Abstract
Background:
Coronary artery disease (CAD), also known as atherosclerotic heart disease, is a leading cause of mortality and morbidity throughout the world. The role of insertion/deletion (I/D) polymorphisms of the angiotensin-converting enzyme (ACE) gene in the etiology of CAD remains to be more completely clarified. The aim of this study was to determine the role of the ACE I/D polymorphism in patients with CAD and to study the association together with traditional risk factors in assessing the risk of CAD.
Methods:
Our study population included 145 Tunisian patients with symptomatic CAD and a control group of 300 people matched for age and sex. All participants in the study were genotyped for the ACE I/D polymorphisms obtained by polymerase chain reaction amplification on genomic DNA.
Results:
Our analysis showed that the ACE D allele frequency (P < 10−3; odds ratio [OR] = 5.2; 95% confidence interval [CI] = 3.6-7.6) and DD genotype (P < 10−3; OR = 6.8; 95% CI = 4.4-10) are significantly more prevalent among patients with CAD than in controls and may be predisposing to CAD. We further found that the risk of CAD is greatly potentiated by several concomitant risk factors (smoking, diabetes, hypertension, dyslipidemia, and a family history of CAD).
Conclusion:
The ACE D allele may be predictive in individuals who may be at risk of developing CAD. Further investigations of these polymorphisms and their possible synergisms with traditional risk factors for CAD could help to ascertain better predictability for CAD susceptibility.
Introduction
Coronary artery disease (CAD) remains a leading cause of mortality in both high- and low-income countries. 1,2 It is characterized by lesions of the coronary artery generated by the phenomenon of atherosclerosis. 3 Coronary artery disease is defined as a coronary obstruction in 1 or more major coronary arteries and documented by quantitative coronary angiography. 4 To date, CAD is considered as a multifactorial disease. 5 In addition to well-known risk factors such as age, gender, hypertension, dyslipidemia, smoking, a family history of disease, and diabetes mellitus (DM), 6 recent research has shown that genetic and environmental factors play important roles in the development of CAD. 7
Notably, the renin–angiotensin system (RAS), which regulates blood pressure, plays a pivotal role in the pathogenesis of CAD. The RAS is one of the major regulatory systems of cardiovascular physiology. It includes cardiovascular remodeling, sodium homeostasis, and maintenance of vascular tone. 8
Several studies have suggested that polymorphisms in the components of the RAS are important in the development and progression of CAD in some individuals. Owing to the key role of angiotensin-converting enzyme (ACE) in RAS, ACE insertion/deletion (I/D) polymorphisms have frequently been investigated. 9,10
The ACE gene is located on chromosome 17q23. It spans 21 kb and consists of 26 exons and 25 introns. The ACE I/D polymorphism (rs4646994) is characterized by the presence (I) or absence (D) of a 287 bp Alu repeat sequence within intron 16, resulting in 3 genotypes—DD, II, and ID. 11
The results from association studies between ACE and CAD have been inconsistent, and several hypotheses have been suggested to explain possible mechanisms. The results from recent Tunisian studies provided conflicting results and did not consider the relationship between the ACE gene and traditional risk factors of CAD. 12,13
Thus, the purpose of this study was to determine the relationship between the ACE I/D polymorphism and the presence of CAD, and further, to study the association and possible synergisms with traditional risk factors in predicting the risk of CAD.
Materials and Methods
Study Populations
Our study population consisted of 145 Tunisian patients with symptomatic CAD (94 males and 51 females, with a mean age of 62.18 ± 13.36 [standard deviation, SD] years), admitted for diagnostic coronary angiography. Patients with significant coronary obstruction determined by coronary angiography were selected consecutively from the Department of Cardiology. Only patients with the evidence of myocardial infarction (MI), angina, a history of prior angioplasty, CAD bypass grafting, or patients with more than 30% stenosis in 1 or more coronary arteries were included. However, patients with cardiomyopathy, valvular heart disease, congenital heart disease, or stroke were excluded from the study. Additionally, the control group consisted of 300 participants (186 males and 114 females, with a mean age of 62.50 ± 10.55 [SD] years) with no history of cardiovascular disease. Age- and gender-matched participants were recruited from diverse regions of Tunisia. Despite the small number, this sample was a good representative of our population as patients and controls are referred to this hospital from all regions of Tunisia.
