Abstract
Polymorphisms in cytochrome P450 (CYP) 2C9 (CYP2C9) gene result in interindividual variability in warfarin dose requirement. There is a need for characterization of genotype frequency distribution in different populations for construction of customized dosing algorithms to enhance the efficacy and reduce the toxicity of warfarin therapy. This study was carried out in Pakistani population to evaluate the contribution of common CYP2C9 polymorphisms to warfarin therapy. A total of 550 stable patients taking warfarin were enrolled after medical history, physical examination, and laboratory investigations. Single blood sample was collected after informed consent. Genomic DNA was extracted, and genotype analysis for CYP2C9*2 and CYP2C9*3 polymorphisms was done by polymerase chain reaction-restriction fragment length polymorphism assay. A number of samples were also analyzed by direct DNA sequencing for validation of the results. Data were analyzed using SPSS version 20. Genotype frequency distribution of CYP2C9*2 and CYP2C9*3 was found to be different from other populations. Of these 2 polymorphisms, CYP2C9*2 did not demonstrate significant effect on warfarin dose requirement, whereas CYP2C9*3 did show significant effect (P value = .012). It is concluded that there is a need to study genotype frequency distribution and their effect on warfarin dose variability among different populations due to diversity in outcome.
Introduction
Warfarin has been the most commonly prescribed oral anticoagulant for prophylaxis and treatment of various thromboembolic conditions. It is administered as a racemic preparation of R- and S-enantiomers. Two enantiomers not only differ in potency but also in their half-lives and metabolism. R-warfarin is mainly metabolized by cytochrome P450 (CYP) 1A2, CYP3A4, CYP2C19, and CYP1A1, whereas S-warfarin mainly by CYP2C9. As S-warfarin is 3 to 5 times more potent than R-warfarin, so changes in blood levels of S-warfarin affect the anticoagulant response significantly. 1,2
The CYP2C9 enzyme is encoded by CYP2C9 gene that has been found to contain at least 57 CYP2C9 variant alleles defined by the human CYP Allele Nomenclature Committee (http://www.cypalleles.ki.se/cyp2c9.htm), but of these CYP2C9*3 and CYP2C9*2 have been well studied because of their significant effect on S-warfarin metabolism. The CYP2C9*2 allele results from single-nucleotide base substitution from C to T at codon 430 located in exon 3 (rs1799853). This single-nucleotide polymorphism (SNP) causes a change in amino acid residue from arginine to cysteine at codon position 144 (Arg144Cys) on the surface of CYP2C9 enzyme. The CYP2C9*3 allele results from substitution from A to C at codon 1075 located at exon 7 (rs1057910). This SNP leads to a change in amino acid residue from isoleucine to leucine at codon position 359 (Ile359Leu) inside the CYP2C9 enzyme. The most commonly present allele is CYP2C9*1, which is regarded as wild type and produces CYP2C9 enzyme with normal activity, whereas the presence of these 2 SNPs results in decrease in the activity of CYP2C9 enzyme that is more with CYP2C9*3 than with CYP2C9*2 allele. 3 –8
The presence of polymorphic alleles of CYP2C9 decreases the S-warfarin metabolic rate resulting in increased levels of S-warfarin which in turn leads to lower doses of warfarin required to produce the therapeutic response without any bleeding risk. 6,9,10
The reported studies have demonstrated that VKORC1 and CYP2C9 gene variants together with demographic factors account for 50% to 60% variance in warfarin dose requirement. 10 –13 Different dosing algorithms have been constructed from data including demographic factors along with the frequencies of common CYP2C9 and VKORC1 genotypes in their populations. The use of pharmacogenetic-based dosing algorithms has shown better outcome in the form of improved efficacy and less adverse effects such as bleeding. 10,14 –17
A comprehensive dosing model that is applicable regardless of ethnicity can be developed by characterizing the frequency of common polymorphisms in different ethnic populations. This will further help in drafting worldwide applicable clinical guidelines for warfarin prescription. Pakistan, although among the top 10 populous countries in the world, has been underrepresented in pharmacogenetic studies. The present study was carried out to determine the genotype frequencies of CYP2C9*2 and CYP2C9*3 polymorphisms in Pakistani population and to determine its effect on warfarin dose requirement.
