 ) were included in this review.
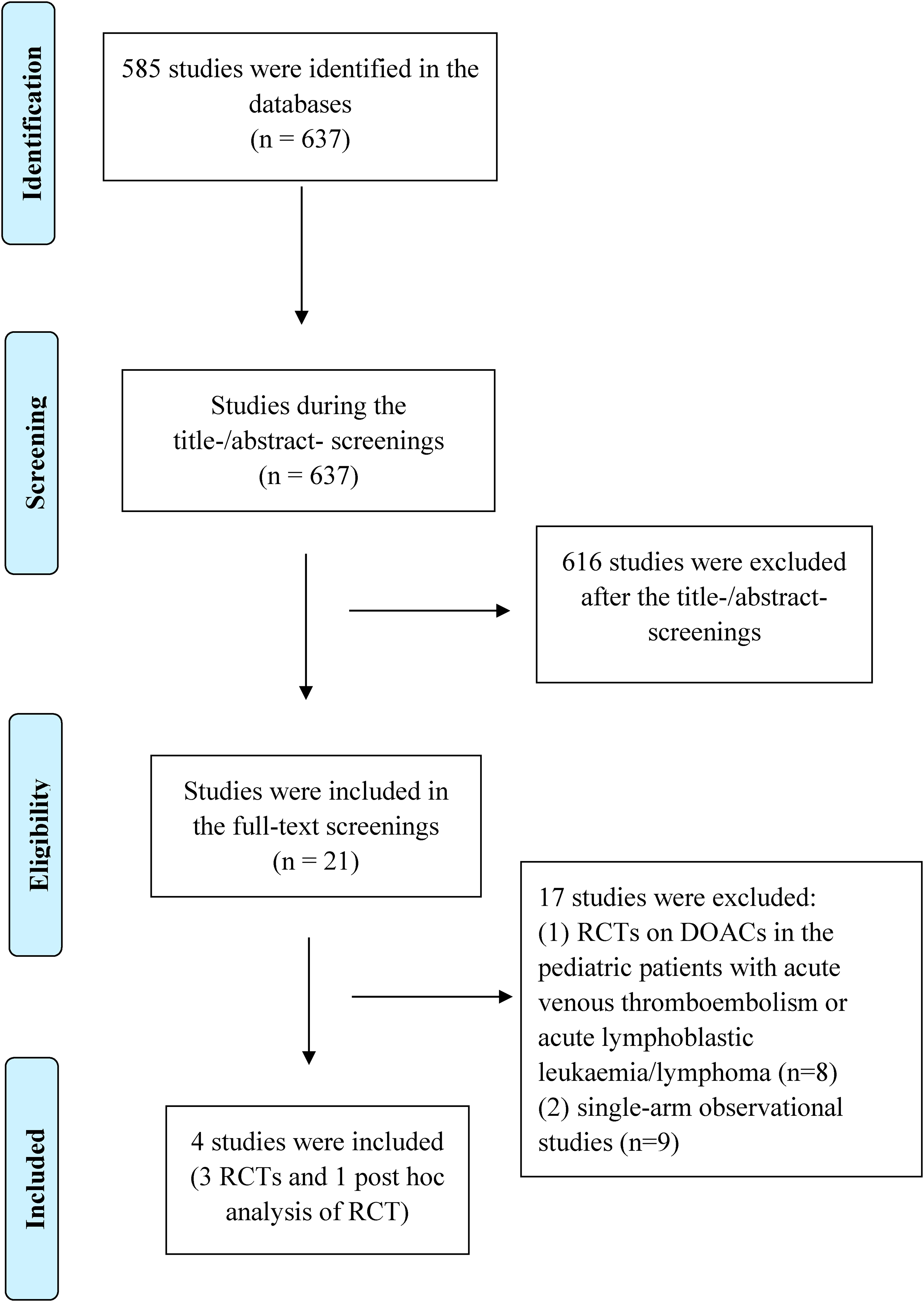
The flow chart of the systematic process in the study selection of this review.
Study Characteristics
Table 1 shows the baseline characteristics of the included studies. There were 3 pivotal RCTs enrolled pediatric patients (age of < 18 years) with cardiac diseases, namely UNIVERSE (rivaroxaban; NCT02846532), SAXOPHONEN (apixaban; NCT02981472), and ENNOBLE-ATE (edoxaban; NCT03395639). The remaining one was a post hoc analysis of the pivotal pediatric trials on dabigatran for the treatment of acute venous thromboembolism (DIVERSITY; NCT01895777) and secondary prevention for venous thromboembolism (NCT02197416). The results of the included studies are summarized in Table 2.
the Baseline Data of the Included Studies in This Review.