Epidemiological and clinical data such as hypertension, noninsulin–dependent type II diabetes mellitus (T2DM), dyslipidemia, smoking, a family history of CAD, and angina type were included in the study. The risk factors considered were hypertension, T2DM, dyslipidemia, cigarette smoking, and a family history of CAD. Information about the conventional risk factors for CAD was obtained from all participants using a standard questionnaire, and the data were validated with reference to hospital case records. Hypertension was defined as a blood pressure of 160/95 mm Hg or greater on repeated measurements and/or the current use of antihypertensive drugs due to a history of arterial hypertension. 14 Diabetes mellitus was defined as hyperglycemia requiring antidiabetic drugs or a fasting blood sugar level exceeding 7.0 mmol/L (126 mg/dL). 15
Dyslipidemia was defined on the basis of the European guidelines on cardiovascular disease prevention in clinical practice that uses total cholesterol (TC) and low-density lipoprotein (LDL-C) levels to guide treatment. In general, TC < 190 mg/dL and LDL-C < 115 mg/dL were regarded as normal (nondyslipidemic). In patients with established cardiovascular disease (MI, acute coronary syndrome, and stroke) or T2DM, thresholds of TC < 175 and LDL-C < 100 mg/dL were applied. Patients exceeding either threshold were considered dyslipidemic. High-density lipoprotein and triglycerides were not used to define dyslipidemia. 16 Cigarette smoking was assessed by asking participants about history of smoking habits. A family history of CAD was defined as having a first degree relative with established CAD before 55 years of age in men and 65 years in women. 17
All participants in the study (or their legal proxies) gave informed consent for giving a blood sample for genetic analysis. The study protocol was approved by the Ethics Committee of the Military Hospital of Tunisia and has, therefore, been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Genotyping
Genomic DNA was extracted from peripheral blood leukocytes with the DNA extraction kit (QIAmp blood kit, Qiagen GmbH; Hilden, Germany) according to the manufacturer’s protocol. The amplification with primers was used to amplify the genomic DNA. To detect the ACE I/D polymorphism, polymerase chain reaction-based protocols were performed. The ACE alleles were visualized as fragments of 490 bp (I) and 190 bp (D). Amplified DNA fragments were detected by agarose gel electrophoresis (2% agarose gel), stained with ethidium bromide, and UV transillumination.
Statistical Analysis
Genotype distribution and allele frequencies were represented as absolute numbers and percentages. Differences between cases and controls were evaluated by using the χ2 test or Fisher exact test for qualitative variables and the Student t test for quantitative variables. The associations between conventional risk factors and genotypes with CAD were reported using odds ratio (OR) with a 95% confidence interval (CI). Multivariate logistic regression was applied to analyze factors associated with the risk of CAD. Probability values P < .05 were considered statistically significant. Genotype distributions were in Hardy-Weinberg equilibrium as assessed by the χ2 test. All association analyses were carried out using SPSS software, version 16.0 (IBM, Armonk, New York).
Results
The demographic characteristics of the 145 patients are shown in Table 1. There were significant differences between the patients with CAD and the control group with regard to DM, hypertension, and dyslipidemia. Using the χ2 test, patients with hypertension (60.7% vs 8.0% controls; P < 10−3; OR = 17; 95% CI = 10-30), DM (53.1% patients vs 3.0% controls; P < 10−3; OR = 36; 95% CI = 17-77), and dyslipidemia (42.1% patients vs 4.0% controls; P < 10−3; OR = 17; 95% CI = 8.9-34) were found to be possible independent risk factors for CAD. However, there were no significant differences between patients and controls regarding gender, age, smoking, and those with a family history of CAD.
