Abstract
Background:
Although the rate of bleeding among patients with atrial fibrillation (AF) taking novel oral anticoagulants in randomized controlled trials is described, the rate of bleeding with “real-world” use is uncertain.
Methods:
We conducted a retrospective electronic medical record interrogation and subsequent chart review among patients within Intermountain Healthcare between October 2010 and November 2012. Patients were included if they had a diagnosis of AF and were receiving either dabigatran or rivaroxaban. Rates of major bleeding were calculated.
Results:
Among 2579 patients, 13 (0.5%) experienced major bleeding (95% confidence interval [CI] 0.23-0.77), 5 (0.19%) experienced intracranial hemorrhage (95% CI 0.02-0.36), and 2 (0.08%) experienced fatal bleeding. Of the 13 patients experiencing a major bleed, 8 (61.5%) would have been excluded from the Randomized Evaluation of Long-Term Anticoagulation Therapy (RE-LY) and Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET AF) clinical trials.
Conclusion:
We observed a rate of major bleeding similar to that reported in randomized clinical trials among patients with AF prescribed dabigatran or rivaroxaban.
Introduction
Atrial fibrillation (AF) is the most common arrhythmia with a prevalence of 1% in the general US population. 1 The risk of ischemic stroke and death is increased in persons with AF and so anticoagulation is recommended for patients with AF at increased risk of stroke. 2 –5 The novel oral anticoagulants dabigatran etexilate, and rivaroxaban are highly effective for stroke prevention in AF but as with all anticoagulants are associated with a risk of bleeding. 6,7 The reported rate of bleeding associated with anticoagulants varies substantially between populations and trials based in large part on the definition used. Using a standardized definition for major bleeding, 8 375 (6.17%) of 6076 patients taking dabigatran in the Randomized Evaluation of Long-Term Anticoagulation Therapy (RE-LY) study 6 and 395 (5.55%) of 7111 patients taking rivaroxaban in the Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET AF) study 7 experienced a major bleeding event.
The novel oral anticoagulants possess favorable characteristics including pharmacokinetics which allow administration of a fixed dose, no routine monitoring of drug effect, and lower rates of major bleeding in clinical trials. For these reasons among others it is likely that clinical practice will lead to the use of novel oral anticoagulants in a broader population not represented in controlled trials. Understanding whether a broader adoption and “real-world” use of the novel oral anticoagulants are associated with a higher rate of bleeding than that observed in clinical trials would be useful in guiding future clinician education and practice.
Recently, there has been certain notoriety, especially in the media, of the possibility of increased bleeding with the novel oral anticoagulants and particularly dabigatran. 9 Increased vigilance is common with recently approved and marketed medications due, in part, to heightened awareness of possible adverse effects and the lack of provider comfort and experience. In some instances this is medicolegally driven with directed advertisements focusing on adverse effects targeting branded drug manufacturers. The objective of this study is to report the rate of major bleeding among patients taking a novel oral anticoagulant in a real-world setting at a major health care organization.
Methods
Study Design
This retrospective electronic medical record (EMR) and chart review was conducted at Intermountain Healthcare, the largest health care organization in Utah, including data from 22 hospitals and more than 200 clinics and pharmacies. Data were extracted from the Enterprise Data Warehouse (EDW) at Intermountain Healthcare between October 19, 2010, and November 2012. The EDW is a central data repository that houses all medical record data for patient encounters at Intermountain Healthcare hospitals, clinics, and pharmacies. No research funding was provided for this study. The protocol was approved by the Institutional Review Board of Intermountain Medical Center.
