Abstract
Background:
Our aim was to identify laboratory assays in order to assess the anticoagulant effects of dabigatran etexilate (DE).
Methods:
Twenty patients with nonvalvular atrial fibrillation treated on DE (110 mg per os twice daily) and 20 on acenocoumarol were studied. Conventional coagulation tests, endogenous thrombin potential (ETP), thromboelastometry (ROTEM), epinephrine-induced light transmission aggregometry (LTA), and Hemoclot Thrombin Inhibitors (HTI) were performed in all patients.
Results:
In ROTEM analysis, the lysis index at 60 minutes was significantly lower in patients receiving DE (P = .011). In LTA, patients on DE showed decreased aggregation compared to those on acenocoumarol, marginally insignificant (P = .068). Regarding ETP, acenocoumarol affected thrombin generation more than dabigatran (area under the curve [AUC], P < .001), while statistically significant associations were detected between dabigatran levels, as determined by the HTI assay, and almost all parameters of ETP assay (AUC, P < .001).
Conclusion:
The role of ETP in estimating anticoagulant activity of dabigatran possibly requires further research.
Introduction
Atrial fibrillation (AF) is the most frequent cardiac arrhythmia worldwide, often complicated by intracardiac thrombus formation, which can subsequently embolize to peripheral arteries, causing serious and/or fatal thrombotic events. Atrial fibrillation and prevention of stroke or systemic thrombosis are extremely important in anticoagulant drug treatment. Ongoing developments in anticoagulant therapy have introduced direct thrombin inhibitors (DTIs) for prophylactic or therapeutic anticoagulation. In Europe and Canada, a new oral DTI, dabigatran etexilate (DE; Pradaxa, Boehringer Ingelheim Pharmaceuticals, Inc; Germany), has been approved for preventing either peripheral arterial embolism after AF 1 or primary venous thromboembolism after major orthopaedic surgery 2 while recently Food and Drug Administration also approved Pradaxa for treatment and reduction in the risk of recurrence of deep venous thrombosis and pulmonary embolism. Dabigatran etexilate is a potent, synthetic prodrug that is converted into active drug in vivo, rapidly acting DTI that specifically and reversibly inhibits both free and clot-bound thrombin by binding to the active site of the thrombin molecule. 3
Direct thrombin inhibitors have several advantages over heparins and vitamin K antagonists (VKAs). They do not require laboratory monitoring and dose adjustment for conventional anticoagulant treatment due to their short half-life, the wide therapeutic window, and the predictable anticoagulant response. However, monitoring may be necessary in certain cases, such as in patients with renal failure, in the elderly individuals, or in case of complications including severe bleeding or thrombosis while on anticoagulant therapy, potential overdose, and emergency surgery. 4 –7
Thrombin, which is inactivated by dabigatran, has a central role in the whole hemostatic process by affecting all of its phases. It activates platelets; amplifies the hemostatic response by activating factors V, VIII, and XI; converts fibrinogen to fibrin; stabilizes the fibrin clot through activation of factor XIII; 8 and further affects fibrinolysis by regulating an important factor in the clot stabilization, the thrombin activatable fibrinolysis inhibitor (TAFI). 9
The effect of dabigatran on the various coagulation assays has been mainly investigated in vitro using plasma spiked with dabigatran, 3,10 –14 so data on the ex vivo impact of dabigatran on routine coagulation assays are limited. 15,16
Taking into account findings revealing clinical concerns about severe hemorrhagic complications in certain groups of patients 17 –19 and arguments regarding the value of monitoring dabigatran levels and adjusting the dose, 20 we herein tried to determine the laboratory assays that could be used to assess its anticoagulant activity. Despite the availability of the Hemoclot test, 21,22 there is still uncertainty about the most proper readily available laboratory test to estimate the degree of anticoagulation with dabigatran. This anticoagulant effect has been compared to that of acenocoumarol, a classical VKA.
