Abstract
Oral anticoagulants (OACs), including vitamin K antagonists and direct-acting OACs, are commonly used for the prevention and treatment of thrombosis. OAC-treated patients may require urgent anticoagulation reversal in circumstances of life-threatening bleeding or emergent surgery. Despite the availability of Food and Drug Administration-approved OAC reversal agents, published treatment guidelines, and institutional protocols, use of OAC reversal products and dosing vary considerably and off-label use is common. We review key factors influencing current clinical practice and examine barriers to the successful implementation of anticoagulation protocols, including discrepancies in the definition of major bleeding, dosing considerations, variability in laboratory testing practices, inconsistent study endpoints, formulary considerations, and the use of OAC reversal agents in special populations. We provide our expert opinion on best practices for oral anticoagulant reversal and an anticoagulation reversal protocol that could be implemented by healthcare institutions to standardize and optimize clinical practice.
Keywords
Introduction
Oral anticoagulants (OACs), including vitamin K antagonists (VKAs) and direct-acting OACs (DOACs), are commonly used for the prevention and treatment of thrombosis for conditions such as atrial fibrillation or venous thromboembolism1–3; however, their use is associated with an increased risk of bleeding.1,3,4 Urgent anticoagulation reversal is necessary in patients presenting with major or life-threatening bleeding, or in those requiring urgent surgery or invasive procedures.1–3,5
There are three Food and Drug Administration (FDA)-approved reversal/hemostatic agents for OACs: idarucizumab (for dabigatran reversal), 4-factor prothrombin complex concentrates (4F-PCC, for warfarin reversal), and phytonadione (vitamin K, for warfarin reversal) (Table 1).2,5–11 Andexanet alfa was approved for rivaroxaban and apixaban reversal but was voluntarily withdrawn from the US market on December 22, 2025, due to thromboembolic event safety concerns 12 ; it remains on the market in multiple countries (eg, in the EU and Canada).13,14 Off-label use of non-specific agents such as 3-factor (3F-), 4F-, and activated-PCC (aPCC) is common and recommended in some anticoagulation reversal treatment guidelines (Table 2).2,5–8,10,11,15–17
Oral Anticoagulant Reversal Agents.

*Andexanet alfa was voluntarily withdrawn from the US market on December 22, 2025; it is still available in other countries and may still be relevant for rivaroxaban and apixaban reversal in certain settings when available. 12
3F-PCC, three-factor prothrombin complex concentrate; 4F-PCC, four-factor prothrombin complex concentrate; anti-FXa: anti-factor Xa; aPCC, activated prothrombin complex concentrate; FDA, Food and Drug Administration; FII, factor II; FVII, factor VII; FIX, factor IX; FX, factor X; FXa, activated factor X; INR, international normalized ratio; IU, international unit; IV, intravenous; MOA, mechanism of action; OAC, oral anticoagulant; T1/2, half-life; TF, tissue factor; TFPI, tissue factor pathway inhibitor; VKA, vitamin K antagonist; VKDF, vitamin K-dependent factor.
Summary of Guidelines for Oral Anticoagulation Reversal.
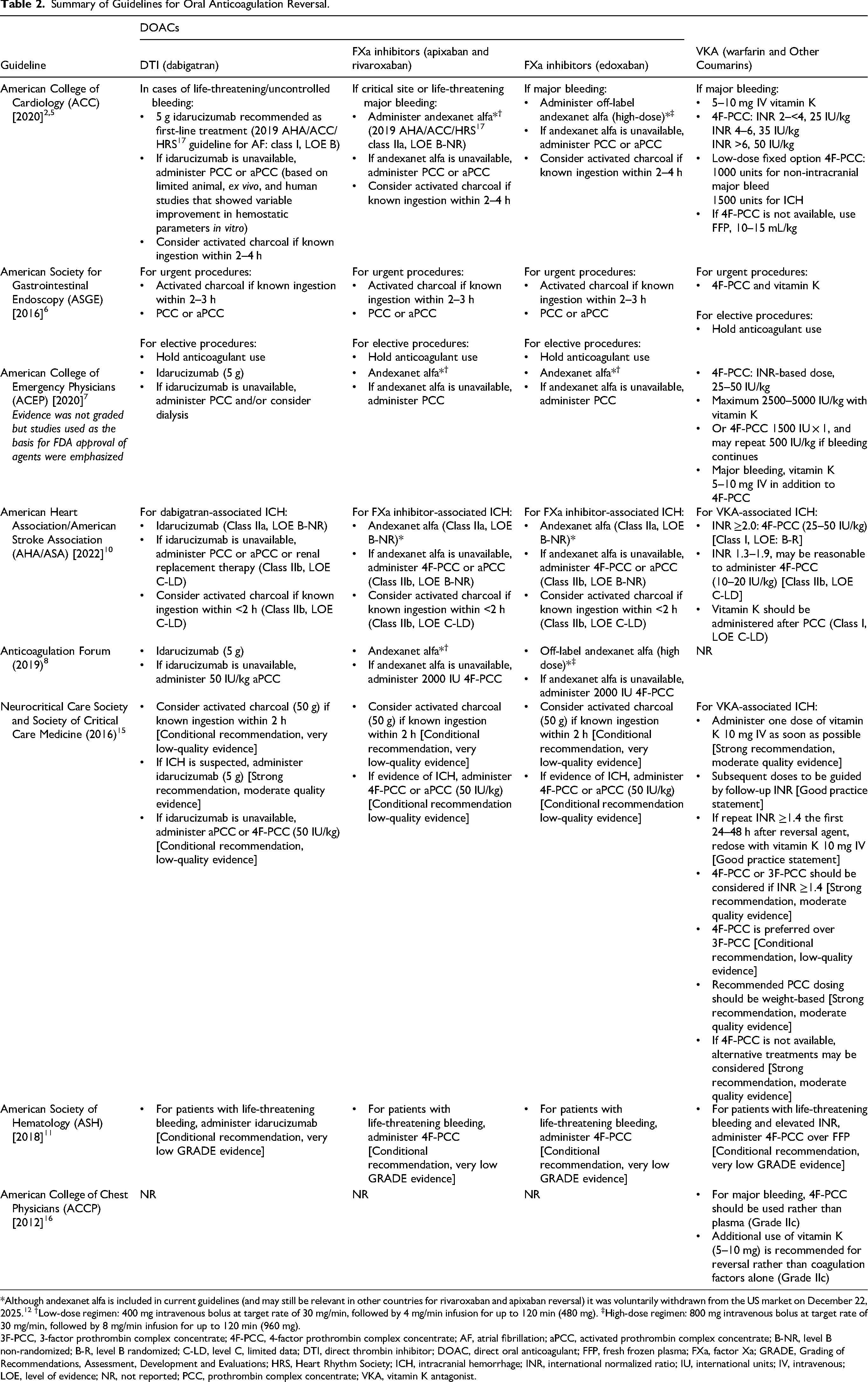
*Although andexanet alfa is included in current guidelines (and may still be relevant in other countries for rivaroxaban and apixaban reversal) it was voluntarily withdrawn from the US market on December 22, 2025. 12 †Low-dose regimen: 400 mg intravenous bolus at target rate of 30 mg/min, followed by 4 mg/min infusion for up to 120 min (480 mg). ‡High-dose regimen: 800 mg intravenous bolus at target rate of 30 mg/min, followed by 8 mg/min infusion for up to 120 min (960 mg).