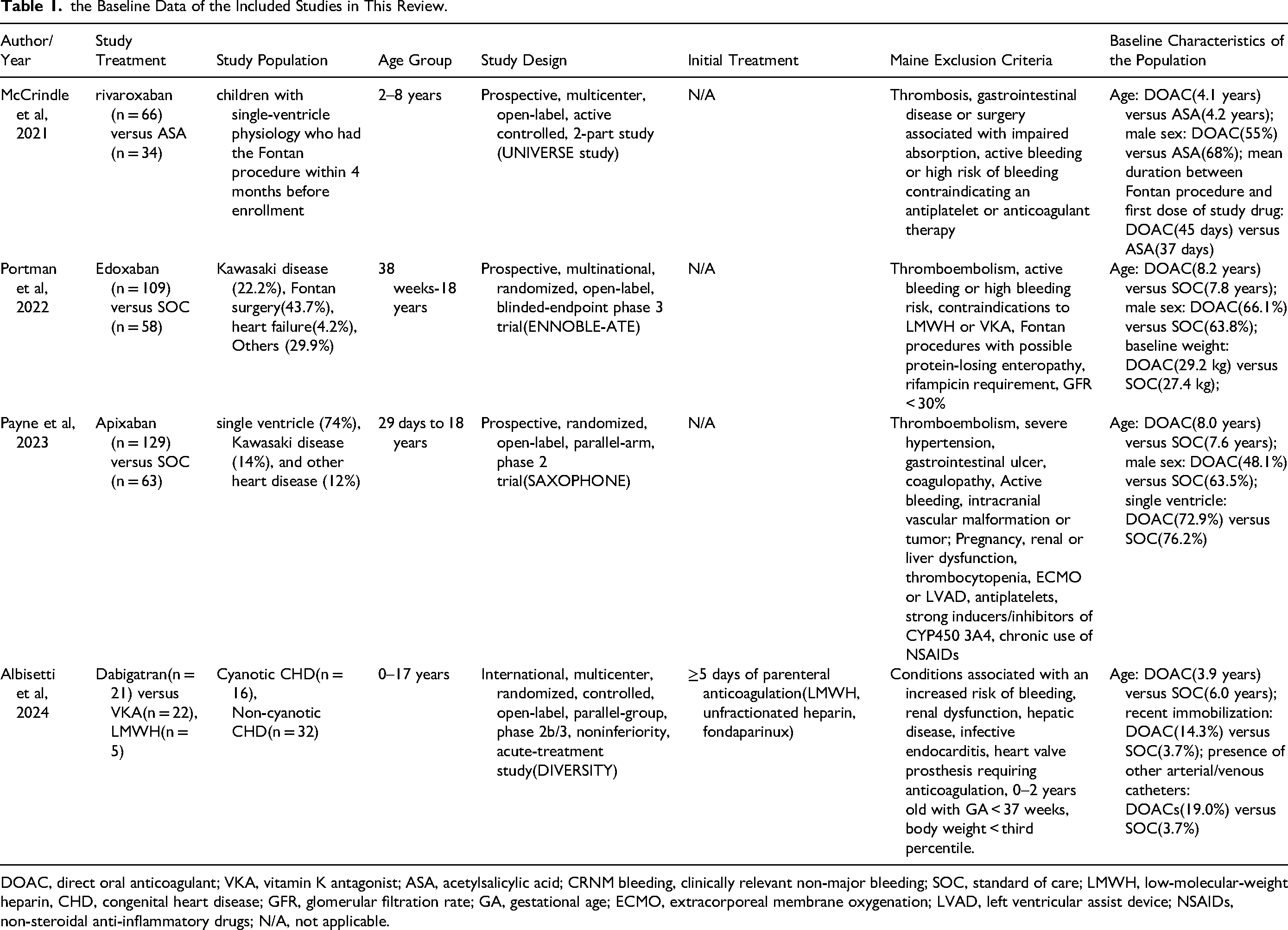
DOAC, direct oral anticoagulant; VKA, vitamin K antagonist; ASA, acetylsalicylic acid; CRNM bleeding, clinically relevant non-major bleeding; SOC, standard of care; LMWH, low-molecular-weight heparin, CHD, congenital heart disease; GFR, glomerular filtration rate; GA, gestational age; ECMO, extracorporeal membrane oxygenation; LVAD, left ventricular assist device; NSAIDs, non-steroidal anti-inflammatory drugs; N/A, not applicable.
