Abstract
Background:
Four direct oral anticoagulants (DOACs) have been licensed for the treatment of atrial fibrillation (AF); efficacy and safety have been shown in clinical trials, but its real use in elderly and very elderly people is still unclear.
Aim:
To evaluate the impact of DOACs in our patients (pts) aged ≥75 years and switched from other treatments.
Methods:
From September 2013 to May 2015, all consecutive pts aged ≥75 years, males and females, in treatment for AF and switched to DOACs are considered in this study. Follow-up (FU) was scheduled after 3 and 6 months by phone and after 12 months by visit.
Results:
Two hundred thirty-two pts in treatment for AF were switched to DOACs, among these 143 (61.6%) pts aged ≥75 years (mean age, 81 years). The medium FU was 9.6 months, during which 4 minor bleedings in 4 different pts and 1 clinically relevant nonmajor bleeding were reported, all treated with temporary cessation or reduction in DOACs. Two strokes occurred in pts in treatment with dabigatran 110 mg, both resolved without serious sequelae; 2.8% of pts had nausea, itching, vomiting, or discomfort, half of these returned to acenocumarol, and the remaining switched to other DOAC. Four pts died, but the deaths were not related to anticoagulation.
Conclusion:
As reported for general people, also in our elderly population, DOACs resulted in a good alternative to old antithrombotic therapies. Efficacy and safety associated with a higher compliance by pts bring these drugs to be the first choice for long-term anticoagulation.
Introduction
Atrial fibrillation (AF) is the most common arrhythmia, and its treatment plays an important role in the public health, causing the continuing increase in new cases. In the world, the prevalence of AF is estimated between 0.5% and 1% in general population. In the early years of this decade, in Italy, the adult population with AF is estimated to be around 1.9%, slightly less than Germany (2.3%) or Sweden (2.9%), 1 but in the people aged >80 years increases over 8%. 2 Atrial fibrillation is not a life-threatening disease, but it can significantly influence quality of life, 1 and in most cases, it is associated with other socioeconomics problems, such as disability, cognitive disturbances, hospitalization, and need for continuous care at home or specialist care. 3 Until the short time ago, the only oral anticoagulant treatment available for patients (pts) with AF was the vitamin K antagonists (VKAs), but now, 4 direct oral anticoagulants (DOACs) are licensed to be used in case of AF: apixaban, dabigatran, edoxaban, and rivaroxaban. 4 –7 The clinical trials carried out with these drugs have shown their safety and efficacy compared with warfarin, but few and unclear data 8 are available about their use in elderly or very elderly pts. Despite the less drug–drug interaction, the less drug–food interaction, and the no need for routine monitoring make it easy for its practical use, 9 the absence of a specific reversal drug in case of bleeding 10 sometimes does not make them the first-choice drugs, especially in case of elderly pts.
In our study, we wanted to evaluate the impact of DOACs on our pts aged ≥75 years and switched from other treatments during 12 months of follow-up (FU). We also wanted to consider the interaction of this new treatment with their preexisting comorbidities and related therapies and the impact with their quality of life, and we have evaluated the difficultly to take the new drugs and the need for care comparing with what happened in the past.
Patients and Methods
Patients
From September 2013 to May 2015, all consecutive pts aged ≥75 years, males and females, previously treated with VKAs, warfarin, and acenocumarol; or low-molecular-weight heparin (LMWH) or fondaparinux; and/or antiplatelet drugs for AF (paroxysmal, persistent, or permanent) and switched to DOACs due to time in therapeutic range (TTR) <50% and/or low compliance to previous treatment and/or difficulty to perform periodic checks were initially considered.
Methods
All data regarding age, sex, renal and hepatic function, previous anticoagulants intake, TTR, CHA2DS2-VASc score to assess thromboembolic risk and HASBLED score to assess bleeding risk, all adverse events, and their management were reported in our database.