3F-PCC, 3-factor prothrombin complex concentrate; 4F-PCC, 4-factor prothrombin complex concentrate; AF, atrial fibrillation; aPCC, activated prothrombin complex concentrate; B-NR, level B non-randomized; B-R, level B randomized; C-LD, level C, limited data; DTI, direct thrombin inhibitor; DOAC, direct oral anticoagulant; FFP, fresh frozen plasma; FXa, factor Xa; GRADE, Grading of Recommendations, Assessment, Development and Evaluations; HRS, Heart Rhythm Society; ICH, intracranial hemorrhage; INR, international normalized ratio; IU, international units; IV, intravenous; LOE, level of evidence; NR, not reported; PCC, prothrombin complex concentrate; VKA, vitamin K antagonist.
Currently, the clinical approach to anticoagulation reversal is based on published guidelines, institutional protocols, and clinician experience 18 ; however, clinical practice frequently differs from these recommendations and varies based upon the institution, availability of reversal agents, and physician specialty.18,19 Moreover, many hospitals may not have comprehensive anticoagulation reversal guidelines that address all clinical scenarios and patient populations. 19 As such, there remains a need for clear and consistent recommendations that are available to multidisciplinary teams and incorporate real-world clinical scenarios. Herein, we aim to review current guidance on oral anticoagulation reversal, discuss the barriers that impact the implementation of an anticoagulation reversal protocol, and describe how a clear decision pathway could be standardized and implemented across all specialties at a healthcare institution. Based on current available evidence and known pharmacologic properties of anticoagulation reversal agents, we propose a treatment algorithm for OAC reversal.
Current Guidance on Anticoagulation Reversal
There are a number of US and international guidelines for anticoagulation reversal (Table 2),2,5–8,10,11,15,16 and various approaches and algorithms for anticoagulation reversal have been published.2,4,5,7,8,20,21 The location and severity of bleeding influences the decision to proceed with anticoagulation reversal, and in published guidelines there is variability in the definition of the types of bleeds that warrant use of a hemostatic agent.2,7,21 These factors should be evaluated in tandem with the thrombotic risk to the patient, as well as other factors including delayed presentation or confirmation of diagnosis. In addition, there is variability in the endpoints selected to demonstrate the efficacy of OAC reversal agents in clinical publications. To determine the best approach to anticoagulation reversal, a number of clinical (Figure 1), logistical, and legal factors should be taken into consideration.

Proposed pathway to determine need for OAC reversal. *Critical site defined as brain, eye, spine, airway, pericardium, etc. †A non-major bleed that requires emergent surgery or procedural intervention may benefit from OAC reversal. OAC, oral anticoagulant; RBC, red blood cell. Reused and adapted with permission from Elsevier, from ACC Expert Consensus Decision Pathway on Management of Bleeding in Patients on Oral Anticoagulants. Tomaselli GF, et al. Journal of the American College of Cardiology, Volume 76, Issue 5, Pages 594–622, Copyright (2020). Permission conveyed through Copyright Clearance Center, Inc.
