Abstract
Rivaroxaban, the first oral direct factor Xa inhibitor, was approved for stroke prevention in nonvalvular atrial fibrillation in 2011. Limited data are available regarding major bleeding in a clinical practice setting. The purpose of this study is to describe the patient characteristics, management, and outcomes of major bleeding events in patients receiving rivaroxaban for atrial fibrillation. This retrospective, single health system study identified patients with rivaroxaban having a major bleeding event between July 2011 and June 2014. Patients were identified through adverse event reporting or by cross-referencing rivaroxaban with International Classification of Diseases, Ninth Revision diagnosis codes for atrial fibrillation and hemorrhage, with and without transfusion. A total of 60 patients were identified. The mean age of patients was 80.3 ± 7.4 years. The most common bleed sites were gastrointestinal (63.3%) and intracranial (26.7%). Higher dose than recommended based on renal function was present in 35% of patients and concurrent antiplatelet therapy occurred in 70%. Activated prothrombin complex concentrate was utilized in 30% of patients and recombinant factor VIIa in 6.7%. A procedure or surgery was performed for bleed management in 10 patients. Anticoagulation was held at discharge in 76% of patients. A total of 6 patients died during hospital admission, 5 of whom experienced an intracranial hemorrhage. In conclusion, patients experiencing a rivaroxaban major bleeding event were elderly, often renally impaired, and receiving concurrent antiplatelet therapy. In-hospital mortality was 10%. The majority of patients (76%) had anticoagulation therapy held at discharge.
Introduction
Rivaroxaban, the first oral direct factor Xa inhibitor, was approved for the prevention of stroke and systemic embolism in patients with nonvalvular atrial fibrillation in August 2011 following completion of the Rivaroxaban Once Daily Oral Direct Factor Xa inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET-AF). 1,2 In this study, rivaroxaban was shown to be noninferior to warfarin for the prevention of stroke and systemic embolism with a similar rate of major bleeding and a significant reduction in intracranial hemorrhage. Despite the favorable clinical trial results, in practice, bleeding adverse events remain a concern for patients on long-term rivaroxaban therapy as no reversal agent is yet available. 3 In real-world practice, the use of rivaroxaban extends beyond the clinical trial populations studied and likely occurs with less structured follow-up. 4,5 Although prospective observational studies with rivaroxaban have reported bleeding rates similar to ROCKET-AF, concerns about the outcomes of those with hemorrhage have been raised. 6,7 In 2012, the Institute of Safe Medication Practices reported a fatal hemorrhage rate with rivaroxaban as twice that of warfarin, 8% versus 4%, respectively. 8 In 2014, while the penetration of rivaroxaban into the marketplace was increasing, more than 1600 hemorrhage adverse events were reported to the US Food and Drug Administration (FDA). 9 With limited data available evaluating rivaroxaban major bleeding in the practice setting, a detailed examination of such events is needed. The National Action Plan for Adverse Drug Event Prevention (ADE Action Plan) developed by the Department of Health and Human Services has also identified evaluation of anticoagulant bleeding in the “real-world” setting as a priority area for research. 10 The purpose of this study is to describe the characteristics, management, and outcomes of patients with atrial fibrillation who experience a rivaroxaban major bleeding event and to identify opportunities for improving the safe use of rivaroxaban in the real-world practice setting.
Methods
This institutional review board–approved, retrospective, single health system study identified rivaroxaban patients aged ≥18 years with a major bleeding event between July 1, 2011, and June 30, 2014. Patients were identified for inclusion through either an internal health system adverse event reporting system or by cross-referencing a list of rivaroxaban patients during the study period with International Classification of Diseases, Ninth Revision (ICD-9) diagnosis codes for atrial fibrillation, hemorrhage, and packed red blood cell (PRBC) transfusion. The list of ICD-9 hemorrhage codes was compiled through a literature review and collaboration with coding personnel (Supplemental Appendix 1). 11 –13 Patients with an ICD-9 code for intracranial hemorrhage did not require the transfusion code. All patients identified through the above processes underwent electronic health record review to determine whether a temporal relationship was present between the bleeding event and rivaroxaban therapy and whether the bleed met major bleeding criteria according to the International Society on Thrombosis and Haemostasis (ISTH). 14,15 All data were verified by 2 study investigators.