The guidelines to the use of DOACs for AF establish: apixaban (Eliquis; Bristol-Myers Squibb/Pfizer, Uxbridge, UK) 5.0 mg twice daily (BID) or 2.5 mg BID in pts at bleeding risk or in pts with at least 2 of following characteristics: age ≥ 80 years, plasmatic creatinine ≥1.5 mg/dL, and/or weight ≤60 kg; dabigatran etexilate (Pradaxa; Boehringer-Ingelheim, Ingelheim am Rhein, Germany) 150 mg BID or 110 mg BID in pts at bleeding risk or in pts ≥80 years with or without moderate renal failure (creatinine clearance [CrCl]: 30-50 mL/min) or pts with weight <50 kg; and rivaroxaban (Xarelto; Bayer, Bayer Pharma AG, Berlin, Germany) 20 mg 4 times daily (QID) or 15 mg QID in pts at bleeding risk or in pts with CrCl 15 to 30 mL/min.
Patients with severe renal failure (CrCl <15 mL/min) or severe hepatic failure were excluded from switch. Due to the difficulty in checking the international normalized ratio (INR) immediately before starting DOACs, such as recommended, considering pharmacologic profile of VKAs and conventional bridging therapy, we have decided to stop warfarin 48 hours and acenocumarol 24 hours before starting DOACs.
Follow-up was scheduled after 3 and 6 months by phone contact and after 12 months by visit at our center. During the last visit, we have orally administered to all pts 5 questions concerning their quality of life: (1) Therapy with DOACs gives reasonable assurance concerning the control of your disease? (2) Is it easy to remember to take this new therapy now as there are only few controls?; (3) The quality of life appears to have improved with these new drugs? (4) The need for help by family members or caregivers is increased or decreased compared to before? and (5) Would return to take VKAs? All responses were recorded but not statistically evaluated.