Challenges in Current Anticoagulation Reversal Guidelines
Defining Indications for Reversal
Major Bleeding and Life-Threatening Bleeding
Several anticoagulation reversal guidelines recommend using reversal agents for major bleeding; however, there is no consistent definition of what constitutes a major hemorrhage (Table 3).2,7,22,23 In line with the International Society on Thrombosis and Haemostasis definition of major bleeding, the American College of Cardiology (ACC) treatment guidelines define a major hemorrhage as bleeding consisting of ≥1 of the following: a) bleeding at a critical site (eg, intracranial hemorrhage [ICH], intraocular bleeding, spinal bleeding), b) hemodynamic instability, or c) bleeding associated with a hemoglobin decrease of ≥2 g/dL or administration of ≥2 units of red blood cells (Table 3). 2 The recommendations from the American College of Chest Physicians (ACCP), 16 however, use the term ‘major bleed’ without providing a definition. It is noted in the ACCP guidelines that different studies use different definitions for the term ‘major bleed’. The use of reversal agents is recommended for ‘life-threatening’ bleeding in the American Society for Gastrointestinal Endoscopy, 6 American College of Emergency Physicians (ACEP), 7 American Society of Hematology, 11 and Anticoagulation Forum 8 recommendations. Of these guidelines, only the ACEP provides a clear definition of life-threatening bleeding, 7 stating that it is a composite of the Bleeding Academic Research Consortium Type 3a and 3b definitions (Table 3). The Anticoagulation Forum guidance recommends reversal for only a subset of ‘major bleeds’, such as bleeding that is life-threatening, into a critical organ, or that is not controlled with maximal supportive measures. 8 Several guidelines mention bleeding at a ‘critical site’; this is defined similarly in the ACC 2 and ACEP 7 guidelines as an organ such as the brain, eye, spine, etc; however, the term is not defined in the recommendations from the Anticoagulation Forum. 8 OAC reversal protocols should be clear as to which bleeds do and do not warrant reversal and use explicit, standardized definitions whenever possible to guide clinical practices. 24 Reversal should be prioritized for life-threatening bleeding and additionally, it may be helpful to provide categorizations for minor bleeding that may not require urgent reversal or intervention.
Summary of Bleeding Definitions.

*Excluding dental, nasal, skin, and hemorrhoid.
ACC, American College of Cardiology; ACEP, American College of Emergency Chest Physicians; BARC, Bleeding Academic Research Consortium; ISTH, International Society on Thrombosis and Haemostasis.
Type of Bleed
ICH is reported as one of the most frequent indications for OAC reversal. 19 Early anticoagulation reversal is associated with improved outcomes in patients with ICH, 25 and the American Heart Association/American Stroke Association recently included time to reversal of 90 min as a performance measure for the care of these patients. 26 Compared with ICH, non-ICH bleeds are often more ambiguous. There are some gray areas in which it is unclear if an OAC-treated patient would require anticoagulation reversal. For example, an OAC-treated patient with a non-life-threatening gastrointestinal (GI) bleed, undergoing a chest tube/central line insertion, or requiring a non-emergent, but still urgent surgery, may not require reversal.
Study Endpoint Variability
Clinical research should aim to improve patient care and reduce morbidity and mortality, and the outcomes studied should reflect real-world settings. Though clinical outcomes are important measures of efficacy, use of surrogate markers (eg, laboratory data, imaging) is common; this, alongside variability in study endpoints, makes it difficult to translate clinical research into practice and policy. Moreover, the definitions of many clinical outcomes are not standardized, and this makes comparing the effectiveness of different reversal agents a challenge. The lack of consensus on what efficacy endpoints should be in clinical studies has led to various surrogate markers of efficacy being measured. For example, in the ANNEXA-I trial of andexanet alfa in ICH, the primary endpoint of hemostatic efficacy was defined as: expansion of hematoma volume by <35% at 12 h post-baseline, an increase of <7 points on the National Institutes of Health Stroke Scale, and no rescue therapy received between 3 and 12 h. Although the primary endpoint was met and hemostatic efficacy was reported to be better with andexanet alfa than standard care, it was also associated with increased thrombotic events and stroke, and no differences in 30-day modified Rankin scale or mortality. 27 Such use of surrogate markers contributes to controversies in implementing reversal agents.
OAC-Specific Reversal Considerations
Warfarin (VKAs)
Guidelines for warfarin reversal recommend the use of PCCs in combination with vitamin K in patients with major bleeds or requiring urgent surgery.2,5–7,10,11,15,16
Dosing of VKA-Reversal Agents
Phytonadione (vitamin K) at an intravenous (IV) dose of 5–10 mg is recommended for reversal of VKA-antagonist-associated life-threatening bleeds.2,6,9,11,16 Some studies have utilized lower doses (<5 mg) to avoid long-term warfarin resistance in patients in whom early re-anticoagulation is anticipated and the ACC guidelines recommend 2–5 mg for non-major bleeding.2,28–30 Due to the delayed onset and peak effect of phytonadione (6 and 24 h, respectively), clotting factors, preferably PCC (over plasma), are recommended for patients with major bleeds in conjunction with vitamin K.2,6,7,10,11,15,16
The original trials that compared 4F-PCC to plasma for VKA-antagonist reversal used a weight-based dosing strategy stratified by international normalized ratio (INR; 25-50 units/kg) which is currently recommended in the package insert.31–35 Compared with weight-based PCC dosing, fixed-dosing regimens are well described in the context of warfarin reversal. 36 While fixed-dose strategies may be facilitated more quickly, they may result in lower rates of goal INR attainment and a higher rate of repeat dosing.36,37 Lower rates of goal INR attainment are more frequently seen with lower doses (eg, 1000 International Units [IU]) rather than higher doses (eg, 1500-2500 IU). 37 The implementation of fixed-dose protocols can result in significant resource savings and potentially a faster time to administration compared with weight-based dosing. 38
A systematic review of PCC dosing regimens for warfarin reversal identified five main dosing principles based on: weight alone; weight and initial INR; weight, initial INR, and target INR; physician decision; or fixed-dose strategy. Regardless of the dosing strategy used, better clinical and INR outcomes were reported when a treatment protocol was utilized, 39 highlighting the value of a standardized approach to reversal. More data are needed in high-risk patients who may require lower doses of PCC, such as those with a left ventricular assist device (LVAD), 40 recent thrombosis, or those requiring re-dosing.