The results of the included studies in this review.
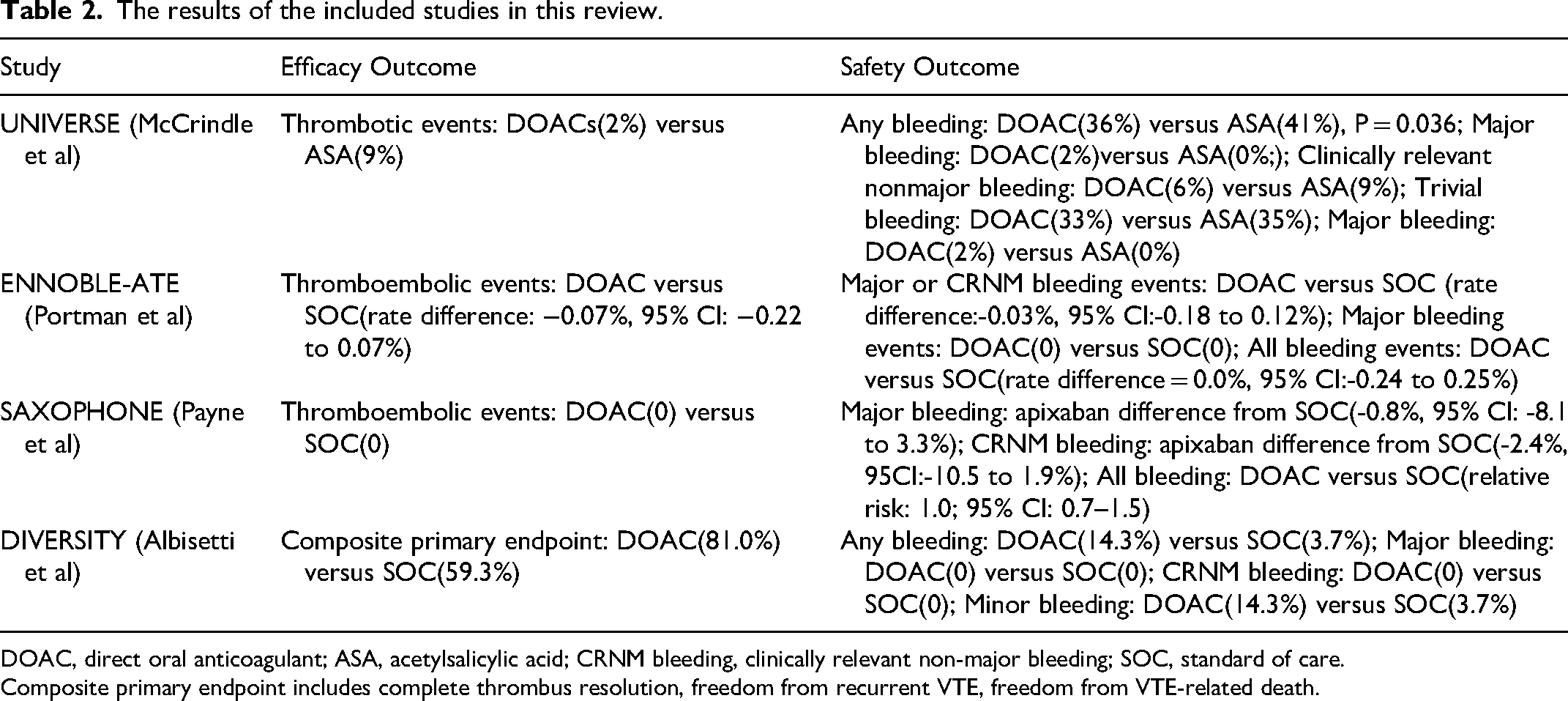
DOAC, direct oral anticoagulant; ASA, acetylsalicylic acid; CRNM bleeding, clinically relevant non-major bleeding; SOC, standard of care.
Composite primary endpoint includes complete thrombus resolution, freedom from recurrent VTE, freedom from VTE-related death.
