Abstract
Background
International guidelines recommend at least three months anticoagulation in all patients after acute venous thromboembolism (VTE) and suggest those with unprovoked events be considered for indefinite anticoagulation if the risk of recurrence is high and the risk of bleeding during treatment non-high. Other authors have recently argued against using a dichotomy unprovoked/provoked events to decide on anticoagulation duration and suggest instead using overall risk factors present in each patient as the basis for deciding.
Aim
This sub-analysis of the WHITE study aimed at assessing the reasons for the treatment decisions taken by doctors in different countries.
Results
1240 patients were recruited in 7 countries (China, Czechia, Poland, Portugal, Russia, Slovakia, and Tunisia). Anticoagulation was extended in 51.7% and 49.3% of patients with unprovoked or provoked events (n.s.); stopped in 15.4% versus 28.9% (P < .0001), and changed to antithrombotic drugs (sulodexide or aspirin) in 32.9% versus 21.8% (P < .0001). In the 430 subjects with isolated distal deep vein thrombosis (IDDVT) anticoagulation was stopped in 34.4%, continued in 37.0% (mainly those with post-thrombotic syndrome [PTS]) and switched to antithrombotics in the balance. High risk of recurrence was the most prevalent reason (>83% of cases) given to continue anticoagulation, regardless of nature and site of the index events, followed by risk of bleeding and presence of PTS signs.
Conclusion
On average, attending physicians estimated the risk of recurrence in real life conditions, and the consequent therapeutic decision, using all the information available, not limiting to the location or nature of the index event.
Introduction
Venous thromboembolic (VTE) events tend to recur. Though recurrent events may occasionally occur even during anticoagulation, their rate increases sharply after anticoagulant treatment is discontinued. The risk of recurrence, however, is not the same for all patients with a first VTE episode. It has been calculated that up to one quarter of patients with deep vein thrombosis (DVT) and/or pulmonary embolism (PE) will experience a recurrent VTE event within the next 5 years. 1 To reduce the risk of recurrence, international guidelines recommend extending anticoagulant treatment beyond the duration indispensable for treating an acute VTE episode, ie the first 3 to 6 months (usually called “maintenance” anticoagulation period), in those patients who are at high risk of recurrences and do not present conditions at high risk of bleeding.2–5,6 However, to estimate a patient‘s risk of recurrence and bleeding if anticoagulant treatment is continued is not an easy job for clinicians. The factors affecting the risk of VTE recurrence are many and vary in nature and kind, and data on their actual role are scanty. Furthermore, the individual risk of bleeding in anticoagulated patients is not easy to assess and most of the currently available predictive clinical tools have turned out to be unsatisfactory.7,8 For all these reasons, guideline recommendations are difficult to translate into clinical practice and the optimal duration of anticoagulation after a first VTE episode is still uncertain. In daily clinical practice, the decision on anticoagulation duration is influenced by many factors, such as the physician‘s experience, confidence in guideline recommendations, and patient characteristics and preferences. Recent reports on management of patients with VTE hailing from different countries point to the wide variety of practices among physicians in this regard.9–11
The aim of the WHITE study was to analyze data on these issues collected from everyday clinical practice in a series of countries with significantly different socio-economic conditions and healthcare systems. The population profile and main results, which showed that the unprovoked or provoked nature of the event was not necessarily the main reason to continue or stop the secondary prophylaxis, was already reported. 12 The present article is a sub-analysis of the WHITE study and reports on the factors and reasons guiding the decisions taken by the treating physicians regarding the therapy, if any, they prescribed patients after an initial and maintenance period of treatment post-acute VTE and the reasons for doing so.
Materials and Methods
Study Design, Participants, Study Population, Data Collection
As detailed elsewhere, 12 WHITE was a multicenter, multinational, observational, non-interventional, investigators-initiated, no-profit, prospective study (ClinicalTrials.gov Identifier: NCT04646993), aimed at analyzing clinicians’ decisions on treatment after maintenance therapy in subjects with a first-ever DVT event of the lower limbs and/or pulmonary embolism. The study was performed in 7 countries (China, Czechia, Poland, Portugal, Russia, Slovakia, and Tunisia), with one Country Coordinator for each country inviting local clinical vascular centers to participate in the study and cooperating with local contract research organizations (CRO) to obtain the authorizations needed to join the study.