A standardized data abstraction form and rule/definition document was used to collect demographics, laboratory values, patient characteristics, bleed diagnosis, bleed management, and patient outcomes. Patient characteristics included renal and hepatic impairment, anemia, thrombocytopenia, hypertension, congestive heart failure, diabetes, stroke/transient ischemic attack, peptic ulcer disease, active alcohol abuse, malignancy, fall risk (defined as any history of previous falls or disease states potentially increasing the risk of falls), history of bleeding, anticoagulant dose, concomitant interacting medications, periprocedural management, transitions between anticoagulants, unintended anticoagulation continued while bleeding, and medication reconciliation errors. Stroke risk assessment scores (CHADS2 and CHA2DS2-VASc) and bleeding risk assessment scores (HEMORR2HAGES and HAS-BLED) were also calculated for each patient. 16 –19
The following data were collected to assess bleed diagnosis and management: bleed-related diagnostic tests, procedures, surgeries, use of blood products (fresh frozen plasma [FFP] or PRBC), hemostatic agents (activated prothrombin complex concentrate [aPCC], prothrombin complex concentrate, recombinant factor VIIa), and phytonadione. Outcome data collected included hospital and intensive care unit length of stay postbleed, change of status to hospice/palliative care, anticoagulation status at discharge, and in-hospital all-cause mortality.
Results
A total of 60 patients with rivaroxaban major bleeding events were identified. Patient information is shown in Table 1. The majority of patients presented to the emergency department with evidence of a major bleed. Patients were at high risk for both thromboembolism (median CHA2DS2-VASc score of 5) and bleeding (median HAS-BLED score of 3). Rivaroxaban 20 mg was prescribed to 58.3% (n = 35) of patients, 15 mg was prescribed to 33.3% (n = 20), and lower or unknown doses were prescribed to the remaining 8.3% (n = 5). Gastrointestinal major bleeding events occurred most frequently followed by intracranial hemorrhage (Table 2). Details of patients experiencing intracranial hemorrhage are provided in Table 3.
Patient Information.
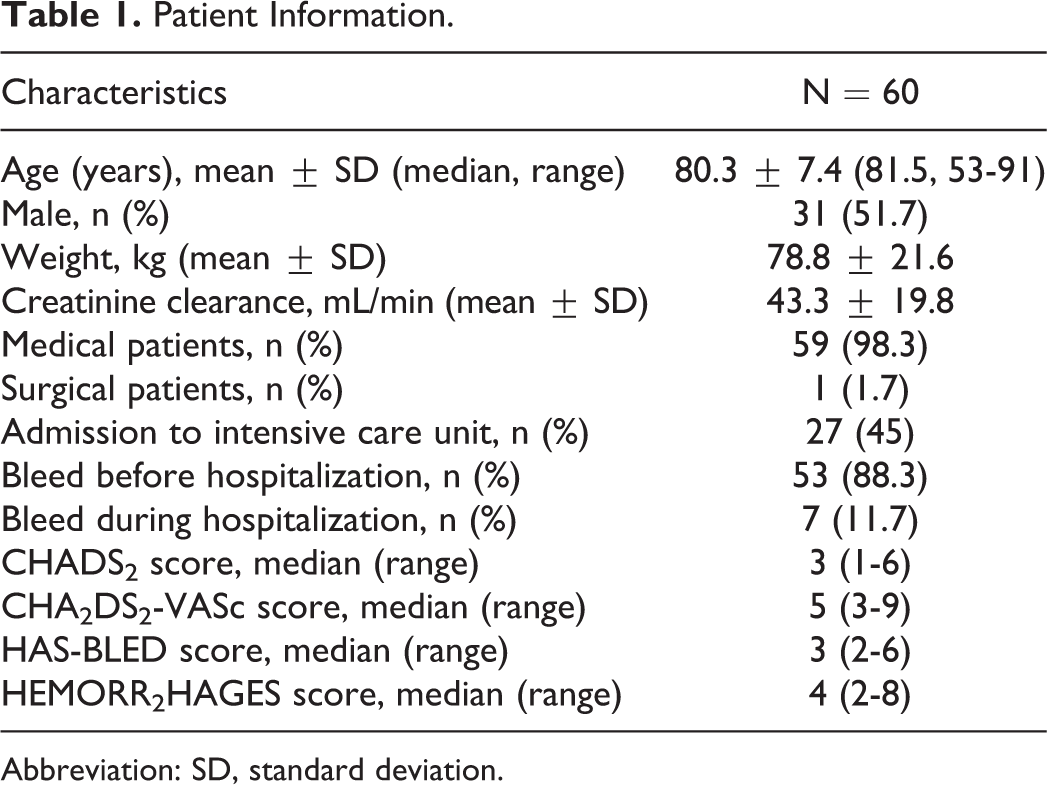
Abbreviation: SD, standard deviation.