Materials and Methods
Clinical Settings and Protocol
The clinical data collection and laboratory investigations were carried out at 2 major clinical setups in Pakistan providing anticoagulation therapy, Armed Forces Institute of Cardiology, Rawalpindi, and National Institute of Cardiovascular Diseases, Karachi. The analytical procedures were carried out at Centre for Research in Experimental and Applied Medicine, Army Medical College Rawalpindi, in collaboration with Institute of Biomedical and Genetic Engineering, Islamabad. The study protocol was approved by ethical committees of institutes.
Study Patients
The study was conducted in accordance with the current Good Clinical Practices 18 and the Declaration of Helsinki. 19 Study patients were adults of either gender between the age of 18 and 65 years who were receiving warfarin therapy. All participants were Pakistani citizens belonging to different regions of Pakistan to provide representation from all areas. Each patient was evaluated by detailed medical history, physical examination, and laboratory tests. Stable patients taking warfarin were recruited in the study after informed consent. A stable patient was defined as the one whose warfarin dose had been constant for at least 3 previous clinic visits over a minimum period of 3 months and had an international normalized ratio of the prothrombin time within the range of 1.5 to 3.5. 14,20 –22 The patients with hepatic and renal disease, any comorbid disease, or taking any concurrent medication or diet, which could have affected warfarin therapy, were excluded.
Genotyping
A total of 550 patients were recruited in the study. A 2 mL of blood sample was collected in EDTA-containing tube and stored at 4°C for genotyping. The genomic DNA from all of the samples was isolated mainly by standard organic method involving chloroform and phenol. 23 The protocol was slightly modified per requirement of the working laboratory. DNA isolation kit was used (QIAamp DNA Mini; Qiagen Inc, Valencia, CA, USA) to extract genomic DNA from some of the blood samples that were either less in quantity or somewhat clotted.
Polymerase Chain Reaction-Restriction Fragment Length Polymorphism Assay
Genotyping of the CYP2C9*2 and CYP2C9*3 polymorphisms was done by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) assays. The sequences of forward (5′-GGAGGATGGAAAACAGAGACTTA-3′) and reverse (5′-TGAGCTAACAACCAGGACTCAT-3′) primers used for CYP2C9*2 and forward (5′-GCTGTGGTGCACGACGTCCAGAGATGC-3′) and reverse (5′-ACACACACTGCCAGACACTAGG-3′) primers for CYP2C9*3 were obtained from a reported study. 24 The optimization of PCR with reproducible results was performed. The PCR was carried out for each sample in a final volume of 20 µL containing initial and final concentrations of the reagents as given in Table 1. The thermal stages with their temperatures and number of cycles are given in Table 2. The amplified DNA fragment harboring the CYP2C9*2 was digested with AVAII restriction enzyme, whereas for CYP2C9*3 restriction enzyme NsiI was used. The digestion was carried out by adding 1 µL of respective enzyme (10 U/µL) and 3µL 10× digestion buffer to 10 µL of the PCR product and adjusting the final volume to 30 µL with dH2O. The mixture was incubated at 37°C for 16 hours. The size of the RFLP bands was depicted with DNA size reference ladder.
Polymerase Chain Reaction (PCR) Reagents With Their Concentrations Used for PCR.