Patients of any ethnicity, male and female, aged ≥18 years, were eligible for the study if treated with anticoagulant therapy for 3 to 12 months following a documented first-ever event of provoked or unprovoked DVT of the lower limbs and/or of PE. Upon protocol approval by the competent Ethics Committee/Institutional Review Board, subjects provided written informed consent before inclusion in the study. Subjects <18 years old with thrombosis in other sites, unable or unwilling to give written informed consent, were excluded from the study (complete list of inclusion/exclusion criteria in the protocol of the study, available on request). Each subject had the right to pull out of the study at any moment without providing any reason.
Information on participants in the WHITE study were recorded and collected in a structured case report form on a secure, web-based central electronic database designed and managed by Officinebit (Switzerland) and were controlled at the study coordinating center (Arianna Anticoagulazione Foundation) by a dedicated study-monitor (C.M.) who – when necessary – sent data queries to participating sites to ensure complete and accurate reporting. Each patient received a unique identifier and personally identifiable data were removed at the central electronic database to ensure anonymity.
The scope of this report was limited to an analysis of the relationship between index VTE event and patient characteristics and clinician decisions for the management of patients regarding the secondary VTE prevention as well as the reasons for such decisions.
The site of index VTE events was classified in a) DVT, when thrombosis involved proximal veins (with/without involvement of calf deep veins) without diagnosed PE; b) PE, when the presentation was as PE, either associated or not with a DVT (proximal and/or distal); c) IDDVT, when thrombosis was limited to deep calf veins.
Statistical Analysis
Data were analyzed with SPSS version 24 integrated for specific items with R version 3.6.1. All variables were summarized with the usual descriptive techniques and compared, when necessary, with the chi square or Fisher‘s test for nominal variables, and ANOVA, integrated where appropriate with the post-hoc pairwise Tukey HSD test, for the continuous variables. The impact of demographic and prognostic factors – including the country – on the choice was tested by chi square or odds ratio estimate and, where appropriate, logistic regression analysis.
Funding
The WHITE study was promoted by the “Arianna Anticoagulazione” Foundation (Bologna, Italy), and managed together with a Core Team of vascular-expert professionals (the detailed list of the study boards and their make-ups is shown in Appendix 1). The Foundation received an unrestricted research grant from Alfasigma (Bologna, Italy), specifically designed to fund this study.
Results
Baseline Characteristics and Index Events
Patients were included in the WHITE study starting from April 2018 to December 2020, when 1240 patients had been recruited in the study by 79 clinical sites (the list of active clinical centers is reported in Appendix 1), active in 7 countries. As shown in Table 1, the highest proportion of all patients was included in Russia (40.4%), and the lowest in Portugal (3.3%). The enrolled patients were relatively young (mean age ± SD = 56.6 ± 15.5), half of them were women (50.7%). Overall, the index events were prevalently proximal (± distal) DVT (52.6%) followed by isolated distal DVT (IDDVT, 34.7%), while PE (either associated with DVT or isolated) was less prevalent (12.7%). The distribution of index event types was significantly different by country (P < .001), essentially because the rate of the reported PE in Czechia patients (30%) was significantly greater than in all other countries (P = .005). The index VTE events were classified as unprovoked in 696 cases (57.8%) and provoked in the rest. The classification differed significantly across countries (P < .001), with the proportion of unprovoked events significantly below average in China and Czechia (Table 1). Table 2 lists the conditions reported as associated with provoked events. Surgery was the most frequently reported factor, followed by bedrest >4 days, trauma, and limb immobilization.
Active clinical sites and enrolled patients in each country, and sites of index events.

*classification as provoked/unprovoked missing in the 35 patients with isolated PE.
[a] ANOVA; [b] chi square; (%w) denotes % within country.