Site of Bleed.

aOther bleed sites include chest wall extravasation, epistaxis, splenic hemorrhage, hematuria, thigh hematoma, and gluteus maximus hematoma.
Patients With Intracranial Hemorrhage.
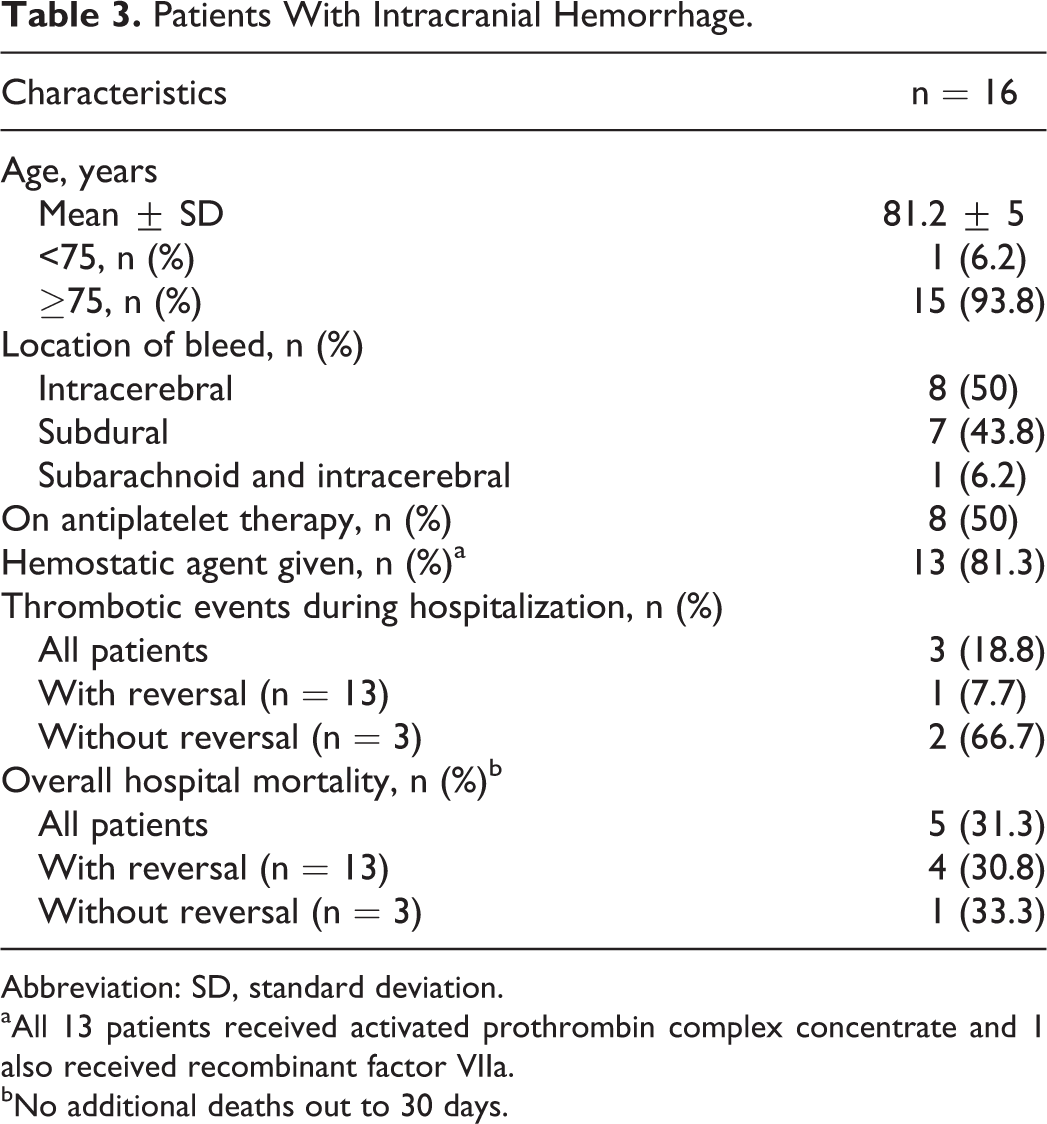
Abbreviation: SD, standard deviation.
