Abstract
The introduction of oral direct anti-Xa anticoagulants apixaban and rivaroxaban has significantly impacted the treatment and prevention of thromboembolic disease. Clinical scenarios exist in which a quantitative assessment for degree of anticoagulation due to these agents would aid management. The purpose of this work was to evaluate the chromogenic antifactor Xa assay calibrated with heparin standards at our institution for assessment of intensity of anticoagulation with rivaroxaban or apixaban in addition to its current use for unfractionated heparin or low-molecular-weight heparin. We also aimed to propose expected steady state peak and trough antifactor Xa activities for these agents based upon dosing regimens approved for nonvalvular atrial fibrillation. Antifactor Xa activity correlated very strongly with apixaban and rivaroxaban concentration in both spiked samples and treated patient plasma samples (r 2 = .99, P < .001). This correlation was observed over a broad range (20-500 ng/mL) of drug concentrations, as sample dilution with pooled normal plasma significantly extended the range of quantitative assessment. Based on drug concentrations previously published in pharmacokinetic studies, the expected steady state peak and trough antifactor Xa activity ranges for apixaban are 1.80 to 2.20 IU/mL and 0.70 to 1.10 IU/mL, respectively. For rivaroxaban, these ranges are 3.80 to 6.20 IU/mL and 0.60 to 1.00 IU/mL, respectively. In conclusion, our findings demonstrate that heparin-calibrated antifactor Xa activity correlates strongly with apixaban and rivaroxaban concentration. The dilution of samples allowed for this correlation to be extended over the majority of on-therapy drug concentrations.
Introduction
The introduction of oral direct anti-Xa anticoagulants has significantly impacted the treatment and prevention of thromboembolic disease. Apixaban and rivaroxaban are frequently utilized direct oral factor Xa inhibitors with relatively predictable pharmacodynamic and pharmacokinetic properties. Due to the therapeutic consistency of these agents, monitoring for intensity of anticoagulation is not routinely performed. However, there are clinical scenarios in which a quantitative assessment of degree of anticoagulation due to oral Xa inhibitors would aid clinical management. Examples include the preoperative state, during an episode of acute bleeding or kidney injury, recurrent thrombosis, and medication overdose.
Currently, practitioners in the United States wanting to utilize readily available coagulation tests to assess anticoagulation with direct oral factor Xa inhibitors face significant limitations. While the prothrombin time (PT) varies in a linear manner over a wide range of oral Xa inhibitor concentrations, there are differences between PT reagents, which vary with the factor Xa inhibitor being tested. 1 –4 For example, while a normal PT with one reagent may indicate a lack of significant anticoagulation due to residual rivaroxaban, a normal value with a less sensitive PT reagent may not. 5 In regards to apixaban, the PT is weakly sensitive in general and depending upon the reagent used may not exceed the reference range for the test at peak drug concentrations. 1
Antifactor Xa assays have been extensively utilized to monitor intensity of anticoagulation for patients receiving unfractionated heparin (UFH) and low-molecular-weight heparin (LMWH) therapies. Chromogenic antifactor Xa activity has been shown to correlate linearly with serum drug concentrations over a wide range for direct Xa inhibitors, and a measurement of antifactor Xa activity is theoretically more specific for the anticoagulant effect of these agents. 6 At this time, there are no Food and Drug Administration (FDA)-approved rivaroxaban- or apixaban-specific antifactor Xa calibration materials available for clinical use in the United States. However, the drug levels of rivaroxaban and apixaban are attainable for research use purposes using FDA-approved chromogenic antifactor Xa kits and research use only calibration material. Commercial antifactor Xa assays with UFH or LMWH calibrators have also demonstrated a linear relationship with direct Xa inhibitor drug concentrations. 4,7 These commercial assays are routinely available at most institutions, can be completed rapidly, and may offer a quantitative assessment of anticoagulation. A validated antifactor Xa assay correlating antifactor Xa activity with drug concentration using a readily available assay with UFH or LMWH calibrators may provide information in scenarios when an assessment of anticoagulation intensity is needed for patients on rivaroxaban or apixaban therapy.