Major bleeding was defined following the International Society for Thrombosis and Hemostasis (ISTH) criteria 10 : fatal bleeding and/or symptomatic bleeding in a critical organ or area such as intracranial, intraspinal, intraocular, retroperitoneal, intra-articular, pericardial, or intramuscular with compartment syndrome and/or bleeding causing a fall in hemoglobin level of 2 g/dL or more or leading to transfusion of 2 or more of units of whole blood or red cells. Clinically relevant nonmajor bleeding (CRNMB) was defined as less acute events, for example, an asymptomatic hemoglobin drop, and bleeding events that result in a temporary cessation of antithrombotic therapy.
Results
From September 2013 to May 2015, a total of 232 pts with AF were switched to DOACs, 143 of them were aged ≥75 years (61.6%; mean age, 81 years). Baseline features are shown in Table 1.
Baseline Features of Patients.
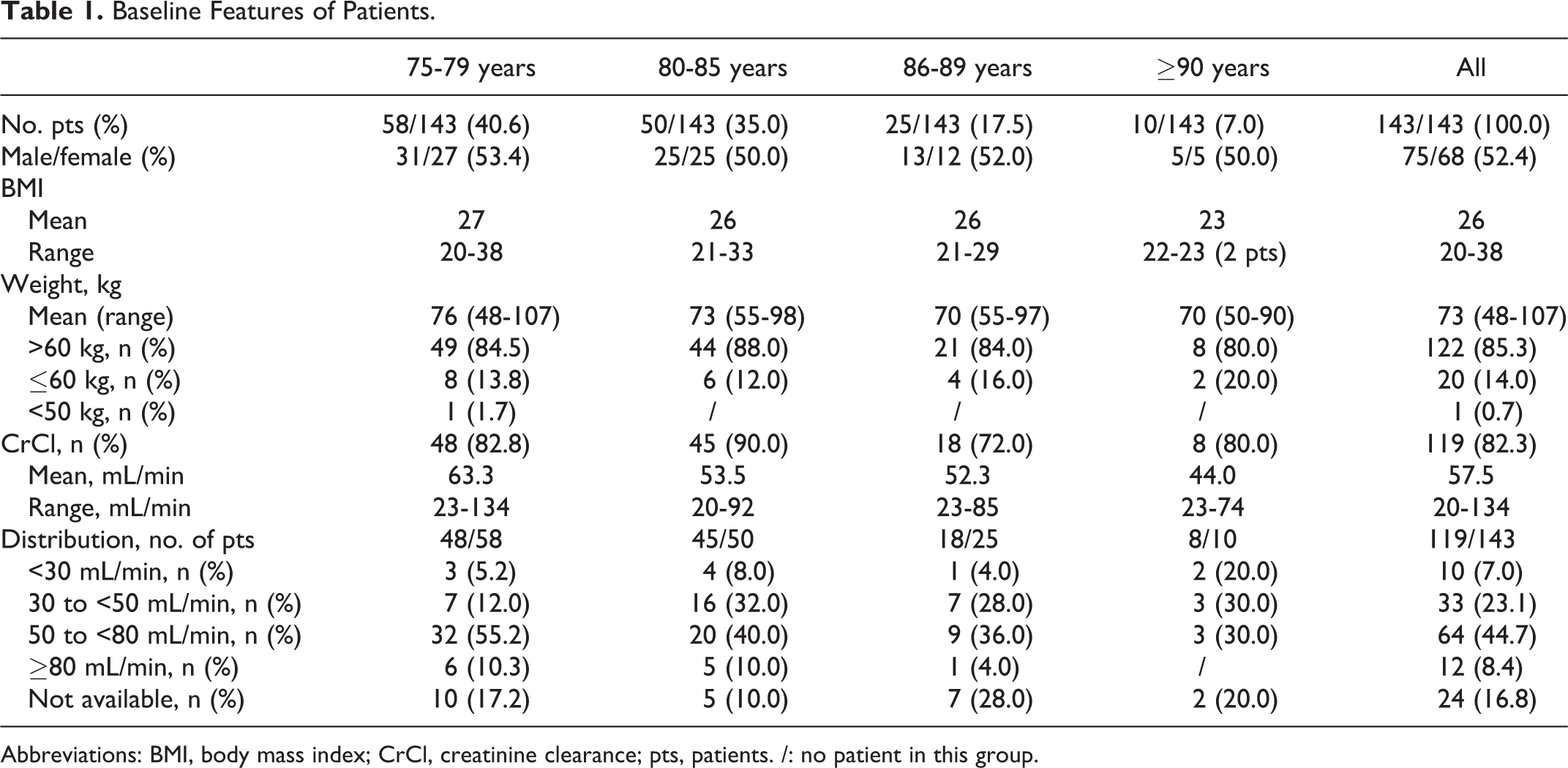
Abbreviations: BMI, body mass index; CrCl, creatinine clearance; pts, patients. /: no patient in this group.
All pts were previously treated for AF, paroxysmal, persistent, or permanent, with anticoagulant drugs (oral or parenteral) and/or with antiplatelets. All 3 DOACs licensed in Italy for AF treatment (apixaban, dabigatran, and rivaroxaban) were used to replace the old therapies such as reported in Table 2. Of note, 61.7% of pts were switched to DOACs due to TTR <50%; 18.2% due to low compliance to previous therapy, generally parenteral treatment with LMWH or fondaparinux, and the remaining 20.1% due to difficultly to perform periodic checks (remoteness from hospital, need for family or caregivers help, difficult venous access, etc).
Previous and New Treatments for AF and Scores.