aAll 13 patients received activated prothrombin complex concentrate and 1 also received recombinant factor VIIa.
bNo additional deaths out to 30 days.
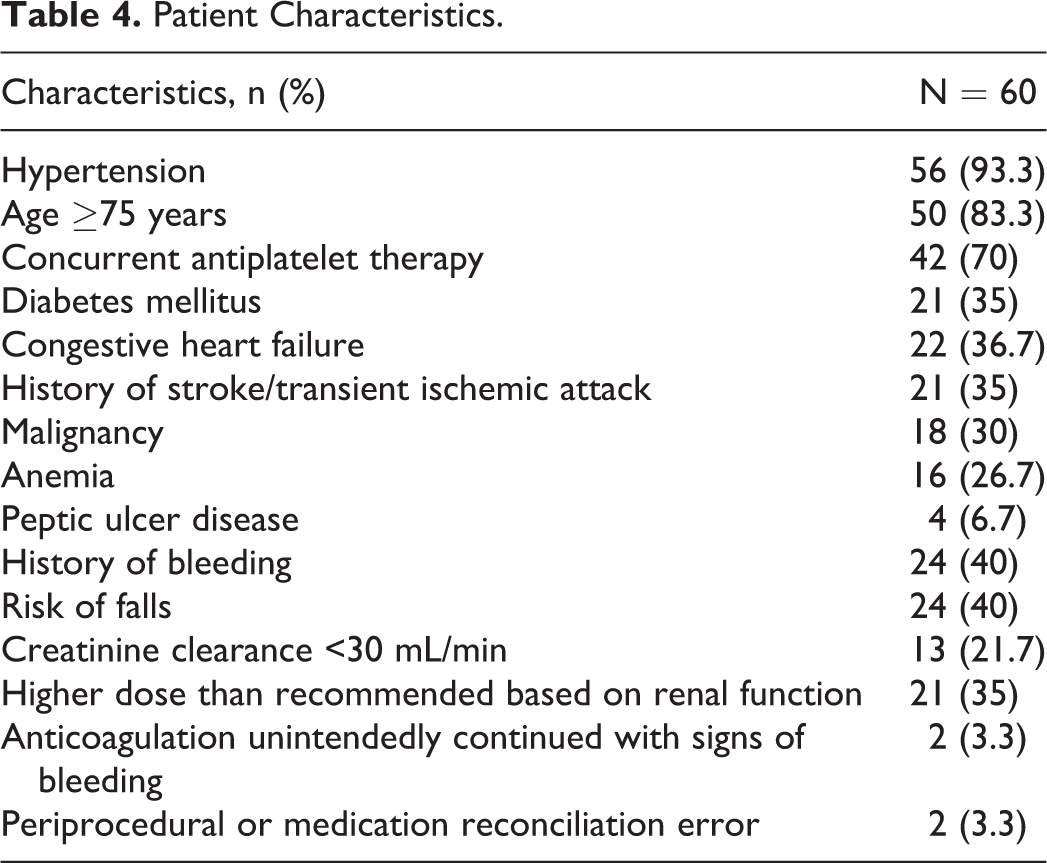
Patients with rivaroxaban major bleeding events often had a history of hypertension (56/60, 93.3%), and the majority were aged 75 years or older (50/60, 83.3%) (Table 4). Concurrent use of antiplatelet therapy was seen in 70% (42 of the 60) of patients, with 6 patients on dual antiplatelet therapy and 1 patient on triple antiplatelet therapy. A history of bleeding was present in 40% (24 of the 60) of patients, with the same number having a fall risk. A creatinine clearance (CrCl) of <30 mL/min was present in 21.7% (13 of the 60) of patients. Higher dose than recommended based on renal function was present in 35% (21 of the 60) of patients. Prior to the bleeding event, 6 patients underwent a procedure within 7 days and 1 patient had a periprocedural error (a patient failed to hold rivaroxaban as instructed prior to the procedure). One medication reconciliation error occurred (a patient admitted to the hospital was started on a higher dose than they were receiving prior to admission). Rivaroxaban was continued on admission in 2 patients with signs of bleeding (rivaroxaban was continued in 1 patient after an order to hold and administered in another patient admitted with a hemoglobin of 6.5 g/dL).