The purpose of this work is to evaluate the antifactor Xa assay calibrated with hybrid standards (UFH and LMWH) at our institution for assessment of intensity of anticoagulation with rivaroxaban or apixaban. We also sought to propose peak and trough antifactor Xa activity levels in patients receiving apixaban and rivaroxaban based on dosing regimens approved for nonvalvular atrial fibrillation.
Methods
This retrospective study was approved by the Colorado Multiple Institutional Review Board. Plasma samples from apixaban- or rivaroxaban-treated patients were collected from August 1, 2014, through April 10, 2015. These patients were receiving a variety of dosing regimens. Any given patient could have as many as 3 samples included for analysis. In order for a sample to be included in this study, the patient must have been between 18 and 89 years of age and had at least 1 coagulation test (PT or activated partial thromboplastin time) ordered by the primary provider during the patient’s hospital admission for routine clinical care. These samples were drawn at various times in relation to apixaban or rivaroxaban administration; thus, peak and trough drug concentrations were not directly measured. Instead, these values were selected from previously published pharmacokinetic data and applied to the linear regression model created within this study. 8,9 The samples were collected in 3.2% sodium citrate tubes and stored in the University of Colorado Hospital clinical laboratory at −70 C until the time of analysis. Patients were excluded if they received additional anticoagulants with the ability to affect either PT or antifactor Xa activity.
The University of Colorado Hospital clinical laboratory performed the chromogenic antifactor Xa and PT testing. All of the chromogenic antifactor Xa tests were carried out using the STA liquid heparin kit (Diagnostica Stago, Parsippany, New Jersey) with hybrid antifactor Xa calibration methods and are reported in IU/mL. The PT tests were performed using the Neoplastin Plus PT reagent (Diagnostica Stago) with a normal range of 11.7 to 14.1 seconds. Serial manual dilutions of patient specimens whose antifactor Xa activity were greater than 1.8 IU/mL were performed by the University of Colorado Hospital clinical laboratory using CRYOcheck Pooled Normal Plasma (Precision Biologic, Dartmouth, Nova Scotia, Canada) as needed to attain antifactor Xa readings within the reportable range (0.0-1.8 IU/mL) of the STA-R coagulation analyzer (Diagnostica Stago). The antifactor Xa activity results obtained using the UFH/LMWH calibrator were compared to plasma rivaroxaban and apixaban concentrations obtained either by spiking normal plasma with known concentrations of the drugs or by utilizing rivaroxaban- or apixaban-specific (research use only) calibration materials (Diagnostica Stago).
Linear regression analysis was used to assess the degree of correlation between PT and antifactor Xa activity with oral direct factor Xa inhibitor drug concentration. A correlation coefficient of .80 or greater was considered indicative of a strong correlation for each test assessed. The statistical tests were performed using correlation scatter plot analysis (Microsoft Excel version 2010, Redmond, Washington). The proposed expected steady state peak and trough antifactor Xa activities were calculated using previously published pharmacokinetic data and the regression model obtained from the patient samples collected within this work. 8,9
Results
Patient Characteristics
The demographic and clinical information of the patients with included samples are described in Table 1. In total, 12 patients with 14 samples and 8 patients with 10 samples were analyzed from the rivaroxaban and apixaban groups, respectively. Dosing regimens varied among both the apixaban and the rivaroxaban groups. None of the included samples were collected from patients receiving the agents for postoperative thromboprophylaxis.
