Abstract
Background:
Rivaroxaban (Xarelto, Bayer HealthCare, Leverkusen, Germany) is a new oral anticoagulant drug. Anticoagulants may cause bleeding, thereby requiring reliable monitoring and efficient therapy. We investigated thromboelastometry versus routine coagulation tests to measure prophylactic and therapeutic concentrations of rivaroxaban and their reversal with prothrombin complex concentrate (PCC) and activated recombinant factor VII (rFVIIa) in vitro.
Methods:
Rivaroxaban was solubilized, and PCC and rFVIIa were added in 2 concentrations to the rivaroxaban-spiked blood samples, and thromboelastometry and measurements were performed.
Results:
Rivaroxaban increased tissue factor–activated clotting time (CTExTEM) dose dependently. Activated partial prothrombin time (aPTT), prothrombin time ratio (PTR), and prothrombin time (PT) were changed significantly in both concentrations. Reversal with PCC in both dosages caused no significant change in the measured parameters. For prophylactic rivaroxaban dosage, rFVIIa changed the PT significantly but not CTExTEM, aPTT, and PTR. For therapeutic rivaroxaban dosage, the CTExTEM was significantly reduced. The other parameters remained unaffected.
Conclusions:
Thromboelastometry can detect rivaroxaban effects. In vitro rFVIIa seems highly effective for reversal in contrast to PCC.
Introduction
Several new oral anticoagulants have been approved recently and launched to the market worldwide. These oral anticoagulants belong either to the group of direct factor Xa inhibitors (rivaroxaban and apixaban) or to the group of direct thrombin inhibitors such as dabigatran. Although the new oral anticoagulants mark the beginning of a new era in anticoagulation, still 2 vital questions remain unanswered. First, how can we assess the effects of rivaroxaban in emergency situations such as bleeding or urgent surgery? Second, which medication can be given to these patients if reversal is needed? To answer both the questions, we first performed an in vitro pilot study.
Since 2008, rivaroxaban was approved for thromboprophylaxis after hip and knee surgery, and In 2011, it was approved for stroke prevention in patients with nonvalvular atrial fibrillation and treatment of deep vein thrombosis by the Food and Drug Administration (FDA) and European Medicines Agency (EMA). 1 The pharmacokinetic profile is characterized by a short half-life of 3 to 9 hours. 2 Rivaroxaban shows a dual way of excretion (one-third excreted unchanged by the kidneys and the remaining two-third metabolized to inactive components and half eliminated by hepatic and another half by renal pathways). 1 Due to the pharmacodynamic characteristics of rivaroxaban and its low risk of accumulation, regular laboratory controls do not seem to be necessary. 3 However, quantitative assessment of rivaroxaban exposure might be indicated to guide clinical decisions in emergency settings and cases of suspected overdose. Parameters such as prothrombin time (PT) and the activated partial thromboplastin time (aPTT) are highly reagent and assay dependent. 4,5 The variability of PT results cannot be standardized with the international normalized ratio system commonly used for vitamin K antagonists. 4 In addition, at low plasma concentrations of rivaroxaban, measuring the antifactor Xa activity using specific calibrators appears to be the most reliable measuring method. 3 –6 Point-of-care techniques such as rotational thromboelastometry in whole blood may offer a clinical advantage due to bedside availability and shorter turnaround times compared to the conventional laboratory parameters.
Previous studies investigated the effects of prothrombin complex concentrate (PCC) and activated recombinant factor VIIa (rFVIIa) and reported differing effects. These authors used only 1 dose of reversal agent, that is, rivaroxaban, or investigated an animal model only. 7 –10 The manufacturer Bayer Health-care gives only global recommendations for bleeding under rivaroxaban therapy. First, transfusion with fresh frozen plasma, red blood cells concentrates, or thrombocytes is suggested. Second, if bleeding continues, the use of procoagulant agents such as PCC, rFVIIa, or prothrombin complex concentrate should be considered. The manufacturer also states that there are no enough clinical experiences to determine the most effective reversal agent. 1 Current publications support this point of view. 11,12
There are 2 aims of this in vitro pilot study: first, to investigate in vitro whether the clotting time (CT) in the ExTEM (tissue factor-activated CT [CTExTEM]) assay of the rotational thromboelastometry correlates better with prophylactic and high prophylactic–therapeutic levels of rivaroxaban compared to standard plasmatic coagulation tests. Second, to study the impact of different doses of coagulation factor concentrates (PCC and rFVIIa) on the in vitro reversal of prophylactic and therapeutic peak plasma levels of rivaroxaban.