The patient population primarily consisted of children with congenital or acquired heart diseases, including conditions like single ventricle physiology, Kawasaki disease, and other heart defects. The study by McCrindle et al exclusively included children with single ventricle physiology who underwent Fontan procedure. Whereas the study by Albisetti et al included children with objectively confirmed VTE with or without CHD, but no information regarding acquired heart conditions were available, we therefore only focused on the CHD population.
In terms of intervention design, the DOACs investigated across the studies included dabigatran, apixaban, and edoxaban and the control groups received SOC which consisted of VKAs and LMWHs, with the exception of the study by McCrindle, in which the control group was treated with acetylsalicylic acid(ASA). Treatment duration and follow-up varied, with some studies observing patients for up to 12 months. The age of participants ranged from 28 days to 18 years. The mean age varied by study and treatment group, from 3.9 to 8.2 years old. Most key characteristics were fairly balanced between the interest group and control group across the studies. Supplemental Table 2 provides a comprehensive evaluation of the RCTs. Notably, all studies included in the analysis demonstrated a low risk of bias. Supplemental Table 3 presents the pharmacologic properties of the included DOACs.
Pivotal Trials on DOACs in Patients with Cardiac Diseases
The UNIVERSE trial was a clinical research study that focused on primary prevention, and thus, the trial compared the effectiveness of rivaroxaban with aspirin. On the other hand, the SAXOPHONEN trial was a phase 2 trial, meaning it was conducted to assess the safety and effectiveness of apixaban compared to the standard of care. Similarly, the ENNOBLE-ATE trial was a phase 3 trial that compared the efficacy of edoxaban with the standard of care, aiming to evaluate whether edoxaban performed better when compared to the currently accepted standard treatments. Additionally, what sets the ENNOBLE-ATE trial apart is the fact that it had an extension arm, aiming to evaluate the sustainability and long-term effects of edoxaban compared to the standard of care.
The UNIVERSE study focused exclusively on post-Pontan procedure children with single ventricle physiology. 100 participants were randomized 2:1 to receive either rivaroxaban or ASA. The study showed rivaroxaban reduced the rate of the thrombotic events to 2%, compared to 9% in the ASA group over a 12-month period. Major bleeding event was seen only in 1(2%) patient in the rivaroxaban group, but not in the ASA group. CRNM bleeding occurred less frequently in the rivaroxaban group than in the ASA group(6% vs 9%). The proportion of patients suffered from any bleeding events appeared to be comparable between both groups.
The SAXOPHONEN study included a total of 192 infants and children, 129 and 63 of which were treated with apixaban and SOC, respectively. No thromboembolic events were reported in either the apixaban or the control group. The apixaban group seemed to be associated with a significantly lower incidence of bleeding events. The reported major bleeding rates were 0.8%(95% CI: 0.0%-4.3%) in the apixaban arm, compared to 1.6%95% CI: 0.0%-8.7%) in the SOC arm. Clinically relevant non-major(CRNM) bleeding also occurred at a much lower rate in the apixaban arm(difference:-2.4, 95% CI: −10.5 to 1.9). Both groups exhibited similar rates of bleeding events, with each arm reporting a rate of 37%.
The ENNOBLE-ATE trial by Portman et al, consisting of 109 patients in the edoxaban arm and 58 patients in the SOC arm, found that during the main treatment period thromboembolic events happened in 0 patients in the edoxaban group and 1 patient (1.7%) in the SOC group. CRNM bleeding occurred in 0.9% of patients in the edoxaban group, as compared to 1.7% in the SOC group, providing annualized rates of 0.04 and 0.07%. No major bleeding was documented in both groups. During the extension period (edoxaban treatment), 1(0.7%) CRNM bleeding event and 2(2.8%) thromboembolic events were observed.
Post hoc Analysis Data on DOACs in Patients with Congenital Heart Disease
The post-hoc sub group analysis of the DIVERSITY trial on patients with CHD by Albisetti et al included 48 patients, 21 of which were treated with dabigatran and 27 with SOC. 16 patients were classified as having cyanotic CHD, where the rest were diagnosed with non-cyanotic CHD. The average age of the dabigatran group was younger than the SOC group. In terms of the effectiveness outcome, the composite primary endpoints defined by complete thrombus resolution, freedom from recurrent VTE, freedom from VTE-related death, were achieved by 81.0% and 59.3% of patients in the dabigatran and SOC group respectively. Major bleeding and CRNM bleeding did not occur in either group, minor bleeding events were seen in 14.3% of patients in the dabigatran group, compared to 3.7% of patients in the SOC group. Patients with cyanotic CHD exhibited poor response to the anticoagulation effect of both dabigatran and SOC compared to patients with non-cyanotic CHD, but dabigatran was still associated with higher rate of achieving the primary endpoint in both subgroups.