Patient Characteristics.a
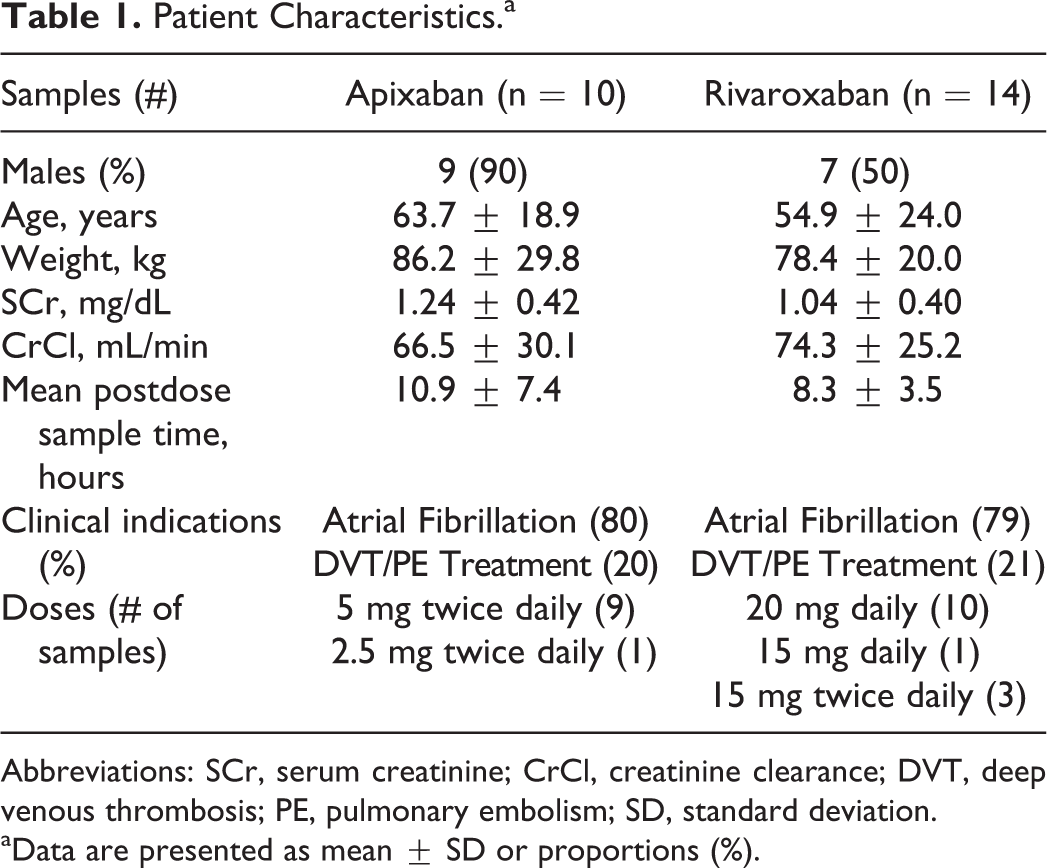
Abbreviations: SCr, serum creatinine; CrCl, creatinine clearance; DVT, deep venous thrombosis; PE, pulmonary embolism; SD, standard deviation.
a Data are presented as mean ± SD or proportions (%).
Spiked Samples of Known Drug Concentration
The Hybrid STA liquid heparin antifactor Xa result correlated strongly with both apixaban and rivaroxaban concentration in spiked samples of known drug concentration (Figures 1 and 2). The correlation coefficient obtained for each agent was above the predefined threshold for strong correlation with an r 2 = .99 (P < .001).
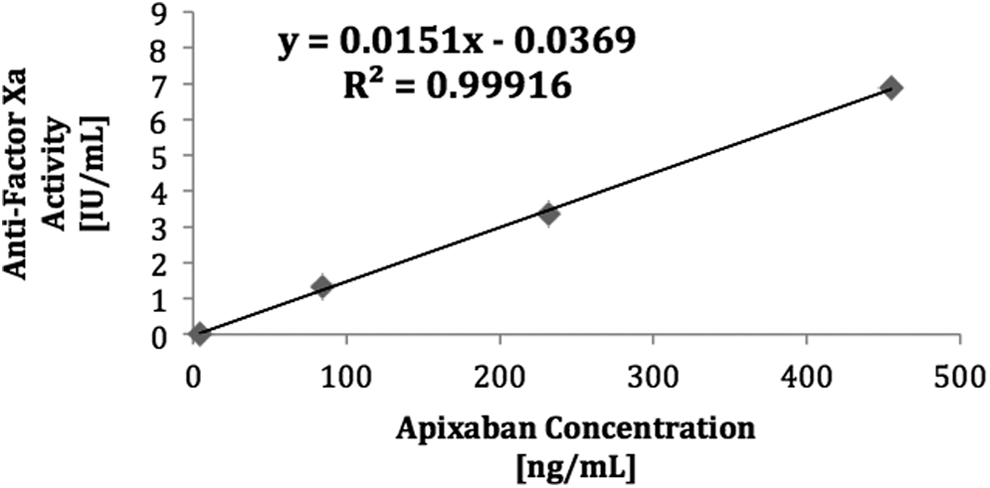
Correlation between STA liquid heparin hybrid antifactor Xa activity and apixaban concentration in spiked samples.
