Abstract
The availability of direct oral anticoagulants has caused a paradigm shift in thrombosis management. The direct thrombin inhibitor dabigatran seems to obstruct tenase complex by inhibiting thrombin generated in the initial phase and feed back to the amplification phase of cell-based coagulation reactions. However, it is still not fully understood if and how dabigatran impact platelet function. This observational study aimed to assess in vitro platelet function in patients with atrial fibrillation receiving dabigatran. Platelet aggregability was tested with platelet-rich plasma using platelet aggregometry (PACKS-4 aggregometer). Blood samples were stimulated with thrombin receptor agonist peptide (TRAP; 32 μmol/L).
Results:
A total of 28 patients with nonvalvular atrial fibrillation were enrolled. The mean age was 71.57 (9.75) years (range: 50-87 years), 16 patients were women, and the mean CHA2DS2VASc score was 3.93 (1.41). All patients began treatment with dabigatran as initial anticoagulant treatment. The minimum term use of dabigatran was 18 days. Dabigatran doses were 110 mg (57.14%) or 150 mg (42.86%) twice daily. The TRAP-induced platelet aggregation was significantly lower 2 hours after taking dabigatran compared to baseline value (79.39 [13.38] vs 90.14 [10.5]).
Conclusion:
The TRAP-induced platelet aggregation was reduced in cardiovascular patients 2 hours after receiving dabigatran. Our findings could have some important clinical implications because platelet aggregation and coagulation cascade are affected at the same time.
Introduction
Dabigatran etexilate is a low-molecular-weight prodrug that exhibits no pharmacological activity. After oral administration, dabigatran etexilate is converted to its active form, dabigatran, a potent, competitive, and reversible direct inhibitor of thrombin. 1 –5 Thrombin has an active site and 2 secondary binding exosites. 6 Dabigatran binds to the active site, thereby inactivating both fibrin-bound and unbound (free) thrombin. 7 Thrombin is an essential enzyme in the coagulation system, but it is also the strongest endogenous platelet agonist. 8 Thrombin’s effect on platelets is mainly mediated by 2 G-protein-coupled receptors, protease-activated receptor (PAR) 1 and PAR4. 9 Mechanistically, thrombin proteolytically cleaves a part of the extracellular loop of these receptors. It mediates platelet aggregation, calcium mobilization, and platelet shape change, ultimately resulting in activation of the platelet GPIIb/IIIa receptor. 10 –12 This means that hemostasis is affected on many levels, which may result in increased risk of bleeding. Therefore, the inhibition of thrombin will affect primary (platelets) and secondary (coagulation factors) hemostasis. This conclusion is indirectly confirmed by recent studies that showed an increased risk of bleeding with dabigatran therapy. Hernandez et al showed that the use of dabigatran is associated with a higher risk of bleeding relative to warfarin, with 30% greater risk of any bleeding event, 58% greater risk of major bleeding, and 85% greater risk of gastrointestinal bleeding. The risk of intracranial hemorrhage is higher among warfarin users, with a hazard ratio of 0.32 (95% confidence interval: 0.20-0.50) for dabigatran compared with warfarin. 13 In 2014, Graham et al showed that dabigatran is associated with 28% greater risk of gastrointestinal hemorrhage compared to warfarin therapy. 14 No increased risk of major bleeding with dabigatran was found. In both analyses, treatment with dabigatran was associated with a significantly lower risk of intracranial hemorrhage, a reduction of about 65% compared with warfarin. Report, which is based on an analysis of the US Food and Drug Administration’s Mini-Sentinel database, showed that there is no difference in rates of gastrointestinal bleeding and intracranial hemorrhage among dabigatran users compared with warfarin-treated patients. 15 This review has serious limitations, as it is based on inpatient diagnosis codes, did not adjust for confounding variables, and did not include a detailed medical record. In the analysis by Hernandez, Zhang, and colleagues, 16 they looked at a 5% random sample of Medicare beneficiaries in 2010 and 2011 from the Centers for Medicare and Medicaid Services (CMS). After adjustment for baseline characteristics using propensity-score weighting, the adjusted incidence of major bleeding was 9.0% in the dabigatran users vs 5.9% in the warfarin group. Intracranial bleeding was significantly lower with dabigatran.