Laboratory Testing
Protocols for managing the reversal of warfarin have been established and are typically guided by readily available INR monitoring with goal INR values often targeted to <1.5. 41 However, a recent study comparing 4F-PCC and plasma for VKA reversal found that 4F-PCC rapidly restored vitamin K-dependent factors to hemostatic levels even in patients with a post-treatment INR >1.5, showing that post-reversal INR may underestimate restoration of vitamin K-dependent factors after 4F-PCC administration. 41 Nonetheless, INR is routinely used as a surrogate marker in patients receiving PCC for warfarin-associated reversal.
Direct-Acting Oral Anticoagulants (DOACs)
For DOAC reversal, current guideline recommendations include idarucizumab2,5,7–11 and andexanet alfa2,5,7,8,10 as the targeted reversal agents for dabigatran and for apixaban or rivaroxaban, respectively (Table 2). The recommendations for use of these specific reversal agents were based on low-level evidence as several guidelines were published before availability of DOAC-specific reversal agents or before publication of results from larger clinical trials.6,11,16 This circumstance, in addition to the recent withdrawal of andexanet alfa from the US market, highlights that guidelines can quickly become outdated. 42 Furthermore, several guidelines recommend aPCC, 3F-PCC, or 4F-PCC when specific reversal agents are not available.2,5–8,10,11,15 However, there is significant heterogeneity in PCC dosing among clinical guidelines. These discrepancies make it difficult for clinicians to interpret the guidelines when developing OAC reversal protocols for use in clinical practice.2,5–11,16
Dosing of DOAC Reversal Agents
Data are limited on the safety and effectiveness of fixed-dose PCC strategies when PCCs are used to reverse factor Xa inhibitors (FXaIs). Most studies of 4F-PCC for the management of FXaI-associated bleeding use a weight-based strategy, and only a few studies have directly compared fixed versus variable dosing. 43 However, a recent meta-analysis that evaluated the effectiveness and safety of fixed versus variable dosing of 4F-PCC for FXaI-related bleeding in 1760 patients reported no differences in hemostatic effectiveness, thromboembolic events, or mortality between dosing strategies. 44 Furthermore, a fixed-dosing strategy was associated with administration of lower doses of 4F-PCC compared with variable dosing. This has led to differing guideline recommendations; for example, the Neurocritical Care Society recommends weight-based dosing (50 IU/kg) 2 while the Anticoagulation Forum recommends fixed dosing (2000 IU). 8 There is less ambiguity in the dosing of idarucizumab that is administered as a 5 g IV push (Table 1).
Laboratory Testing
Reversal protocols should also describe how to obtain and utilize quantitative tests of anticoagulant effect when making decisions for DOAC reversal. Standard assays such as INR, prothrombin time, activated partial thromboplastin time, and heparin-calibrated anti-FXa should be available at most institutions. Collaboration with hematology and pathology can be considered to explore the availability of specialized assays such as FXaI-calibrated anti-FXa assays45–48 and dilute thrombin time, which can aid in the decision-making surrounding reversing DOACs. However, most institutions do not have access to specialized assays, and thus a standardized approach to the interpretation of ‘conventional’ coagulation assays and qualitative aspects of anticoagulation (ie, time of last dose) is essential for reversal protocols (Table 4).2,8,20,27,49
Coagulation Assays and the Impact of OACs on Their Results. 2
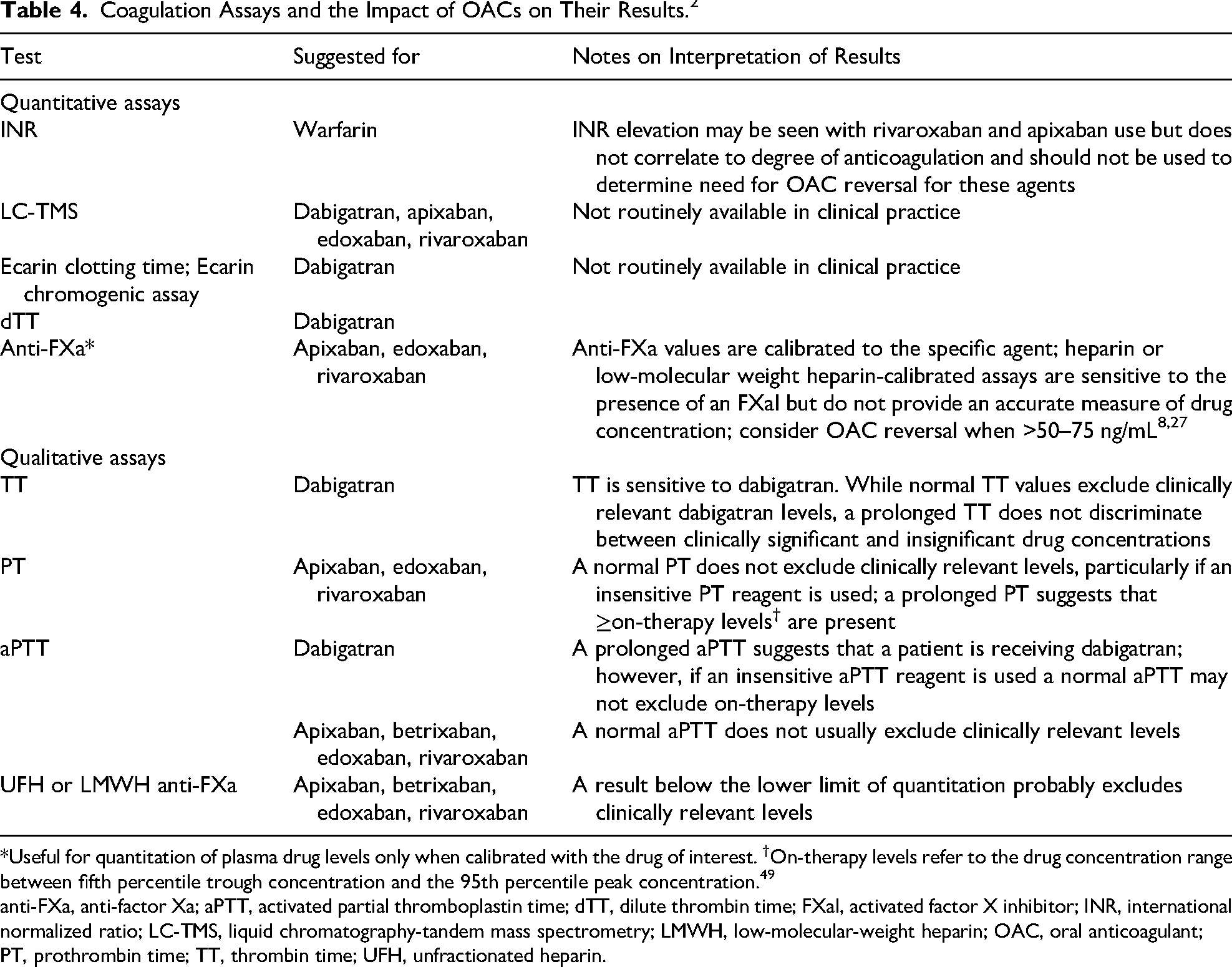
*Useful for quantitation of plasma drug levels only when calibrated with the drug of interest. †On-therapy levels refer to the drug concentration range between fifth percentile trough concentration and the 95th percentile peak concentration. 49