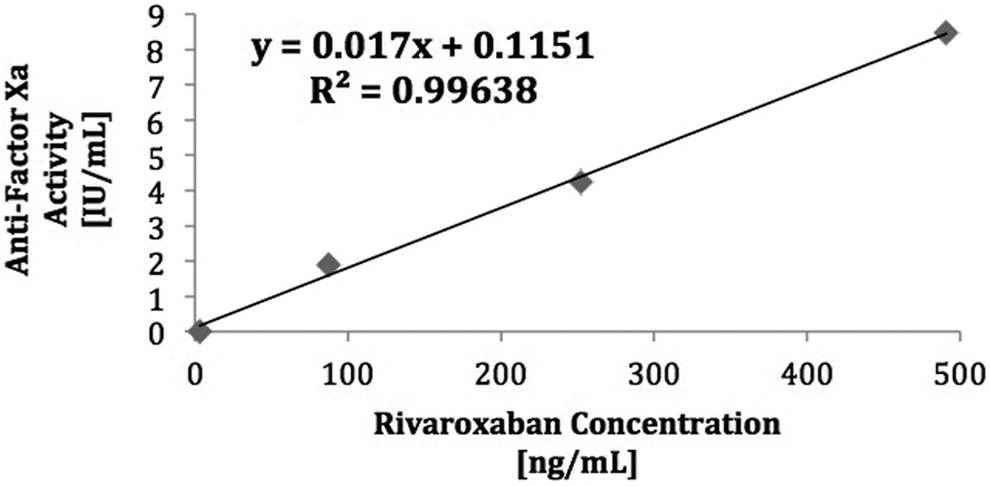
Correlation between STA liquid heparin hybrid antifactor Xa activity and rivaroxaban concentration in spiked samples.
Plasma Samples From Rivaroxaban- or Apixaban-Treated Patients
Prothrombin times performed with Neoplastin Plus prothrombin reagent correlated with both apixaban (r 2 = .77; Figure 3) and rivaroxaban concentration (r 2 = .91; Figure 4) as determined by the use of rivaroxaban and apixaban calibrators for the anti-Xa assay. The correlation observed between PT and apixaban concentration was below the predefined threshold indicative of a strong correlation.
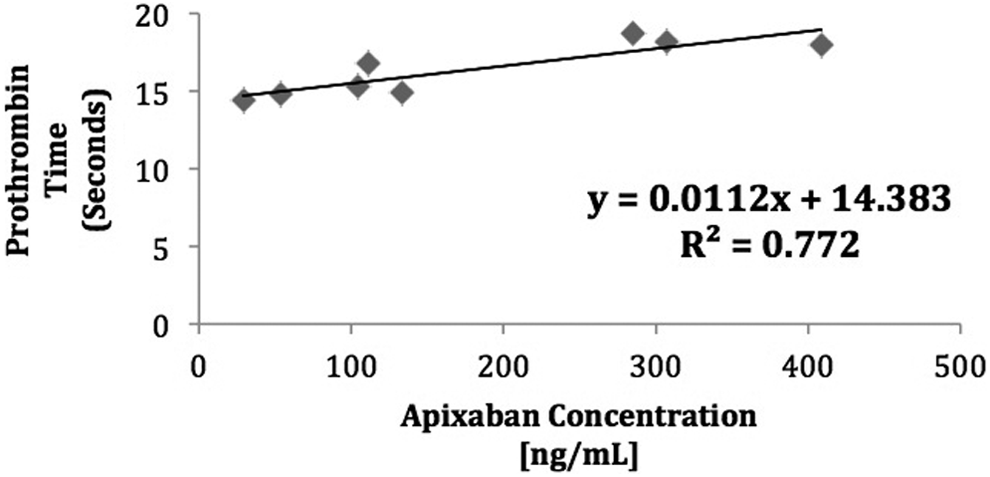
Correlation between prothrombin time and apixaban concentration in treated patient samples.