Clinical Characteristics of Study Participants.a
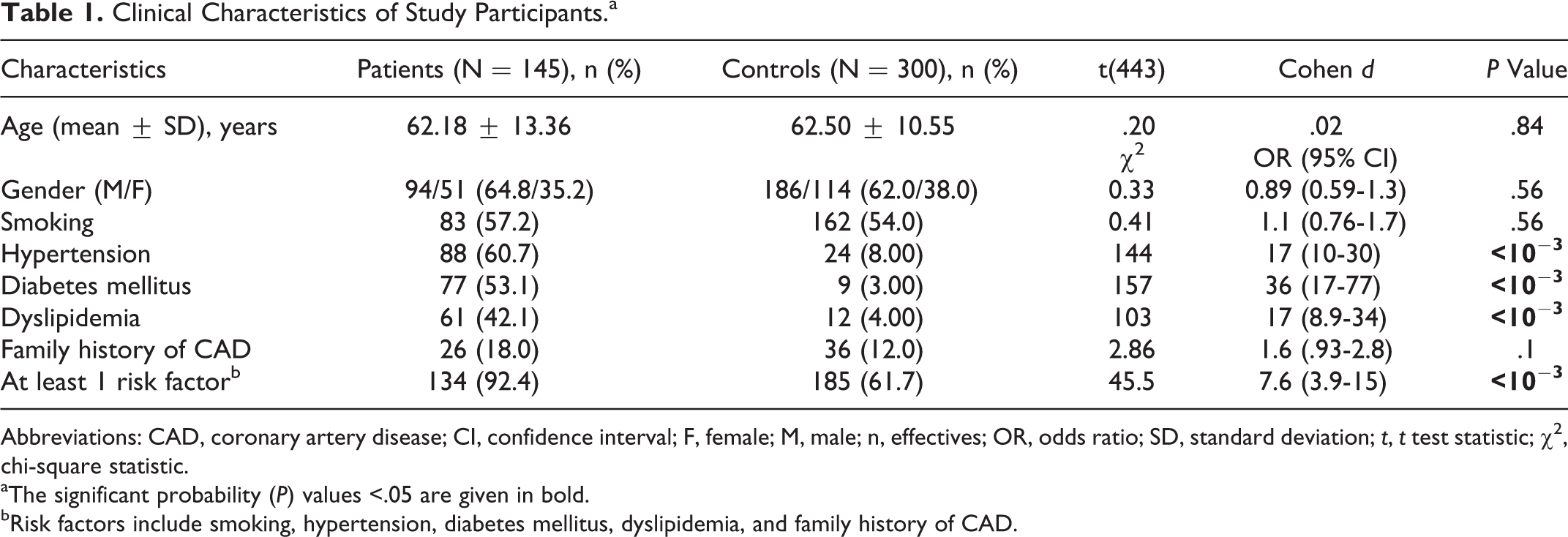
Abbreviations: CAD, coronary artery disease; CI, confidence interval; F, female; M, male; n, effectives; OR, odds ratio; SD, standard deviation; t, t test statistic; χ2, chi-square statistic.
aThe significant probability (P) values <.05 are given in bold.
bRisk factors include smoking, hypertension, diabetes mellitus, dyslipidemia, and family history of CAD.
The distribution of the ACE genotypes in the study population (Table 2) was in Hardy-Weinberg equilibrium (patients: χ2 = .14, P = .71; controls: χ2 = .01, P = .92). The ACE D allele frequency (86.90% patients vs 56.2% controls; P < 10−3; OR = 5.2; 95% CI = 3.6-7.6) and DD genotype (75.9% patients vs 31.7% controls; P < 10−3; OR = 6.8; 95% CI = 4.4-10) were significantly more prevalent among patients with CAD than in controls.
Distribution of ACE I/D (Genotypes and Alleles) in Coronary Artery Disease for Patients and Controls.a
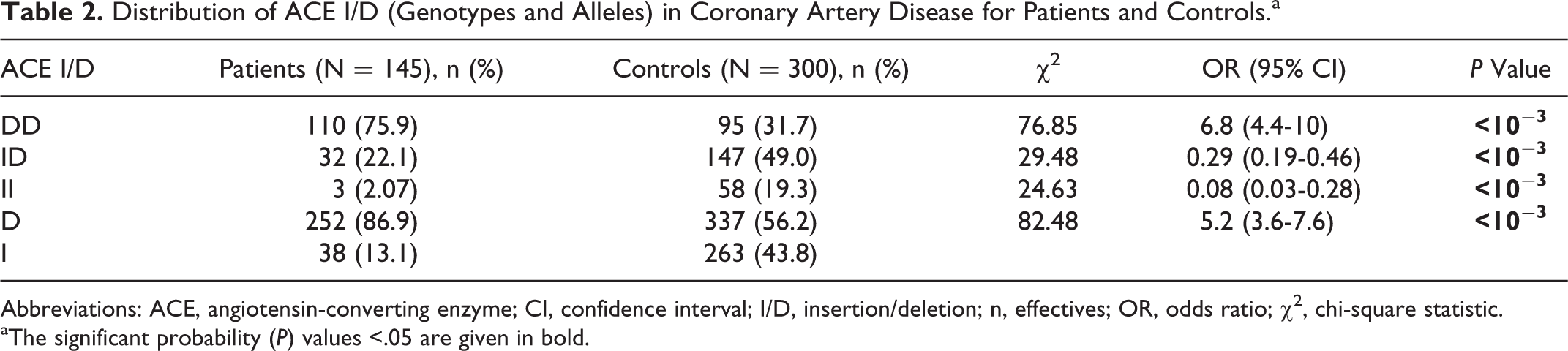
Abbreviations: ACE, angiotensin-converting enzyme; CI, confidence interval; I/D, insertion/deletion; n, effectives; OR, odds ratio; χ2, chi-square statistic.