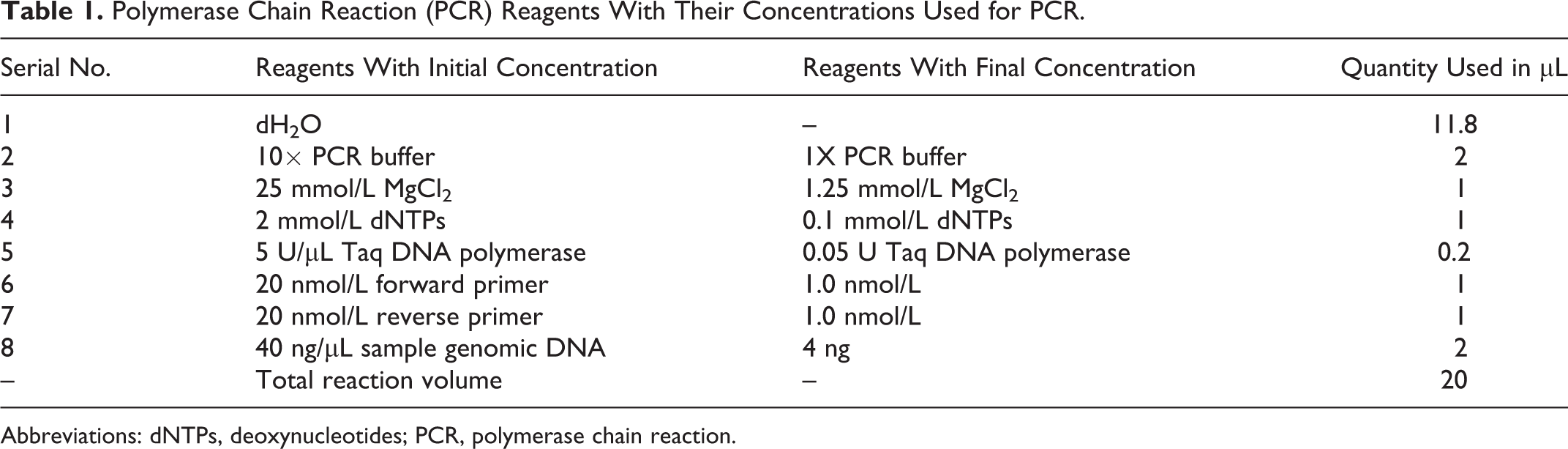
Abbreviations: dNTPs, deoxynucleotides; PCR, polymerase chain reaction.
Thermal Profile for the PCR for CYP2C9 Alleles.
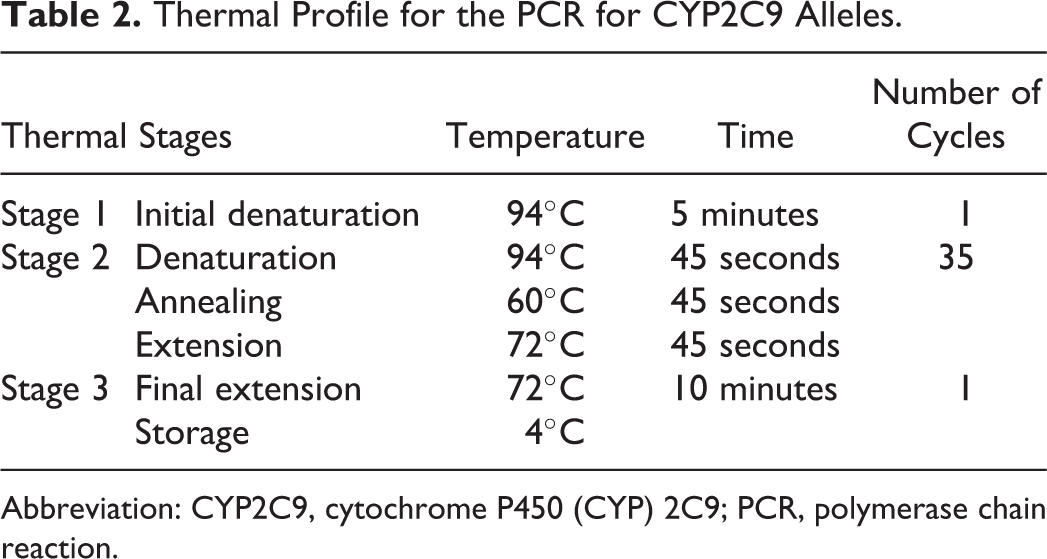
Abbreviation: CYP2C9, cytochrome P450 (CYP) 2C9; PCR, polymerase chain reaction.