Materials and Methods
Study Population and Sample Collection
We recruited 10 healthy adult volunteers without preexisting illness, pregnancy, or drug therapy. The volunteers donated 50 mL of peripheral venous blood 3 times. The blood was sampled into 10-mL citrate tubes spiked with 1 mL 3.13% citrate and an EDTA tube for a full blood count. The samples were shaken gently after sampling. Before spiking with rivaroxaban or an antidote, the samples were divided into 5-mL aliquots.
Preparation of Rivaroxaban Solution
Rivaroxaban was solubilized with dimethyl sulfoxide (DMSO) in order to establish a liquid preparation. According to the manufacturer’s manual, we dissolved the lyophilized rivaroxaban with 100% DMSO to a stock solution with 500 µg/mL. The plasma samples were prepared and spiked with rivaroxaban according to the publication of Mueck et al 13 to achieve prophylactic and therapeutic rivaroxaban concentrations similar to the blood samples at peak levels (2 hours after drug administration). The first plasma sample called Rivaproph consisted of a prophylactic concentration of 80 ng/µL equivalent to the plasma level in a prophylactic dosage of 10 mg/d. The second plasma sample called Rivather consisted of a therapeutic concentration of 200 ng/µL equivalent to the plasma level in a therapeutic dosage of 20 mg/d.
To achieve the estimated solutions, we diluted the stock solution as follows:
Rivather: 80 µL stock solution + 120 µL DMSO 100% = 200 ng/µL;
Rivaproph: 80 µL Rivather solution + 120 µL DMSO 100% = 80 ng/µL.
To achieve a more practicable dilution for exact pipetting, we diluted both rivaroxaban solutions with human plasma in a 1:9 ratio.
Preparation of PCC Solution
We used Octaplex (Octapharma GmbH, Langenfeld, Germany) as a PCC concentrate. Octaplex 600 IU was standardized according to the content of factor IX only and contains the following coagulation factors in average: factor II 800 IE, factor IX 615 IE, factor VII 275 IE, factor X 800 IE, protein C 700 IE, protein S 300 IE, sodium (75-125 mg), and heparin (100-250 IU). The lyophilized PCC was diluted with aqua dest to a concentration of 1.4 IE/100 µL. To achieve a true life dosage, we estimated it in a 75-kg patient with 70 mL blood per kilogram body weight (BW) that received 3750 IE at a dose of 50 IE/kg (0.7 IE/mL) and 1875 IE at a dose of 25 IE/kg (0.35 IE/mL).
Preparation of rFVIIa Solution
We added rFVIIa in a 90-µg/kg BW and a 180-µg/kg BW dosage. This is equivalent to 0.5 and 1 µg/mL whole blood. 14 We diluted 1 mg of rFVIIa with Tris chloride buffer to a concentration of 0.2 µg/µL. The rFVIIa was provided as a unrestricted gift by NovoNordisk, Bagsvaerd, Denmark.
Measurement Methods
Rotational Thromboelastometry With the Ex-TEM Assay
Rotational thromboelastometry is a viscoelastic method to measure the formation and strength of the clot ex vivo 15,16 and is carried out simultaneously on 2 ROTEM delta devices (courtesy of TEM International GmbH, Munich, Germany), according to the manufacturer’s instructions, using the recommended test kits provided by the manufacturer. We chose the CT as the major measurement parameter. CTExTEM is the time between start of the measurement and the inception of clot formation. All samples were analyzed within 1 hour after venipuncture with EXTEM, using tissue factor as an activator. The run time was 15 minutes at 37°C.
Plasmatic Measurements
The coagulation assays were measured using a mechanical coagulometric method on the STA-R analyzer and reagents from Roche (Mannheim, Germany). For the aPTT, we used the STA aPTT reagent (reference range 26-40 seconds); the prothrombin time ratio (PTR) was measured with the STA Neoplastin plus reagent (reference range 70%-130%). Fibrinogen was measured using the method of Clauss (reference range 1.6-4.0 g/L).