aThe significant probability (P) values <.05 are given in bold.
Three multivariate logistical models, for all participants and by gender, indicate that the DD genotype (all: OR = 8.5; males: OR = 9.2; females: OR = 9.0), hypertension (all: OR = 6.6; males: OR = 10; females: OR = 4.4), diabetes (all: OR = 22; males: OR = 61; females: OR = 9.4), and dyslipidemia (all: OR = 5.6; males: OR = 6.4; females: OR = 5.6) are statistically significant independent risk factors for CAD. Smoking (males: OR = 5.3) was found to be a risk factor only for males (Table 3). The adjusted OR for CAD due to the DD genotype and dyslipidemia is commensurate across all 3 models; however, the adjusted OR for CAD in male with diabetes is more than 6-fold than that of women with diabetes, and the adjusted OR of CAD in males with hypertension is 2-fold than that of women with hypertension. Diabetes is the most significant risk factor for both genders. The adjusted risk for women with diabetes is on par with the risk associated with the DD genotype, and the risk associated with hypertensive males is similar to the risk of DD carriers.
Multivariate Logistic Regression Models Showing Significant Risk Factors Having Association With CAD (Ensemble, Male, and Female Models).a,b
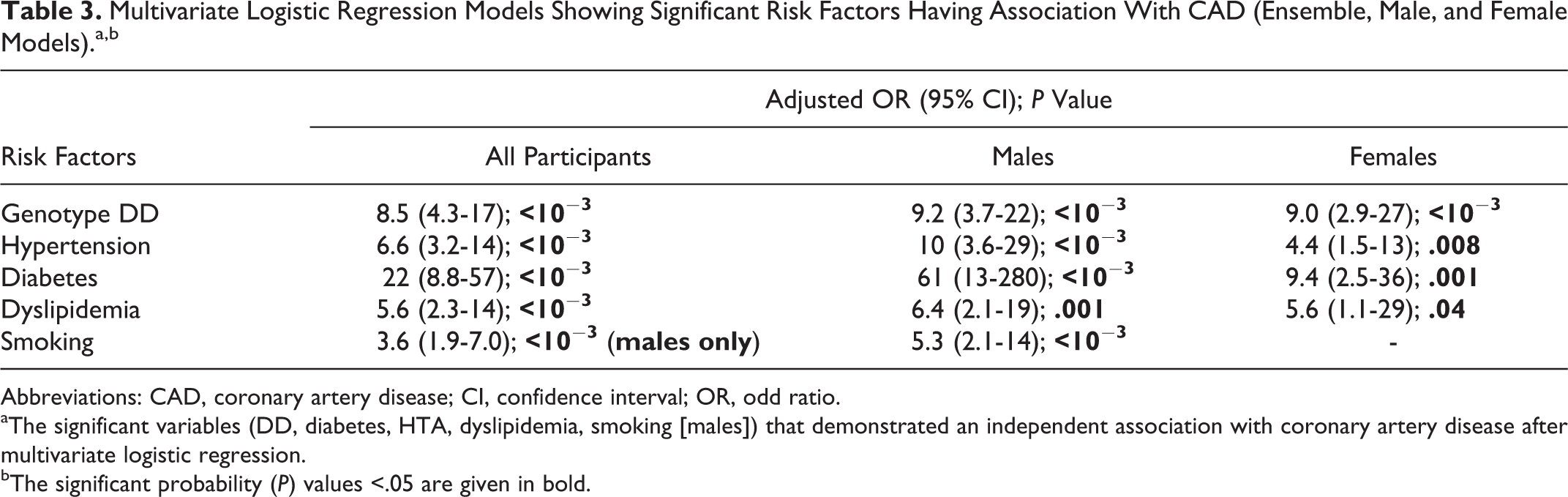
Abbreviations: CAD, coronary artery disease; CI, confidence interval; OR, odd ratio.
