Abstract
Objective:
Heparin-induced thrombocytopenia (HIT) occurs in up to 5% of patients exposed to unfractionated heparin for 5 or more days. Direct thrombin inhibitors (DTIs) are currently the only Food and Drug Administration (FDA)-approved agents for the treatment of HIT. The purpose of this study is to determine whether fondaparinux is an appropriate first-line alternative anticoagulant in patients with suspected or confirmed HIT.
Methods:
A retrospective study was conducted by identifying all patients who received a DTI or fondaparinux during a 5 year period, August 2009-August 2014. Patients were included if they had a HIT panel/serotonin-release assay analysis (regardless of the result) and were initiated on a DTI or fondaparinux for alternative anticoagulation. The primary outcome was new, recurrent, or progressive thromboembolic event. Secondary outcomes included bleeding events, platelet count recovery, and hospital stay.
Results:
A total of 1022 patients were evaluated, and 47 patients met the inclusion criteria. Twelve patients were HIT positive and 35 were HIT negative. Seven (14.9%) of the 47 patients experienced a new thrombosis, none of whom were on fondaparinux only (FONDA). There were 4 new minor bleeds, with 1 bleed as a result of being on fondaparinux. FONDA treatment resulted in a slightly shorter median duration of hospital stay compared to the DTI-only group and the DTI followed by fondaparinux group. There is a potential for cost savings with fondaparinux due to the ease of administration and availability to be given in the outpatient setting.
Conclusion:
In this small retrospective review, fondaparinux appeared similarly efficacious and safe compared to DTIs for the treatment of suspected HIT.
Introduction
Heparin-induced thrombocytopenia (HIT) occurs in up to 5% of patients who are exposed to unfractionated heparin for 5 or more days. Due to early recognition and discontinuation of heparin therapy, the mortality rate in response to HIT has decreased from 20% to 2%. 1 Heparin-induced thrombocytopenia can be diagnosed via the enzyme-linked immunosorbent assay (ELISA) immunoassay or the serotonin-release assay (SRA) functional assay. The SRA functional assay remains the gold standard diagnostic test with 95% sensitivity and specificity. However, the SRA functional assay is usually reserved for definite confirmation of antibody presence after receiving a positive result on an ELISA immunoassay test (HIT panel). 2,3
The use of alternative anticoagulants in patients with suspected HIT remains an unanswered question. The available agents for use in patients with suspected HIT are the direct thrombin inhibitors (DTIs): argatroban, bivalrudin reserved for patient’s undergoing percutaneous coronary intervention), and previously lepirudin (discontinued in May 2012). 4 Fondaparinux, a factor Xa inhibitor, has been used for the treatment of patients with HIT but has not gained Food and Drug Administration (FDA) approval for this indication. Once patient’s platelet counts recover to >150 000 cell/mm3, they can then be switched to warfarin therapy if continued anticoagulation is warranted. 5 Although argatroban is the only FDA-approved agent available for use in the general patient population, it must be given as a continuous intravenous infusion. This leads to a longer length of hospital stay and increased medication and administration costs. On the other hand, fondaparinux can be administered subcutaneously and can lead to quicker transition of care to the outpatient setting in an outpatient setting. There are also observed differences in half-life, recommended renal dosage adjustments, and effect on international normalized ratio (INR) between the DTIs and fondaparinux (Table 1).
DTI Versus Fondaparinux.