The measurements of the rivaroxaban levels have been performed using a specific chromogenic assay for the antifactor Xa activity (COAMATIC Heparin) with rivaroxaban calibrators (all from Haemochrom, Essen, Germany). In brief, the chromogenic substrate sticks to factor Xa. The content of factor Xa is directly proportional to the change in optical density. 3
Measurement Order
Citrated blood of 50 mL was divided into ten 5-mL aliquots. The aliquots were prepared and measured as follows:
blank sample: 5 mL whole blood + 50 µL plasma–DMSO mixture (5 µL DMSO + 45 µL plasma);
Rivaproph: 5 mL whole blood + 50 µL Rivaproph preparation;
Rivaproph + 25 µL PCC preparation or 25 µL rFVIIa preparation;
Rivaproph + 50 µL PCC preparation or 50 µL rFVIIa preparation;
Rivather: 5 mL whole blood + 50 µL Riva 2 preparation;
Rivather + 25 µL PCC preparation or 25 µL rFVIIa preparation;
Rivather + 50 µL PCC preparation or 50 µL rFVIIa preparation.
Statistical Analysis
Due to the small number of samples, the nature of this study is exploratory. The significance level with Bonferroni correction was found to be 0.01 (5 variables). The Friedman test for multiple groups with paired samples was applied. Wilcoxon test for paired samples was used for the 2-group relation if the level of significance was below 0.01. The nonparametric design was used due to the small number of samples.
Results
Effects of Prophylactic and Therapeutic Rivaroxaban Levels on the CTExTEM
Rivaroxaban prolonged the CTExTEM in prophylactic dosage (80 ng/mL) by 35 seconds (P = .005). The therapeutic concentrations (200 ng/mL) increased the CTExTEM by 61 seconds (P = .005). The rivaroxaban concentrations achieved in our samples are not exactly 80 ng/mL, respectively, 200 ng/mL but display a normal variation. The CT did not correlate with either prophylactic or therapeutic rivaroxaban levels. All results are shown in Table 1.
Results for Measurement of Prophylactic and Therapeutic Rivaroxaban Dosage.a
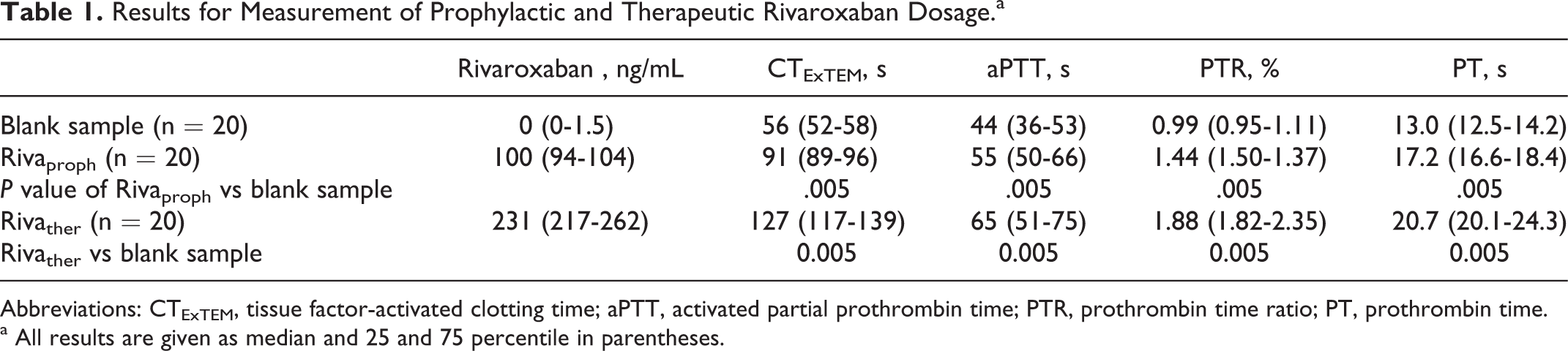
Abbreviations: CTExTEM, tissue factor-activated clotting time; aPTT, activated partial prothrombin time; PTR, prothrombin time ratio; PT, prothrombin time.
a All results are given as median and 25 and 75 percentile in parentheses.