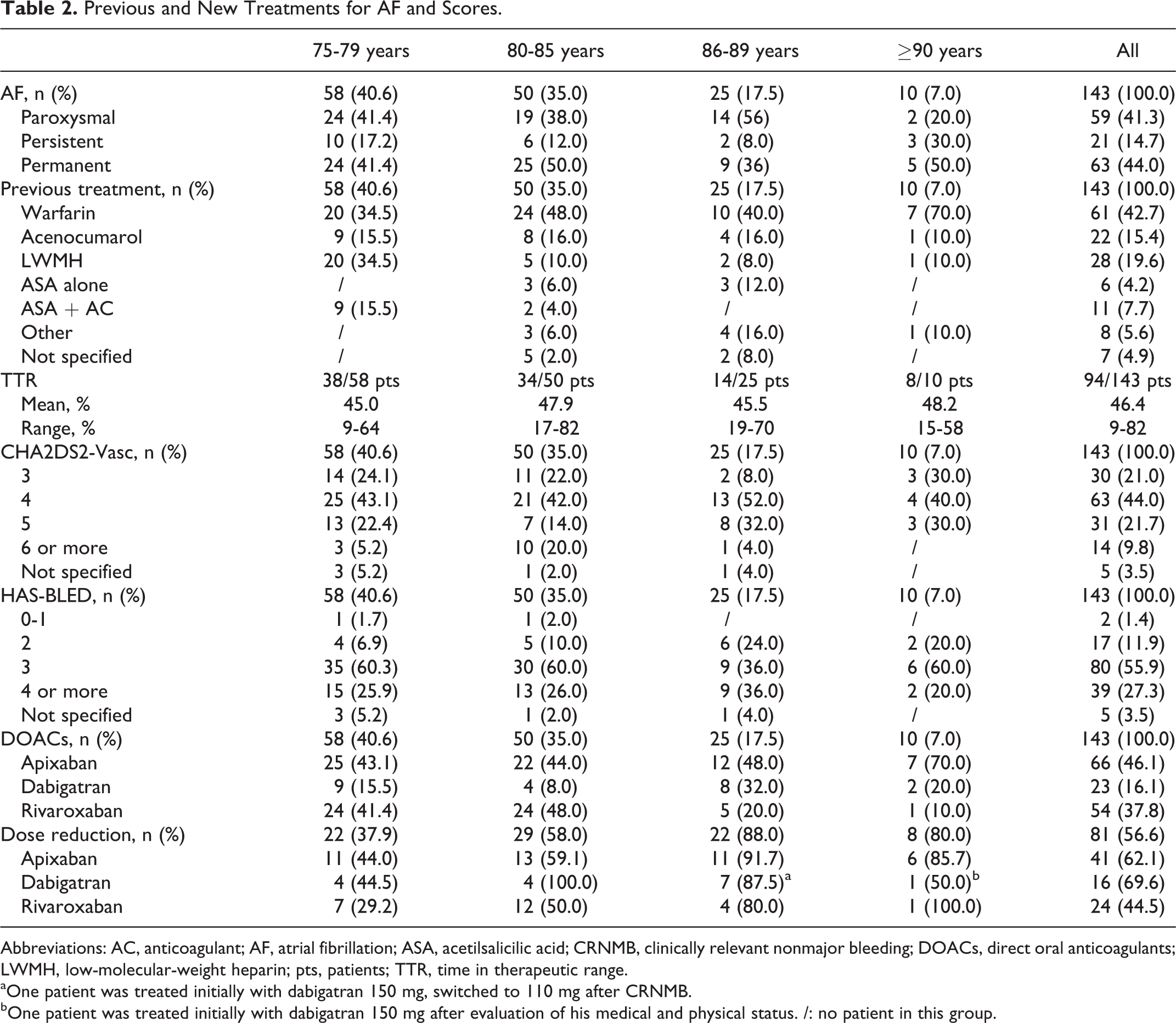
Abbreviations: AC, anticoagulant; AF, atrial fibrillation; ASA, acetilsalicilic acid; CRNMB, clinically relevant nonmajor bleeding; DOACs, direct oral anticoagulants; LWMH, low-molecular-weight heparin; pts, patients; TTR, time in therapeutic range.
aOne patient was treated initially with dabigatran 150 mg, switched to 110 mg after CRNMB.
bOne patient was treated initially with dabigatran 150 mg after evaluation of his medical and physical status. /: no patient in this group.