anti-FXa, anti-factor Xa; aPTT, activated partial thromboplastin time; dTT, dilute thrombin time; FXaI, activated factor X inhibitor; INR, international normalized ratio; LC-TMS, liquid chromatography-tandem mass spectrometry; LMWH, low-molecular-weight heparin; OAC, oral anticoagulant; PT, prothrombin time; TT, thrombin time; UFH, unfractionated heparin.
Viscoelastic testing (VET) with rotational thromboelastometry (ROTEM®) or thromboelastography (TEG®) could potentially be valuable indirect measures of clinically relevant levels of DOACs, but studies measuring anticoagulant activity using TEG® and ROTEM® have reported poor correlation compared with specialized targeted coagulation parameters. Thus, commercially available assays without DOAC-calibrated cartridges should not be used to guide whether reversal is indicated. 2 Recently, measurements recorded with TEG®6 s DOAC-specific cartridges have demonstrated a significant correlation with dabigatran, rivaroxaban, and apixaban blood concentrations. 50 When available, this technology may be useful for urgent assessment of coagulation status in DOAC-treated patients who require emergent surgery or have uncontrolled bleeding, 51 although its clinical utility has been questioned and limited real-world experience exists to guide use. 52
Indications for Reversal
DOAC-Associated ICH
ICH is a critical site bleed for which DOAC reversal is generally appropriate, but there is nuance in that decision. Despite ICH being one of the most frequent indications for OAC reversal, a recent study of DOAC-associated ICH treated with PCC versus conservative management showed that PCC use was not associated with improved neurological outcomes or hematoma expansion, and that a higher Glasgow Coma Scale (GCS) on admission was associated with a higher likelihood of good neurological outcome. 53 Moreover, although better control of hematoma expansion was reported with andexanet alfa compared with standard care in the ANNEXA-I trial, andexanet alfa was also associated with increased thrombotic events; there was no difference in GCS between patient groups at baseline. 27 Similar results were observed in a 6-year retrospective study of reversal of FXaI treatment in patients with mild traumatic brain injury (TBI) where 4F-PCC did not impact ICH progression, neurological decline, or the need for neurosurgical intervention. 54 In a 2025 retrospective study comparing the efficacy and safety of andexanet alfa versus PCC for DOAC-associated ICH, 87.8% of andexanet alpha recipients and 81.8% of PCC recipients achieved excellent or good hemostasis (p = 0.048); however, andexanet alfa treatment was associated with nearly twice the rate of thrombotic events (7.9% vs 4.2%; p = 0.014). There was no difference in GCS between patient groups at baseline. 55 Although further research is needed, this evidence reflects that the clinical benefit of reversal can depend on the anticoagulant used, the chosen reversal agent, bleeding etiology, and timing of reversal. Evidence has indicated that in DOAC-treated patients, the incidence of ICH after head trauma is relatively low.56,57 This may be due to the majority of suspected TBI cases in DOAC-treated patients being older patients who have had a low-energy mechanism of injury (ie, a ground-level fall). Although reversal agents are not recommended in situations of trauma in the absence of bleeding, 8 computed tomography (CT) scans should be initiated when patients present with neurological complaints to assess for any evidence of ICH 58 that may require administration of a reversal agent or further medical attention. It should be noted that DOAC reversal in ICH should not follow a ‘one-size-fits all’ approach; rather, protocols should account for bleed severity, timing of administration, and clinical deficits when making decisions between agents and whether to reverse. Further research is needed to determine whether reversal of DOAC-associated ICH reduces hematoma expansion and improves clinical outcomes, and to determine the optimal patient population.