Effects of Prophylactic and Therapeutic Rivaroxaban Levels on Plasmatic Coagulation Tests
Addition of rivaroxaban increased the aPTT (RIVAproph + 4 seconds, P = .169; RIVAther + 21 seconds, P = .005) and the PTR (Rivaproph 0.99 vs 1.44, P = .005, Rivather 1.88, P = .005). The PT increased in both rivaroxaban concentrations: Rivaproph + 4.2 seconds, P = .009 and Rivather + 7.7 seconds, P = .005. Neither plasmatic coagulation parameter showed a reliable correlation with the rivaroxaban levels in both groups (Table 1).
Reversal of Rivaroxaban Anticoagulation
Effects of the Reversal With PCC on Thromboelastometry on the CTExTEM
The addition of PCC showed no significant effect on CTExTEM in both rivaroxaban concentrations and PCC doses (details given in Table 2).
Results for Measurement Order With PCC for Reversal of Prophylactic and Therapeutic Rivaroxaban.
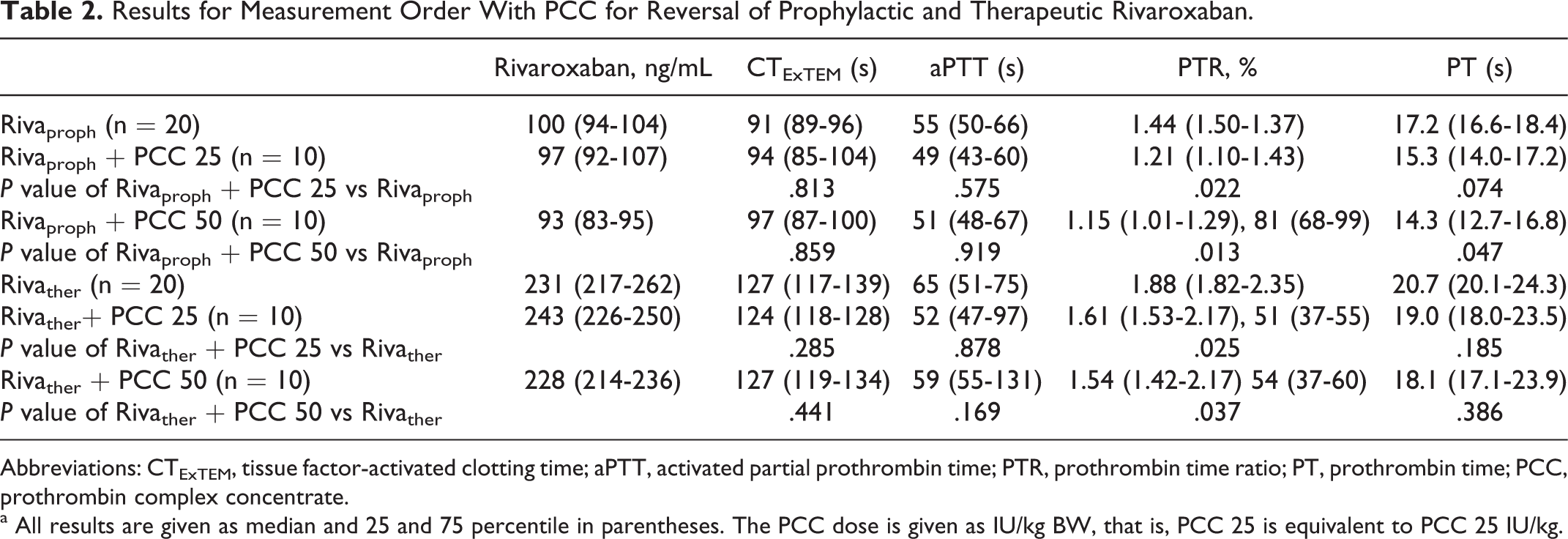
Abbreviations: CTExTEM, tissue factor-activated clotting time; aPTT, activated partial prothrombin time; PTR, prothrombin time ratio; PT, prothrombin time; PCC, prothrombin complex concentrate.
a All results are given as median and 25 and 75 percentile in parentheses. The PCC dose is given as IU/kg BW, that is, PCC 25 is equivalent to PCC 25 IU/kg.
Effects of Reversal With PCC on Plasmatic Coagulation Tests
The plasmatic measurements showed no significant changes after addition of PCC dose (see Table 2).