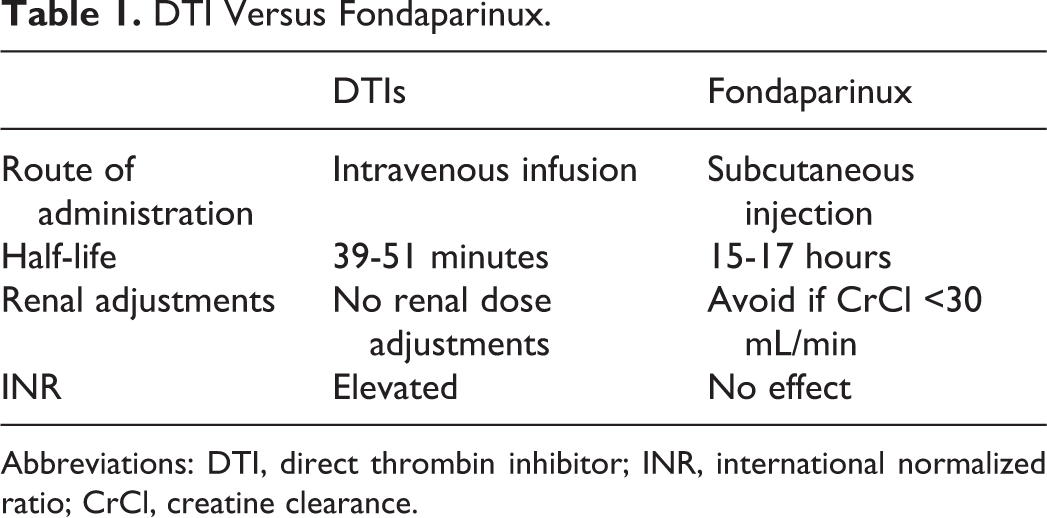
Abbreviations: DTI, direct thrombin inhibitor; INR, international normalized ratio; CrCl, creatine clearance.
It has been shown that there is a low likelihood of cross-reactivity between heparin and fondaparinux, which makes fondaparinux a possible HIT treatment alternative once heparin is discontinued. 1,6 Fondaparinux also offers a cost saving when compared to argatroban due to lower drug costs, decreased administration costs, and shorter duration of hospital stay. 3 Therefore, the question arises as to whether or not fondaparinux should be used as an alternative agent to argatroban for the treatment of HIT.
Various case reports and case studies concluded that fondaparinux may be an effective alternative anticoagulation agent for the treatment of HIT. 7,8 When compared to FDA-approved agents, patients in these reports who received fondaparinux did not experience new, recurrent, or progressive thromboembolic events and had a platelet count recovery that was comparable to the other agents. However, these reports have a limited sample size and did not address further end points such as bleeding risk, duration of hospital stay, and cost of therapy. This study thus aims to address the efficacy and safety of fondaparinux as an alternative first line agent for the treatment of suspected or confirmed HIT as compared to argatroban and lepirudin.
Methods
Study Population
A retrospective chart review was conducted at VAPHS to determine whether fondaparinux is an appropriate alternative to DTIs for the treatment of HIT. Patients were identified by generating lists for those with inpatient orders for a DTI or fondaparinux between August 2009 and August 2014. This list was cross-referenced with laboratory orders for an HIT panel and/or SRA, listed heparin allergy, and International Classification of Diseases, Ninth Revision codes for HIT (289.84). Patients met inclusion criteria if they had a HIT panel/SRA sent for analysis (regardless of whether the antibody test results were positive or negative) and if they received fondaparinux, a DTI, or both as alternative anticoagulation. The analysis was completed on all patients who received an alternative anticoagulant once heparin was discontinued. Patients were excluded if a HIT panel or SRA results were unavailable or if they were not started on an alternative anticoagulant.
Data Collection
Data collection was performed using the Computerized Patient Record System (CPRS), the Veteran Affairs electronic medical record. Data were collected on patient demographics, history of thromboembolic event (in the past 5 years), history of bleeding event, the heparin agent and dose (prophylactic or treatment) prior to HIT suspicion, the specific alternative anticoagulant and dose (prophylactic or treatment) prescribed at the time of HIT suspicion, the patient’s 4T score at time of HIT suspicion, if patients had an active thrombosis, platelet counts, days on heparin therapy prior to discontinuation, and duration of hospital stay.
Definitions
An ELISA assay for the diagnosis of HIT incorporates the use of coated wells with PF4 complexed with heparin. Prediluted plasma containing heparin–PF4 antibodies will bind to the wells if present. A second conjugated affinity-purified antihuman immunoglobulin (Ig) G, M, and A will bind to the complex, and the intensity of observed coloration is directly proportional to the antibodies present in the test sample.