Discussion
In our systematic review, data from 3 RCTs and one post-hoc analysis of an RCT were gathered and examined to assess the effectiveness and safety of DOACs in paediatric patients with heart disease. The results indicate that DOACs are at least effective, if not more so, compared to the traditionally used antithrombotic agents including aspirin, VKAs and LMWHs in reducing the rate of thromboembolic events. Furthermore, the incidence of bleeding events appeared to be comparable between DOACs and SOC, with major and CRNM bleeding remaining low in both groups.
Several types of congenital or acquired cardiac diseases frequently occur in children. Repair of single ventricle congenital heart disease with a Fontan procedure may result in the risk of thromboembolism, which needs antithrombotic agents. Acquired cardiac disease such as Kawasaki disease with giant coronary artery aneurysms or dilated cardiomyopathy may also be at high risk of thromboembolic events. However, anticoagulation therapy remains an urgent clinical issue in these scenarios. For adult congenital heart disease with Fontan circulation, although prior observational studies and meta-analysis have demonstrated that the effectiveness and safety of DOACs are comparable to VKAs or LMWH,28–31 the evidence level is relatively low due to the lack of RCT data. In children with heart diseases, our current systematic review included the prospective UNIVERSE (rivaroxaban), ENNOBLE-ATE (edoxaban), and SAXOPHONEN (apixaban) trials24–26 with a total of 471 participants, suggesting that DOACs at least had similar efficacy and safety compared to VKAs or LMWH in the prevention of thromboembolism. In the SAXOPHONEN trial, the pharmacokinetics and pharmacodynamics of apixaban were also assessed, and their data were consistent with adult NOAC trials.24,32 Further study should include more sample size to confirm our findings in the pediatric clinical practice. In addition, the dose and cost-effectiveness of NOAC in children need further examination. 33
RCTs ensure the balance of findings between the treatment groups and acquire a fair assessment of the trial treatment effect. However, the study participants enrolled in RCTs sometimes are not equal to those in real-world settings. A prior meta-analysis has indicated that both data from observational studies and NOAC trials consistently supported the use of DOACs superior to VKAs for stroke prevention in adult patients with atrial fibrillation.32,34–38 In children, whether similar findings of DOACs in RCTs are observed in observational studies remains unclear. After searching the databases, we found that several observational single-arm studies have assessed the effectiveness and safety profiles of DOACs in pediatric patients with cardiac diseases such as Kawasaki disease with giant aneurysms,20,23 congenital heart disease with thrombosis,15,17,18,21 and children awaiting heart transplantation. 39 Most of these studies were case series with no control group, limiting the comparison between RCT and real-world evidence of DOACs in children. Nevertheless, current real-world evidence still supports systemic anticoagulation with DOACs in pediatric patients with cardiac diseases. Observational data regarding the comparison between DOACs and standard of care (VKAs or LMWH) are still extremely limited. Therefore, further multicenter real-world studies with larger sample sizes are still required to investigate the effects of DOACs compared with VKAs or LMWH in pediatric patients with a wider range of patient characteristics and a more extended follow-up period.
Safety Aspects Pertaining to the use of DOACs in Paediatric Patients
Although the occurrence of bleeding events in patients treated with DOACs, especially major and CNRM bleedings remained low or similar compared to the SOC groups, the DIVERSITY trial however reported a significantly higher rate of minor bleedings in the DOAC arm. Therefore, it is crucial to identify the underlying comorbidities, drug-drug interactions (particularly with the inducers and inhibitors of CYP3A4 and/or P-glycoprotein) that could potentially predispose the patients to a higher bleeding risk. Pediatric heart patients are often critically ill, requiring invasive procedures, therefore understanding the appropriate timing for perioperative interruption of DOACs is vital for optimal patient outcome. For low bleeding risk procedures, it is recommended to discontinue DOACs 24 h before surgery and restart on the same day if no bleeding is observed. For high bleeding risk procedures, a 48-h DOAC-free window is suggested before surgery, with a restart 24 h afterward if no bleeding occurs. 40
Age based dosing strategies are necessary to cater specifically to paediatric patients. For example, rivaroxaban dosing in the Einstein Jr phase 3 trial was adjusted based on body weight to match the pharmacokinetic profile achieved in adults. Younger children and infants, particularly neonates, often require tailored dosing due to their distinct pharmacokinetic profiles, characterized by faster clearance rates. These dosing strategies are critical in managing the balance between efficacy and safety, especially in populations like neonates where standard dosing regimens may not be applicable or safe.