Study Population
Patients taking either dabigatran or rivaroxaban with a diagnosis of AF were eligible for inclusion. Patients were defined as taking dabigatran or rivaroxaban if they (1) were administered a dose of either medication in the hospital (identified in the EMR electronic medication administration record), (2) had record of dabigatran or rivaroxaban reported in the system-wide online prescription tracking module, (3) were dispensed dabigatran or rivaroxaban from an Intermountain Healthcare pharmacy, or (4) had either dabigatran or rivaroxaban documented in the EMR identified as present under “active medications.” Atrial fibrillation was defined using International Classification of Diseases, Ninth Revision (ICD-9) code 427.3 or as documented on electrocardiogram via verbal or electronic dictation. To assure that patients included were actively receiving a novel oral anticoagulant and had not been initially provided a prescription for a novel oral anticoagulant then were switched back to warfarin, patients with an international normalized ratio (INR) of ≥1.8 in the 90 days following initiation of either dabigatran or rivaroxaban were excluded from the final analysis.
Outcomes
Our primary outcome was the rate of major bleeding defined as fatal bleeding, bleeding into a critical organ or organ space including intracranial, intraspinal, intraocular, intra-articular, peritoneal, and pericardial, or other bleeding in the setting of transfusion of ≥2 units of packed red blood cells. This included bleeding into the gastrointestinal or genitourinary tracts. To identify patients experiencing a major bleeding event, 2 techniques were used. First, the EMR was interrogated for ICD-9 codes diagnostic for major bleeding. A list of all ICD-9 codes can be found in Appendix A. Second, EMR interrogation for the administration of blood products (≥2 units of packed red blood cells) was performed. One author (GVF) performed a manual chart review to assure the occurrence of active bleeding in the setting of active novel oral anticoagulant therapy and to capture baseline demographic information. We refrained from including in our definition of major bleeding a solitary drop in hemoglobin of ≥2 mg/dL in the absence of clinically overt bleeding due to the lack of specificity (eg, hemoglobin changes can occur for reasons other than bleeding, such as hydration). All major bleeding was verified with manual electronic chart review.