Methods
The study population consisted of 40 patients with nonvalvular AF on anticoagulation recruited from the Second University Department of Cardiology and Department of Haemostasis in the “Attiko” University Hospital in Athens, Greece. The 20 patients in group A received the standard dose of Pradaxa commercially available in Greece (110 mg per os twice daily) for patients with high congestive heart failure, hypertension, age ≥75 years, diabetes mellitus, stroke/transient ischemic attack, vascular disease, age 65-75 and female sex (CHA2DS2-VASc) score. Group B included 20 patients, matched on age and sex with those in group A, taking VKAs (acenocoumarol) with target international normalized ratio (INR) between 2.0 and 3.0. The study was performed in accordance with the Declaration of Helsinki and approved by the hospital’s institutional review board (Approval Number: 11, November 9, 2012). Informed consents were obtained from all patients.
Individuals with platelet count <100 × 109/L and active thrombosis or bleeding, patients under simultaneous anticoagulant and antiplatelet therapy or under VKA treatment with INR out of the defined limits, those with renal and/or hepatic and/or thyroid dysfunction, with malignancy or with chronic infectious or autoimmune diseases, and patients with active infection during initiation of the study were excluded.
For each participant, we calculated the CHA2DS2-VASc score, obtained detailed personal medical history (in respect to thrombotic and/or bleeding complications), and performed thorough clinical examination. At least 7 days after study enrollment, the following laboratory examinations were performed in all study participants including, complete blood count, thrombin time (TT), activated partial thromboplastin time (aPTT), prothrombin time (PT) and INR, fibrinogen,
The last dose of antithrombotic medication was administered approximately 3 hours before blood sampling. Light transmission aggregometry, ROTEM analysis, and conventional coagulation tests were carried out on the same day, within 2 hours of sampling. Complete blood counts were performed on a Sysmex XE-2100 analyzer (Roche, Illinois). Thrombin time, aPTT, PT and INR, fibrinogen, and
Blood samples for HTI analysis were anticoagulated with 0.109 mol/L trisodium citrate (9:1, v/v blood anticoagulant) and immediately centrifuged at 2500g for 20 minutes. For the ETP test, the blood specimen was centrifuged at 1500g for 20 minutes, the supernatant was removed and was then centrifuged again. Plasma was snap frozen in small portions and stored at –20°C until the assays were performed. Both tests were also performed on BCS XP System Haemostasis analyzer.
Hemoclot Thrombin Inhibitors
Hemoclot Thrombin Inhibitors (Hyphen BioMed, Neuville-sur-Oise, France) is an in vitro diagnostic device for the quantitative measurement of dabigatran in human citrated plasma, with a clotting method based on the inhibition of a constant and defined concentration of thrombin. For measuring DTI in plasma, first, the diluted plasma (1:8 to1:20) was mixed with a normal pooled human plasma. Clotting was then initiated by adding a constant amount of highly purified human thrombin. The clotting time measured is considered to be directly related to the concentration of assayed DTI in the tested plasma. Intra-assay coefficient of variation for HTI was 7.88%.
Endogenous Thrombin Potential
INNOVANCE ETP (Siemens Healthcare Diagnostics) is a global hemostasis function test to assess the ETP of plasma samples. The incubation of plasma with phospholipids and activator and calcium ions leads to initiation and propagation of the coagulation processes, eventually resulting in the generation of thrombin. Thrombin generation (TG) and the subsequent inactivation were recorded by monitoring conversion of a specific slow-reacting chromogenic substrate at a wavelength of 405 nm over time. The assay was performed using BCS XP System Hemostasis.
The estimated parameters of the TG curve included area under the curve (AUC), also referred to as ETP; the Lag time (tlag) that describes the time from the initiation of the reaction until TG is being observed; the time to peak (Tmax) which is the time from the initiation of the reaction until the maximum TG is being observed; and finally the maximum TG depicted by peak height (Cmax). The intra-assay coefficient of variation for AUC was 1.45%.
Fibrin aggregation inhibitor prevents the interference of fibrin with the optical detection of the chromophore. A mathematical algorithm was applied to correct the substrate conversion curve. Therefore, the activity of a 2 macroglobulin bound thrombin, which has no known biological activity, but is still capable of cleaving small chromogenic substrates, was subtracted from the substrate conversion curve.
Light Transmission Aggregometry
The whole blood specimen was collected in 3.8% trisodium citrate and centrifuged at 200g for 10 minutes to obtain platelet-rich plasma (PRP). The remaining specimen was recentrifuged at 2000g for 15 minutes to obtain platelet-poor plasma (PPP). The platelet count was adjusted to lie between 200 × 103/μL and 300 × 103/μL with PPP.