Patient Characteristics.
The approach to bleed management and patient outcomes are outlined in Table 5. Diagnostic testing was performed in 85% (51 of the 60) of patients. Tests included esophagogastroduodenoscopy, computed tomography scan, nuclear medicine red blood cell scan, colonoscopy, sigmoidoscopy, and cystoscopy. A procedure or surgery was performed for bleed management in 16.7% (10 of the 60) of patients. Procedures included ablation, argon plasma coagulation, and hemorrhoid reduction. Surgeries included decompressive craniotomy and splenectomy. Blood products were utilized in 78.3% (47 of the 60) of patients. Hemostatic reversal agents were used in more than one-third (21 of the 60, 35%), with 13 of these patients experiencing an intracranial hemorrhage. Patient status change to hospice or palliative care and in-hospital mortality were both 10% (6 of the 60). Of the 6 patients who died in hospital, 5 experienced an intracranial hemorrhage.
Bleed Management and Patient Outcomes.
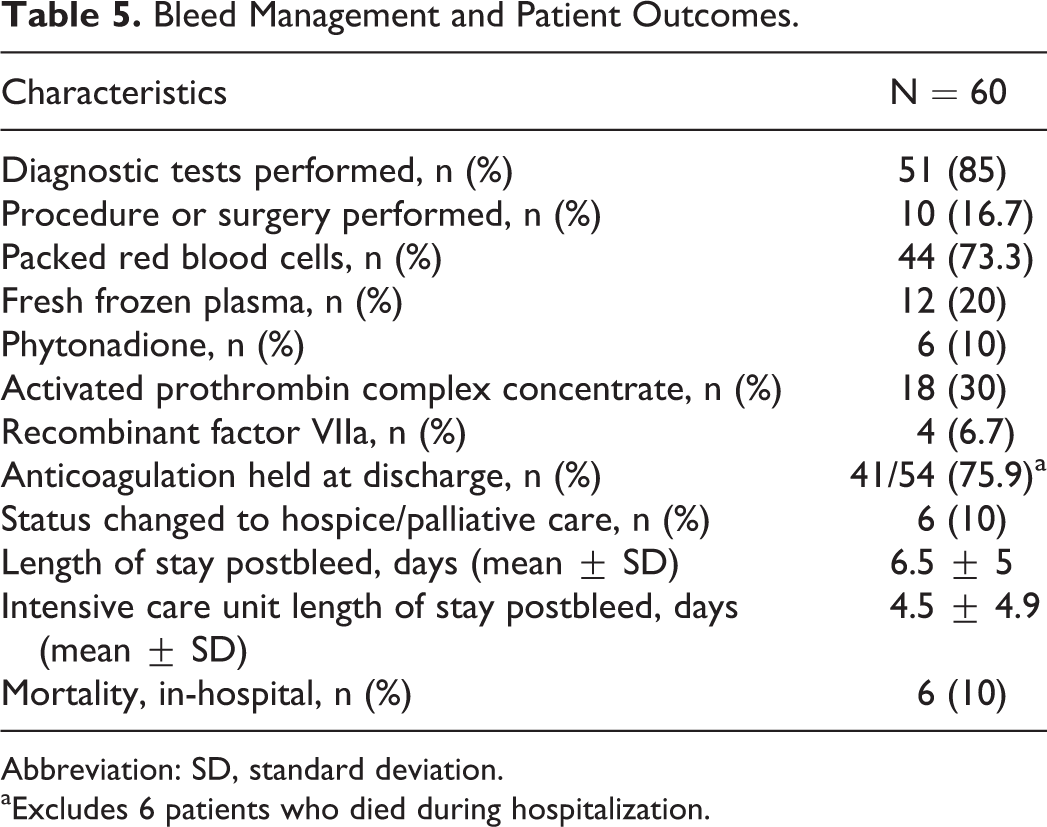
Abbreviation: SD, standard deviation.
aExcludes 6 patients who died during hospitalization.