Surgery
Various factors need to be considered when patients on DOACs require surgery, including the procedure-specific bleeding risk and whether the surgery is elective, emergent, or urgent. In cases of elective surgery, discontinuing the DOAC, regardless of bleeding risk, may be sufficient. 59 In these cases, it is important to establish a standardized protocol that stipulates when to stop/restart the DOAC depending on variables such as bleeding risk or type of surgery/procedure, without the need for laboratory testing or bridging. In cases of urgent or emergent surgery, laboratory testing (see DOACs Laboratory Testing section) may be useful to determine preoperative DOAC levels and therefore inform whether DOAC reversal is necessary before surgery. 59
Concomitant Medications and Comorbid Conditions
While the half-life of DOACs2,60 should be considered in instances of severe bleeding where the relevant laboratory tests are unavailable or are not obtainable within a timely manner, the metabolic impact that concomitant medications and/or comorbidities can have on circulating DOAC levels must also be considered. In patients with these confounding factors, the decision on the time since last dose to when reversal is still indicated should be made on an individualized basis. Drug–drug interactions can affect an individual's DOAC level and drug clearance, 60 particularly as a large number of DOAC-treated patients have comorbid conditions that require medication. DOACs are a substrate for p-glycoprotein (P-gp), and rivaroxaban and apixaban undergo cytochrome P450 (CYP)-mediated metabolism; therefore, reviewing for patient exposure to common medications that inhibit or induce P-gp and CYP-mediated metabolism should be considered. 61
Renal and hepatic dysfunction can also lead to complex clinical scenarios with regards to anticoagulation. Patients with impaired renal function who receive dabigatran, which is primarily renally excreted, have shown an increased risk of bleeding and thrombosis. 62 Though dabigatran has the greatest amount of renal elimination comparatively, all DOACs undergo some degree of renal elimination and use is cautioned in patients with end-stage renal disease. Furthermore, in patients with chronic kidney disease, apixaban levels have been detected 10 days on from last known dose.63,64 Therefore, these patients may require reversal even if their last dose of DOAC was more than 48 h prior. The liver also plays a key role in the metabolism of direct FXaIs; however, these anticoagulants have not been well studied in patients with severe hepatic dysfunction and standard laboratory tests may not reliably assess hemostatic function in this patient population. 2 There may also be a role for utilizing DOAC-calibrated anti-FXa levels; although specific thresholds for reversal have not been elucidated at this time, this could be an opportunity for future research.
Considerations for Standardization and Protocolization
Special Populations
Older Population
DOACs are widely used in the older population and it is estimated that 70% of patients who have atrial fibrillation are between 65 and 85 years of age. 65 Studies have demonstrated that patients aged 75 years and above are at higher risk of ICH and GI bleeding with DOACs compared with younger patients,65,66 in addition to being at higher risk for acute renal failure and chronic kidney disease. 65 A retrospective analysis highlighted the challenges with prescribing and maintaining FDA-recommended DOAC doses in the older population, stating individualized treatment that considers renal function, comorbidities, and concomitant antiplatelet therapy would be more appropriate than standard treatment. 65 For example, rivaroxaban has a prolonged half-life in the geriatric population compared with patients aged 20-45 years (11-13 h vs 5-9 h), 67 and therefore, the therapeutic window for appropriate reversal may vary between the geriatric and general population. Decision pathways that govern OAC reversal in older patients should include these considerations to optimize reversal strategies.
Pediatrics
Apixaban, dabigatran, and rivaroxaban are approved for pediatric use; however, there is a paucity of data on the use of DOAC-specific reversal agents in this patient population, with most supporting data coming from adults.68,69 Case studies have provided some evidence of use of DOAC reversal agents in dabigatran overdose 70 and for rivaroxaban-associated ICH 71 ; however, more research is needed to understand what the optimal reversal approach is in these patients, both to inform clinical practice and to ensure that pediatric hospitals have access to these reversal agents.68,72
Patients with LVADs and Heart Transplant
Patients with LVADs require anticoagulation to prevent thromboembolic complications and are at increased risk of bleeding due to physiologic changes attributed to the LVAD. Currently, the American Association for Thoracic Surgery and the International Society for Heart and Lung Transplantation guidelines only recommend warfarin in these patients. 73 In patients with an LVAD and ICH, the prognosis is poor, especially in patients who require surgical intervention. In general, anticoagulation should be reversed or stopped for these high-risk patients. 74 4F-PCC has been shown to reverse the effects of warfarin with no apparent risk of thromboembolism in patients with an LVAD73,75; however, further studies are needed in this area due to the limited number of patients and the variability in the type of device included in existing studies.
For warfarin reversal prior to heart transplantation, data have shown that a PCC-based reversal strategy reduced the consumption of fresh frozen plasma without impacting other clinically important surgical outcomes 76 ; however, unplanned intraoperative use of anticoagulation reversal agents (mostly vitamin K) to minimize bleeding complications associated with mechanical circulatory support device explantation during heart transplantation has been associated with a higher risk of thromboembolic events. 77 Further research into the safety of anticoagulation reversal in this patient population is required.
Pregnancy and the Peripartum
The risk of major bleeding is increased in the peripartum period, with hemorrhage being the leading cause of maternal death worldwide. 78 Venous thromboembolism in pregnancy is common and many women require thromboprophylaxis through pregnancy and postpartum. 79 Clinical guidance for anticoagulation reversal during pregnancy is limited; however, postpartum hemorrhage clinical guidelines recommend that management of bleeding in pregnancy, including reversal of anticoagulants, should not differ from non-pregnant patients on antithrombotics. 79 However, oral anticoagulants are not commonly used during pregnancy due to teratogenicity risk with warfarin and lack of data for DOACs.