History of a bleeding event was defined as a gastrointestinal bleed, intracranial hemorrhage, or hematuria within the past 5 years. A patient at high risk of bleeding was defined as a consistent hemoglobin level <9 mg/dL.
Baseline platelet count was defined as the platelet count immediately prior to initiation of any form of heparin. Platelet count recovery was defined as a return to baseline platelet count or higher.
When assessing for efficacy and evaluating the primary end point, a new, recurrent, or progression of thromboembolic event was confirmed with vascular doppler ultrasound or chest computed tomography.
A minor bleeding event was defined as being hemodynamically stable, but required hospitalization, a physician visit, or evaluation by a provider. A major bleeding event was defined as being hemodynamically unstable, requiring a transfusion of ≥2 units of blood, retroperitoneal bleed, intracranial hemorrhage, or death.
A successful bridge to warfarin therapy was defined as patients reaching a therapeutic INR and discontinuation of the parenteral anticoagulant.
Outcomes
The primary endpoint was to determine whether patients had a new, recurrent, or progressive thromboembolic event while on fondaparinux. Secondary endpoints evaluated for a recovery in platelet counts, minor and major bleeding events, if patients were successfully transitioned to warfarin therapy, and duration of hospital stay.
The data in this article were derived from a quality assessment and quality improvement (QA/QI) activity as determined per VHA Handbook 1058.05. 9 This handbook identifies QA/QI activities as “Operations Not Constituting Research,” and the activity was approved as such.
Results
A total of 1022 patients were evaluated, with 47 patients meeting inclusion criteria after analysis. Reasons for exclusion included patients who did not have a HIT panel sent, received fondaparinux for a non-HIT indication, were not initiated on alternative anticoagulation, or had a nonthrombocytopenic reaction to heparin. Of the 47 included patients, 12 patients were HIT positive and 35 patients were HIT negative. Patient demographics, anticoagulant dosing, and testing descriptions are shown in Table 2.
Patient Demographics.
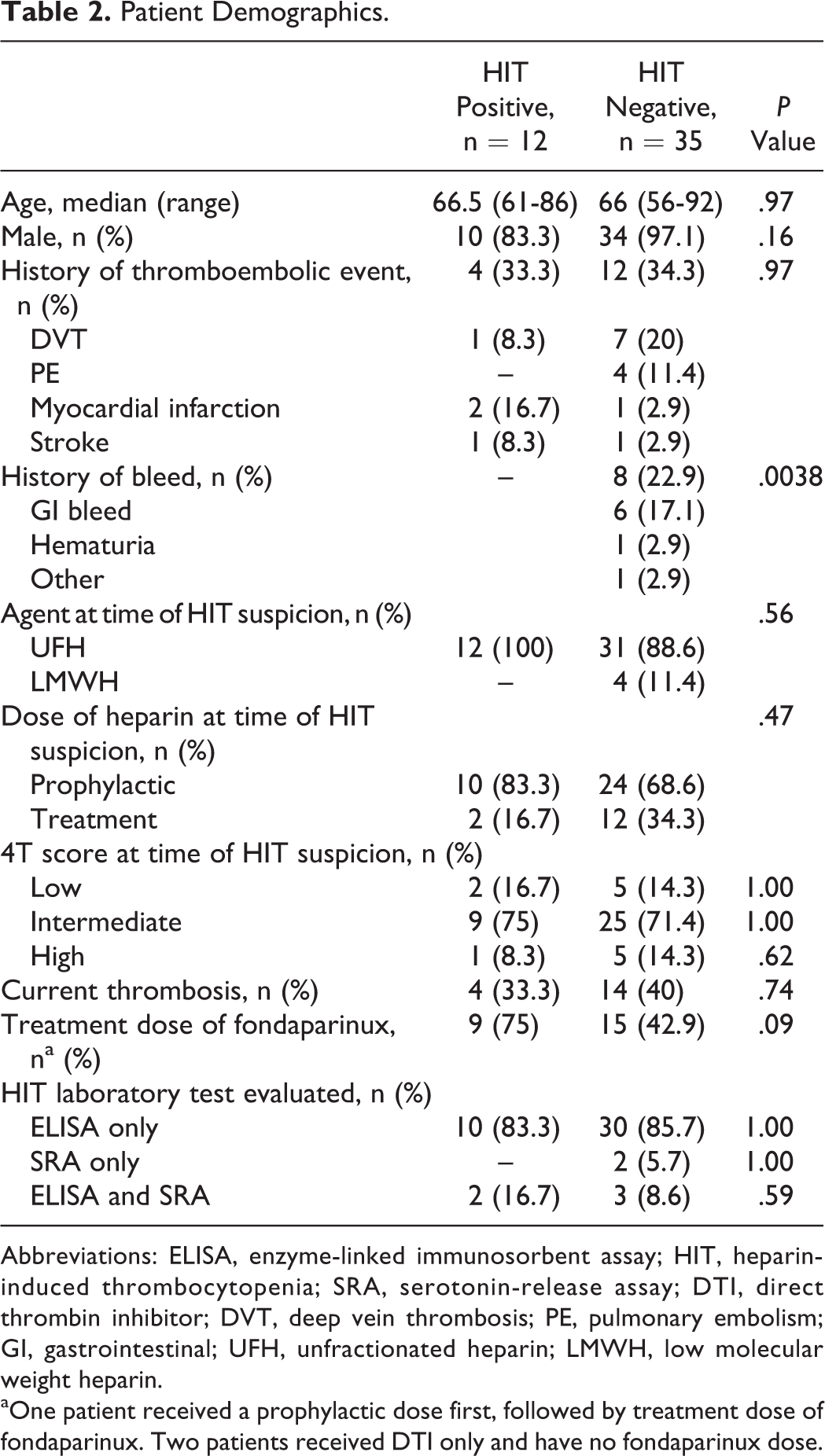
Abbreviations: ELISA, enzyme-linked immunosorbent assay; HIT, heparin-induced thrombocytopenia; SRA, serotonin-release assay; DTI, direct thrombin inhibitor; DVT, deep vein thrombosis; PE, pulmonary embolism; GI, gastrointestinal; UFH, unfractionated heparin; LMWH, low molecular weight heparin.
aOne patient received a prophylactic dose first, followed by treatment dose of fondaparinux. Two patients received DTI only and have no fondaparinux dose.
Of those patients who were found to be HIT positive, the exact ELISA immunoassay quantity is expressed in Table 3. If no ELISA result was available, it was determined that they had an SRA completed (Table 3).
Test Results for HIT-Positive Patients.