Among patients identified as experiencing major bleeding, we sought for concomitant use of a medication that might interact with the anticoagulant and increase the risk of bleeding. These medications included clopidogrel, prasugrel, and ticagrelor, nonsteroidal anti-inflammatory drugs including ibuprofen, naproxen, meloxicam, indomethacin, ketorolac, sulindac, etodolac, celecoxib, and any dose of aspirin. This information was gathered from the system-wide online prescription tracking module. The percentage of patients experiencing a major bleed receiving an interacting medication was calculated. Additionally, the exclusion criteria used in defining study populations in the RE-LY and ROCKET AF trials 6,7 were cross-referenced with patients admitted with major bleeding. The percentage of patients who would have been excluded from either trial was calculated. We calculated the CHADS2 and HAS-BLED 10,11 scores for each patient who experienced a major bleed. The HAS-BLED score can be found in Table 1. Results are compared with those documented in large randomized controlled trials 6,7 when appropriate.
The HAS-BLED Score.a
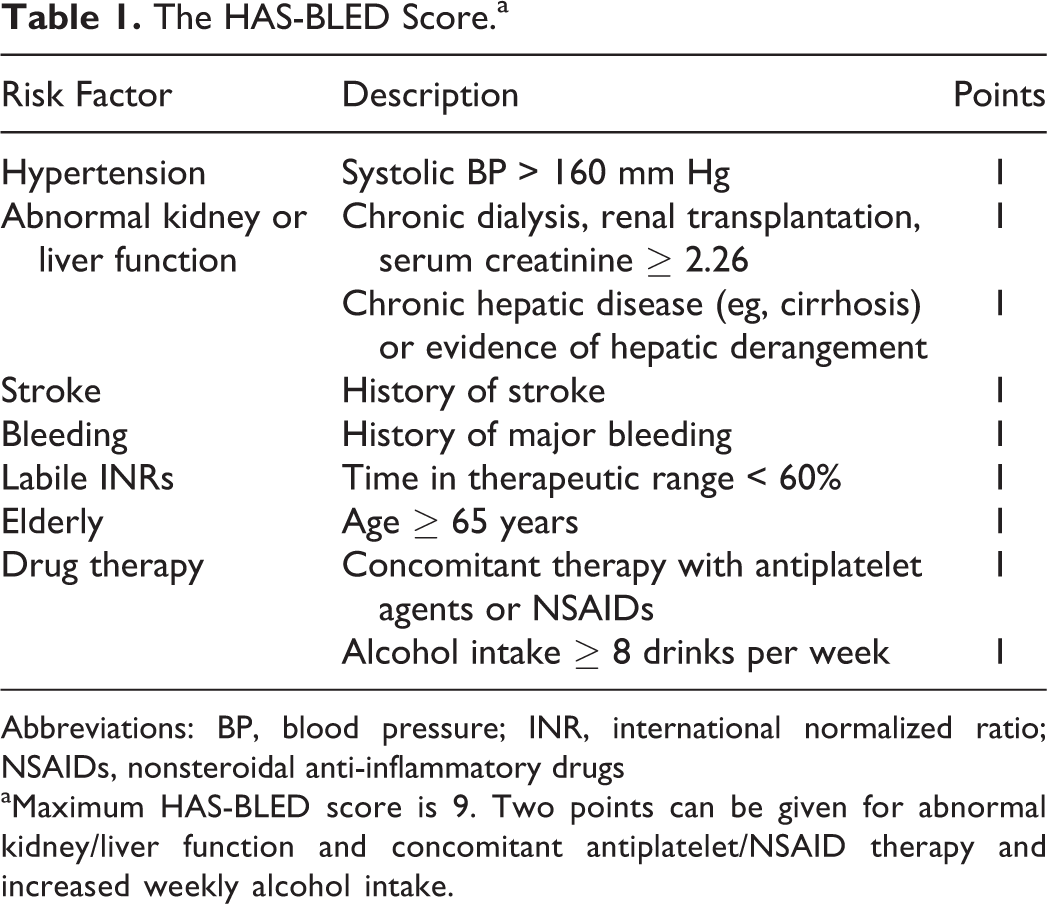
Abbreviations: BP, blood pressure; INR, international normalized ratio; NSAIDs, nonsteroidal anti-inflammatory drugs
aMaximum HAS-BLED score is 9. Two points can be given for abnormal kidney/liver function and concomitant antiplatelet/NSAID therapy and increased weekly alcohol intake.
Statistical Analysis
The primary analyses are descriptive in nature with continuous data displayed as means with standard deviations (SDs) and categorical data with percentile.
Results
Patients
There were 6910 separate encounters of patients on either dabigatran or rivaroxaban. Of these, 1951 encounters were removed because of patient duplication, leaving 4959 individual patients. Atrial fibrillation had been diagnosed in 3075 patients. When verifying whether patients were initiated and continued on the study medications, using the above-mentioned definition, 487 patients were excluded from the group not experiencing major bleeds, leaving 2566 for final analysis. Nine patients were excluded from the group experiencing a major bleed upon manual chart review (Figure 1). Reasons for exclusion included major bleeding while not taking a novel oral anticoagulant within the previous 7 days (n = 2), major bleeding after transitioning back to warfarin therapy (n = 5), or no evidence of major bleeding on manual chart review (n = 2). The final study cohort consisted of 2579 consecutively enrolled patients.