aThe significant variables (DD, diabetes, HTA, dyslipidemia, smoking [males]) that demonstrated an independent association with coronary artery disease after multivariate logistic regression.
bThe significant probability (P) values <.05 are given in bold.
Stratification of participants analyzed the influence of the genotypes with or without cardiovascular risk factors (Table 4). Carriers of the DD genotype were found at increased risk with female participants (OR = 9.3; 95% CI = 4.1-21) as compared to male participants (OR = 5.9; 95% CI = 3.4-10). When the cardiovascular risk factors were excluded from the analysis, the association of the DD genotype with CAD was more pronounced in participants without hypertension (OR = 6.2; 95% CI = 3.3-12) than in participants with hypertension (OR = 5.7; 95% CI = 2.2-15). The association of the DD genotype with CAD was also stronger in nondiabetics (OR = 8.3; 95% CI = 4.4-16) than in diabetics (OR = 5.33; 95% CI = 1.22-23). In contrast, DD carriers among smokers (OR = 8.5; 95% CI = 4.7-16) and those with dyslipidemia (OR = 8.2; 95% CI = 2.1-32) were at a greater risk as compared to DD carrier nonsmokers (OR = 5.9; 95% CI = 2.9-12) and nondyslipidemic DD carriers (OR = 5.7; 95% CI = 3.3-9.9). The DD genotype was also found to convey a higher risk with CAD in participants having a family history of CAD (OR = 12; 95% CI = 3.5-45) than in participants without a family history of CAD (OR = 6.1; 95% CI = 3.7-9.9).
DD Versus (ID + II) by Stratum for Patients Versus Controls.a
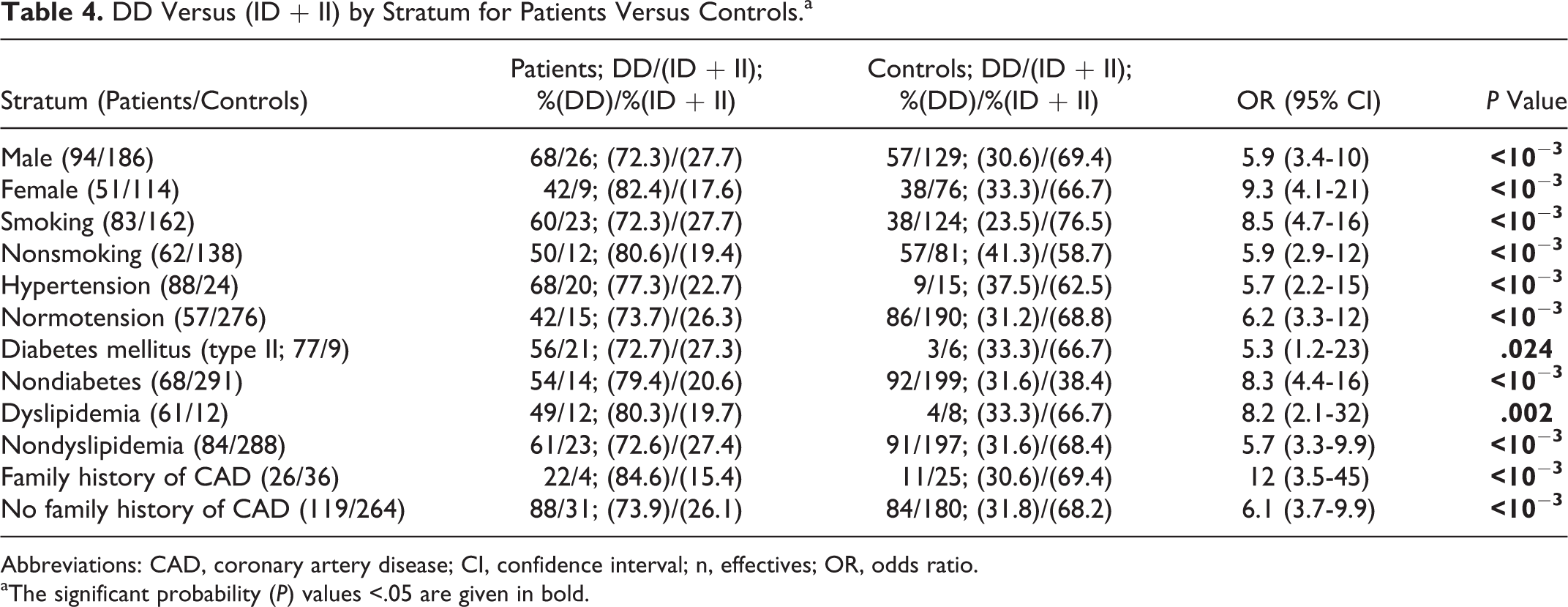
Abbreviations: CAD, coronary artery disease; CI, confidence interval; n, effectives; OR, odds ratio.