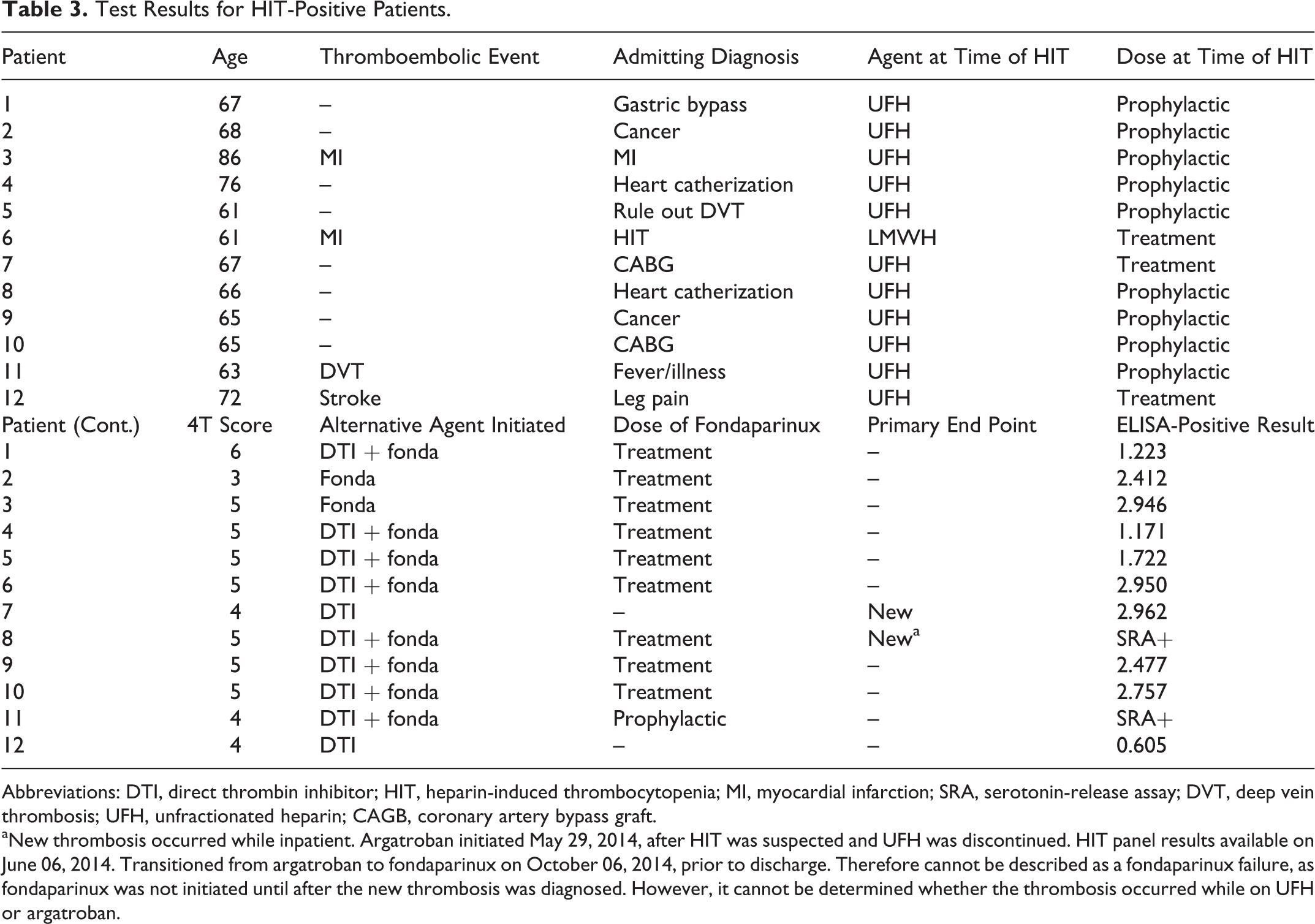
Abbreviations: DTI, direct thrombin inhibitor; HIT, heparin-induced thrombocytopenia; MI, myocardial infarction; SRA, serotonin-release assay; DVT, deep vein thrombosis; UFH, unfractionated heparin; CAGB, coronary artery bypass graft.
aNew thrombosis occurred while inpatient. Argatroban initiated May 29, 2014, after HIT was suspected and UFH was discontinued. HIT panel results available on June 06, 2014. Transitioned from argatroban to fondaparinux on October 06, 2014, prior to discharge. Therefore cannot be described as a fondaparinux failure, as fondaparinux was not initiated until after the new thrombosis was diagnosed. However, it cannot be determined whether the thrombosis occurred while on UFH or argatroban.
New/Recurrent/Progressive Thromboembolic Events
Of the HIT-positive patients (N = 12), 2 were treated with DTI only (DTI), 2 with fondaparinux only (FONDA), and 8 received a DTI initially then were transitioned to fondaparinux (DTI + FONDA) prior to discharge. For the primary outcome, there were no recurrent or progressive thromboembolic events in any of the 12 HIT-positive patients, including the 2 patients who received FONDA. Two (16.7%) patients experienced a new thrombosis, 1 patient on argatroban and 1 patient on a DTI followed by fondaparinux. No new, recurrent or progressive thromboembolic events were noted in HIT positive while on fonda alone.