DNA Sequencing
In order to validate the PCR-RFLP genotypes; direct sequencing of 100 samples was done through automated capillary sequencing method. Both SNPs were amplified using the same primers as mentioned in PCR. The sequencing reaction product was purified and loaded to ABI Genetic Analyzer 3130 (Applied Biosystem, Life Technologies, Carlsbad, California) for sequencing. The samples for DNA sequencing were selected randomly among all samples giving positive results for PCR-RFLP so that the results can be validated.
Data Analysis
Data were analyzed using SPSS version 20.0. The genotypes and allele frequencies were estimated from the observed numbers of each specific allele, and 95% confidence interval was calculated. The expected Hardy-Weinberg (H-W) frequencies for genotypes were calculated using H-W Equilibrium equation. Genetic data for deviation from H-W equilibrium were tested using χ2test. Analysis of variance (ANOVA) was applied for comparison of different CYP2C9 genotypes with warfarin dose requirement. A P value of <.05 was taken as statistically significant. Analysis of variance was followed by post hoc Tukey test for pairwise comparison if ANOVA gave a P value of <.05.
Results
The amplified PCR product was resolved at 396 base pair (bp) in case of CYP2C9*2 polymorphism. The PCR fragment containing C allele was digested into 2 fragments of 223 bp and 173 bp, whereas T allele was not digested and resolved at 396 bp. The amplified PCR product was resolved at 298 bp for CYP2C9*3. The PCR fragment containing A allele was digested into 2 fragments of 274 bp and 24 bp, whereas C allele was not digested and resolved at 298 bp. The results obtained on gel electrophoresis were confirmed on DNA sequencing electropherogram. The results obtained from both methods were in 100% concordance.
A total of 509 samples gave successful results for CYP2C9*2 and 527 for CYP2C9*3 polymorphism. The allele and genotype frequency distribution along with expected H-W frequencies for both SNPs are given in Table 3. A P value of more than .05 in both SNPs implies that observed frequencies did not deviate from H-W equilibrium. The effect of CYP2C9*2 and CYP2C9*3 polymorphisms on warfarin dose was determined, and the comparison has been summarized in Table 4. There was statistically significant effect of different CYP2C9*3 genotypes on warfarin dose requirement (P value <.05), whereas CYP2C9*2 polymorphism did not have any significant effect on warfarin dose (P value >.05).On the basis of pairwise comparison of different CYP2C9*3 genotypes (Table 5), it has been inferred that patients possessing homozygous polymorphic CYP2C9*3/*3 genotype required lesser warfarin dose when compared to homozygous wild-type CYP2C9*1/*1 genotype (AA), as the difference in dose requirement was statistically significant (P value <.05).
Allele and Genotype Frequency Distributions of CYP2C9*2 and CYP2C9*3 Polymorphisms.
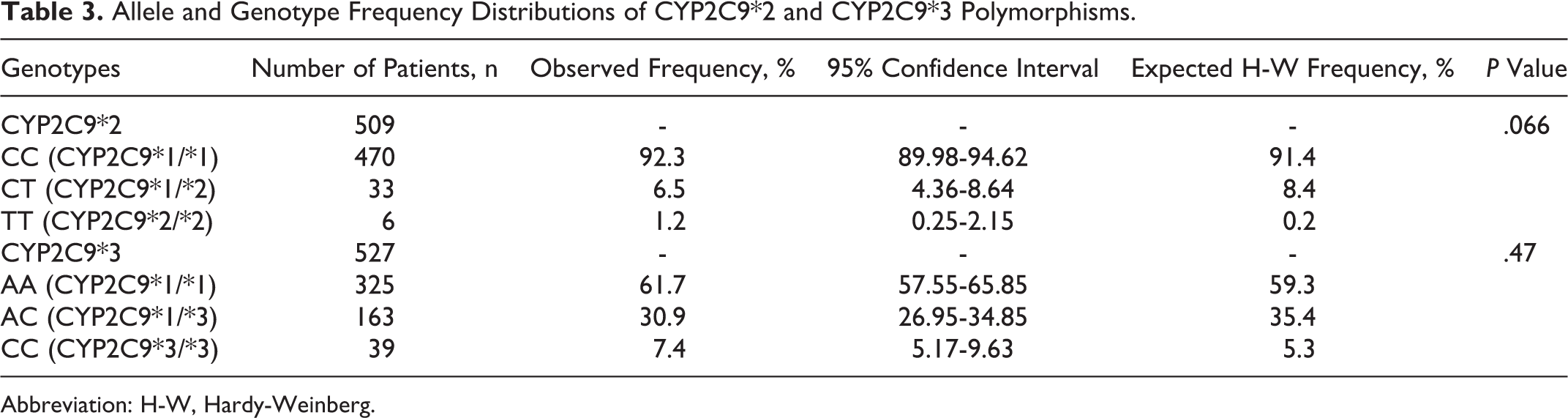
Abbreviation: H-W, Hardy-Weinberg.