Non-Anticoagulated Patients
Anticoagulation reversal agents have been reported to be used in patients presenting with coagulopathy or bleeding but not receiving anticoagulation therapy. Recombinant activated factor VII has been used in non-anticoagulated patients with ICH and, while hematoma volume was reduced, an increased frequency of arterial thromboembolic events versus placebo was reported without improvements in functional outcomes. 80 PCC has been used in non-anticoagulated ICH patients with cirrhosis, though in this cohort, a single dose of PCC did not significantly improve the rate of stable head CT versus standard therapy and thus may not provide clinical benefit. 81 In patients with hepatic dysfunction, a previous retrospective study reported an increased 30-day mortality rate in 4F-PCC-treated patients with coagulopathy who did not receive VKAs compared with those who received VKAs. 82 However, the incidence of hepatic dysfunction was higher in the cohort of patients who did not receive VKAs, therefore placing them at a higher baseline risk for mortality. 82
INR is often elevated in patients with liver failure and thus not an appropriate measure of coagulation status in these patients. 83 The use of VET with ROTEM® or TEG® may be more predictive of coagulation status and bleeding risk than INR. 83
Patients who Decline Blood Products
Other special patient populations to consider include Jehovah's Witness patients, who may reject transfusion of blood components such as PCCs due to their religious beliefs. Patients may also decline treatment with blood products due to concerns over infection or immune reactions. Shared decision-making by having a thorough discussion with the patient or their caregiver regarding the options for anticoagulation reversal is necessary, as each patient may have a different level of comfort with receiving blood products or blood components. Factor VII, idarucizumab, tranexamic acid, and vitamin K are treatments that are likely acceptable for these patients.
Cost
PCCs and idarucizumab are associated with a high cost and can be thousands of dollars per dose.84–86 One US institution performed a retrospective study of off-label 4F-PCC for FXaI-associated bleeding and reported the average 4F-PCC cost per reversal to be $6647. 84 The chosen dose can also significantly impact overall cost. Dose rounding of PCC, where the calculated dose is rounded to the closest vial size, has been conducted across various institutions.85,86 Compared with not dose rounding, this approach has been shown to improve cost-effectiveness for 4F-PCC with no clinical implications and has significantly reduced the time to achieve the target INR. 85
Expert Opinion on the Development and Effective Implementation of Anticoagulation Reversal Protocol
The evidence discussed in the earlier sections highlights that the variability in available guidance and current recommendations impacts the implementation and adoption of new clinical practices set out in anticoagulation reversal protocols. As mentioned, factors such as clinical presentation (location/severity of bleed; need for surgery), time since last dose of OAC, comorbidities, concomitant medication use, cost, and availability of reversal agents further complicate the decision-making process.2,4,8,60 We have considered this evidence and propose a decision tree that may be implemented to circumvent the challenges faced when trying to design and implement an anticoagulation reversal protocol.
While standardized definitions of the types of bleeding that require reversal would be beneficial, the reversal agent-specific factors we have described also need to be taken into consideration. In Figure 2, we propose considerations for institutions to take into account when developing and implementing an anticoagulation reversal protocol.
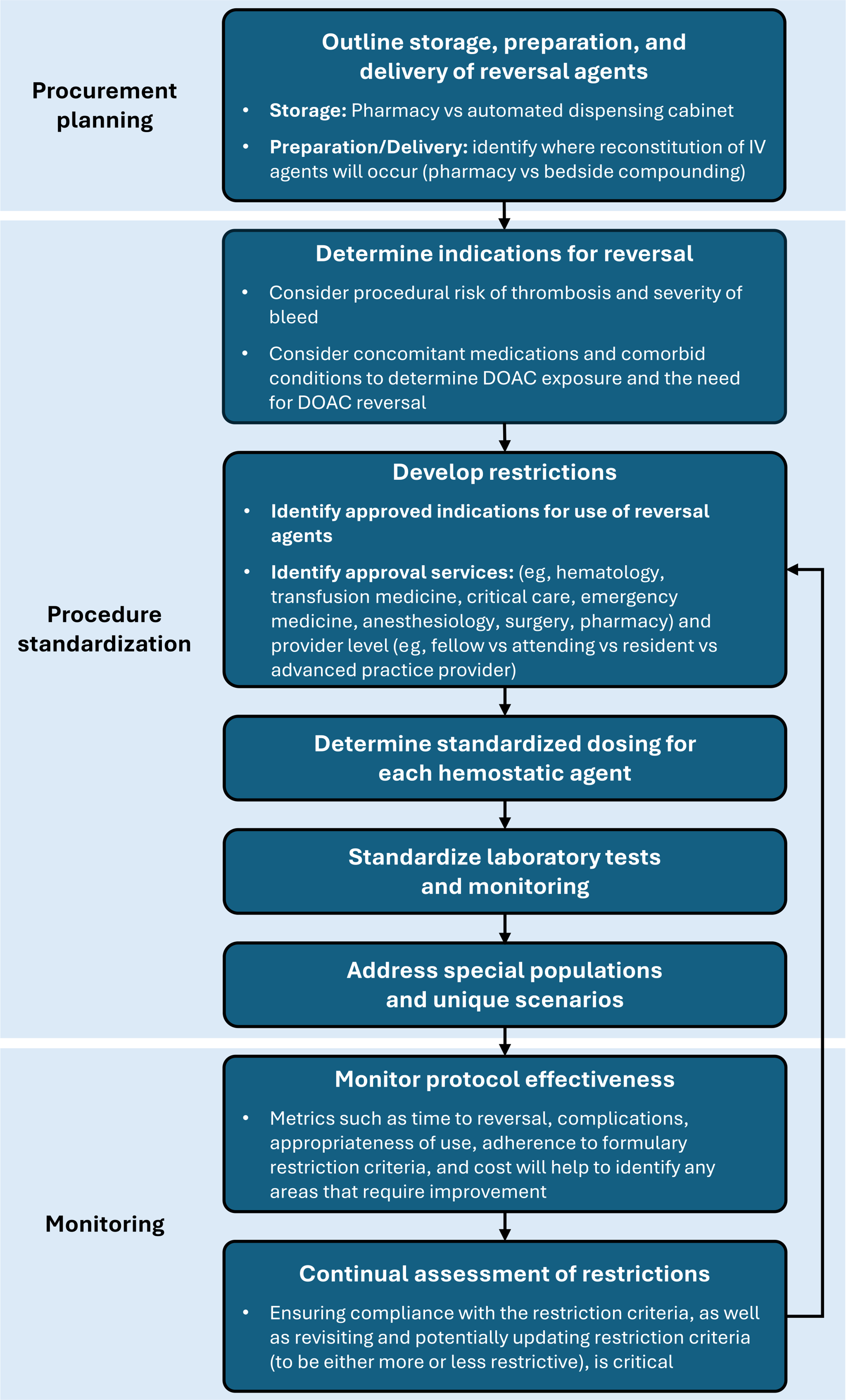
Institutional considerations for implementation of an OAC reversal protocol. DOAC, direct-acting oral anticoagulant; IV, intravenous; OAC, oral anticoagulant.
Proposed Anticoagulation Reversal Pathway