Effects of Reversal With rFVIIa on the CTExTEM
Addition of either dose of rFVIIa decreased the CTExTEM in the prophylactic concentration of rivaroxaban to the blank sample values but failed to reach the level of significance of .01 (rFVIIa 90 µg/kg: −35 seconds, P = .028; rFVIIa 180 µg/kg: −40 seconds, P = .028). In the therapeutic rivaroxaban concentration, CTExTEM was significantly decreased close to the normal values (blank sample 56 seconds, with 90 µg/kg rFVIIa = 64 seconds, P = .005, and 180 µg/kg rFVIIa = 60 seconds, P = .005; Table 3).
Results for Measurement Order with rFVIIa for Reversal of Prophylactic and Therapeutic Rivaroxaban.a
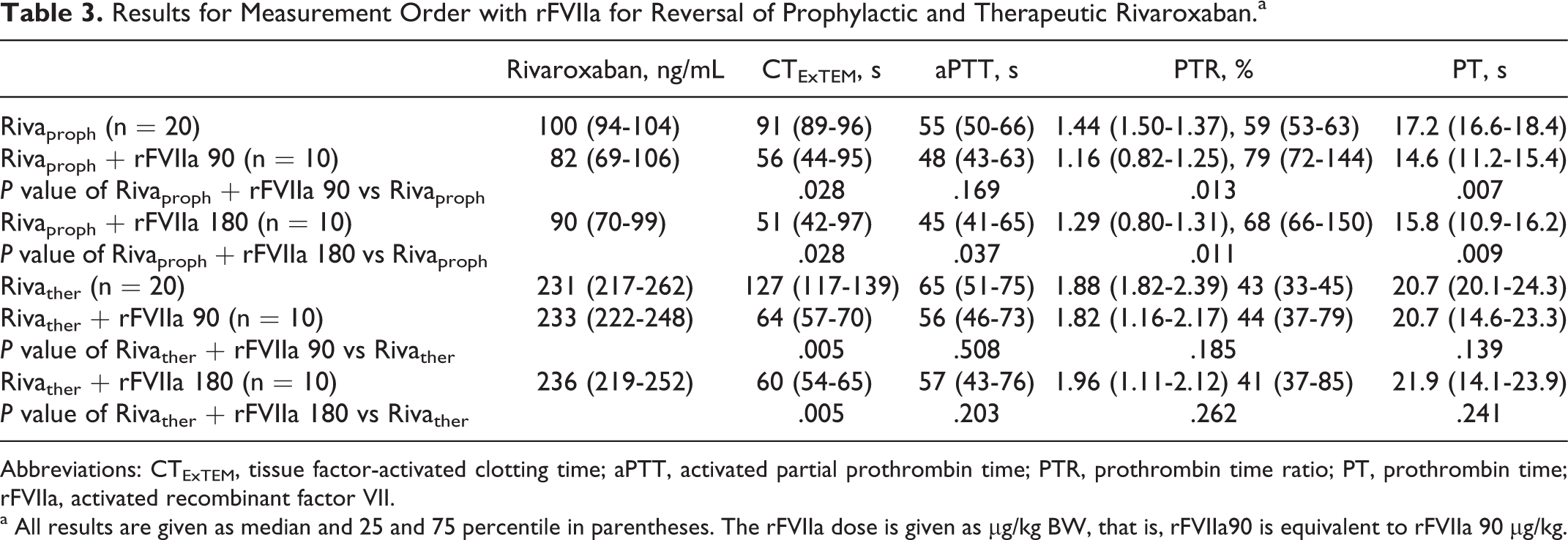
Abbreviations: CTExTEM, tissue factor-activated clotting time; aPTT, activated partial prothrombin time; PTR, prothrombin time ratio; PT, prothrombin time; rFVIIa, activated recombinant factor VII.
a All results are given as median and 25 and 75 percentile in parentheses. The rFVIIa dose is given as µg/kg BW, that is, rFVIIa90 is equivalent to rFVIIa 90 µg/kg.
Effects of Reversal With rFVIIa on Plasmatic Coagulation Tests
The results are given in Table 3. Both dosages of rFVIIa significantly reversed the effects of prophylactic rivaroxaban as measured by the PT. The aPTT decreased in the therapeutic rivaroxaban dosage but not significantly. The PTR did not change after the intervention (see Table 3).