The test was conducted as described previously. 23 Aggregation was performed using a Biodata-PAP-4 aggregometer (Bio/Data Corp, Pennsylvania). The 100% line was set using PPP and a 0% baseline established with PRP before addition of the agonist. Epinephrine 5 μmol/L (CHRONO-LOG Corporation, Havertown, Pennsylvania) was the agonist used. The test procedure included the transfer of 0.45 mL PRP into a cuvette incubated at 37°C for 3 minutes. Subsequently, 25 μL of epinephrine were added into the PRP and the aggregation pattern was allowed to generate for 10 minutes.
Thromboelastometry
Viscoelastic measurements were done using rotational ROTEM (Tem Innovations GmbH, Munich, Germany). Recalcified blood was analyzed on the ROTEM analyzer (Tem Innovations GmbH) using the nonactivated TEM (NATEM) assay. The NATEM test is a semiquantitative in vitro diagnostic assay used on the ROTEM delta Thromboelastometry System to monitor the coagulation process, contact activated by the surface of the measurement cell, in citrated whole blood specimens. The resulting assay is very sensitive toward almost any kind of change in the balance of the coagulation system.
The following TEM variables were measured: the clotting time (CT [seconds]) that was determined by the time elapsed from the start of measurement until the formation of a clot 2 mm in amplitude. The clot formation time (seconds) was the time elapsed from the end of the CT (amplitude of 2 mm) until a clot firmness of 20 mm was achieved. The α angle (αo) was the angle between the central line (x-axis) and the tangent of the TEM tracing at the amplitude point of 2 mm describing the kinetics of clot formation. The maximum clot firmness (MCF [mm]) reflects the final strength of the clot. The lysis index at 60 minutes (LI 60) is defined as the percentage of remaining clot stability in relation to the MCF following the 60-minute observation period after CT and indicates the speed of fibrinolysis.
Statistical Methods
Descriptive statistics are presented as means ± standard deviations, medians and interquartile ranges (IQR), or percentages when appropriate. Because most of the variables were nonnormally distributed, nonparametric tests (the Fisher exact test and the 2-sample Wilcoxon rank-sum [Mann-Whitney] test) were used for the statistical evaluations. Correlations were assessed by the Spearman rank correlation coefficient and the respective P value. Spearman ρ of <.20 is considered to indicate very weak correlation, .21 to .40 indicates weak correlation, .41 to .60 indicates moderate correlation, .61 to .80 indicates strong correlation, and >.81 indicates very strong correlation. For hypothesis testing, a probability level of <.05 was considered to be statistically significant. All statistical tests were 2 sided. Stata software was used for all statistical analyses (Stata Corp, College Station, Texas).
Results
The descriptive characteristics, the associated comorbidities, and the hematological and biochemical parameters of both patient groups are presented in Table 1. The 2 groups were similar without any statistically significant differences. In all, 3 patients in group A and 1 in group B had CHA2DS2-VASc scores <2. The measurements of HTI in patients receiving dabigatran (median: 0.07[μg/mL]; IQR: 0.035-0.14) were nonnormally distributed.