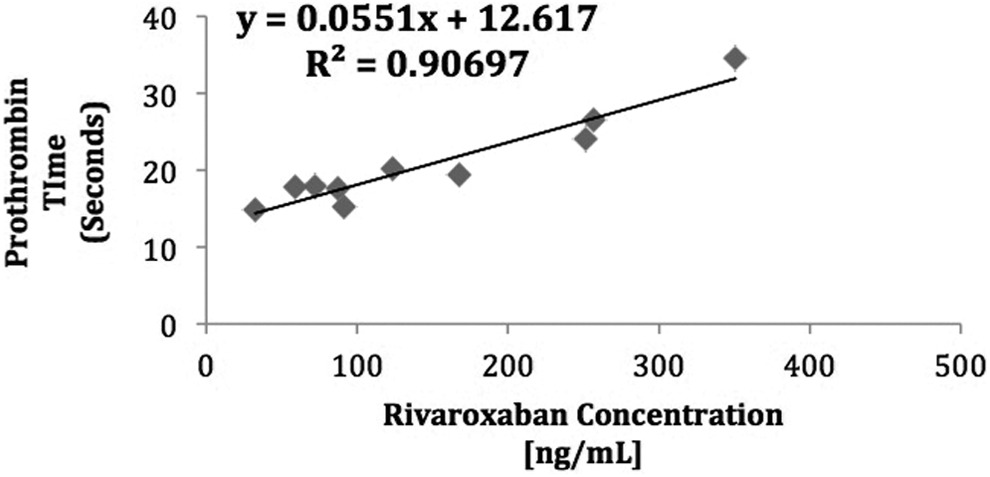
Correlation between prothrombin time and rivaroxaban concentration in treated patient samples.
The hybrid STA liquid heparin antifactor Xa levels correlated strongly with both apixaban and rivaroxaban concentration over a broad range (20-500 ng/mL) in patients treated with these agents (Figures 5 and 6). The correlation coefficient obtained for each agent was above the predefined threshold for strong correlation with an r 2 = .99 (P < .001).
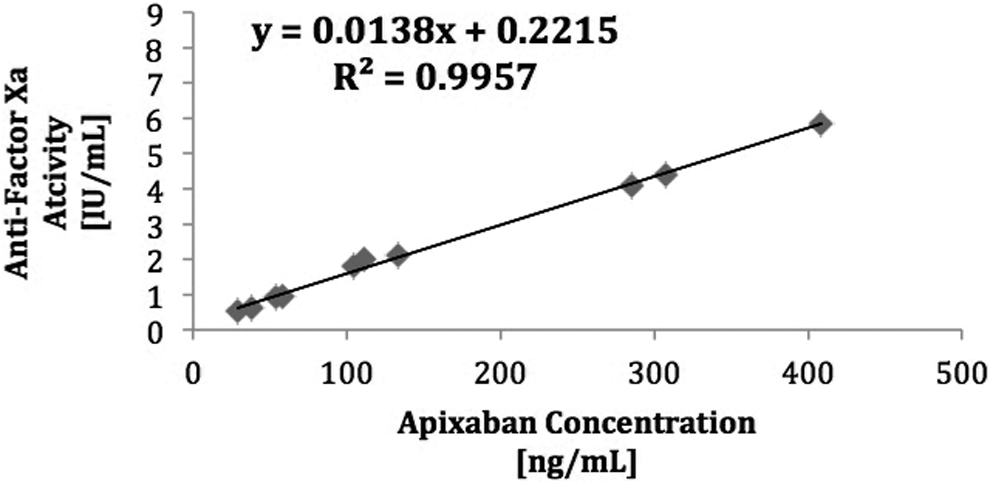
Correlation between STA liquid heparin hybrid antifactor Xa activity and apixaban concentration in treated patient samples.