Monitoring of Rivaroxaban Content
According to the previous work of Mueck et al, the estimated ranges for the peak values of rivaroxaban in prophylactic and therapeutic ranges were 80 to 100 ng/mL and 200 ng/mL, respectively. 13 In both groups, the targeted ranges were reached (see Tables 1–3).
Discussion
It has to be expected that the number of critical bleeding events will increase in the next years related to the rising number of patients treated with rivaroxaban. This will in turn increase challenges for surgeons and anesthesiologists. 17,18 The availability of an effective reversal agent is essential for the safe performance of emergency surgeries in patients treated with rivaroxaban or reversal of rivaroxaban-induced bleeding.
Our study investigated whether thromboelastometry-derived parameters and standard coagulation parameters are reliable for the monitoring of prophylactic and therapeutic levels of rivaroxaban. Furthermore, PCC and rFVIIa in 2 doses were assessed for the reversal of prophylactic and therapeutic levels of rivaroxaban. There are 2 main findings of this investigation. First, the CTExTEM of the rotational thromboelastometry shows a concentration-dependent prolongation after the addition of escalating concentrations of rivaroxaban but does not correlate with prophylactic or therapeutic rivaroxaban levels or standard coagulation tests. Second, addition of rFVIIa mainly reduced the CTExTEM, whereas the addition of increasing doses of PCC did not affect neither the CTExTEM nor the standard coagulation assays.
Monitoring of Rivaroxaban in Prophylactic and Therapeutic Dosage With Thromboelastometry and Standard Coagulation Parameters
To the best of our knowledge, we have shown that the CTExTEM detects the anticoagulant effects of prophylactic and therapeutic levels of rivaroxaban. These results allow the interpretation that thromboelastometry as a point-of-care monitoring detects the qualitative effect of rivaroxaban at the bedside. This is helpful especially in emergency situations when shorter turnaround times of the point-of-care monitoring results may influence treatment decisions. However, the CTExTEM does not correlate with quantitative (that is prophylactic and therapeutic) rivaroxaban levels. This limits the clinical usefulness of thromboelastometry for rivaroxaban as a bedside drug monitoring. Therefore, monitoring of rivaroxaban anticoagulation with the CTExTEM underlies the same limitations as with the PT, aPTT or PTR. On the contrary, the specific chromogenic antiXa assay does not display exactly the anticoagulant effect of rivaroxaban as it primarily reflects the plasma levels of the drug. This raises the question whether plasma levels of rivaroxaban can be used to guide the use of reversing agents. Future investigations, for example, using an ex vivo design, are required to answer this question.
Previous studies described the anticoagulant effects of rivaroxaban using standard coagulation tests but did not measure correlation with the rivaroxaban levels using a rivaroxaban calibrated antifactor Xa levels 9,10 but with PT, aPTT, bleeding time, blood loss, and endogenous thrombin generation. In our study, the PTR using the Neoplastin plus assay decreased after the addition of rivaroxaban in prophylactic and therapeutic dosing, which confirms the earlier data from Mani et al. 5 This is in accordance with the findings of Lindhoff-Last et al who reviewed different standard laboratory measurements 3 and concluded that the reductions in the PTR are assay dependent and showed variable sensitivities. However, it has to be noted that the PTR using Neoplastin plus did not correlate well either in prophylactic or in therapeutic rivaroxaban plasma levels. This is in contrast to the results published previously 19 but might be explained by the fact that earlier data were measured over a broad range of rivaroxaban plasma levels (prophylactic and therapeutic); whereas our data show that this correlation is no more acceptable when prophylactic and therapeutic concentrations are analyzed separately. The aPTT is prolonged less sensitively by rivaroxaban and seems to have no benefit for clinicians to monitor the anticoagulant therapy with rivaroxaban or reversal treatment. 3,20 Our results confirm that the aPTT is less affected by rivaroxaban and no concentration dependency can be identified.