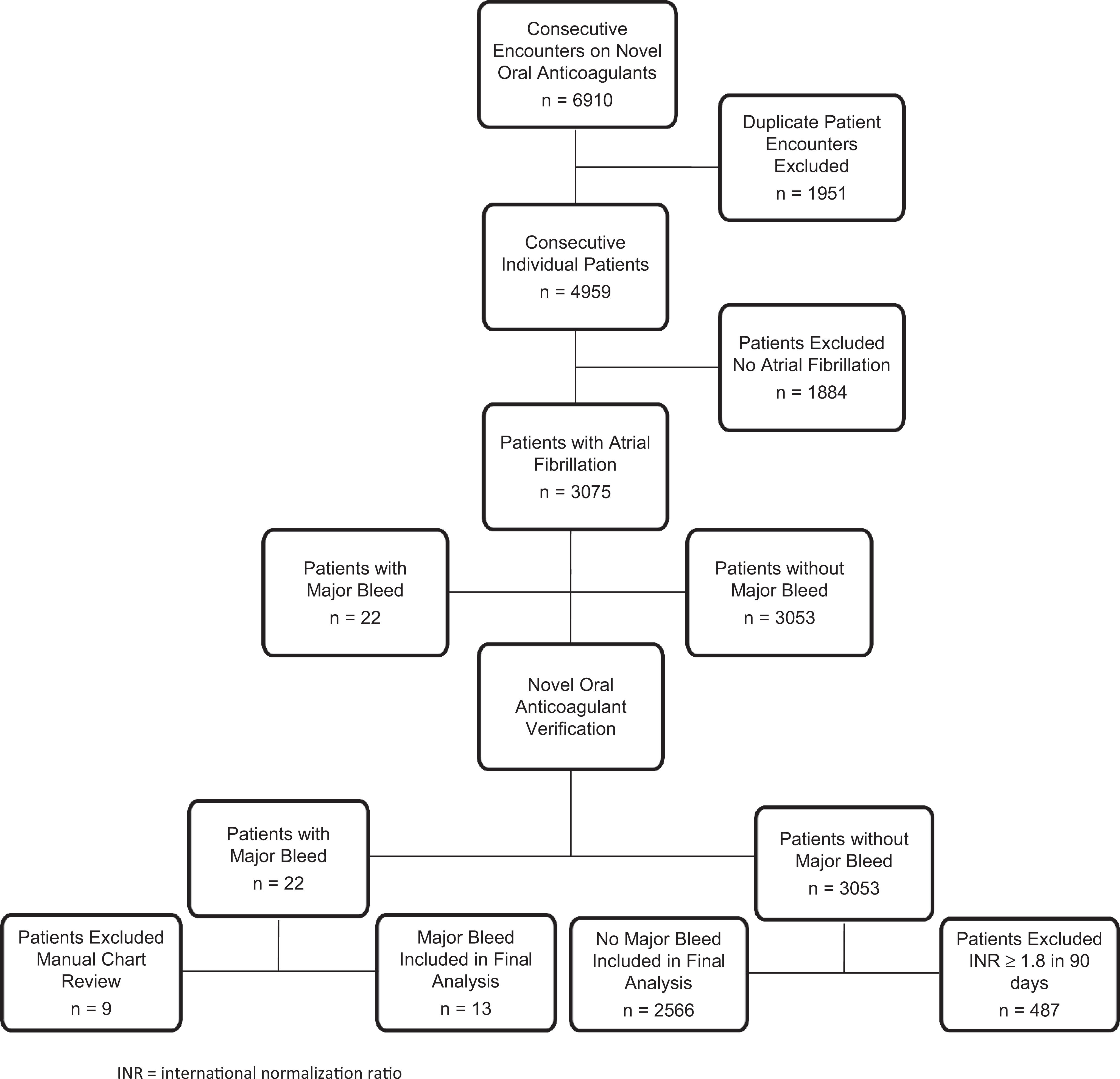
Study design: A graphic of patient enrollment and exclusion. INR indicates international normalization ratio.
Major Bleeding Outcomes
Of the 2579 patients, 13 (0.5%) experienced a major bleed (95% confidence interval [CI] 0.23-0.77) over the 2-year time frame. Fatal bleeding occurred in 2 (0.08%) patients: one taking dabigatran and one taking rivaroxaban. Ten major bleeds occurred in patients taking dabigatran (0.52%) and 3 occurred in patients taking rivaroxaban (0.43%). Intracranial bleeding occurred in 5 (0.19%) patients (95% CI 0.02-0.36). Other sites of major bleeding included gastrointestinal (3 patients), intraocular (2 patients), retroperitoneal (1 patient), pericardial (1 patient), and genitourinary (1 patient). All bleeding events were found to be medically related after manual chart review, and thus no included events were secondary to surgical procedures or deemed related to such procedures. Patient characteristics of those experiencing a major bleed are found in Table 2.