New Bleeding Events
When evaluating the total population (N = 47) for new bleeding events, there were 4 new minor bleeds. Those with minor bleeds were hemodynamically stable but required a hospital or physician visit. Bleeding events occurred in 3 (16.7%) patients on a DTI and 1 (10%) patient on FONDA. No patients who received a DTI + FONDA experienced a new bleeding event. These results were not found to be statistically significant (P > .99; Table 4).
Bleeding Events.

Abbreviations: DTI, direct thrombin inhibitor; FONDA, fondaparinux.
aAll bleeding events were minor bleeds (ie, hemodynamically stable but required hospital/provider visit with/without intervention).
Days to Platelet Count Recovery
In patients who were HIT positive (N = 12), there was no difference in the median days to platelet count recovery (FONDA = 10.5 days, DTI + FONDA = 9 days, P = 1.0). The median number of days for the DTI group was unable to be calculated, as 1 patient was lost to follow-up and the other patient never had a decrease in platelet count (Table 5).
Platelet Count Recovery.

Abbreviations: DTI, direct thrombin inhibitor; FONDA, fondaparinux.
Days on Warfarin Bridge
Eight HIT-positive patients were bridged to warfarin therapy, with 6 patients being bridged from FONDA. There were no patients in the DTI + FONDA group being bridged to warfarin. The median days on bridge therapy were similar between the 2 groups (DTI: 6, FONDA: 8.5; P = .64). Two patients who received FONDA were excluded from the median number of days because they discontinued warfarin therapy prior to reaching a therapeutic INR (Table 6).
Warfarin Bridging.
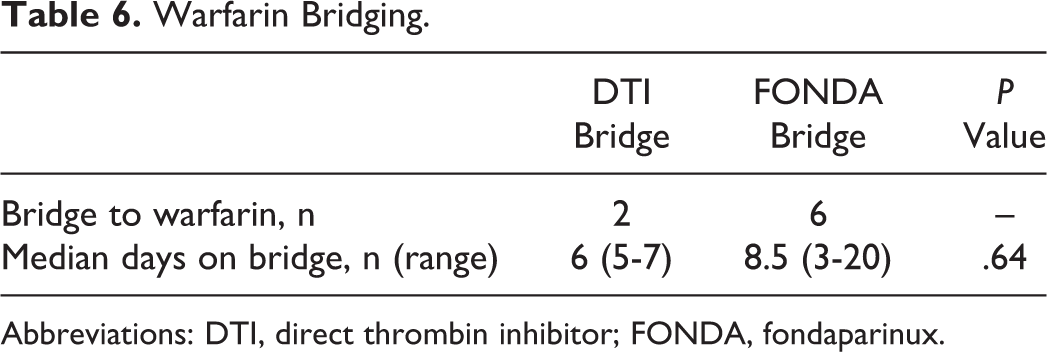
Abbreviations: DTI, direct thrombin inhibitor; FONDA, fondaparinux.
Duration of Hospital Stay
FONDA treatment resulted in a slightly shorter median duration of hospital stay at 17.5 days (range 5-40 days) compared to 19.5 days (range 5-40 days) in the DTI group and 18 days (range 2-65 days) in the DTI + FONDA group. However, fondaparinux can be administered in the outpatient setting, which also leads to improved quality of life and patient satisfaction (Figure 1).
