Abstract
Empiric management in suspected heparin-induced thrombocytopenia (HIT) is challenging due to imperfect prediction models, latency while awaiting test results and risks of empiric therapies. When there is high clinical suspicion for HIT, cessation of heparin and empiric non-heparin anticoagulation with FDA-approved argatroban is recommended. Alternatively off-label fondaparinux or watchful waiting have been utilized in clinical practice. Outcomes of patients empirically managed for HIT have not been compared directly in clinical trials and patients that ultimately do not have HIT are often overlooked. Clinicians need studies investigating empiric management to guide decision making in suspected HIT. In this study, adverse events (AE) were categorized and compared in patients being evaluated for HIT while undergoing empiric management by non-heparin anticoagulation with argatroban or fondaparinux, both at therapeutic or reduced doses, or watchful waiting with or without heparin. AE were defined as new thrombosis confirmed on imaging or new bleeding event after HIT was first suspected. A retrospective chart review of 312 patients tested for HIT at an academic hospital was conducted. 170 patients met inclusion criteria. Patients were excluded if the 4Ts score was < 4. The 4Ts score is a pretest probability for HIT based on thrombocytopenia degree, timing, alternative causes and presence of thrombosis. Included patients were divided according to management groups and compared with logistic regression analysis. Bleeding risk significantly differed between management groups (p = 0.002). Despite adjustment for bleeding risk, fondaparinux was associated with increased AE, (p = 0.03, OR = 5.81), while argatroban was not. There was no difference in AE based on time to initiation of empiric treatment and no advantage to reduced dosing with either anticoagulant. These findings challenge assumptions surrounding empiric HIT management.
Keywords
Introduction
Heparin-induced thrombocytopenia (HIT) is a rare but potentially fatal prothrombotic drug reaction that occurs in 1-3% of patients exposed to heparin. 1 Historically, HIT has been under-diagnosed and recognized late in the disease process, leading to high thrombosis-related morbidity and mortality. 1 –3 However, advances in diagnostic testing paired with increased disease awareness has caused a paradigm shift in clinical practice, such that now HIT is being widely suspected and empirically treated at rates far exceeding the true disease incidence. 2,3 Early treatment can reduce the risk of thrombosis by 50-70%. 4 Meanwhile, empiric HIT treatment exposes patients to expensive alternative anticoagulants associated with bleeding risk and that lack reversal options. 2,3 Additionally, simply withholding heparin may put patients at risk for thrombosis. 2
The diagnosis of HIT is a challenging multi-step process that often requires days and is convoluted by real-world barriers. 1,2 The 4Ts score can be difficult to calculate due to incomplete clinical information and because the differential diagnosis for thrombocytopenia is broad. 2 Delays obtaining timely test results are common, as many clinical laboratories batch samples, are limited to processing on business days, and send samples to outside facilities. 2 Further, patients with critical illness or multiple comorbidities are more likely to have a false positive heparin-platelet factor 4 (H-PF4) assay, yet are also often at greater risk of an adverse event from empiric HIT treatment. 3,5 Due to these barriers, clinicians frequently face challenging initial management decisions while awaiting definitive laboratory results. Current expert opinion recommends empiric HIT treatment until results of a confirmatory functional assay, such as the serotonin release assay (SRA), are known in those with an intermediate to high clinical suspicion of HIT and a positive anti-H-PF4 screening immunoassay. 2 Empiric treatment includes cessation of all heparin products and initiation of non-heparin anticoagulation, specifically direct thrombin inhibitor argatroban. 5 Fondaparinux, a subcutaneously delivered pentasaccharide factor Xa inhibitor, is commonly used as off-label treatment. 3 Additionally, some clinicians discontinue heparin but opt for “watchful waiting” (WW) or reduced-intensity dose (RID) anticoagulation instead of therapeutic dose (TD) when perceived bleeding risk is high. 3
The objective of this study was to identify, categorize and explore the correlation between clinically significant bleeding episodes and new or progressive thrombotic events with various regiments of empiric management for suspected HIT, while awaiting the results of in-house H-PF4 immunoassay testing and send-out SRA confirmatory testing at an academic center.
Methods
A retrospective chart review was performed of all patients who underwent laboratory testing for HIT at a large academic medical center from April 2017 to April 2019. Electronic medical records were screened and a single reviewer collected data in a pre-designed excel worksheet. An attending physician reviewed cases in which discrepant H-PF4 and SRA results existed to determine HIT diagnoses based on the documentation and clinical determination of consultants at the time of diagnosis. Patients were excluded from the study if H-PF4 or SRA results were missing or indeterminate. Patients were excluded if a prior diagnosis of HIT had been made or if the calculated 4Ts score was < 4.
Regarding laboratory testing, patient blood samples were collected according to the manufacturer’s instructions. H-PF4 antibody testing was performed in-house with Immucor Lifecodes H-PF4 Enhanced kit from April 2017 to January 2019 and an IgG-specific H-PF4 enzyme-linked immunosorbent assay was utilized January 2019 to April 2019. Results of this assay were considered positive if the optical density (OD) was
Cases were grouped and categorized according to the regimens of empiric HIT management: argatroban TD (
Excel and Statistical Package for the Social Sciences (SPSS) were used for data analysis. Empiric treatment groups were analyzed with univariate analysis to compare baseline patient characteristics. Logistic regression analysis was used to compare AE that occurred in treatment groups. P-values < 0.05 were considered significant.
Results
Data from 312 patients for whom HIT testing was sent between April 2017 and April 2019 were analyzed. Of these, 170 patients met inclusion criteria (Figure 1). 62.7% of the patients were male and 37.1% were female. The average age of patients was 61.1 ± 16.1 years (Table 1). 11.8% of patients were ultimately diagnosed with HIT. Table 1 contains a comparison of baseline characteristics categorized by treatment group.