Characteristics of the 13 Patients Experiencing a Major Bleed.a
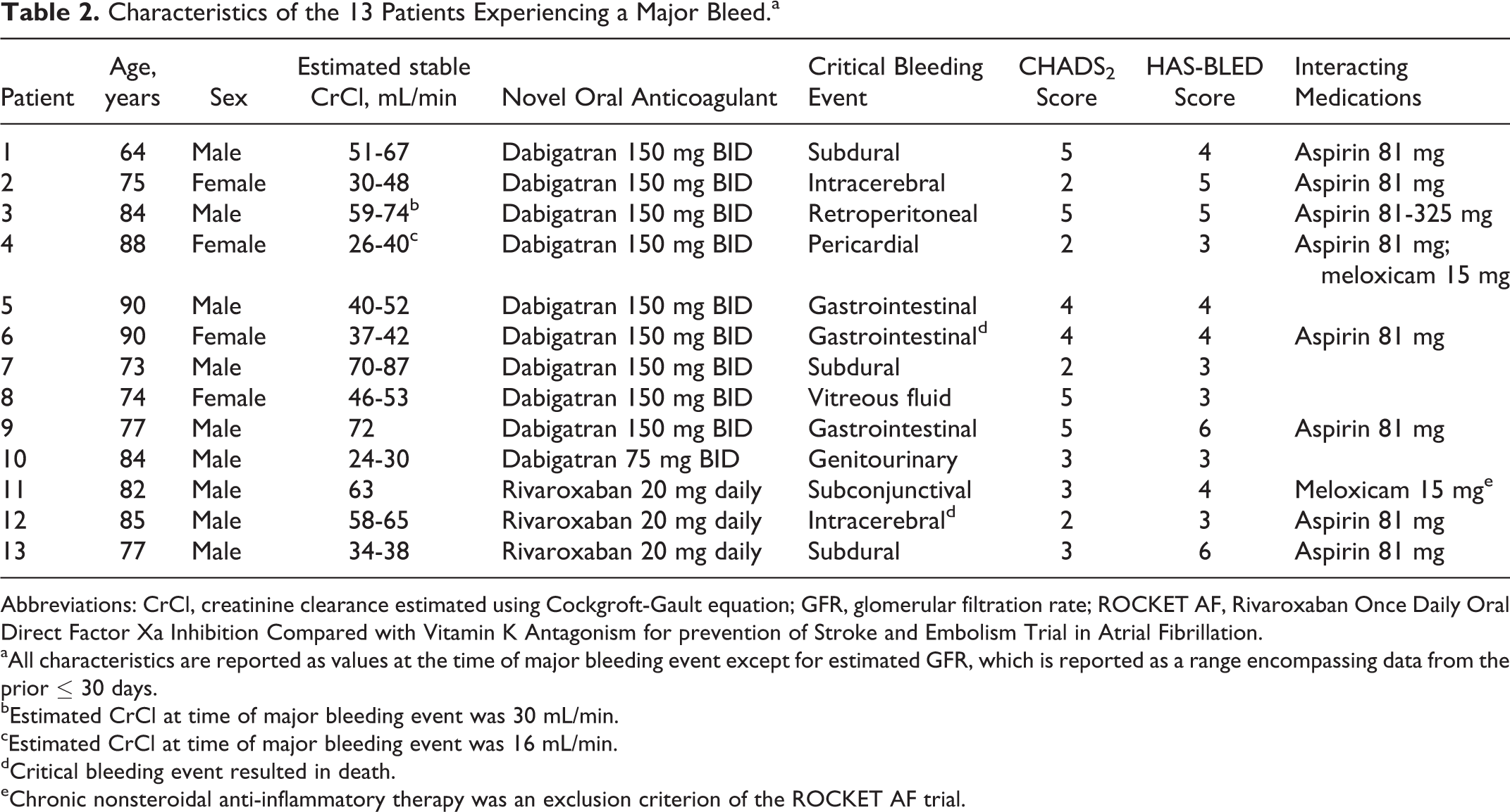
Abbreviations: CrCl, creatinine clearance estimated using Cockgroft-Gault equation; GFR, glomerular filtration rate; ROCKET AF, Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for prevention of Stroke and Embolism Trial in Atrial Fibrillation.