Conditions reported as associated with provoked events. The table lists all the conditions indicated by the attending physician as possibly associated with provoked events.

Decisions on Treatment Extension
On average, maintenance anticoagulation lasted approximately 6 months, during which time patients were treated with direct oral anticoagulants (DOACs) in 86 to 94% of cases in China, Poland, Portugal, Russia, and Czechia, and in 52% in Slovakia; the remainder received vitamin K antagonists (VKAs), as was the case for all Tunisian patients. Anticoagulation duration was slightly longer in patients with unprovoked versus provoked events, but the difference was statistically significant only in Czechia (P = .005) and Slovakia (P = .005; Supplemental Table S-1).
We already reported [12] that the attending physicians decided to stop anticoagulant treatment in 255 (20.6%) patients while extended anticoagulation (whatever the drug used) was prescribed in 636 (51.3%), and antithrombotic drugs - principally sulodexide (N = 273) or antiplatelets (N = 84) - in the remaining 349 (28.1%) patients (Table 3). Decisions differed significantly among countries. All specific treatments were stopped in only a very few patients in Slovakia (3.1%), but in up to 40% of patients in Czechia (Figure 1). Extended anticoagulation was prescribed in a range stretching from 25.7% of patients in Czechia to 68.3% in Portugal. Replacement of anticoagulants with antithrombotic agents was prescribed in a minority of patients in Portugal (4.9%) and China (16,1%) but involved one-third of patients in the other countries, reaching 60.2% of subjects in Slovakia; we have already evidenced [12] that the impact of the unprovoked or provoked nature of the index event on the therapeutic decisions differed from one country to another (Supplemental Table S-3). Overall, anticoagulation was stopped in 15.4% and 28.9% and continued in 51.7% and 49.3% of patients with unprovoked or provoked events, respectively. Anticoagulation was stopped in a minority of cases (about 13%) and extended in more than half of the patients when the index event was proximal DVT or PE (Figure 2). Anticoagulation was stopped in one-third and extended in a similar proportion of IDDVT cases. Prescription of antithrombotics was similar (about one-fourth of patients) in all types of events.