Characteristics of Patients on Dabigatran (group A, n = 20) and Patients on Acenocoumarol (group B, n = 20).a
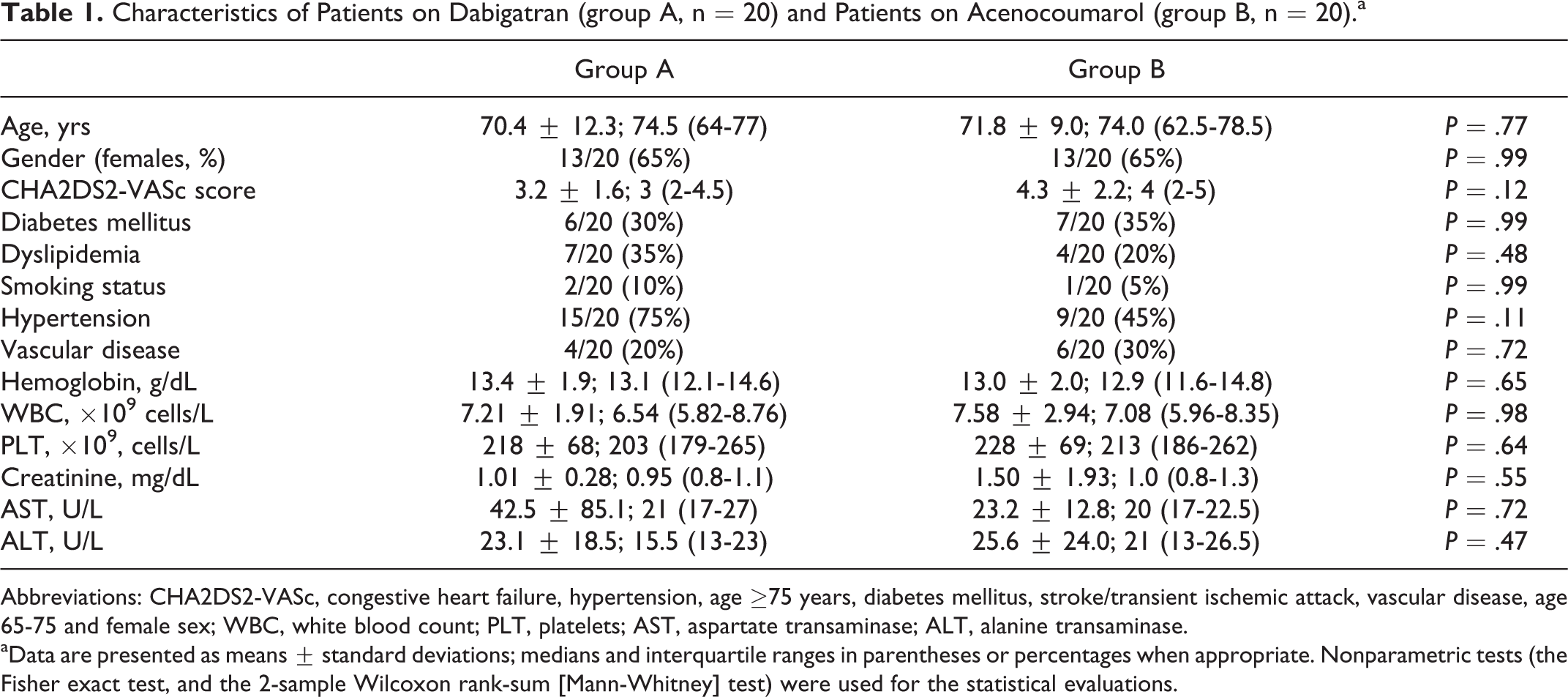
Abbreviations: CHA2DS2-VASc, congestive heart failure, hypertension, age ≥75 years, diabetes mellitus, stroke/transient ischemic attack, vascular disease, age 65-75 and female sex; WBC, white blood count; PLT, platelets; AST, aspartate transaminase; ALT, alanine transaminase.
aData are presented as means ± standard deviations; medians and interquartile ranges in parentheses or percentages when appropriate. Nonparametric tests (the Fisher exact test, and the 2-sample Wilcoxon rank-sum [Mann-Whitney] test) were used for the statistical evaluations.
The results of all coagulation assays are shown in Table 2. Regarding conventional coagulation tests, patients on DE exhibited statistically significantly prolonged TTs (P < .001) compared to patients taking acenocoumarol. On the other hand, as expected, INR values were higher in the latter group (P < .001). The aPTT values were similar across the 2 patient groups.