aThe significant probability (P) values <.05 are given in bold.
Discussion
Coronary artery disease is a polygenic disease that involves complex interactions among several pathophysiological pathways with multiple genes and environmental risk factors. 18,19 We analyzed several traditional cardiovascular risk factors in patients with CAD. This analysis revealed that diabetes, hypertension, and dyslipidemia have significant effects on the presence of CAD. These results are consistent with studies performed in Egypt, Iran, Spain, and Germany. 11,19 –21
There are several pathophysiology mechanisms that explain diabetes, hypertension, and dyslipidemia in the contribution of CAD. Abnormal processes such as damage to the arterial wall, metabolic disorders, oxidative stress, and endothelial dysfunction can lead to arterial sclerosis, lesions of the coronary artery, and increase in the risk of development of atheromatous plaque. 22,23
Our study revealed a strong association between the ACE D allele, in general, and the DD genotype, in particular, to the risk of CAD. In comparison with other studies, our results are well in concordance with other studies. 10,19,24 –26 However, the results of others studies in Iran and Germany did not find any association between the ACE I/D polymorphisms and CAD. 11,27 The etiologies by which ACE I/D genotypes may predispose an individual to the development of CAD remain incomplete. The observation that major coronary risk factors are associated with coronary endothelial dysfunction even before stenoses are visible has led to the concept that endothelial dysfunction occurs at the early stages of atherosclerosis. 28 The ACE I/D polymorphisms have been implicated in the pathogenesis of atherosclerosis and play an important role in endothelial function. 29,30 Angiotensin-converting enzyme, a key enzyme in the RAS system, converts angiotensin I to the potent vasoconstrictor angiotensin II. The chronic influence of higher levels of angiotensin II in the ACE D allele carriers in comparison with the angiotensin I allele carriers may concur to the development and/or progression of atherosclerosis. 31 An increase in the ACE activity will increase angiotensin II, affecting cell growth and proliferation by stimulating a variety of cytokines and growth factors that cause endothelial dysfunction by reducing nitric oxide bioavailability. 32,33 The enhanced ACE expression in macrophages and smooth muscle cells of coronary artery plaques has been previously reported to indicate that ACE activity in lesions contributes greatly to the progression of atherosclerosis. 31 The ACE DD genotype has been associated with higher serum ACE activity, and numerous studies have investigated the association between the D allele and atherosclerosis. 28 –30,34
In our logistic model, 4 independent variables were associated with the occurrence of coronary atherosclerosis—DD genotypes, hypertension, diabetes, and dyslipidemia. The logistic regression analysis confirmed that the ACE DD genotype is an important risk factor that would increase the odds for CAD by more than 8-fold over those without the DD genotype while holding other risk factors constant. Furthermore, the DD genotype and the D allele, concurrent with hypertension, seem to promote the risk of CAD. This result is consistent with previous reports, Kato et al had found that the DD genotype of the ACE I/D polymorphism is a major risk factor for cerebral and cardiovascular events such as CAD and stroke in Japanese patients with hypertension. 35 Other studies also reported strong associations between the DD genotype and the risk of hypertension in their study populations. 36,37 In regard, it appears that the ACE DD genotype acts synergistically with increased blood pressure to increase the risk of coronary disease. However, it also appears that the ACE I/D polymorphism may be a promoter by itself in the development of hypertension. Multiple maladaptive pathways play roles in this process. Angiotensin-converting enzyme D allele carriers have been found to have higher tissue ACE messenger RNA expression, increased concentrations of circulating ACE, and increased bradykinin degradation. These may impair endothelium-dependent vasodilatation that generates damaged blood vessels and contributes to hypertension. 35,36,38,39