Distribution by country of the decision for the continuation of prophylaxis. The solid lines indicate the average proportion of decision in the total sample. “Stop” denotes the decision to interrupt whatever specific pharmacological treatment; “anticoagulation” denotes the decision to continue with the same or other anticoagulant at full or reduced dose; “antithrombotic” denotes the decision to replace the anticoagulant with a non-anticoagulant antithrombotic, mostly sulodexide or ASA.
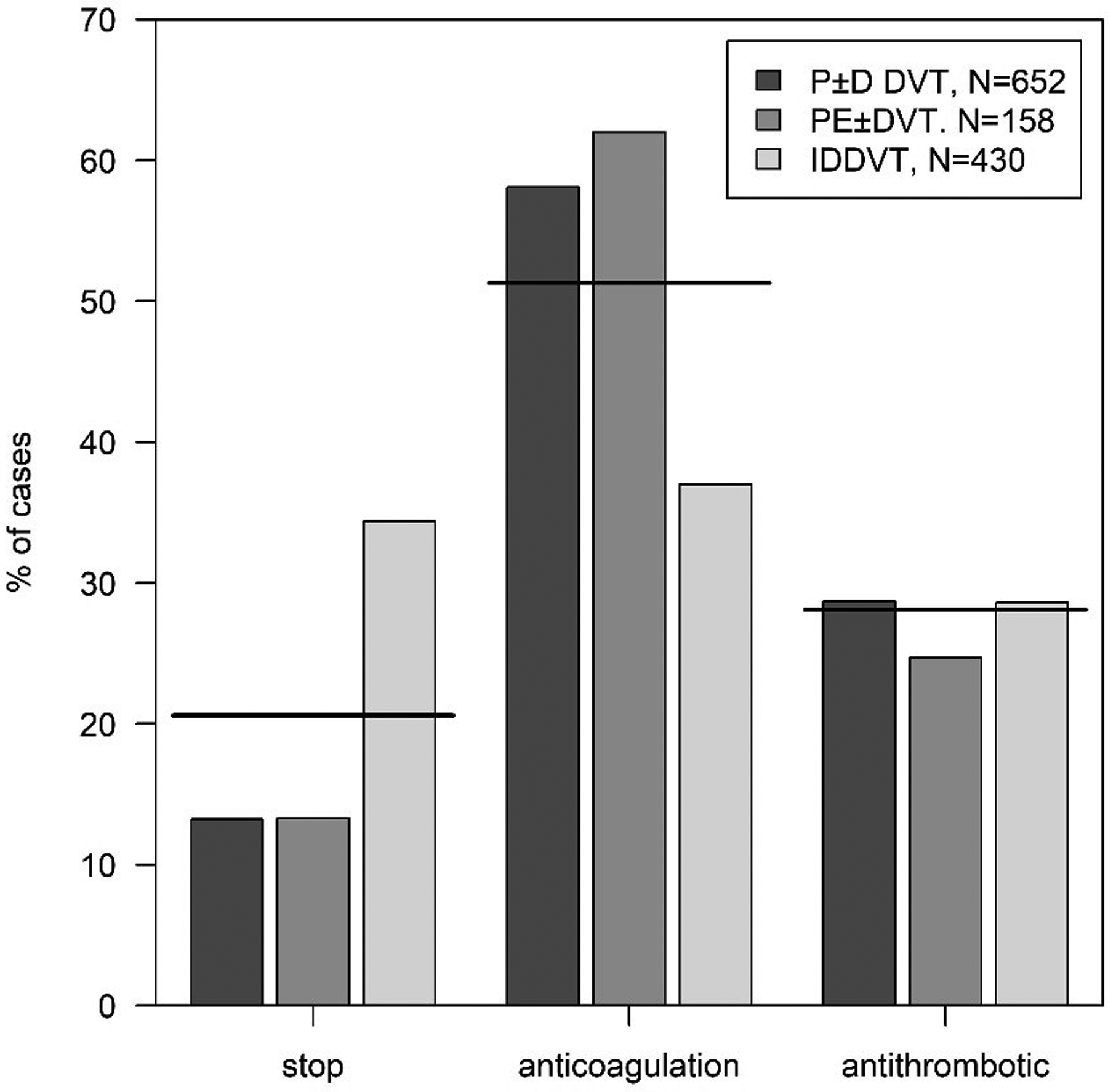
Distribution of the decision for the continuation of prophylaxis stratified by index event. The solid lines indicate the average proportion of decision in the total sample (N = 1240). “Stop” denotes the decision to interrupt whatever specific pharmacological treatment; “anticoagulation” denotes the decision to continue with the same or other anticoagulant at full or reduced dose; “antithrombotic” denotes the decision to replace the anticoagulant with a non-anticoagulant antithrombotic, mostly sulodexide or antiplatelets. P ±D DVT indicates proximal or proximal plus distal DVT; PE ± DVT indicates pulmonary embolism with or without DVT; IDDVT indicates isolated distal DVT.
We investigated the potential impact of factors such as sex, age (<65 /65-74/ ≥75 years), nature (unprovoked/provoked) and site (IDDVT/DVT/PE) of index event, concomitant diseases (no/yes), cardiovascular risk factors (absent/present) and country on the odds of deciding for extended anticoagulation or treatment with other antithrombotics rather than discontinuing treatment (Supplemental Table S-4). Age, sex, or the presence of cardiovascular risk factors did not significantly affect the decision. The odds for continuing anticoagulation rather than stopping it were 2.71 [1.59 to 4.61; P < .001] in subjects with PE versus those with DVT, 1.49 [1.07 to 2.06; P = .018] in subjects with concomitant diseases versus those without, and 3.98 to 19.49 in all countries (except Czechia) with reference to Tunisia (the only country where DOACs were not available). When continuing with the same anticoagulant, the dose was maintained in 83% of cases, and reduced in just 17%. In approximately 85% of cases the anticoagulants used for long term treatment were DOACs.