Relationship of Warfarin Dose With CYP2C9 Genotypes.
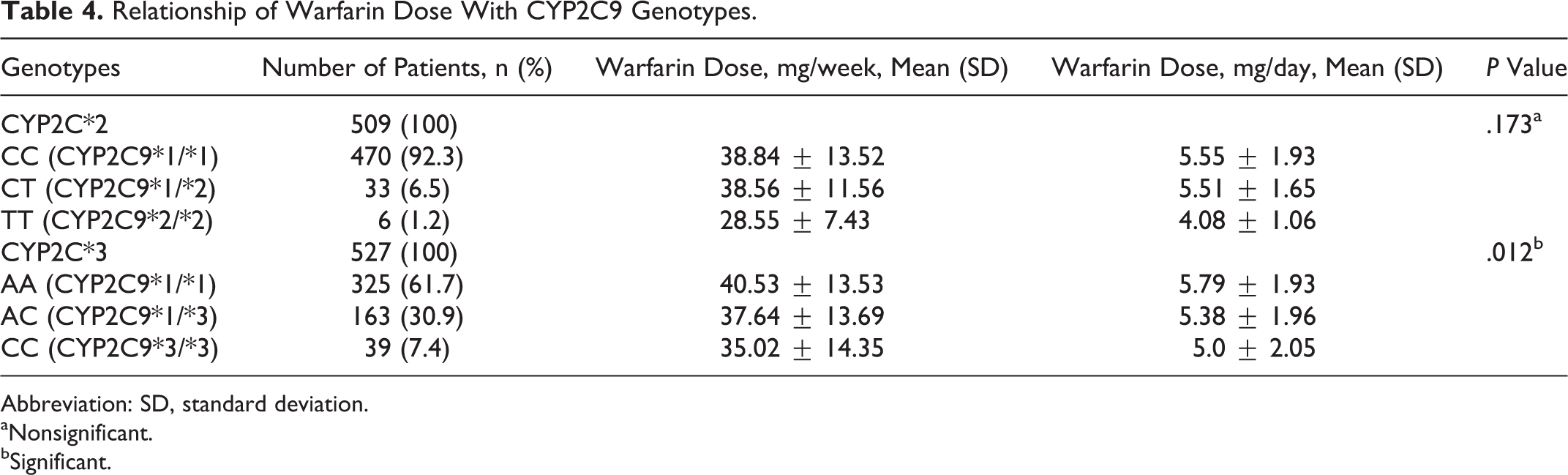
Abbreviation: SD, standard deviation.
aNonsignificant.
bSignificant.
Pairwise Comparison of Warfarin Dose Among CYP2C9*3 Genotypes.