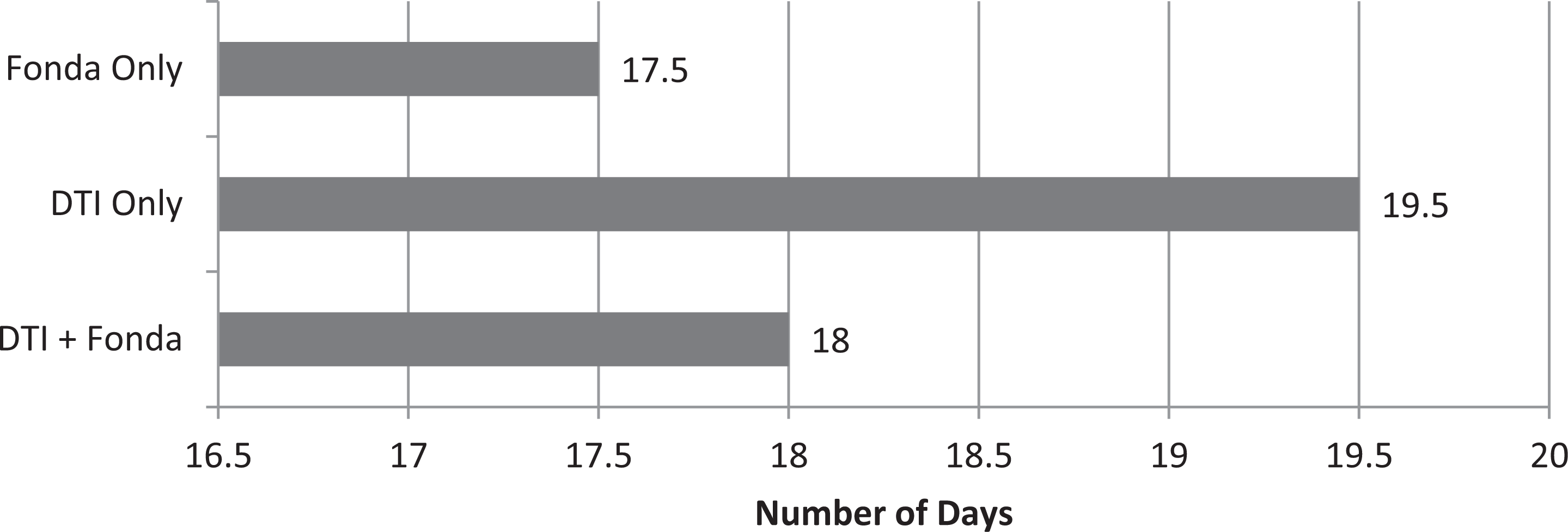
Median duration of hospital stay.
Cost Avoidance
There is the potential for cost savings when using fondaparinux for bridge therapy (compared to argatroban) due to its ease of administration, lower cost of acquisition, and the availability to be administered in the outpatient setting, leading to decreased length of hospital stay. Due to lower medication costs, administration costs, and overall health-care costs, there is a potential to save approximately $416.17 per day when fondaparinux is used (Table 7).
Drug Acquisition Cost Expressed in U.S Dollars.a

aCosts per day are based on a 75-kg patient; treatment dose fondaparinux 7.5 mg daily and argatroban 2 μg/kg/min.
Discussion
There is limited data evaluating the use of fondaparinux for the treatment of suspected or confirmed HIT. Currently, only argatroban and bivalirudin are available for use in patients with suspected/diagnosed HIT. However, due to administration restrictions and the cost of therapy, it is important to assess other possible treatment options. This retrospective review adds to the limited evidence looking at fondaparinux as a possible alternative treatment option for HIT. The results of this review are comparative to the results that were reported in various other case reports and case studies. 10 –12
Warkentin and colleagues completed a retrospective study with 16 patients who were SRA positive for HIT and presented with clinical features according to the 4T score; 56% of patients had an active HIT-associated thrombosis (proven by a venous Doppler scan) at the time of initiation of alternative treatment. All patients received fondaparinux (treatment dose) for a total of 30 days. At the 30-day end point, no patient developed a new, recurrent, or progressive thrombosis, and only 1 patient developed a major bleed (defined as a calf hematoma). All patients included within the study experienced platelet count recovery during fondaparinux treatment (median of 4 days). All 13 of the patients who required continued anticoagulation with warfarin were successfully transitioned to warfarin, with a median of 5 days to therapeutic INR. This study concluded that fondaparinux shows promise for the treatment of patients with SRA-positive acute HIT. 13 The study by Warkentin is comparable to this review when looking at the sample size and outcomes. In both of these studies, there were no new, recurrent, or progressive thromboembolic events, all patients had a recovery in platelet counts, and all patients were successfully transitioned to warfarin from fondaparinux.