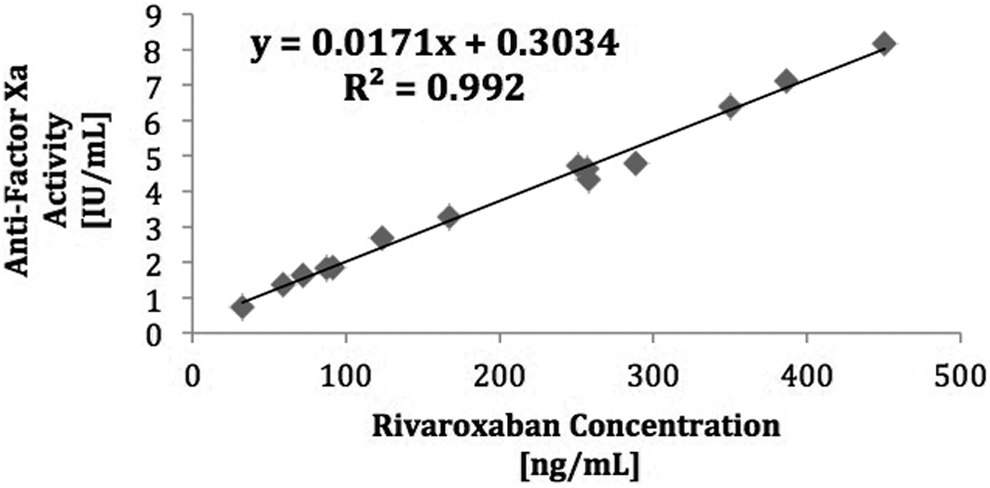
Correlation between STA liquid heparin hybrid antifactor Xa Activity and rivaroxaban concentration in treated patient samples.
The expected steady state hybrid STA liquid heparin antifactor Xa activity ranges for patients therapeutically treated with apixaban or rivaroxaban with regimens approved for nonvalvular atrial fibrillation are displayed in Table 2. Calculated expected steady state peak and trough antifactor Xa activity ranges for apixaban 5 mg twice daily are 1.80 to 2.2 IU/mL and 0.70 to 1.10 IU/mL, respectively. For rivaroxaban 20 mg once daily, these ranges are 3.80 to 6.20 IU/mL and 0.60 to 1.00 IU/mL, respectively.
Expected Steady State Peak and Trough STA liquid Heparin Hybrid Antifactor Xa Activity for Apixaban- and Rivaroxaban-Treated Patients Using Dosing Regimens for Nonvalvular Atrial Fibrillation.a

Discussion
The findings of our study demonstrate that chromogenic antifactor Xa activity obtained from a heparin calibrated antifactor Xa assay correlates very strongly in a linear manner with apixaban and rivaroxaban concentration. The correlation was observed over a broad (20-500 ng/mL) spectrum of drug concentrations and for both spiked and treated patient samples. Unique to our analysis, we demonstrated that this strong correlation exists above the normal upper limit of quantification (ULQ) for the STA Liquid Heparin Hybrid assay, requiring that serial dilutions of the sample are performed to extend to the quantifiable range of the assay to antifactor Xa activities that correlate with on-therapy drug concentrations for apixaban and rivaroxaban. We also found that PT performed with Neoplastin Plus reagent correlated strongly with rivaroxaban concentration but did not correlate as strongly with apixaban concentration in treated patient plasma samples.