Therefore, the aim of the present study was to assess the effects of dabigatran on in vitro platelet aggregation in patients with nonvalvular atrial fibrillation.
Materials and Methods
The local ethical committee of the Jessenius Faculty of Medicine in Martin approved this study (EK 1702/2015). All study participants agreed to participate in the project and signed a written informed consent in accordance with the Declaration of Helsinki.
Dabigatran was administrated twice daily (at 7:00
Light transmission aggregometry (LTA) was performed using the international protocol for the laboratory investigation of platelet function. 17 We want to emphasize that testing was performed on patients without any antiplatelet or nonsteroidal anti-inflammatory drugs (10-14 days before measurement) and with normal platelet count (≥150 × 109/L). The antecubital venous blood was collected into tubes containing 3.2% buffered sodium citrate (anticoagulant–blood ratio 1:9) to assess platelet aggregation. Platelet aggregability was tested with platelet-rich plasma using platelet aggregometry (PACKS-4 aggregometer; Helena Laboratories, Texas, USA). Blood samples were stimulated with thrombin receptor agonist peptide (TRAP; 32 μmol/L).
All patients on dabigatran treatment included in our study were hospitalized on the First Department of Internal Medicine during July and November 2016, and there were no selection criteria. Data are presented as numbers with frequencies for categorical variables and means with standard deviations for continuous variables. For comparison of the different groups, the closed test principle was used. An overall comparison was performed, followed by pairwise comparison if the results were significant. All tests were 2 tailed, and P values less than .05 were considered statistically significant. Data were analyzed with SPSS 21.0.0.0 (SPSS Inc, Chicago, Illinois).
Results
Table 1 presents full clinical baseline characteristics of the patients. A total of 28 patients with nonvalvular atrial fibrillation were enrolled. The mean age was 71.57 (9.75) years (range: 50-87 years), 16 patients were women, and the mean CHA2DS2VASc score was 3.93 (1.41). All patients began treatment with dabigatran as initial anticoagulant treatment. The minimum term use of dabigatran was 18 days. Dabigatran doses were 110 mg (57.14%) or 150 mg (42.86%) twice daily. The mean dabigatran concentration was 90.56 (77.61) ng/mL in sample 1 and 143.27 (103.62) ng/mL in sample 2 (see Figure 1). The TRAP-induced platelet aggregation did not correlate significantly with dabigatran levels (see Table 2 and 3).
Clinical Baseline Characteristics of the Patients.
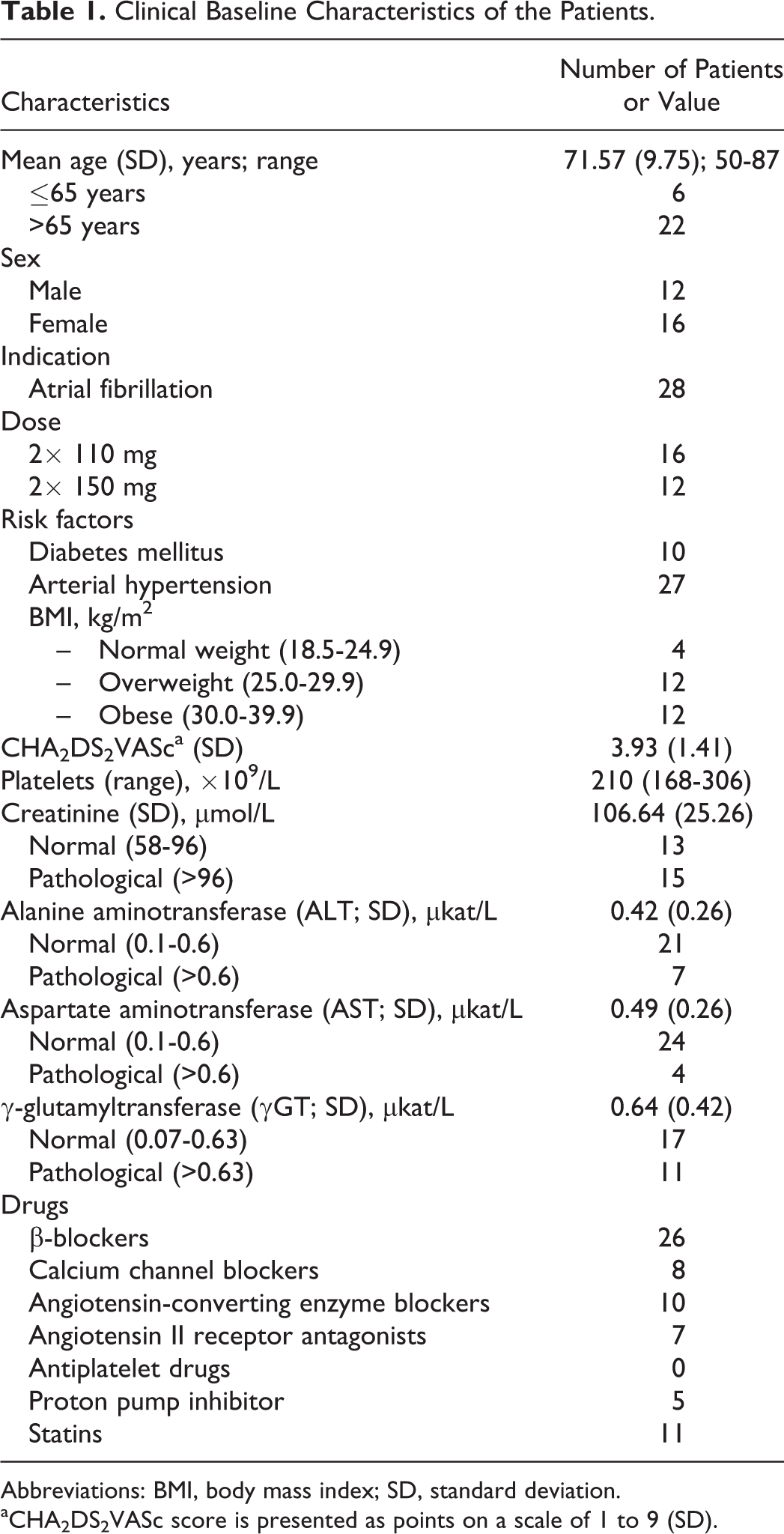
Abbreviations: BMI, body mass index; SD, standard deviation.
aCHA2DS2VASc score is presented as points on a scale of 1 to 9 (SD).
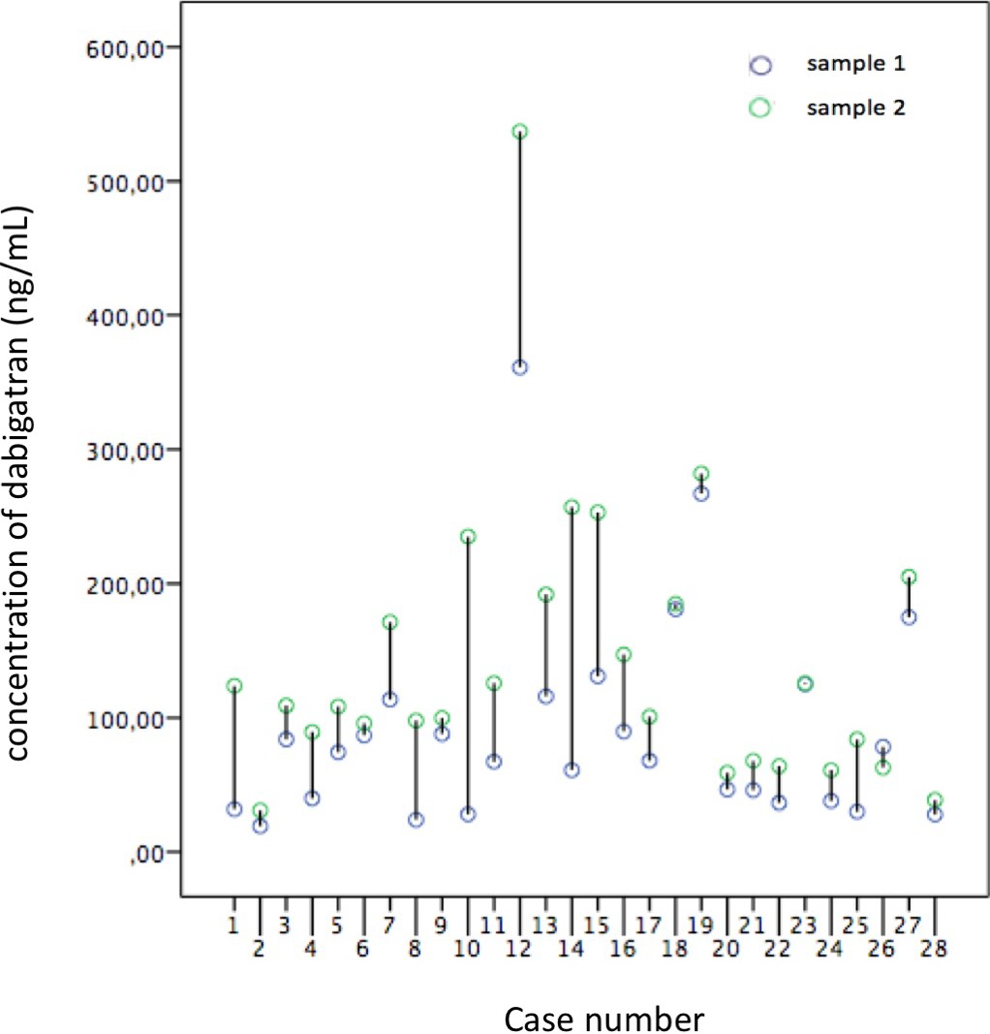
Concentration of dabigatran for each patient.
Correlation Between TRAP-Induced Platelet Aggregation and Anti-Factor IIa in Sample 1.a
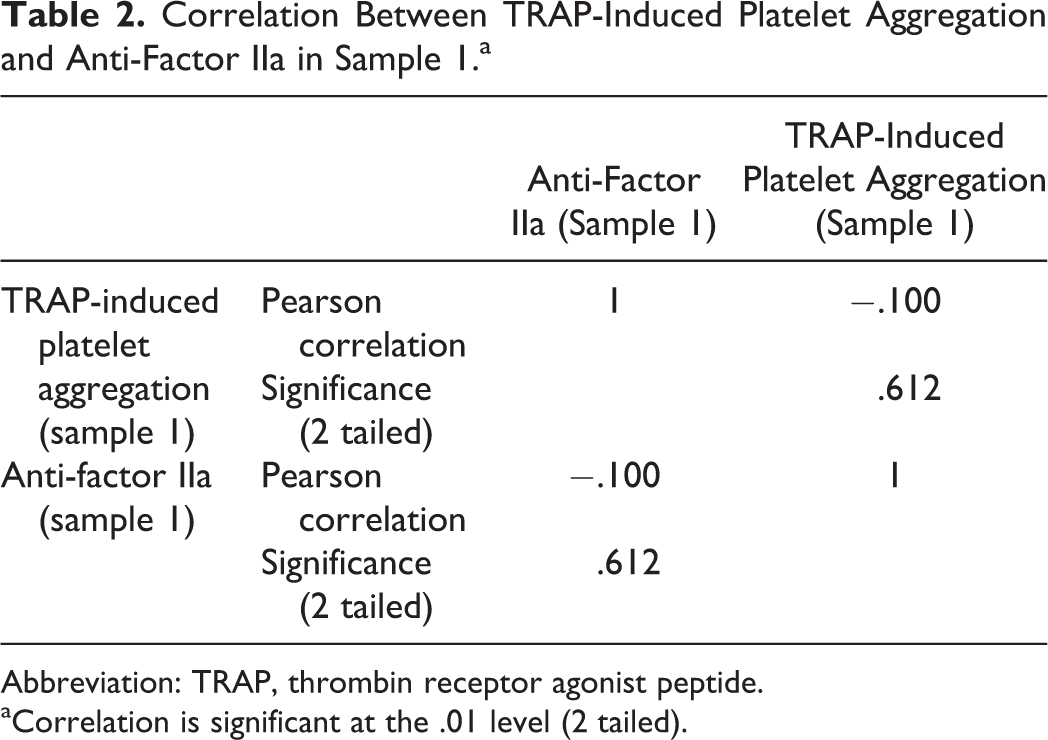
Abbreviation: TRAP, thrombin receptor agonist peptide.