All patients had interruption of anticoagulant therapy. Thrombotic events during the index hospitalization occurred in 11.7% (7 of the 60) of patients. Anticoagulation remained on hold in 5 of these patients at the time of the thrombotic event. Three (14.3%) of 21 patients receiving a hemostatic reversal agent experienced a venous thrombotic event during hospitalization. All 3 events occurred more than 5 days after hemostatic agent administration and while anticoagulation therapy was on hold. Anticoagulation was held at discharge in over 75% (41/54) of patients and remained on hold at 90 days in 20 of 41 patients. Five patients experienced a thrombotic event within 30 days postdischarge, 4 of whom remained off anticoagulation at the time of event.
Discussion
This study evaluated the characteristics, management, and outcomes of patients receiving rivaroxaban who experienced a major bleeding event over a 3-year period. Patients were captured using a systemic process of cross-referencing ICD-9 codes with manual chart verification of events. Although other clinical trials and evaluations of real-world practice have reported on bleeding events, this study undertook a comprehensive, individual patient-level review of bleeding events in the real-world atrial fibrillation population.
Patients experiencing a rivaroxaban major bleeding event were at high risk for both thromboembolism (median CHA2DS2-VASc score of 5) and bleeding (median HAS-BLED score of 3). The majority of patients were elderly with a median age of 81.5 years, with 83.3% being older than 75 years. In the ROCKET-AF trial, the median age of those with major bleeding was 76 years, with increasing age cited as an independent risk factor for both thromboembolism and major bleeding. 20 –22 Observational data from the practice setting have also reported an older age in those with rivaroxaban major bleeding compared to those without. 6,7,23 Renal impairment (defined as CrCl <50 mL/min) was frequent in the current study (73.3%), with 13 (21.7%) patients having a CrCl <30 mL/min. Despite an FDA-approved rivaroxaban dose for patients having atrial fibrillation with a CrCl of 15 to 30 mL/min, such patients were excluded from ROCKET-AF, and safety data in this population are lacking. The Dresden Registry on rivaroxaban major bleeding, which included patients with both atrial fibrillation and venous thromboembolism, found that a CrCl <50 mL/min occurred more often in those with major bleeding as compared to those without, 22% versus 10.4%, respectively (P = .0048). 7 This study found a higher dose than recommended based on renal function in 35% of patients, similar to findings from a prospective international registry of rivaroxaban use. 6
One of the more striking findings of the current study was that 70% of patients were on concomitant antiplatelet therapy (95% aspirin), which is twice the rate reported in ROCKET-AF. 21 In ROCKET-AF, aspirin was a risk factor for rivaroxaban major bleeding. Other observational studies of rivaroxaban major bleeding have reported a much lower frequency of concurrent aspirin therapy ranging from 2.9% to 8.5% of patients, perhaps in recognition of this risk. 7,23 Indications for concurrent aspirin and rivaroxaban therapy are limited. 24 To raise awareness of the bleeding risk with aspirin therapy, the European Heart Rhythm Association recommends the bleeding risk of aspirin in elderly patients to be considered equal to that of the vitamin K antagonists. 25 Patient selection for rivaroxaban therapy should include a comprehensive assessment of modifiable risk factors for bleeding. In addition to evaluating the need for concurrent antiplatelet therapy, other opportunities for improving the safe use of rivaroxaban were also recognized (Table 6).
Opportunities to Improve Safe Use of Rivaroxaban.