Several studies have reported antifactor Xa methods that accurately determine oral direct anti-Xa anticoagulant concentration when agent-specific calibration materials are utilized. 6,11 –14 However, currently, there are no FDA-approved, agent-specific calibration materials available for clinical use in the United States, and determination of oral direct anti-Xa inhibitor concentration in a patient care setting remains problematic. Our study is in agreement with currently available evidence that demonstrates antifactor Xa assays calibrated with UFH/LMWH standards demonstrate a concentration-dependent linear relationship with apixaban and rivaroxaban. 4,7,15 –17
In previous work, the assessable range of oral direct Xa inhibitor concentrations via heparin-calibrated antifactor Xa assays has been significantly limited by the great sensitivity of the assays to the presence of oral direct Xa inhibitor agents. 17 The limitations of the commonly used ULQ for the various antifactor Xa assays has only allowed for quantitative assessment of antifactor Xa activity for a small portion of potential drug concentrations in treated patients. Gosselin et al reported a rivaroxaban concentration of approximately 80 ng/mL corresponded to the ULQ for the STA liquid heparin hybrid calibration, which is an antifactor Xa activity of 1.80 IU/mL. 17 Similarly, our linear regression model correlates an antifactor Xa activity of 1.80 IU/mL to a rivaroxaban concentration of 87 ng/mL. In our study, the use of serial dilutions successfully extended the assessable range of rivaroxaban concentration up to 500 ng/mL, which is the upper limit of the validated range for the research use only, rivaroxaban-specific calibration material utilized for drug quantification in this work.
Gosselin et al also observed a high degree of sensitivity to rivaroxaban with the STA liquid heparin antifactor Xa assays and proposed that the assays may serve as a viable method to exclude the presence of rivaroxaban. 17 The research use only, rivaroxaban-specific calibration material limited our lower limit of quantification (LLQ) to 25 ng/mL. This low rivaroxaban concentration correlates with an antifactor Xa activity of 0.73 IU/mL using the STA liquid heparin hybrid antifactor Xa assay regression model developed in this study. Therefore, it appears likely that an STA liquid heparin hybrid antifactor Xa assay result of <0.1 IU/mL would exclude the presence of a clinically relevant concentration of rivaroxaban.
Becker et al used a chromogenic antifactor Xa assay calibrated with LMWH standards to assess correlation of antifactor Xa activity with apixaban drug concentration. 7 Consistent with findings of our study, the authors found that antifactor Xa activity strongly correlated with apixaban plasma concentrations in a linear manner. This correlation was observed at low (<100 ng/mL), intermediate (100-200 ng/mL), and high (>200 ng/mL) apixaban plasma concentrations. 7 In their study, the LWMH LLQ of 0.3 IU/mL correlated with an apixaban concentration of approximately 60 ng/mL. Becker et al suggested apixaban-mediated anticoagulation can be detected at very low plasma concentrations using a standard laboratory chromogenic anti-Xa assay with LMWH. 7 In our study, the research use only, apixaban-specific calibration material limited our LLQ to 20 ng/mL. This low apixaban concentration correlates with an antifactor Xa activity of 0.50 IU/mL using the STA liquid heparin hybrid antifactor Xa assay regression model developed in this study. Therefore, it again appears likely that an STA liquid heparin hybrid antifactor Xa assay result of <0.1 IU/mL would exclude the presence of a clinically relevant concentration of apixaban.
It is important to note that the expected steady state peak antifactor Xa activities proposed within this study are difficult to utilize in a quantitative manner in clinical practice without sample dilution as the values suggested exceed the normal ULQ for the assay. In contrast, the proposed expected steady state trough antifactor Xa activities fall within the standard reportable range for the assay allowing for a quantitative assessment of drug concentration. Indeed, emerging evidence exists suggesting a relationship between plasma drug concentrations and clinical outcomes such as bleeding for direct oral anticoagulants; however, this concept requires further validation in well-designed prospective clinical trials prior to widespread implementation. 18 –21 Therefore, although chromogenic antifactor Xa assays calibrated with heparin standards appear to provide a quantitative assessment for the degree of anticoagulation due oral direct Xa inhibitors, a lack of outcomes data with corresponding drug concentrations may reduce test utility in some instances.
When comparing the proposed antifactor Xa activity ranges for patients therapeutically treated with apixaban or rivaroxaban within this study, there is a marked difference between the antifactor Xa activities expected at peak drug concentrations between the two agents, despite relatively similar expected antifactor Xa activities at trough drug concentrations. This observed difference in peak antifactor Xa activity is also described within a randomized, open-label, crossover study in healthy participant conducted by Frost et al. 22 The investigators concluded that apixaban dosed 2.5 mg twice daily demonstrated smaller peak-to-trough fluctuations in plasma drug concentration and antifactor Xa activity when compared to rivaroxaban dosed 10 mg once daily and thus may suggest more constant anticoagulation with the apixaban regimen. The clinical significance of this finding is yet to be determined.