aCorrelation is significant at the .01 level (2 tailed).
Correlation Between TRAP-induced Platelet Aggregation and Anti-Factor IIa in Sample 2.a
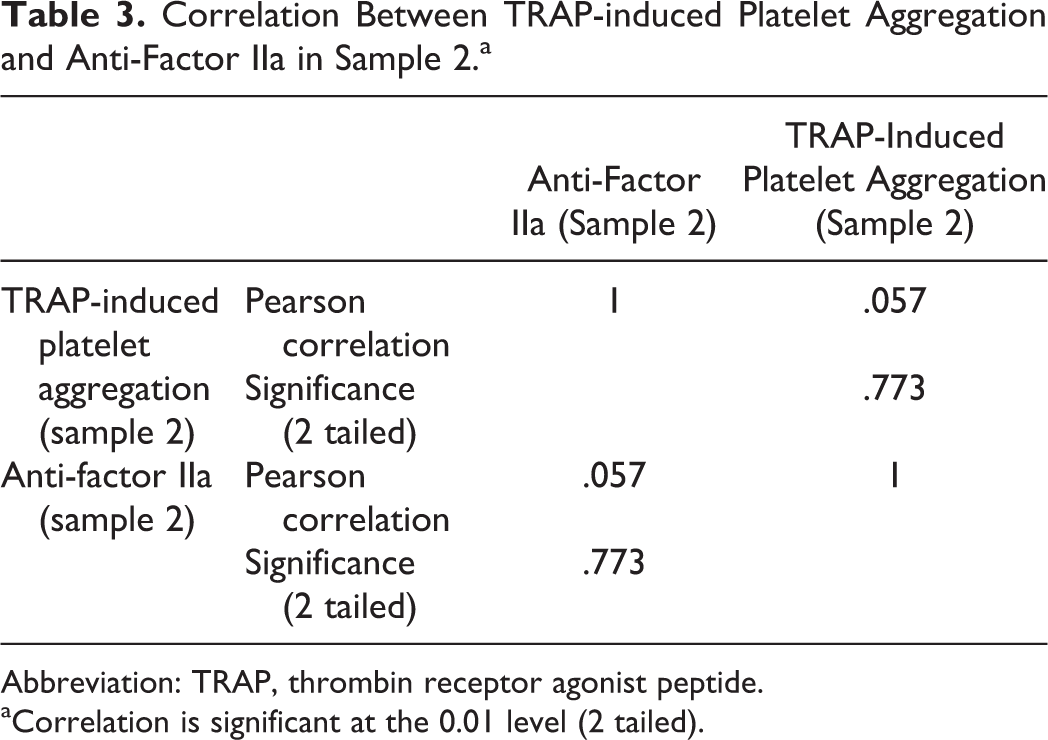
Abbreviation: TRAP, thrombin receptor agonist peptide.
aCorrelation is significant at the 0.01 level (2 tailed).
The TRAP-induced platelet aggregation was analyzed independently of concomitant treatment. As shown in Figure 2, the TRAP-induced platelet aggregation by LTA was significantly reduced in sample 2 compared with sample 1 (79.39% [13.38%] vs 90.14% [10.5%]; P = .000). Aggregation results for each patient are shown in Figure 3. We have done 15 subgroup analyses in order to determine the impact on aggregometer measurement (see Table 4). We did not find any significant difference between the groups.