aAll characteristics are reported as values at the time of major bleeding event except for estimated GFR, which is reported as a range encompassing data from the prior ≤ 30 days.
bEstimated CrCl at time of major bleeding event was 30 mL/min.
cEstimated CrCl at time of major bleeding event was 16 mL/min.
dCritical bleeding event resulted in death.
eChronic nonsteroidal anti-inflammatory therapy was an exclusion criterion of the ROCKET AF trial.
Among patients who experienced a major bleed, the average age was 80.1 years (SD 7.62). The mean CHADS2 and HAS-BLED 10,11 scores were 3.46 (SD 1.3) and 4.08 (SD 1.12), respectively. A total of 9 (69.2%) patients were actively taking interacting medications. In all, 8 patients were taking concomitant aspirin at a dose of 81 mg daily and 2 patients were on chronic meloxicam therapy. One patient was taking both aspirin and meloxicam. Eight (61.5%) patients would have been excluded from the randomized control trials cited. 6,7 The reasons for exclusion are found in Table 3.
Reasons for Exclusion From RE-LY and ROCKET AF.a

Abbreviations: NSAID, nonsteroidal anti-inflammatory drug; ROCKET AF, Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for prevention of Stroke and Embolism Trial in Atrial Fibrillation; RE-LY, Randomized Evaluation of Long-Term Anticoagulation Therapy.
aDepending on whether a patient was receiving dabigatran or rivaroxaban, the RE-LY or ROCKET AF trial exclusion criteria were used, respectively.
Discussion
We report the real-world rate of major bleeding observed in patients receiving either dabigatran or rivaroxaban for the indication of AF over 2 years upon retrospective review. Our study showed a major bleeding rate of 0.5% (95% CI 0.23-0.77). This is not dissimilar to outcomes reported in prospective clinical trials. 6,7 Critical bleeding in ROCKET AF, a definition separate from major bleeding and similar to our definition of major bleeding, defined as bleeding into a critical area (intracranial, intraocular, pericardial, intraarticular, intramuscular with compartment syndrome, or retroperitoneal) occurred in 91 (1.28%) of 7111 patients in the rivaroxaban group. 7 The rate of intracranial bleeding we report is 0.19% (95% CI 0.02-0.36). The ROCKET AF 7 reported intracranial hemorrhage in 55 (0.77%) patients taking rivaroxaban and RE-LY 6 reported 36 (0.59%) intracranial bleeds in those receiving dabigatran.
Recently, a prospective nationwide cohort study in Denmark was undertaken to assess the safety and efficacy of dabigatran in treatment-naive patients with AF. The cohort included 4978 patients receiving dabigatran and used a propensity-matched cohort of 8936 patients receiving warfarin as the comparator. 12 The risk of intracranial bleeding was lower with dabigatran 150 mg twice daily versus warfarin (adjusted hazard ratio: 0.08, 95% CI: 0.01 to 0.40), and no increased risk of major hemorrhage was noted in patients taking dabigatran even when followed for more than 1 year.
Similar results were found in a retrospective study using insurance claims and administrative data from the US Food and Drug Administration Mini-Sentinel database. 9 Gastrointestinal bleeding per 100 000 days of drug therapy was lower in the dabigatran group than in the warfarin group (1.6 vs 3.5, respectively). Intracranial bleeding was also lower in patients prescribed dabigatran than those prescribed warfarin (0.8 vs. 2.4).