Comparison of Coagulation Parameters Between Patients on Dabigatran (group A, n = 20) and Patients on Acenocoumarol (group B, n = 20).a
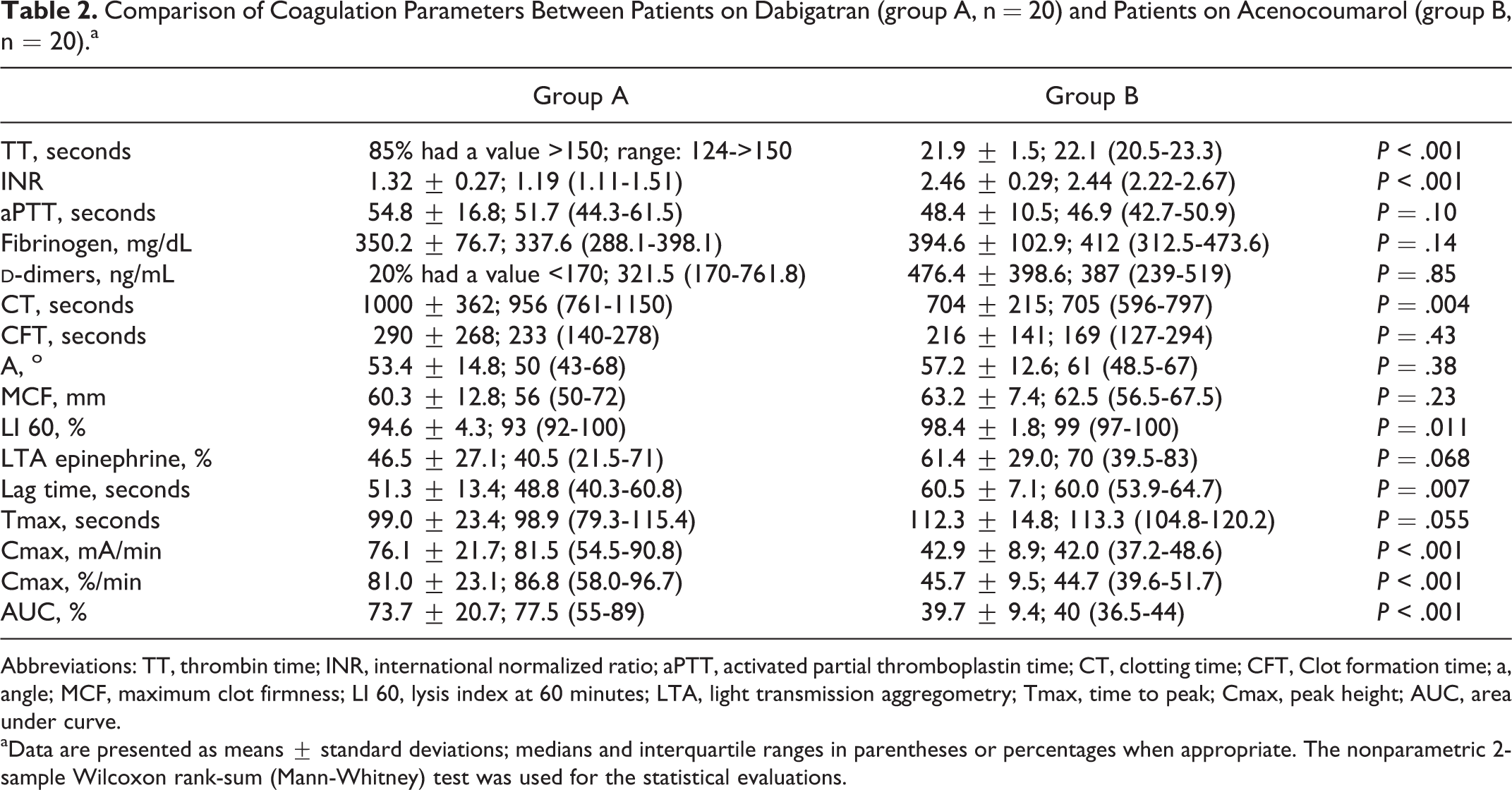
Abbreviations: TT, thrombin time; INR, international normalized ratio; aPTT, activated partial thromboplastin time; CT, clotting time; CFT, Clot formation time; a, angle; MCF, maximum clot firmness; LI 60, lysis index at 60 minutes; LTA, light transmission aggregometry; Tmax, time to peak; Cmax, peak height; AUC, area under curve.
aData are presented as means ± standard deviations; medians and interquartile ranges in parentheses or percentages when appropriate. The nonparametric 2-sample Wilcoxon rank-sum (Mann-Whitney) test was used for the statistical evaluations.
Viscoelastic measurements on the ROTEM analyzer showed a greater impact of dabigatran on procoagulant activity than acenocoumarol. These results were not statistically significant, except for CT (P = .004). Regarding fibrinolysis, the LI 60 was measured in 19 and 17 patients in group A and group B, respectively, due to the absence of an optimal reaction curve and was found significantly lower in patients receiving DE compared to those treated with acenocoumarol (P = .011). These curves of ROTEM analysis in groups A and B are shown in Figures A1 and A2, respectively.