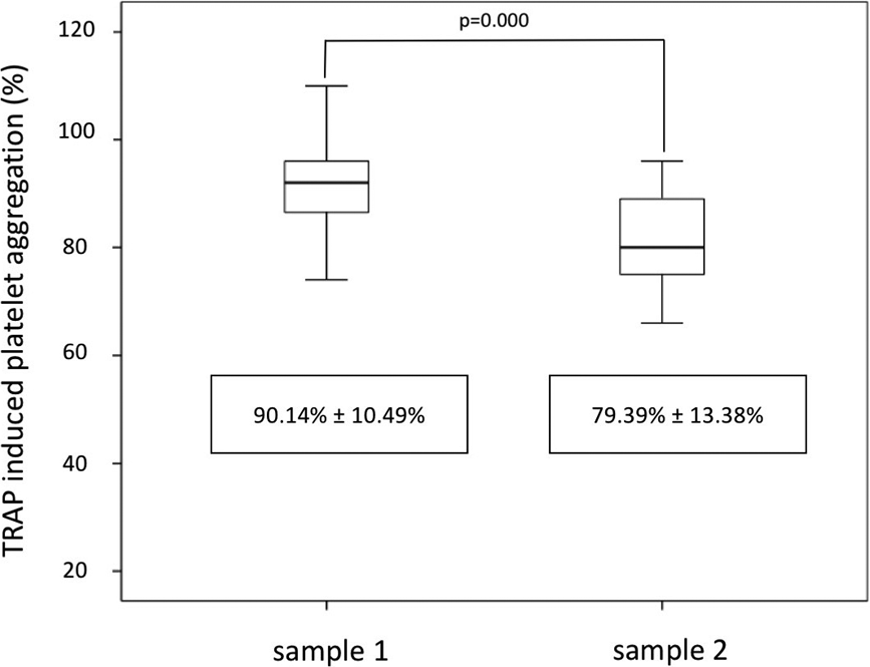
Results of thrombin receptor agonist peptide (TRAP)–induced platelet aggregation.
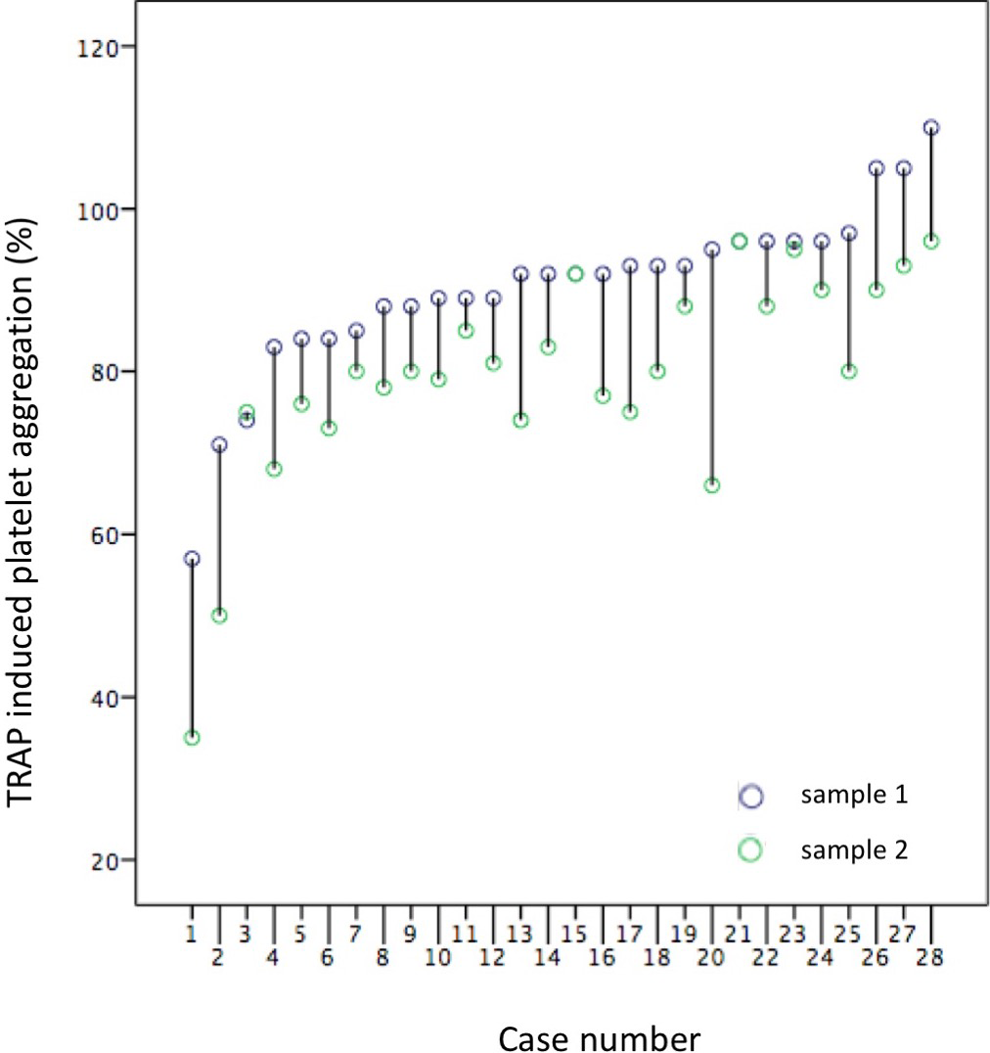
Aggregation results for each patient.
Subgroup Analysis.
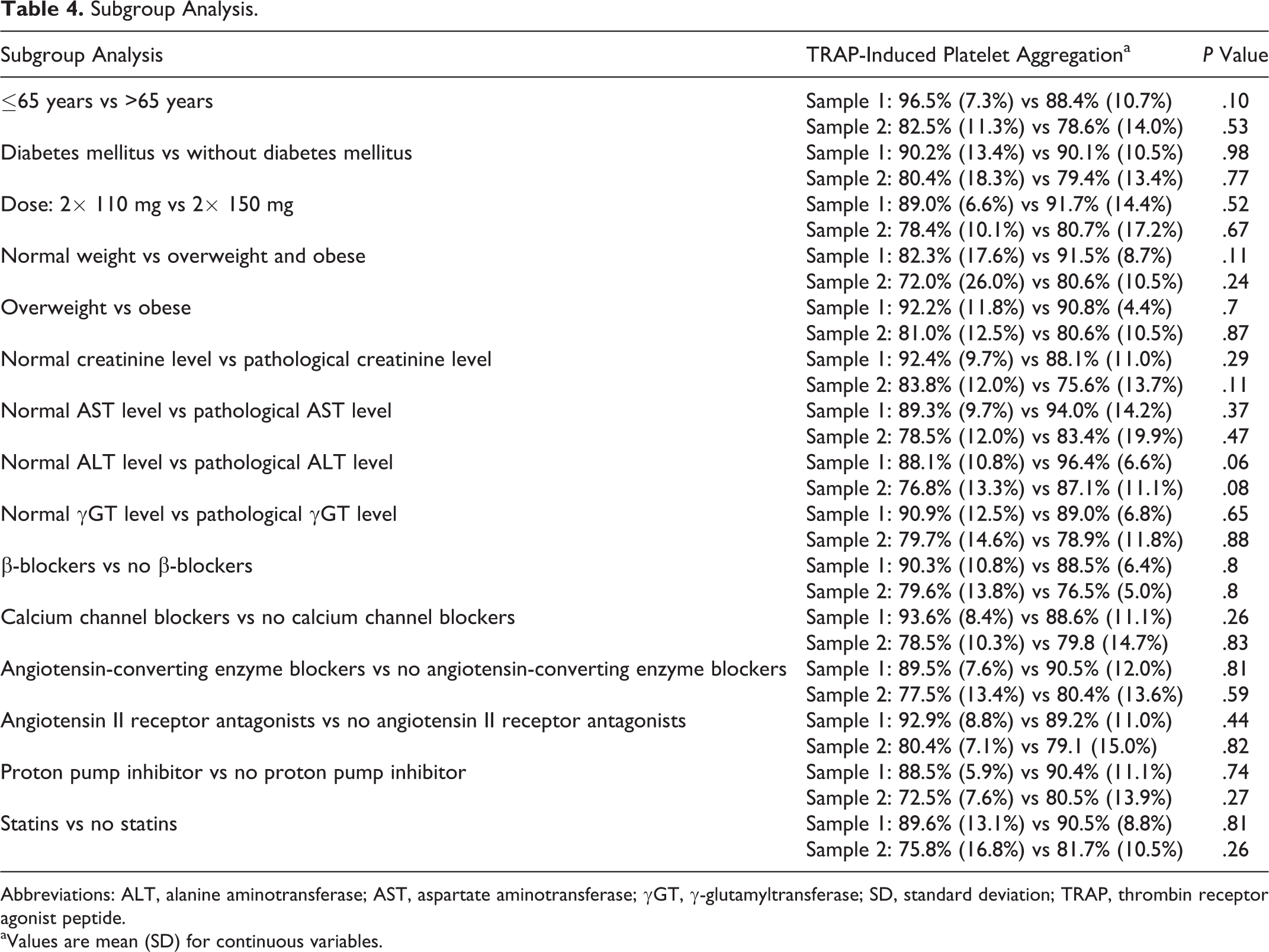
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; γGT, γ-glutamyltransferase; SD, standard deviation; TRAP, thrombin receptor agonist peptide.
aValues are mean (SD) for continuous variables.
Discussion
To our knowledge, this is the first prospective comprehensive study to test whether the dabigatran affects TRAP-induced platelet aggregation. Thrombin is not only a key protein in the cascade of fibrin clot formation but also a potent inducer of platelet aggregation. 17 –19 Dabigatran is a competitive reversible nonpeptide antagonist of thrombin. Most of the actions of thrombin are inhibited in vitro by dabigatran. However, the effects of dabigatran on platelet aggregation are still not fully understood, while there is controversial finding for dabigatran and platelet function. 20 –22