The results of our systematic review underscore the potential of DOACs in pediatric patients with congenital or acquired heart conditions to enhance therapeutic outcomes. By offering a comparable or similar effectiveness without increasing the risk of bleeding, DOACs may offer additional advantages compared to traditional approaches and possibly serve as a new alternative. This could lead to simplification of the treatment protocol, reduced need for monitoring and enhanced adherence, which are particularly beneficial in this population as where treatment compliance are often challenging.
Future Directions
Current trial data on DOAC use in pediatric populations are limited, particularly for neonates, premature infants, and patients with specific conditions like caval, renal, or portal vein thrombosis, arterial thrombosis, arterial ischemic stroke, and known antiphospholipid syndrome. These groups require more data due to unique developmental hemostasis, pharmacology, and thrombosis risk factors. Future research should determine if DOACs can be safely and effectively started immediately upon VTE diagnosis without initial heparin-based treatment and establish the appropriate duration of stability in oral feeds before initiating DOACs in infants and postoperatively. Evaluating the potential role of pharmacodynamic monitoring in pediatric populations is crucial, particularly regarding thromboembolic efficacy, bleeding safety, and off-target effects impacting overall safety. Developing clear guidelines for perioperative interruption of DOACs, specifying procedure types and durations for withholding DOACs, and understanding when and how DOAC reversal agents should be used in pediatric patients are essential. Long-term safety outcomes, particularly concerning growth, neurodevelopment, and physiological processes like angiogenesis, should be studied through phase IV trials, postmarketing studies, and long-term registries. Potential long-term sequelae, such as osteoporosis or neurodevelopmental impacts, must be thoroughly investigated. Addressing these gaps will likely require shifting from traditional multicenter randomized controlled trials (RCTs) to well-designed and well-resourced cooperative cohort studies and registries. Initiatives like the ThromPeds Registry through the International Pediatric Thrombosis Network (IPTN) offer a pragmatic approach to gathering real-world data. 41
Cost-Effectiveness of DOACs
DOACs are stereotypically known for its higher cost due to its status as a newer generation of anticoagulants. The economic perspective of DOACs is of great concern in actual clinical practice as the use of which could potentially harden or alleviate the burden on patients, their families and the health care system. Existing literatures42,43 evaluating the cost-effectiveness of anticoagulants in non-valvular atrial fibrillation patients suggest DOACs are associated with higher quality-adjusted-life-year(QALY) improvement as well as lower cost as compared to warfarin. Among the DOACs, apixaban stands out as the most cost-effective option.
Limitations
We acknowledged that this systematic review was not previously registered in the PROSPERO, an international database of prospectively registered systematic reviews. In addition, there were several other limitations within this review that should be acknowledged. Firstly, the number of studies included in our study and overall sample sizes are relatively small, which may limit the generalizability of the results. Secondly, the studies included vary in their design especially their study populations and the specific types of DOACs examined, making direct comparisons challenging. Thirdly, there is also a lack of long-term data on the effectiveness and safety of DOACs in this patient population, which is crucial for chronic conditions typically seen in pediatric cardiac patients. Lastly, this study did not evaluate the cost-effectiveness of DOACs as compared to other anticoagulant agents in pediatric patients.
Conclusions
These findings suggest that DOACs have the potential to serve as an effective and safe alternative to the other agents in preventing and treating thromboembolism in paediatric patients with heart conditions, potentially improving patient outcomes and overall satisfaction. Further results from larger scale RCTs with more inclusive study designs and diverse patient subgroups are needed to confirm our findings.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241271974 - Supplemental material for Systematic Review of Randomized Clinical Trials on Direct Oral Anticoagulants in Pediatric Heart Diseases
Supplemental material, sj-docx-1-cat-10.1177_10760296241271974 for Systematic Review of Randomized Clinical Trials on Direct Oral Anticoagulants in Pediatric Heart Diseases by Chaokun Guan, Linjuan Guo and Shucheng Liang in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contributions
All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