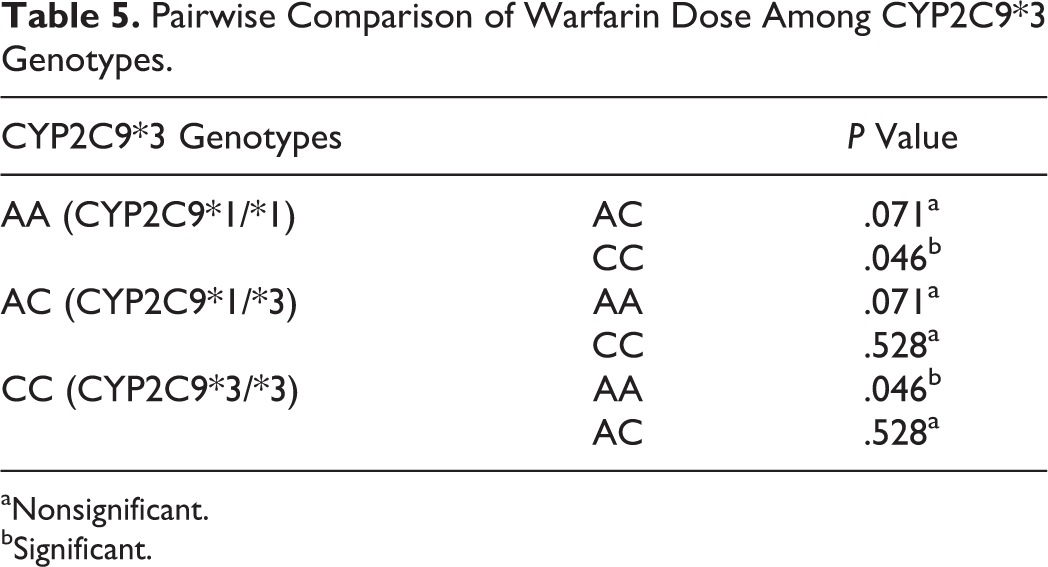
aNonsignificant.
bSignificant.
Discussion
This study was the first large-scale study in Pakistan to report the population-based frequencies of CYP2C9*2 and CYP2C9*3 genotypes and their impact on warfarin dose requirement. This study has pointed to the predominance of wild-type genotype that is more evident in CYP2C9*2 when compared to CYP2C9*3 in Pakistani population. The genotypes frequency distribution in Pakistani population was different from other reported studies conducted in different populations as shown in Table 6. It is evident that these 2 polymorphisms have diverse prevalence and effect in different populations. 24,36,38,43,50 –52 In view of these observations, every population has to have its own local frequency data. Even if we compare our results with those populations surrounding our geographical boundary, the diversity is obvious. It is also noticeable from studies conducted in different Asian countries that the word “Asian population” cannot be reflective of one homogenous population. This observation has been confirmed by some multiethnic studies conducted in Asia including patients belonging to different Asian countries. 38,53,54 So each region should be treated as an individual entity. A small part of human genome is playing its part in this genetic diversity, as the major part has been found to be similar among humans. Different evolutionary changes have played their part over the period of centuries that have led to population heterogeneity across the globe. 55 –57
Minor Allele Frequency (MAF) Distribution of CYP2C9*2 and CYP2C9*3 Genotypes in Different Populations.