This single-center study quantifies platelet aggregation in 28 patients treated with dabigatran by LTA. The TRAP-induced platelet aggregation was significantly lower 2 hours after taking dabigatran compared to baseline value (79.39 [13.38] vs 90.14 [10.5]). Thus, dabigatran reduces platelet aggregation by 12% at the time of maximum plasma concentration. No significant differences were found in the subgroup analysis. Thrombin is capable of activating platelets, which is mediated primarily by the hydrolysis of a G-protein-coupled receptor on the platelet membrane, referred to as PAR-1, and a second receptor (PAR-4) that expresses a lower sensitivity to thrombin. However, dabigatran-bound thrombin is unable to cleave and activate PAR-1. It is also suggested the dabigatran acts upstream of the PAR-1 thrombin receptor by directly binding and inactivating thrombin. 23
Our findings could have some important clinical implications because platelet aggregation and coagulation cascade are affected at the same time. Several studies have shown that the use of dabigatran is associated with a higher risk of bleeding relative to warfarin. 13 –16 The situation could be worse during concomitant administration of antiplatelet or anticoagulant agents. Concomitant use of these drugs should be used with utmost care.
There were several limitations in our study. The small number of participants may have limited the ability to detect small drug effects on platelet function. Secondly, this study was not powered for clinical outcome. Therefore, it cannot be concluded that, for example, combination of antiplatelet therapy and dabigatran is not safe. Thirdly, platelet aggregability is greatly affected by preanalytical issues, and therefore, interpretation of platelet hyperaggregability is potentially accordingly adversely influenced.
Conclusion
In conclusion, our study showed significant effect of dabigatran on platelet aggregability. Further, larger investigations are required to confirm our hypothesis-generating work and correlate them with thrombotic and bleeding clinical events.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants VEGA 1/0187/17.