The medium FU was 9.6 months (range, 1-21 months). During this period, 4 minor bleedings occurred in 4 different pts, respectively, in therapy with rivaroxaban (2 pts): hematoma in the muscle tissue of the left arm in a man aged 78 years (CrCl 71.88 mL/min) treated with rivaroxaban 20 mg and epistaxis in a man aged 86 years (CrCl 57 mL/min) treated with rivaroxaban 15 mg; with dabigatran (1 pt): hematoma in the muscle tissue of the right thigh in a woman aged 88 years (CrCl 31.80 mL/min) treated with dabigatran 110 mg; and with apixaban (1 pt): hematoma in the muscle tissue of the right arm in a man aged 81 years (CrCl 67.30 mL/min) in therapy with apixaban 2.5 mg. All these pts with minor bleeding were immediately treated with a brief cessation of DOACs and in 1 case with a dose reduction in rivaroxaban up to event resolution, when the previous dosage was restored. Only 1 gastrointestinal CRNMB in a pt treated with dabigatran 150 mg was reported, and the event was immediately treated with a temporary cessation of antithrombotic therapy and, when the bleeding was resolved, with a dose reduction in oral anticoagulant, maintained over time. Incidence of all bleedings is 41.6 per 100 pt/years. Stroke occurred in 2 pts, both ≥80 years, in treatment with dabigatran 110 mg, and after resolution, without serious sequelae, the pts were switched to rivaroxaban 20 mg to prevent other thromboembolic events; incidence is 16.7 per 100 pt/years. Four (2.8%) pts reported minor adverse events such as nausea, itching, vomiting, or discomfort; 2 of the pts returned to acenocumarol, and the remaining switched to other DOAC. Four pts died during FU, but no deaths were related to antithrombotic therapy. In effect, also due to advanced age, the majority of our pts had some serious comorbidities, as reported in Table 3.
Concomitant Diseases.
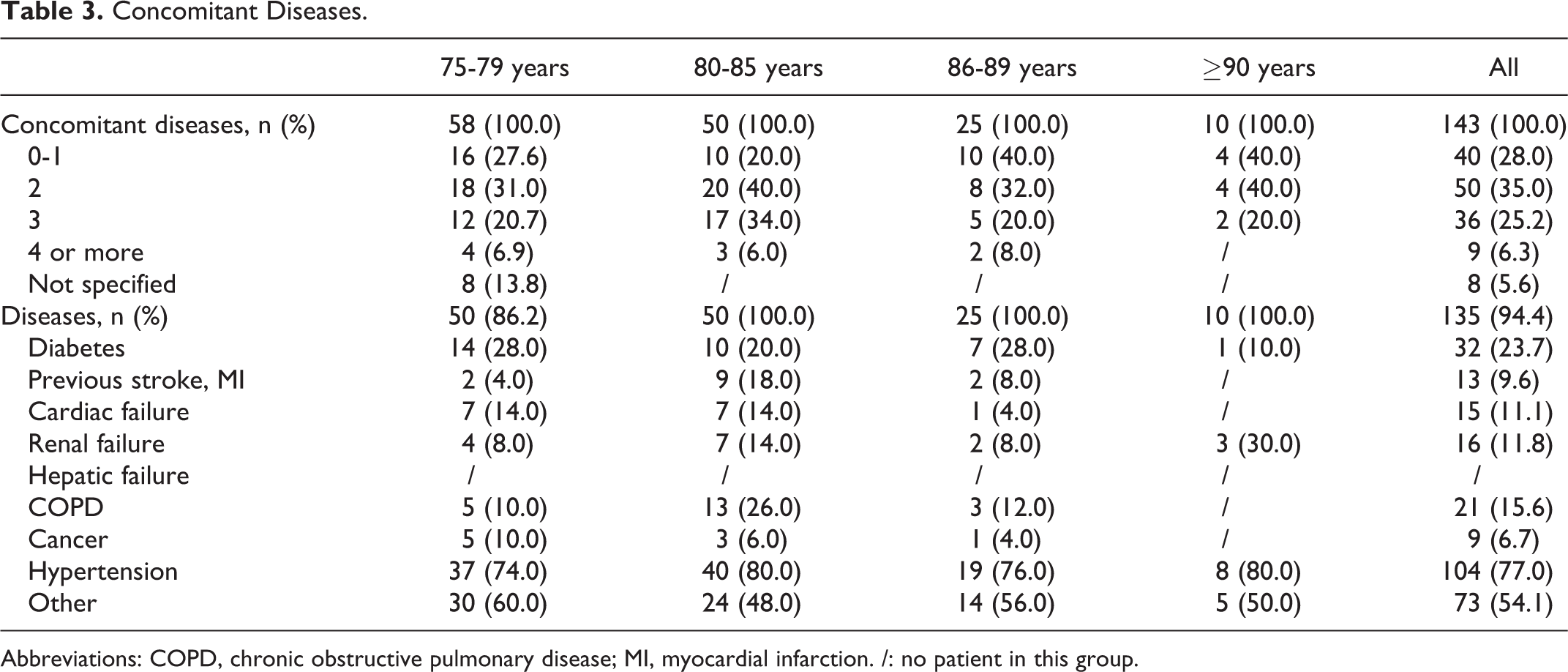
Abbreviations: COPD, chronic obstructive pulmonary disease; MI, myocardial infarction. /: no patient in this group.
Follow-up was performed by phone contact at 3 months to 88.1% of the pts and at 6 months to 67.8% of the pts and by visit at our center after 12 months (37.8% of the pts). All the surveyed pts were administered 5 oral questions concerning their quality of life. The responses showed that most pts appreciate the new treatment, it is considered easy to take, and it has reduced the need for help from family members or caregivers. No pt believes this treatment is worse than the last or want to return to VKAs. Only few pts (<10%) consider the 2 treatments stackable.
Discussion
Atrial fibrillation is associated with a higher incidence of mortality, stroke, or coronary events that can significantly affect the quality of life of pts with AF, also increasing other socioeconomics problems. 1 Atrial fibrillation is a rapid cardiac rate that may cause tachycardia and related cardiomyopathy. Several pharmacological treatments are commonly used for its therapy, but especially in older pts, the gold standard in treatment of AF has been given so far by VKAs. Patients with chronic or paroxysmal AF at high risk for stroke were usually treated with long-term warfarin or acenocumarol to achieve an INR of 2.0 to 3.0. 