The definition of major bleeding in our study included fatal bleeding, bleeding into a critical organ or organ space, or other bleeding in the setting of transfusion of ≥2 units of packed red blood cells. Although this definition differs from the definition of major bleeding used in some large randomized control trials, 6,7,13 it is similar to the definition of critical bleeding used in ROCKET AF. 7 The major bleeding definition endorsed by the International Society of Thrombosis and Haemostasis reported by Schulman and Kearon 8 and used in recent major studies was proposed in order to make appropriate comparisons between studies with regard to major bleeding. For our study, the decision was made to exclude an isolated fall in hemoglobin level of 2 g/dL in the absence of an identified clinically overt bleeding event to avoid misattribution.
Patients experiencing a major bleed in our study were older (80.1 years [SD 7.62]) than the populations receiving dabigatran and rivaroxaban in RE-LY (71.5 years [SD 8.8]) 6 and ROCKET AF (73 years [interquartile range 5]). 7 The mean CHADS2 score of 3.46 (SD 1.3) in patients experiencing major bleed in our study is comparable to the mean CHADS2 score of 3.48 (SD 0.94) for patients receiving rivaroxaban in ROCKET AF 7 but greater than the mean CHADS2 score of 2.2 (SD 1.2) reported for patients taking dabigatran in RE-LY. 6 The annual risk of stroke associated with a CHADS2 score of 3 to 4 is estimated to be 5.9% to 8.5%, without antithrombotic therapy. 14 The mean HAS-BLED score for patients experiencing a major bleed in our study was 4.08 (SD 1.12). A HAS-BLED score of 4 in 1 study had an annual major bleeding risk of 8.9%. 10 Another earlier study documented an annual major bleeding rate of 8.7% in patients with a HAS-BLED score of 4. It should be noted that these studies were conducted in patients receiving warfarin. 11 Scant evidence exists regarding the risk of bleeding among patients taking a novel oral anticoagulant and correlation with a formalized bleeding risk score. 15,16 Acknowledging these limitations, those patients in our study who experienced a major bleed would be characterized as being at high risk of both bleeding and thrombosis. Among patients with an indication for anticoagulation to prevent stroke yet at high risk of bleeding, guidelines presently recommend anticoagulation with close monitoring and a heightened attentiveness to management. 2 The use of an anticoagulant that is equally efficacious yet associated with a decreased risk of major bleeding may represent a favorable choice among these patients.
To assure that patients we identified as taking a novel oral anticoagulant in fact were actively receiving novel oral anticoagulant therapy, and not simply provided a prescription or a few doses but did not continue the drug in a clinically meaningful fashion, we sought INR values within 90 days of novel oral anticoagulant therapy initiation. If in the follow-up period an INR of ≥1.8 was observed, the patient was excluded from further analysis. We felt that an INR elevated to this degree would be unlikely among patients taking a novel oral anticoagulant should an INR be obtained and more likely representative of return to warfarin therapy. In this fashion, we placed a high value on including only patients who were taking a novel oral anticoagulant regularly and were not simply “bridged” with a novel oral anticoagulant to outpatient therapy with warfarin—a scenario that while not studied in clinical trials is sometimes employed in real-world practice. In addition, written or electronic prescriptions are often given to patients for novel oral anticoagulants prior to medication initiation. If a prescription was documented in the online prescription-tracking system, however never filled (perhaps due to cost or for other reasons), these patients were excluded. Also, if a patient was given samples of a novel oral anticoagulant and these samples had been documented in the EMR but the patient subsequently transitioned back to warfarin as defined earlier by an INR ≥ 1.8, these patients were excluded. Given the target INR range of 2 to 3 for the indication of AF, we considered an INR of ≥1.8 high enough to exclude patients who had transitioned from novel oral anticoagulant therapy to warfarin.