A second study completed by Al-Rossaies and colleagues evaluated 12 patients who had confirmed HIT. Five patients were put on fondaparinux and 7 patients received lepirudin. The HIT-related thromboembolic events were equal between the 2 groups at the initiation of the study, with each group having 2 patients with active thrombi. No patients in either group experienced a recurrent thromboembolic event, and time to platelet count recovery was similar between the groups (median of 4 days). However, 3 patients in the lepirudin group required platelet transfusions for resolution of thrombocytopenia. One patient on fondaparinux experienced a major bleed, whereas 2 in the lepirudin group experienced a major bleed. This study concluded that fondaparinux is a potential alternative treatment for HIT and is comparable to lepirudin. 14 When comparing the study by Al-Rossaies et al to this review, the sample sizes were similar and the treatment arms compared fondaparinux to a DTI. When looking at outcomes between the study and this review, no patients experienced a recurrent thromboembolic event, and platelet count recovery was similar in the 2 studies.
A very recent study by Kang and colleagues matched 133 patients to 60 controls using propensity score matching based on age, gender, 4T score, creatinine, and comorbidity index. They then compared treatment with fondaparinux, argatroban, and danaparoid. The study outcomes were thrombosis or thrombosis-related death and bleeding. In those patients who received fondaparinux, 60% of the patients were on prophylactic doses. Thrombosis occurred in 22 (21.4%) patients in the matched group and 13 (21.4%) patients in the control group. When looking at bleeding, 28 (21.1%) patients in the matched group on fondaparinux had a major bleeding event compared to 12 (20%) patients in the control group. This study concluded that fondaparinux showed similar effectiveness and safety as argatroban and danaparoid in patients with suspected HIT. The secondary conclusion was that prophylactic fondaparinux doses seem to be effective if no indication for full anticoagulation exists. 8
In comparison to currently available studies, this small retrospective review showed that fondaparinux is equally safe and efficacious in the veteran population when compared to DTIs for the treatment of HIT. No patient in either group experienced a new, recurrent, or progressive thrombosis while on FONDA. Fondaparinux led to <9% bleeding rate of minor and major bleeding events and showed no delay in platelet count recovery. Of those patients who were bridged to warfarin for continued anticoagulation, fondaparinux showed a comparable duration of time to therapeutic INR when compared to argatroban. However, because fondaparinux is administered in the outpatient setting, follow-up time is extended which could account for the increased duration of days on bridge therapy noticed in the fondaparinux group. Those patients who received fondaparinux had a decreased length of hospital stay, and there is high potential for cost avoidance. Given these results, future studies can further explore the use of fondaparinux for HIT in an expanded population and can broaden the sample size to assess the safety and effectiveness of fondaparinux for HIT.
As with all studies, there are a few limitations that are important to address. This was a small, retrospective chart review that assessed 12 HIT-positive patients. Therefore, the results were limited by the small sample size but are still comparable to previous studies. It was difficult to identify patients for inclusion in the analysis due to the way lists are generated from the VA system. The process required cross-referencing between lists to exclude duplicate patients. It was also difficult to identify patients who had an HIT panel sent for analysis as the laboratory tests are ordered using various names. Finally, the documentation was limited by the individual providers and may have the potential to lead to bias.
In conclusion, fondaparinux is an alternative for the treatment of suspected or confirmed HIT unless the patient has a contraindication to fondaparinux: creatinine clearance ≤30 mL/min, the patient is at high risk of bleeding, the patient is critically ill, weight <50 kg, active bleeding, or infective endocarditis. Further randomized, controlled trials evaluating the use of fondaparinux in HIT are warranted.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