In epinephrine-induced LTA, the dabigatran group exhibited marginally insignificant lower values in epinephrine-induced LTA (P = .068). Aggregation patterns in groups A and B are shown in Figures A3 and A4, respectively. Concerning ETP assay, the procoagulant activity significantly decreased in patients taking acenocoumarol than in those treated with dabigatran (AUC, P < .001). The correlations of HTI values with those of conventional clotting parameters, global coagulation assays, and aggregometry are presented in Table 3.
The Correlation of Dabigatran Levels as Assessed by “Hemoclot Thrombin Inhibitor” With the Following Hemostatic Parameters.
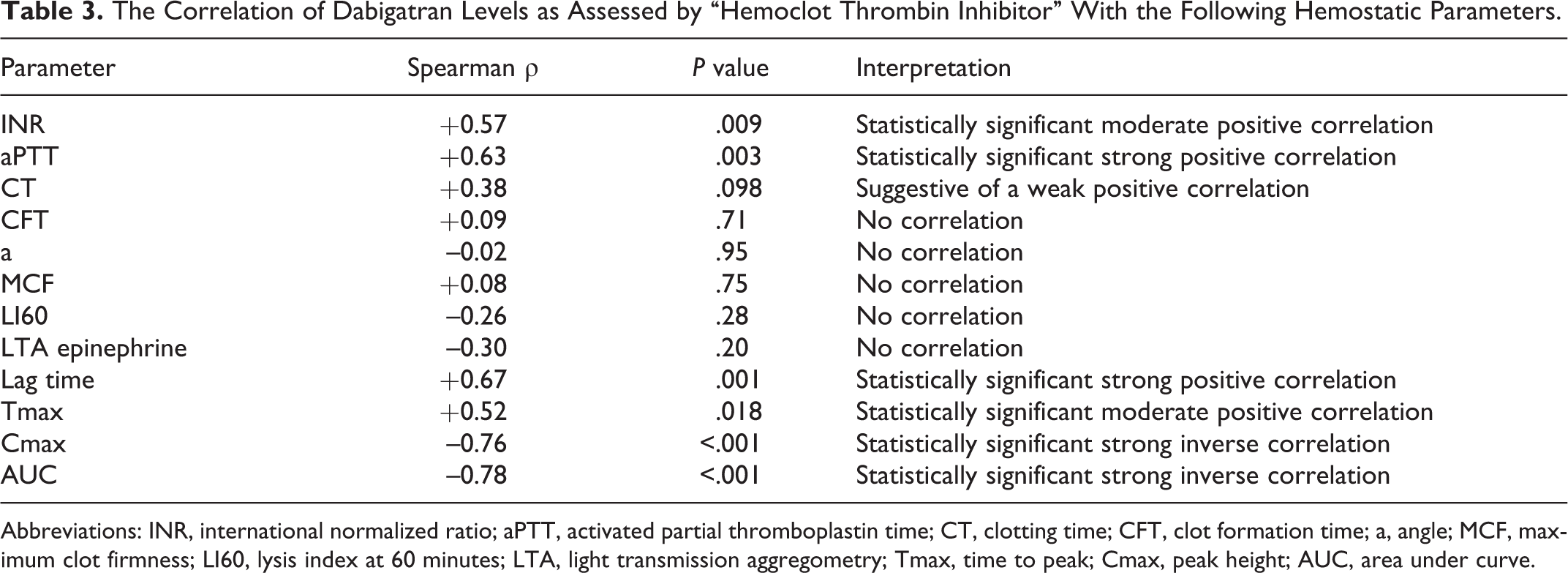
Abbreviations: INR, international normalized ratio; aPTT, activated partial thromboplastin time; CT, clotting time; CFT, clot formation time; a, angle; MCF, maximum clot firmness; LI60, lysis index at 60 minutes; LTA, light transmission aggregometry; Tmax, time to peak; Cmax, peak height; AUC, area under curve.