Strengths and Limitations
A strength of our study is the manual chart review for validation of all major bleeding events. Additional strengths include our use of a broadly adopted definition for major bleeding, and excluding patients with an INR ≥ 1.8 to prevent the misattribution of warfarin-induced major bleeding events to the dabigatran or rivaroxaban groups. Our study limitations include the following. First, the retrospective design is associated with inherent limitations including inability to assess drug adherence and continuation of novel oral anticoagulant therapy. We attempted to mitigate this limitation by including only those patients for whom active novel oral anticoagulant was verified and for whom electronic chart review did not demonstrate a clinical picture consistent with reinitiation of warfarin therapy (eg, INR ≥ 1.8). We acknowledge that patients could have been lost to hospitals outside the Intermountain Healthcare network. The possibility exists that patients who experienced a major bleed were emergently taken to out-of-network hospitals and thus these events would not have been captured in our study. However, SelectHealth, the primary insurance provider for Intermountain Healthcare facilities, insures greater than 20% of Utah’s population and refers patients primarily to Intermountain Healthcare hospitals and clinics. Additionally, Intermountain Healthcare provides care for approximately one-half of the Utah population. It is possible that fatal bleeding occurred among patients who received care outside our system or who never sought medical attention (eg, major bleed leading to death at the patient’s home); however, our observed rate of fatal bleeding was similar to that observed in clinical trials (0.08% compared with 0.2% in ROCKET AF) and suggestive that we did not miss events. Our data set does not allow for reporting rates of major hemorrhage among patients managed on warfarin anticoagulation which would be additively informative. As the aim of this study was to report the incidence of major bleeding and the data of patients experiencing these events, baseline demographic and outcome data of the overall population such as CHADS2 and HAS-BLED scores were not calculated. The exclusion criteria used in defining study populations in RE-LY and ROCKET AF were not cross-referenced with patients who did not experience a major bleed, and it is unknown what percentage of these patients were taking interacting medications yet never presented with a major bleed.
Recently, 4 consensus guidelines have been updated to include recommendations for novel oral anticoagulants in patients with AF requiring anticoagulation. 17 –20 Since the approval of dabigatran in the United States in 2010, the American College of Cardiology Foundation (ACCF), American Heart Association (AHA), and Heart Rhythm Society (HRS) joint task force recently recommended it as an alternative to warfarin for stroke prevention in patients with nonvalvular AF. 17 The most up-to-date guideline released in 2014, supported by the AHA/ACC/HRS, recommends dabigatran, rivaroxaban, apixaban, or warfarin as first-line anticoagulants for AF. In patients unable to maintain a therapeutic INR with warfarin, the novel oral anticoagulants are recommended rather than warfarin. 21 The American College of Chest Physicians and the Canadian Cardiovascular Society (CCS) recommend dabigatran in preference to warfarin except in certain populations. 2,18 The European Society of Cardiology (ESC) guideline focused update published in 2012 also recommends dabigatran in preference to warfarin. 19 Moreover, both the ESC and CCS have published guideline updates recommending rivaroxaban in preference to warfarin for the prevention of stroke in patients with AF. 19,20 With this in mind, and the fact that utilization of novel oral anticoagulants is on the rise and will continue to be studied and recommended in expanding roles for anticoagulation, given the limitations of warfarin therapy determining the real-world risk to benefit ratio of using these medications is critical.
Conclusion
In conclusion, this retrospective study describing rates of major and intracranial bleeding in patients receiving dabigatran or rivaroxaban in the setting of AF found similar outcomes to those of major randomized controlled trials. The rate of major and intracranial bleeding was actually lower in our population. The use of novel oral anticoagulants in a real-world setting seems to confer no additional risk of major bleeding, despite anecdotal reports of this occurring. Additional large-scale and head-to-head comparative effectiveness studies are needed to compare the relative bleeding risk of dabigatran, rivaroxaban, apixaban, and edoxaban with warfarin to determine the true incidence of bleeding with the novel oral anticoagulants in real-world populations.
Footnotes
Appendix A
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