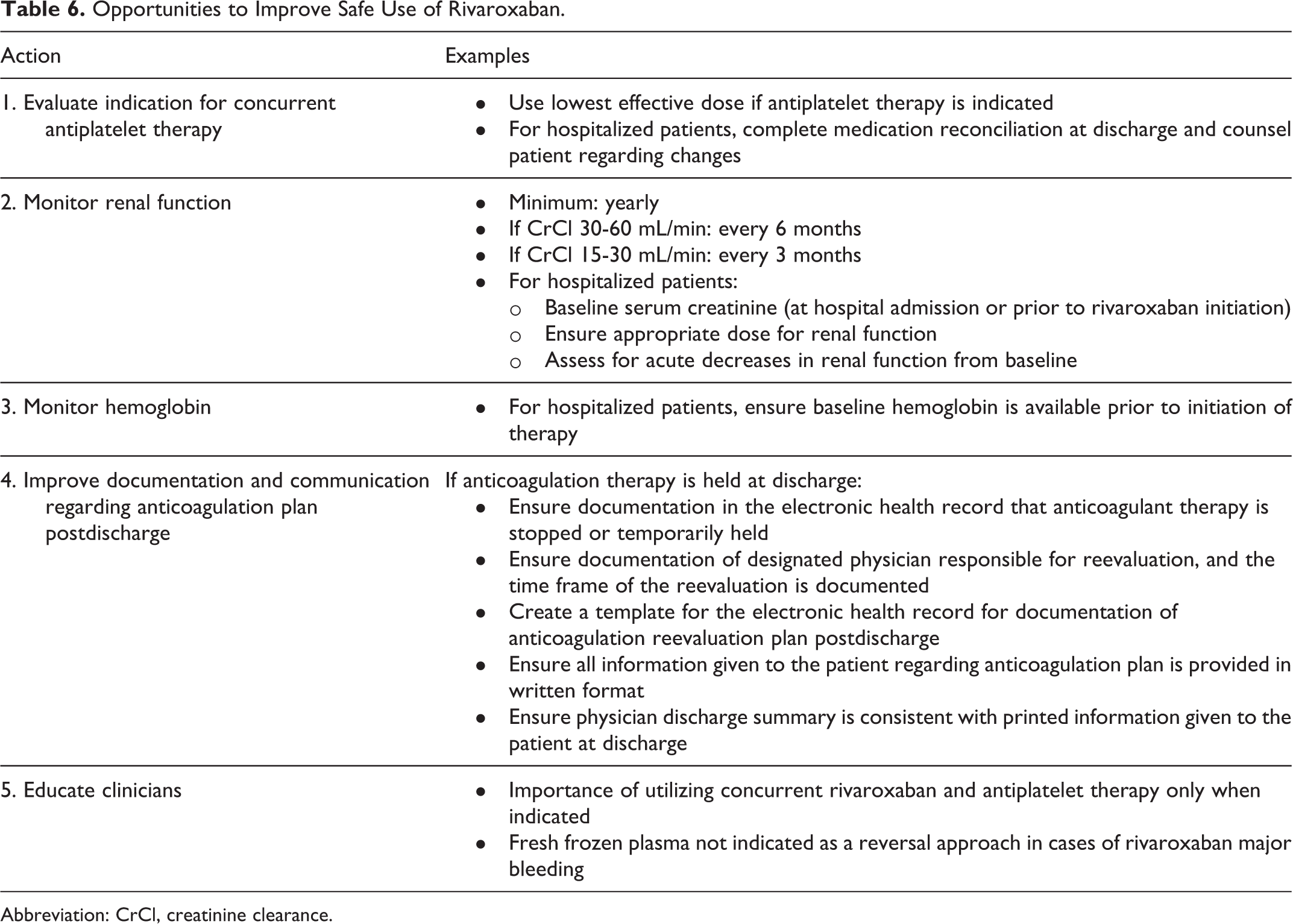
Abbreviation: CrCl, creatinine clearance.
In the current study, gastrointestinal hemorrhage was the most common site of bleeding, while intracranial hemorrhage accounted for more than a quarter of all major bleeding events (27%). This was a somewhat unexpected finding as the major advantage of direct-acting oral anticoagulants (DOACs) over warfarin is a significant reduction in intracranial hemorrhage. 26 Several inception cohort studies of warfarin major bleeding have reported a lower proportion of intracranial major bleeds with warfarin than were found in this rivaroxaban study. 25 In ROCKET-AF, 12.8% of all major bleeding events were intracranial. 2 The majority of observational studies on rivaroxaban use report a lower proportion of intracranial major bleeding events (ranging from 6.1% to 8.1%). 7,23,27 Only 1 international observational study showed a higher proportion of intracranial rivaroxaban major bleeding events (20.3%) than reported in ROCKET-AF. 6 However, the frequency of concurrent antiplatelet use in that observational study was not reported. In a subanalysis of the Randomized Evaluation of Long-Term Anticoagulation Therapy trial, aspirin therapy was found to be an independent risk factor for the development of intracranial hemorrhage. 28 The high frequency of aspirin use in the current study may have had a role in the development of rivaroxaban intracranial hemorrhage. Another potential explanation is that in ROCKET-AF, intracranial hemorrhage was not reduced compared to warfarin in patients aged >75 years, which is different from that of other DOACs. 22,29,30 In the current study, 93.8% of intracranial hemorrhage events occurred in patients aged >75 years.