Based on our findings, antifactor Xa testing provides clinically useful insight into the coagulation status of patients on rivaroxaban or apixaban therapy in several clinical scenarios. These scenarios include situations where clinicians may desire to assess degree of anticoagulation or drug clearance, such as prior to initiating an invasive procedure, during an active hemorrhage, and/or when anticoagulant effects could remain beyond what is anticipated due to impaired renal drug clearance. If efforts are taken to dilute patient samples, serial antifactor Xa activities could be utilized to determine a patient-specific elimination half-life, which could allow for an assessment of risk for excess drug accumulation and subsequent excessive anticoagulation. Antifactor Xa testing could also be used in a qualitative sense to identify or exclude the presence of drug, which could aid in assessing patient adherence or complete drug clearance. Additionally, with reversal agents for oral direct anti-Xa anticoagulants on the horizon, the quantitative assessment offered by antifactor Xa testing could theoretically have a role in helping determine the necessity, dose, or duration/need for repeated dosing of reversal therapy. 23 –25
Based on our findings, Neoplastin Plus PT could also provide clinically useful insight into the scenarios described earlier. However, for patients receiving apixaban, the Neoplastin Plus PT was less sensitive and correlative with the presence of drug. Thus, the use of antifactor Xa testing is likely more helpful than Neoplastin Plus PT for assessing degree of anticoagulation for patients receiving apixaban. Neoplastin Plus PT was considerably more sensitive to the presence of rivaroxaban and may be helpful to clinicians in the aforementioned scenarios for patients receiving rivaroxaban. Practically speaking, both the antifactor Xa test and the PT can be performed rapidly (30-60 minutes at our institution), while the PT offers the economic advantage of being modestly cheaper than the antifactor Xa test. Antifactor Xa testing has the benefit of not being altered by underlying liver disease, and multiple different antifactor Xa tests have been shown to correlate with rivaroxaban concentration, whereas degree of correlation appears more variable among available prothrombin reagents. 5,17 At an institution in which both the Neoplastin Plus PT and antifactor Xa testing are readily available, a case could be made for drawing either or both tests for a patient receiving rivaroxaban depending upon the clinical scenario.
There are several important considerations to weigh when interpreting the results of our study. Considerable variability is present when comparing commercially available chromogenic antifactor Xa assays calibrated with heparin standards, and therefore extrapolating the data presented within this work to other commercial antifactor Xa assays may not be accurate or valid. 17 Second, the samples analyzed within this study were small in number and were drawn at random times throughout the dosing interval. Third, the majority of these patients were taking either apixaban or rivaroxaban prior to hospital admission and were presumably at steady state concentrations. However, verification of multiple doses prior to sampling was not performed and the possibility that these patients were not at steady state or were recently transitioned from a different dosing regimen cannot be excluded. Finally, the majority of on-therapy drug concentrations assessed correlate with antifactor Xa activity above the standard ULQ. Without further dilution of samples, it is likely that the reported antifactor Xa activity will exceed the upper limit of the reportable range for the test, thus eliminating any potential quantitative assessment.
In conclusion, our study demonstrates that heparin-calibrated antifactor Xa activity correlates strongly with apixaban and rivaroxaban concentration. The serial dilution of samples allowed for the correlation of antifactor Xa activity to be extended over the majority of on-therapy drug concentrations observed in clinical practice. The STA liquid heparin antifactor Xa assays appear to be extremely sensitive to the presence of rivaroxaban and apixaban which may be advantageous in the setting of assessing complete drug clearance. Further research is needed to establish drug concentrations and antifactor Xa activities associated with clinical outcomes for rivaroxaban- and apixaban-treated patients.
Footnotes
Acknowledgments
The calibration, reagent, and control materials used to determine apixaban and rivaroxaban concentration within this article were provided by Diagnostica Stago.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