Reversal of Rivaroxaban
Reversal of Rivaroxaban With PCC
Addition of 25 or 50 IU/kg PCC had no significant reversal effect on the CTExTEM in both groups. This is not consistent with the findings of Godier et al 10 who investigated the effects of PCC and rFVIIa on rivaroxaban anticoagulation in a rabbit model. In their study, PCC reduced the CTExTEM significantly (CTExTEM −43%, P < .05) but was considered not suitable for rivaroxaban reversal because it did not reduce bleeding time in vivo. These results are not fully comparable to our findings due to the different study designs and drug concentrations (40 IU/kg) but suggest that the effect of PCC to reverse the anticoagulation achieved with rivaroxaban is questionable. Eerenberg et al investigated the reversal with PCC (50 IU/kg) in healthy volunteers taking 20 mg of rivaroxaban twice daily for 2½ days. 9 This dosage is not approved for patient treatment. In contrast to our and Godier’s results, a complete reversal of the rivaroxaban effect using PCC was observed. 10 The efficacy of the reversal therapy was monitored using the PT (PT 15.8 ± 1.3 seconds vs 12.3 ± 0.7 seconds after reversal with PCC) and the endogenous thrombin potential (ETP 50% ± 63% vs 91% ± 205%). It was concluded that 50 IU PCC per kilogram BW reversed the effect of rivaroxaban. Potential drawbacks of this work are that ETP is not available in clinical routine of most hospitals and that the plasma level of rivaroxaban prior to the administration of PCC was not measured. This leads to lack of clarity as to the effective plasma concentration of rivaroxaban anticoagulation PCC. Our study investigated the peak levels of prophylactic or therapeutic dosage of rivaroxaban according to the previously published evidence. 13 These peak levels and the differences in study design (in vitro vs in vivo) might explain the differences in efficacy of PCC to reverse rivaroxaban.
Reversal of Rivaroxaban With rFVIIa
The addition of rFVIIa decreased the CTExTEM to subnormal levels in prophylactic rivaroxaban concentration and to nearly normal levels in therapeutic concentration with almost no difference between the higher and lower dosage (Table 3). This might suggest that the lower dose of rFVIIa (90 µg/kg) is as effective as the higher dose (180 µg/kg) for reversal of prophylactic and therapeutic rivaroxaban therapy in vitro. These results are, in part, supported by the findings of Godier who reported a 43% reduction in CTExTEM after addition of 150 µg/kg rFVIIa in a rabbit model of induced hepatosplenic bleeding, which might be explained by the known thrombin-generating effect of rFVIIa10mü.
Study Limitations
Due to study setting as an in vitro model, the data allow limited conclusions for the clinical setting. First, the in vitro model does not regard in vivo pharmacokinetic effects such as metabolism or elimination of rivaroxaban. Second, the results from this in vitro setting cannot be directly translated to the clinical efficacy of the reversal agents, although in conjunction with other data from the literature, our results may give an idea to the clinician regarding which factor concentrates could be effective.
Conclusions
This study indicates the use of thromboelastometry as a point-of-care method for urgent measurement of rivaroxaban effects. Our study indicates that the CTExTEM shows a concentration-dependent prolongation after the addition of rivaroxaban but failed to show a reliable correlation with the plasma levels of rivaroxaban. The PT and PTR showed almost the same pattern, indicating that especially at higher rivaroxaban levels conventional coagulation parameters as well as thromboelastometry do not yield reliable results to determine the rivaroxaban concentration.
The evaluation of the efficacy of PCC and rFVIIa to reverse the peak levels of rivaroxaban after prophylactic and therapeutic concentrations showed a potent effect of rFVIIa, whereas PCC did not significantly influence the CTExTEM or conventional coagulation parameters. Therefore, our data support the manufacturers reversal recommendations only for rFVIIa. Our results indicate that 90 µg/kg of PCC are as effective as high doses of rFVIIa (180 µg/kg) for reversal of prophylactic and therapeutic rivaroxaban concentrations. These results need to be validated in an in vivo study to overcome methodological limitation of in vitro testing.
Footnotes
Authors’ Note
Parts of the data of this study were presented at the GTH meeting 2012 in St Gallen, Switzerland, ASA meeting 2012 in Washington, USA, and the German Anesthesia Congress (DAC) 2012 in Leipzig.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:CvH has received honoraria for consultancy work and lectures from Boehringer Ingelheim, Pfizer, Bayer Healthcare, Novo Nordisk, Baxter GmbH and CSL Behring and research grants from Bayer Healthcare and Boehringer Ingelheim. MK received travelling grants for congress participation and is delegate of the Thrombosis Experts Academy, sponsored by Pfizer and Bristol-Myers Squibb. EL, SZ and EP have no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Bayer Healthcare GmbH, Leverkusen, Germany.