Limiting the analysis to the 1205 subjects with DVT, the odds of continuing with anticoagulation rather than stopping it were 3.57 [2.56 to 4.99; P < .001] in those with proximal DVT or DVT + PE versus those with IDDVT, 1.72 [1.24 to 2.38; P = .001] in subjects with unprovoked versus provoked DVT, and 4.41 [2.76 to 7.05; P < .001] in subjects with signs/symptoms of post-thrombotic syndrome (PTS) – assessed using the Villalta score an average 174 days after the index event – after accounting for sex, age, concomitant diseases, cardiovascular risk factor, and country. The odds of continuing with other antithrombotics rather than stopping treatment was 1.68 [1.14 to 2.48; P = .009] in subjects with concomitant diseases versus those without, 2.46 [1.71 to 3.55; P < .001] in those with proximal DVT or DVT + PE versus those with IDDVT, 2.44 [1.70 to 3.51; P < .001] when DVT was unprovoked versus provoked, and 2.48 [1.50 to 4.12; P < .001] in subjects with PTS versus those without.
Focusing on patients with IDDVT, we examined (using contingency tables; Supplemental Table S-5) whether certain factors might have influenced the decision to continue anticoagulation in 37% of subjects. We excluded a significant effect of the classification of the index event as provoked or unprovoked (P = .059), of age (P = .855), sex (P = .123), concomitant disease (P = .183), cardiovascular risk factors (P = .064), duration of maintenance anticoagulation (≤ 6 vs > 6 months; P = .074) and the nature of anticoagulant used during maintenance period (P = .313 for DOACs; P = .279 for VKA).
PTS on the other hand was a strong predictor for continued anticoagulation (P < .001): the subjects with PTS had an odds ratio of 2.63 [1.49 to 4.68] for continuing anticoagulation, versus those without. A significant factor was country (P < .001). China and Slovakia continued anticoagulation in a substantial proportion of IDDVT patients (49.3% and 47.6%, respectively), Russia, Poland, and Portugal in an appreciable proportion of them (35.6%, 30.9% and 30.8%), while the other countries in only a limited number of cases: 20.0% in Tunisia, and none in Czechia (Figure 3).

Proportion of patients with isolated distal DVT, who were prescribed continued anticoagulation at the end of the maintenance period, stratified by country. The distribution is significantly different (P < .001).
Reasons Justifying the Decisions
The investigators failed to give reasons for their decisions in 104 subjects. A few reasons were indicated in over one-tenth of subjects: risk of recurrence (73%), risk of bleeding (42%), no need for further treatment (24%), patient choice (23%), negative D-dimer assay (20%) and presence of PTS signs (17%). Patient age (9%), cost for patient (6%), positive D-dimer (6%), presence of thrombophilic alterations (5%) and active cancer (5%) were indicated in 5 to 10% of subjects. The more frequent reasons physicians gave for their therapeutic decisions (more than one reason in > 60% of patients) are listed in Table 4. For patients who were asked to extend anticoagulation, a high risk of recurrence was the most prevalent reason, reported in > 83% of cases, regardless of nature and site of the index events, followed by risk of bleeding and presence of PTS signs. No need for further treatment was the most frequent reason given in patients stopping anticoagulation, followed by risk of recurrence and patient choice. A high risk of recurrence was the main reason given in patients to be treated with antithrombotics, followed by a high risk of bleeding and negative D-dimer results.
Distribution of clinical decisions for the treatment of all patients beyond the maintenance phase.

The most frequent reasons for justifying treatment decisions; N (%).*

*The reported proportion is relevant to the number of subjects in the specific groups for whom the reason was indicated.
**The information on unprovoked/provoked was not collected in the 35 patients with isolated PE.
Treating physicians in Portugal, Czechia, and Poland (78.0%, 64.3% and 58.6%, respectively) more frequently involved patients when taking their therapeutic decisions (Figure 4). Conversely, only rarely (11.5% overall) – and 2.4% to nil in Portugal, Slovakia, Czechia, and Tunisia – were family doctors involved in the decision.