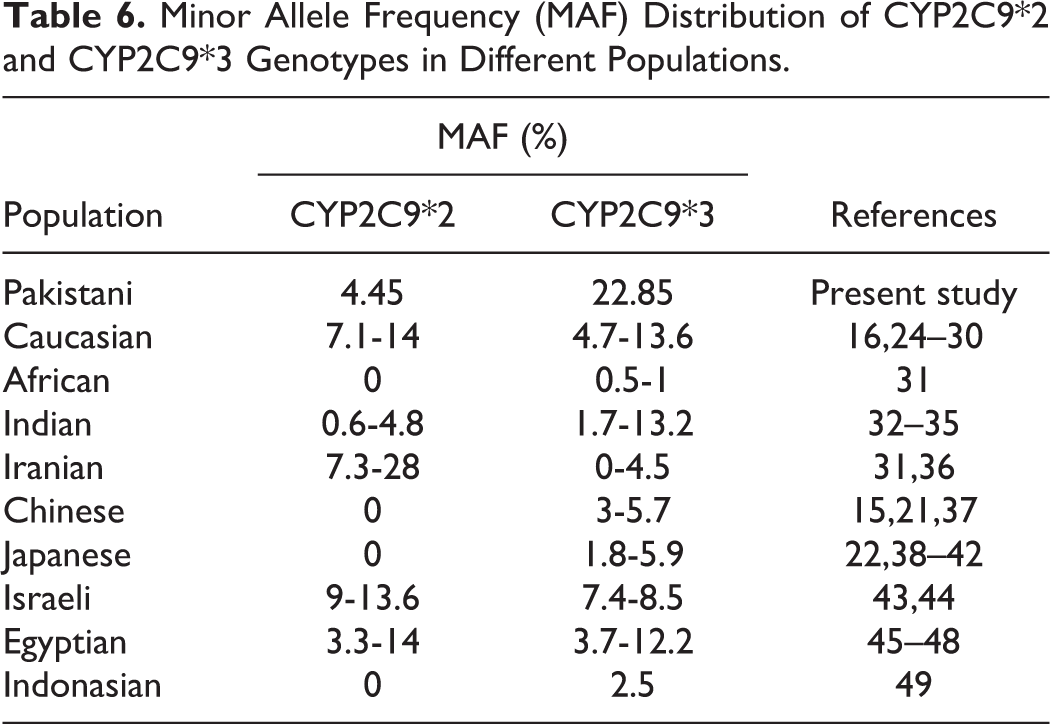
Although warfarin dose was less in patients with homozygous variant genotype for CYP2C9*2 polymorphism, there was no statistically significant difference in dose requirement among different genotypes. As regards the effect of CYP2C9*2 variant allele on warfarin dose requirement is concerned, the results are not based on the diversity of genotype frequency distribution but on the basis of the relative contribution of this variant allele on warfarin anticoagulant response. Some of the studies have demonstrated significant effect on warfarin dose that is reduced in patients possessing variant alleles. 16,25,45,58,59 Other studies have reported similar results as ours, where CYP2C9*2 variant alleles did not show any significant effect on warfarin dose requirement. 10,17,32,43,46,50,60,61 Even within the same population, different studies presented different results, some exhibiting effect, others not. 17,45,46,61,62 This implies that although CYP2C9*2 is a contributor to warfarin dose variance because it decreases CYP2C9 enzymatic activity to some extent, 50,51,63 but if this effect is not that strong, at times it is weighed down or modified by other genetic and nongenetic factors that act as more observable contributors.
In our study, warfarin dose requirement for patients with homozygous variant genotype for CYP2C9*3 (*3/*3) polymorphism was significantly lower than homozygous wild type (*1/*1). But the dose requirement of heterozygous variant genotypes was not significantly different from either homozygous wild type or homozygous variant genotypes. Apart from the fact that different genotype frequency distributions are reported in different populations, the effect of CYP2C9*3 variant allele on warfarin dose was found to be significant in almost all the studies with the exception of few. 39,64 Same significant effect was seen in patients in the present study as well. The patients with variant allele achieved therapeutic anticoagulant response with significantly less dose of warfarin when compared to those having wild-type genotype. This implies that CYP2C9*3 variant allele is an effectual contributor to warfarin dose variability by decreasing the CYP2C9 enzymatic activity leading to reduced S-warfarin clearance. This aspect can be further elucidated in the future studies by analyzing S- and R-warfarin levels in plasma and their relationship with different CYP2C9 genotypes.
Conclusion
The frequency distributions of CYP2C9*2 and CYP2C9*3 genotypes in Pakistani population were different from those reported in other world populations. CYP2C9*2 polymorphism was not found to be affecting warfarin dose, whereas presence of CYP2C9*3 polymorphic alleles reduced the dose requirement. This implies that CYP2C9*3 variant allele is an effectual contributor to warfarin dose variability.
Footnotes
Acknowledgment
The authors are grateful to Higher Education Commission (HEC) of Pakistan for funding this project. The authors are thankful to National University of Sciences and Technology (NUST) for facilitating to carry out this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Project was funded by Higher Education Commission (HEC) Pakistan.