A strong positive correlation was shown between plasma dabigatran levels, as determined by the HTI assay, and aPTT (P = .003). Hemoclot test values also showed a moderate positive correlation with INR (P = .009). Among ROTEM variables, only CT showed a weak positive correlation with dabigatran plasma concentrations (Spearman ρ: +0.38, P = .098).
No association was found between LTA measurements and HTI values. Regarding TG, strong and moderate positive correlations were observed between dabigatran levels and lag time (P = .001), and Tmax (P = .018). Both AUC (P < .001, Figure 1) and Cmax (P < .001) were inversely correlated with HTI values. A significant inverse association was also detected between AUC and aPTT (P = .009).
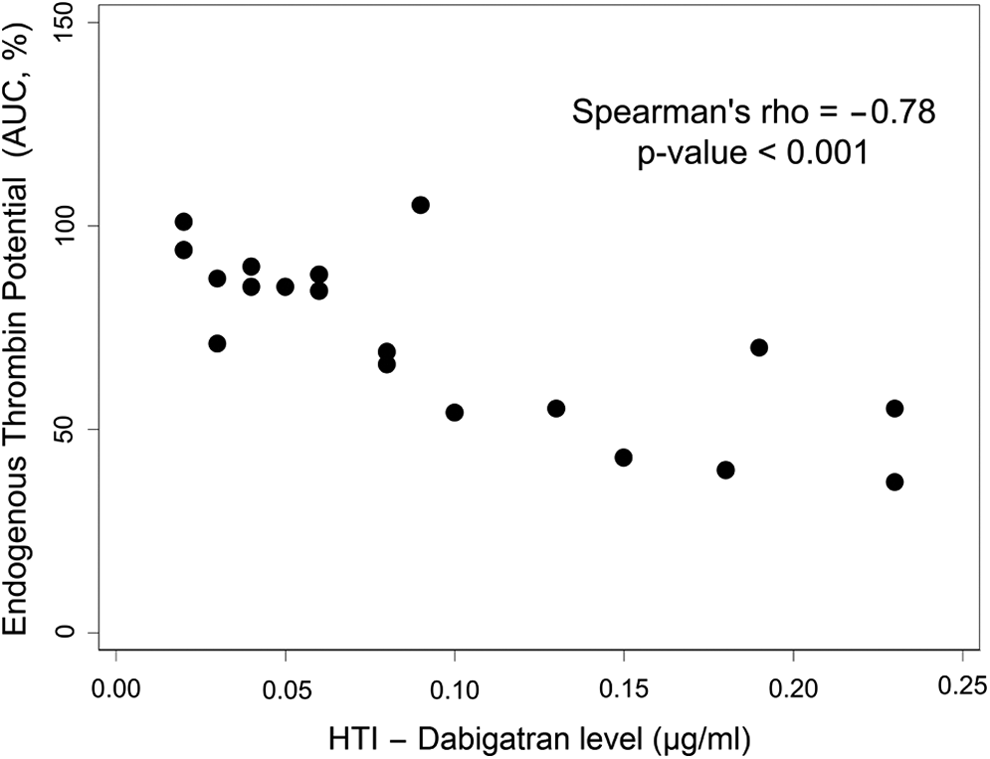
Relationship between endogenous thrombin potential (AUC, %) and plasma dabigatran level (μg/mL) as determined by the Hemoclot Thrombin Inhibitor assay. AUC indicates area under curve; HTI, Hemoclot Thrombin Inhibitor.
Discussion
In the present study, we have shown a strong relation between the values of ETP assay and the measurements of HTI test suggesting that ETP might have a role in assessing the anticoagulant effect of dabigatran in vivo in certain clinical settings.