Distribution of the proportion of involvement of the patient and of the patient's family physician (GP) in the continuation decision. The dot size is proportional to the number of cases; the thick solid line represents the overall prevalence in the whole sample.
Discussion
The current study analyzed data collected in the WHITE study on decisions for long term treatment of patients with a first VTE episode managed in real-life conditions in clinical centers active in seven countries, presenting significant differences as per geographical, socio-economic, and health-care conditions. One of the main results of the analysis was that the unprovoked or provoked nature of the index VTE event was not the leading driver behind the participant physician‘s decision on duration of anticoagulation. This approach was consistent across countries. Though with some differences, overall, the proportion of patients who continued with anticoagulation was similar in patients with unprovoked or provoked index events (about 50%). While international guidelines still recommend extending anticoagulation for secondary prevention only to patients with unprovoked events,2,3,5,6 exactly which patients should be prescribed long-term anticoagulant therapy is still a complex decision in clinical practice. The present results, fully consistent with others recently reported in Italian real-life conditions, 11 confirm that the unprovoked/provoked nature of events is not the only, and sometimes not the main, reason informing treating physician decisions on duration of anticoagulation. Our results indicate that in every-day clinical practice in different countries, physicians look beyond the simple dichotomy of unprovoked/provoked index events and instead try to assess individual patient risk of recurrence using a variety of factors. The probability of being prescribed extended anticoagulation was, in fact, no different in patients with unprovoked or provoked events; it was significantly higher in patients presenting with PE, concomitant disease and in countries where DOACs were available for treatment. The most frequently reported reason for extending anticoagulation was risk of recurrence (reported in >83% of cases), assessed by the attending physician based on nature of index event, course of maintenance anticoagulation, residual thrombi at maintenance termination, and individual classification of recurrence risk. 13
Though with big differences across countries (from almost 50% in China to 0% in Czechia; see Figure 3), the proportion of IDDVT patients prescribed extended anticoagulation was very large (in average 37%). This finding may be surprising since it is well known that the risk of recurrence is lower after IDDVT than after proximal DVT.14–16 For this reason, guidelines recommend limiting anticoagulation in these patients to 3 months. 2 However, in contrast with the guidelines, recent studies show a general and growing trend to give IDDVT patients long anticoagulant treatment. Ageno & Co. 17 found that 27.9% of IDDVT patients included in the prospective XALIA study were treated for >180 days. Schellong & Co. 18 analyzed IDDVT patients included in the prospective GARFIELD-VTE study and found that 60% of them were anticoagulated 6 months after follow-up and 40% 12 months. Similar results were found in a recent Italian prospective study, in which >40% of IDDVT patients received anticoagulation for more than 6 months. 19
This trend towards extending anticoagulation is not limited to patients with IDDVT and is common in real-life clinical practice. The present study, performed in seven different countries, showed that about 60% of all patients with proximal DVT or with PE were prescribed extension of anticoagulation beyond the first 180 days. This result is fully consistent with a recent real-life study performed in Italy where data coming from the prospective observational START-Register showed that more than 67% of patients with proximal DVT or PE (those with IDDVT were excluded) were anticoagulated for >180 days. 20
Interestingly, the present study shows a large use of antithrombotic agents (mainly sulodexide, followed by low-dose aspirin) in place of anticoagulants after maintenance phase. The prescription of these agents after a first period of anticoagulation ranged from 21.8% to 32.9% in patients with provoked or unprovoked events, respectively. Low-dose aspirin 21 and full-dose sulodexide, 22 given after standard anticoagulation treatment, proved effective in reducing the risk of recurrence versus placebo in VTE patients, with low or no risk of major bleeding. However, in the years when the above studies were performed the only alternative treatment available was anticoagulation with vitamin K antagonists, a treatment usually considered at high risk of bleeding complications. Current therapeutic conditions are very different thanks to the availability and large use of DOACs at standard or even at lower dosage which are usually associated with high efficacy and safety regarding VTE recurrence prevention. In all countries involved in the WHITE study (with exception of Tunisia), DOACs were available and effectively used in more than 80% of patients during the maintenance phase. Why did the treating physicians prefer to shift treatment from DOACs to these antithrombotic agents in around one-third of patients instead of continuing DOAC use or stopping treatment? This is an interesting issue and deserves comment. It seems plausible to believe that doctors are aware that all patients with a first VTE are at some, albeit varying, risk of recurrence; however, individual risk is not easy to predict. It may be easy to identify patients at very high risk and to prescribe them extended anticoagulation with DOACs, accepting the associated persistent risk of bleeding. However, the participant physicians seemed worried about completely stopping treatment in all remaining patients and so limited this decision only to patients considered at very low risk of recurrence. They did however often look to hedge against the risk of recurrence by using different drugs such as sulodexide, which is associated with lower risk of bleeding than anticoagulation even if still effective.22,23 Besides patient preference, which may have had a role in decision making, another possible explanation is the cost of the different drugs, especially if patients are asked to share it.