The European Heart Rhythm Association has called to attention the potential to capture nonclinically relevant major bleeding events in trials, even with the use of standardized definitions such as the ISTH criteria. 25 The significance of major bleeding events captured in this study may be greater than those in ROCKET-AF. In the current study, 88% of patients presented to the emergency department with evidence of bleeding, whereas in ROCKET-AF, only 23.4% of major bleed patients required hospitalization. 2 Furthermore, 85% of patients underwent diagnostic testing and 16.7% underwent an invasive procedure or surgery for management of the bleed. Transfusion of PRBC (73.3%), administration of FFP (20%), and the use of hemostatic agents (36%) were more common than in ROCKET-AF and other observational studies. 6,7,23 Our institution has a protocol suggesting aPCC for emergent or life-threatening bleeds with rivaroxaban, and thus, the frequency of intracranial hemorrhage was thought to influence aPCC use, with 81.3% of patients with intracranial hemorrhage receiving aPCC. The anticipated future availability of direct factor Xa inhibitor reversal agents such as andexanet alfa may change the standard of care for life-threatening bleeds, removing an obstacle to the use of direct factor Xa agents and potentially increasing their use. 3
Patients with rivaroxaban major bleeding had an in-hospital all-cause mortality of 10%. Over 80% of deaths occurred in those with intracranial hemorrhage. The overall mortality rate in those with intracranial hemorrhage was 31.3%. This is similar to the 26.9% rate reported in an international registry of rivaroxaban use, where hemostatic agent use was rare. 6 Approximately half of patients in the current study spent time in the intensive care unit postbleed and 10% had a level of care change to hospice. An important finding for practitioners is that 76% (41 of 54) of patients had anticoagulant therapy held at discharge and anticoagulant therapy remained on hold at 90 days in nearly 50% (20/41) of patients. Opportunities for improvement in the documentation regarding the reassessment of anticoagulant therapy postdischarge for those with therapy held at discharge were identified (Table 6). These findings highlight the need for coordination of care during transitions between providers as noted in the ADE Action Plan. 10
The limitations of this study include retrospective design, small sample size, and data from a single health system. Patient identification involved ICD-9 codes. The potential consequence of improper coding is limited to the potential for missed cases of rivaroxaban bleeding as each major bleed was verified by comprehensive chart review. As this was a descriptive study without a control group, true risk factors for rivaroxaban major bleeding could not be determined. Patient characteristics in those with major bleeding were described.
Patients experiencing rivaroxaban major bleeding in practice were elderly, often renally impaired, and on concurrent antiplatelet therapy. More than 25% of all major bleeds were intracranial, with more than one-third of patients receiving a hemostatic agent. In-hospital all-cause mortality was 10%, with the majority of patients having anticoagulant therapy held at discharge. This focused evaluation of rivaroxaban major bleeding events allowed for the recognition of initiatives that can improve the safe use of rivaroxaban therapy. Greater attention to the plan for anticoagulant therapy is needed during the transitions of care for those with therapy held at discharge.
Footnotes
Authors’ Note
Presented at the Society of Critical Care Medicine Congress as a research snapshot presentation; February 2016; Orlando, Florida.
Acknowledgments
The authors acknowledge Lihua Qu, MD, MIS, Director of Outcomes Research, Beaumont Research Institute for assistance in data querying of research patients from the electronic medical records system and Alex Chaben, PharmD, Wayne State University and Peter Szatkowski, PharmD, Wayne State University for assistance in data extraction of research patients.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Smythe reports personal fees from Portola Pharmaceuticals.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