Furthermore, recent findings, as well as this research here within, have shown an association between the ACE D allele, diabetes, and the risk of developing cardiovascular disease. 40 However, some studies failed to discern any relationship between the ACE gene polymorphism and CAD in patients with diabetes. 41,42 A possible mechanism suggests that the ACE DD genotype is involved in increasing the vasoconstrictor angiotensin II as well the degradation of the vasodilator bradykinin, which increases resistance to insulin. 43
In consideration of research indicating possible synergisms between traditional CAD risk factors and the ACE I/D polymorphism, the study population was subgrouped by risk factors (smoking, diabetes, hypertension, dyslipidemia, and a family history of CAD). The distribution of genotypes (DD vs ID + II) for patients versus controls was subsequently studied for statistical associations. Our study revealed that the ACE DD genotype acts in concert with traditional CAD risk factors (smoking, diabetes, hypertension, dyslipidemia, and a family history of CAD) to potentiate the risk of CAD as compared to the reference group. We noted that the association of the DD genotypes with CAD was confirmed in the subgroup of patients without any known risk factor.
In our study, the DD genotype and D allele increased the risk of coronary disease in smokers. However, previous studies had demonstrated that there may be an association between the ID genotype and cigarette smoking in patients with CAD. 31,42 Indeed, it has been noted that smoking and the D allele increase the superoxide anion formation and degradation of the nitric oxide that cause endothelial dysfunction favoring the occurrence of coronary disease. 44,45 Our results also showed that the DD genotype was associated with an increased risk of CAD in patients with dyslipidemia. Other studies noted that hyperlipidemia and the D allele have been associated with a higher risk of CAD. 42,46,47 This may suggest that the ACE gene plays certain roles in lipid metabolism. 48
This current research also determined a mediating/potentiating effect of the DD genotype on those with a family history of CAD. Although the risk of CAD (regardless of the ACE genotype) in those with a family history of CAD was moderate, the risk of CAD in the subgroup containing both a family history of CAD and DD carriers was greatly enhanced. We observed incremental risk of DD genotypes with family history of CAD compared to those without family history of CAD. An Australian study of children found that the ACE I/D polymorphism is associated not only with a parental history of MI but also with CAD in the second-degree relatives. Significant differences in the genotype distribution were detected only in those children with 2 or more affected grandparents. 49 This research is suggestive that a family history of CAD and ACE I/D genotype are bidirectionally associated, whereas our data show that the DD genotype and a family history of CAD are independent risk factors for CAD, and further, that the DD genotype may increase the risk to those who also share a family history of CAD. Since most CAD is believed to have a multifactorial genetic basis involving both genes and interacting environmental factors that determine whether a person will develop the disease, it is difficult to parse which factors producing disease in a family history are acting in concert with the ACE I/D polymorphism to impact an increased risk of CAD.
Our work has several limitations. First, the results here are preliminary owing to the limitation of the small number of patients. Second, ACE plasma concentrations were not determined, and it would be of interest in future studies to better attribute the ACE plasma level with genotypes.
Conclusion
Coronary artery disease is a multifactorial pathogenesis; several risk factors and a genotype association of the ACE I/D polymorphism have been proposed for the development of this disease. The ACE D allele may be predictive in individuals who may be at risk of developing CAD. Furthermore, additional information with regard to the presence of any of the several traditional risk factors (smoking, diabetes, hypertension, dyslipidemia, and a family history of CAD) appear to potentiate the risk of CAD when present in ACE DD carriers. The results here are preliminary owing to the limitation of the small number of patients. However, further investigations of these polymorphisms and their possible synergisms with traditional risk factors for CAD could help to ascertain better predictability for CAD susceptibility.
Footnotes
Acknowledgments
The authors would like to thank Dr Christian Winchell for his precious help in correcting this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