Warfarin has long been the standard of anticoagulation therapy in atrial fibrillation. Recent evidence shows that dabigatran has similar or lower bleeding risk than warfarin. 1,24 Moreover, unlike warfarin, frequent blood sampling to monitor the degree of anticoagulation is considered unnecessary with dabigatran. Nevertheless, there are some issues of concern including the unknown value of monitoring dabigatran levels in certain clinical cases, especially in patients with changing renal function or in elderly people 25 ; the absence of an antidote; and the argument that the same dose of an oral direct inhibitor can result in significant differences in TG inhibition, making bleeding more likely in high responders and thrombosis more likely in low responders. 26
All clotting parameters in rotational ROTEM were affected more by dabigatran than by acenocoumarol, but this effect did not reach statistical significance, except for CT. Thus, a greater impact on procoagulant activity by dabigatran could be assumed. It is also noteworthy, the significant reduction of LI 60 index in patients on dabigatran compared to those on acenocoumarol, possibly indicating an increased fibrinolytic activity in the former group. Although ROTEM is not the most sensitive and proper assay to study fibrinolysis, this finding should be further investigated because a potential underlying mechanism may be involved. Thrombin activates factor XIII, which stabilizes the fibrin clot by creating cross-links that render it more resistant to plasmin-driven degradation and, in parallel, regulates TAFI, which limits plasmin activity by removing from fibrin aminoacids that are essential for plasmin binding. 9
On the contrary, based on ETP, acenocoumarol showed a significantly greater suppression on TG than on dabigatran. This finding is in accordance with that of Dale et al 27 who reported a greater reduction in peak TG and ETP in plasma from warfarin-treated patients than in dabigatran-containing plasma. Considering HTI assay as the gold standard to measure dabigatran levels in plasma, significant positive correlations were observed between HTI values and lag-time and time to peak, supporting the hypothesis suggested by Wagenvoord et al 28 that these parameters can be used to study the effect of DTIs. Moreover, the statistically significant strong inverse correlation between dabigatran plasma levels and AUC observed in our work is noteworthy, since it was absent in a recent study investigating the in vivo impact of prophylactic dabigatran on the TG assay. 15 The different time of blood collection in the 2 studies could explain this discrepancy, since Green et al worked trough levels of dabigatran, while our analysis focused on peak levels. In keeping with this, low concentrations of the direct thrombin inhibitors, melagatran and dabigatran, were recently found to enhance TG and hypercoagulability, possibly via inhibition of the protein C system. 29 Thus, the clinical usefulness of this index in assessment of anticoagulation induced by dabigatran treatment probably needs further evaluation.
The significant associations found between HTI values and conventional coagulation tests (aPTT and INR) confirmed the existing knowledge. However, the limited number of our measurements did not allow the demonstration of curvilinear correlations. The TT was too sensitive to quantify dabigatran levels, since 85% of patients had TTs >150 seconds. Its utility as a screening test to confirm the absence of dabigatran was shown again.
Our findings regarding assessment of the anticoagulant effect of dabigatran and acencoumarol are rather intriguing. Results seem to be assay dependent. Endogenous thrombin potential and ROTEM yielded opposite findings about which of the two drugs supresses more efficiently hemostatic mechanisms. But taking into consideration the significant correlations between dabigatran plasma levels and almost all parameters of ETP assay, the role of ETP in measuring anticoagulant activity of dabigatran possibly requires further research. The impact on ETP could probably account for the differences in the safety profile of dabigatran etexilate versus warfarin, that is, less intracranial bleeding, more myocardial infarctions. 30 However, controversial findings have also been reported regarding dabigatran efficacy and safety compared to warfarin. For instance, myocardial infarctions were found to be lower with dabigatran compared with warfarin in a recent prospective cohort study. 31 Since the safety and efficacy profile of dabigatran related to warfarin has not yet been clearly established, the availability of an appropriate assay to reliably estimate its anticoagulant activity might be useful, especially in certain clinical settings. Furthermore, a potential role of ETP to assess reversibility of dabigatran has been recently investigated. 32,33
The main limitations of our study included the small sample size and the absence of long-term clinical data to investigate the clinical relevance of laboratory parameters tested. However, the use of ex vivo samples, the performance of many laboratory assays, and the investigation of the impact of dabigatran on hemostatic parameters that have not been studied before are helpful in an effort to elucidate several uncertainties regarding the assessment of the anticoagulant effect of dabigatran and its relation with calculating plasma concentrations. In conclusion, our findings showed statistically significant correlations between dabigatran levels and almost all parameters of ETP assay, the role of which in measuring anticoagulant activity of dabigatran in certain clinical settings possibly requires further research.
Footnotes
Appendix A
Author’s Note
The data set is available at the ResearchGate platform, doi:10.13140/2.1.4625.5681.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JL reports personal fees from Bayer and Pfizer.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