11 In the last years, 4 new DOACs were licensed for AF treatment: apixaban, dabigatran, edoxaban, and rivaroxaban. All phase 3 trials performed in different countries showed that DOACs are noninferior to warfarin to reduce thromboembolic events and bleeding, 4 –7 however, few data are available about elderly or very elderly people. 8 The purpose of this study was to evaluate the impact of DOACs (edoxaban excluded because in Italy it is not yet licensed) on our pts aged ≥75 years and switched from other treatments. Conversely, the clinical trials 4 –7 where the number of elderly pts with AF was between 25% 7 and 40.1%, 6 in our case, the percentage was much higher (61.6%) and female sex more represented with 42.7% in our general population, rising to 47.6% in the population older than 75 years. Evaluating the 2 scores, CHA2DS2-VASc and HAS-BLED, and the TTR in case of pts in previous treatment with VKAs, we can point out that the mean CHA2DS2-VASc in our pts was 4.2 and in 31.5% of them was ≥6.0, very high if we consider the phase 3 trials, where, however, the mean was calculated for total study participants. Naturally, this datum is closely related to the age of our pts. In fact, many of them had several major comorbidities, and among these, the most represented were hypertension and diabetes. The mean HAS-BLED was 3.2, without significant difference among the 4 age-related groups considered. The mean TTR was 46.2%, decidedly lower than reported in clinical trials. 4 –7 Despite the major difference found with the Aristotle study, 4 when mean CHA2DS2-VASc was 2.1 and mean TTR was 62.2%, both calculated on the total participants in the trial, but following the statement made by LaHaye et al 12 about the selection criteria of DOACs, recently confirmed by Deitelzweig et al 13 in the US people, we chose apixaban for AF treatment for many of our elderly pts to prevent stroke or systemic embolism without increasing bleeding risk. In fact, also reported by Schulman, 14 only evaluating data reported by clinical trials, but without a direct comparison among DOACs, apixaban may be associated with better profile of efficacy and safety, consideration which must still be proven in the future. International normalized ratio should be tested at the end of VKA therapy, and as recommended, rivaroxaban should be started when INR is below 3.0, whereas dabigatran and apixaban when INR is below 2.0. But as shown by the Dresden NOAC Registry, 15 where only 75% of the pts was testing INR before starting DOACs, there are some difficulties to obtain these data, therefore, to overcome this, considering pharmacologic profile of VKAs, we have established to stop warfarin 48 hours and acenocumarol 24 hours before starting DOACs. No bleeding events occurred during the switch. During this study, 4 minor bleedings occurred in 4 different pts and 1 CRNMB was reported, following ISTH recommendations, 10 and all these events were treated with temporary cessation or reduction in DOACs. Two strokes occurred in pts in treatment with dabigatran 110 mg, both resolved without serious sequelae. Given a low number of pts in treatment with dabigatran, it is difficult to compare this result with that of RE-LY trial. 