This study has important limitations and merits. The participating clinical centers operated in countries that differed one from the other on a wide range of issues that were impossible to adjust for. The number of patients included in each country does not allow general conclusions to be drawn on clinical practice in each country. Finally, this was an observational study, and all the therapeutic decisions were left to the attending physicians with patient information being collected in a prospective registry. For this reason, the interpretation of our findings requires caution. We believe, however, that in today‘s “globalized” world an effort to assess what is the prevalent approach to management of VTE patients in the real-world setting of different countries is of value; furthermore, comparison of everyday therapeutic procedures used in the countries helps standardizing clinical practice.
In conclusion, this study provided information prospectively collected on the management of patients with recent VTE from clinical centers operating in seven countries with widely differing systems. After the maintenance period, anticoagulant drugs were continued in half of patients, while treatment was stopped in 20%; treatment was continued with antithrombotic drugs (especially sulodexide and aspirin) in many patients. Despite differences across countries, the same proportion of patients with unprovoked or provoked index events continued anticoagulation which was extended in more than one third of patients included for isolated distal DVT occurrence, in this case driven by the presence of PTS signs.
Supplemental Material
sj-docx-1-cath-10.1177_10760296211049402 - Supplemental material for Anticoagulation Duration After First Venous Thromboembolism: Real-Life Data From the International, Observational WHITE Study
Supplemental material, sj-docx-1-cath-10.1177_10760296211049402 for Anticoagulation Duration After First Venous Thromboembolism: Real-Life Data From the International, Observational WHITE Study by Gualtiero Palareti, Angelo A. Bignamini, Michela Cini, Young-Jun Li, Tomasz Urbanek, Juraj Madaric, Kamel Bouslama, German Y. Sokurenko, Giuseppe M. Andreozzi, Jiří Matuška, Armando Mansilha and Victor Barinov in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgment
We thank Dr Stephen Jewkes for his revision of the English.
Declaration of Conflicting Interests
Gualtiero Palareti has received consulting fees from Alfasigma. Angelo Bignamini has received consulting fees from Bayer Healthcare and Alfasigma. German Sokurenko has received lectures fees from Alfasigma, Bayer Healthcare, Pfizer. Tomasz Urbanek has received consulting and lectures fees from Alfasigma. The other authors declare no conflict of interest. The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The WHITE study was promoted by the “Arianna Anticoagulazione” Foundation (Bologna, Italy), and managed together with a Core Team of vascular-expert professionals (the detailed list of the study boards and their make-ups is shown in Appendix 1). The Foundation received an unrestricted research grant from Alfasigma (Bologna, Italy), specifically designed to fund this study. The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
The study protocol was approved by the local ethics committees and was conducted in accordance with the Declaration of Helsinki.
Informed Consent
Informed consent Written informed consent was collected according to local practice.
Trial Registration
ClinicalTrials.gov Identifier: NCT04646993
Supplemental Material
Supplemental material for this article is available online.
APPENDIX 1
List of Participating Clinical Centers (in order of decreasing valid cases recruited in each country)
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