The approach to anticoagulation reversal requires careful consideration of the risks and benefits of each strategy.2,5,20 Clinicians caring for patients on OACs should be aware of the optimal strategies for rapid reversal to enable them to choose the most appropriate protocol based on the type of anticoagulant used and the patient's condition. 4 To support clinicians, we propose a treatment algorithm for warfarin, dabigatran, and oral FXa inhibitor reversal (Figure 3).27,87

Recommendations for OAC reversal in patients with bleeding. *Consider higher INR threshold (>2.0) for less severe bleeds and non-neurologic bleeds. †Consider higher dose (2000 units) for patients with INR >6.0 and/or ICH. Retest INR 30 min post-dose. If INR above goal, consider redosing. ‡If for reversal for an intermediate procedure (after which the patient will immediately be re-anticoagulated), consider intermediate dose of vitamin K or omit vitamin K. §For LVAD, reversal may depend on the device and a more specific risk-benefit assessment may be required. ||If patient is not at aspiration risk, consider activated charcoal if known recent ingestion (within 2-4 h). ¶Patients with renal insufficiency may require reversal even if the last dose was >24 h prior. **Dialysis is no longer recommended for dabigatran reversal. ††Edoxaban is listed here as 20 patients in the ANNEXA-I trial received andexanet alfa, despite it not being FDA-approved for reversal of edoxaban. 27 ‡‡4F-PCC is preferred for surgical patients, patients requiring heparin, and patients >18 h since last dose. §§If calibrated FXa level is available, consider using a threshold for eligibility. ||||Andexanet alfa may also be considered in markets where it is available. 4F-PCC, 4-factor prothrombin complex concentrate; aPCC, activated prothrombin complex concentrate; FDA, Food and Drug Administration; FXa, activated factor X; ICH, intracranial hemorrhage; INR, international normalized ratio; IV, intravenous; LVAD, left ventricular assist device; OAC, oral anticoagulant; PCC, prothrombin complex concentrate; ROTEM®, rotational thromboelastometry; TEG®, thromboelastrography.
Conclusion
This review highlights that the current approach to anticoagulation reversal varies despite the availability of treatment guidelines and published treatment algorithms. Implementation of treatment protocols is challenging due to the evolving treatment landscape and continual publication of new evidence, and factors such as variability in definitions of major or life-threatening bleeding and major surgery, the cost of reversal agents and any required testing, patient comorbidities, and logistical and legal considerations. Protocols should be based on evidence and expert opinions from providers who manage patients receiving OACs.
Footnotes
Acknowledgments
Medical writing and manuscript submission support was provided by Bioscript Group, Macclesfield, UK, in accordance with Good Publication Practice guidelines, and funded by CSL Behring.
ORCID iDs
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from CSL Behring. This work was also supported by the USA Department of Veterans Affairs Health Systems.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Karen Berger reports speaker's bureau for CSL Behring (Kcentra) and Octapharma (Balfaxar); and reports participation in an advisory board for Serb Pharmaceuticals. Andrew J. Webb reports consultant for Grace Therapeutics. Alyssa D. Rabon, Laura E. Celmins, Shannon Lovett, and Alexandra Dixon report no conflicts of interest with respect to the authorship and publication of this article. Adam Cuker reports serving as a consultant for MingSight, Pfizer, Sanofi, and Synergy; and has received authorship royalties from UpToDate. Megan A. Rech reports receiving a research grant from the Emergency Medicine Foundation/AstraZeneca; and has participated in an educational video for CSL Behring.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed in this review.
Disclaimer
The findings and conclusions in this document are those of the authors, who are responsible for its content, and do not necessarily represent the views of the VA or of the US Government.