5 Of note, 2.8% of pts had nausea, itching, vomiting, or discomfort in the 3 first months from switch, half of these returned to acenocumarol, and the remaining switched to other DOAC. Four pts died, but the deaths were not related to anticoagulation but to their preexisting diseases. Vitamin K antagonists require frequent laboratory controls due to numerous drug–drug and food–drug interactions, 16 and the relationship to these controls, need to maintain INR in therapeutic range, and the efficacy/safety of treatment are now proven, 17,18 but in several cases, the distance to hospital and the need to be accompanied by a family member or by a nurse cause discomfort in the pts and make difficult the controls, conversely, the DOACs have wide therapeutic window, few drug interactions, no frequent controls, or dose adjustments. All our pts switched to DOACs and followed at 12 months have orally expressed their appreciation for this new treatment.
As reported in clinical trial and registries for general people, also in our little experience in a narrow and selected group of pts, DOACs resulted in efficacy and safety associated with a higher compliance. For this reason, in our center, in the near future, the DOACs will surely the drugs that will be considered first for the treatment of very elderly people in case of long-term anticoagulation, but more studies and more treated pts are needed to confirm our opinion.
Limitations
In our study, the major limitation is given by small sample of pts and by short FU, only 37.8% of pts at 12 months, but in our country, DOACs were licensed much later than other countries, European and non-European, and the experience with these drugs was up to now still limited. All the pts considered were pts switched from other drugs taking several years with different difficulties. The selection of these pts over than others is due only to little number of pts evaluated in a short period of time, in the future, a selection among anticoagulated pts with AF must be made dividing the pts depending on the drugs taken and the time of initiation therapy comparing these data with compliance, side effects, and need to switch. Very important for these elderly pts is the impact that these new drugs can have on their quality of life, and only few oral questions are not sufficient to establish it, a written questionnaire statistically evaluated could be undoubtedly a better choice.
Footnotes
Authors’ Note
The authors SP, AB, UV, and GB contributed equally to conception of the study, interpretation of data, and drafting the article. All authors had full access to all the data and takes responsibility for the accuracy of the data and analysis. This is a retrospective, spontaneous study, not supported by public or private companies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