Inclusion and exclusion flow chart.
Baseline Characteristics Among Treatment Groups.

Abbreviations: Therapeutic dose (TD); reduced intensity dose (RID); watchful waiting (WW); standard deviation (SD); IMPROVE bleed risk assessment model (RAM); International Normalized Ratio (INR); heparin-platelet factor 4 (H-PF4); serotonin release assay (SRA). Asterisk (*) mark statistically significant p-values (<0.05).
τ5 patients had borderline positive SRA tests, classified as positive (2 patients were diagnosed with heparin-induced thrombocytopenia (HIT), 3 were not diagnosed with HIT).
There were no significant differences among treatment groups with respect to age, sex, creatinine, prothrombin time (PT)/international normalized ratio (INR), platelet count, and time to H-PF4 and SRA test results (Table 1). Although not a statistically-significant finding (p = 0.06), the highest average 4Ts scores were observed in the TD argatroban group [5.4 ± 1.2] and the lowest average 4Ts scores were in the WW on heparin group [4.5 ± 0.7]. The IMPROVE bleeding RAM score was significantly different between groups (p = 0.002). The highest average score [9.4 ± 4.7] was seen in WW off heparin group. Higher RAM scores on average were observed in RID compared with TD for both argatroban and fondaparinux.
AE occurred in 7.1% of patients (Table 2). AE were more common among those with confirmed HIT, occurring in 25.0%, compared to 4.67% of adverse events in those ultimately not diagnosed with HIT. 50% of all AE were thrombotic and 50% were bleeding. 50% of all AE occurred in patients treated with fondaparinux. Logistic regression analysis controlled for age, sex, 4Ts score and RAM bleeding risk revealed fondaparinux was associated with more overall AE than WW [p = 0.03; OR = 5.81, 95% CI (1.19, 28.43)] (Table 3). However, argatroban was not associated with more overall AE compared to WW [p = 0.48; OR = 1.88, 95% CI (0.33, 10.54)]. Intensity of anticoagulation and time to treatment initiation also were not associated with a significant difference in overall thrombotic and bleeding events. Qualitative data regarding AE is described in Table 4. One patient had both a thrombotic and bleeding AE.
Adverse Events Among Treatment Groups.

Abbreviations: Therapeutic dose (TD); reduced intensity dose (RID); watchful waiting (WW).
Logistic Regression Analysis Among Treatment Groups.

Abbreviations: Adverse events (AE); therapeutic dose (TD); reduced intensity dose (RID); confidence interval (CI). Note: Above logistic regression models control for patient age, sex, IMPROVE bleed risk assessment model (RAM) scores and 4Ts score. Asterisk (*) mark statistically significant p-values (<0.05).
Qualitative Adverse Event Data.
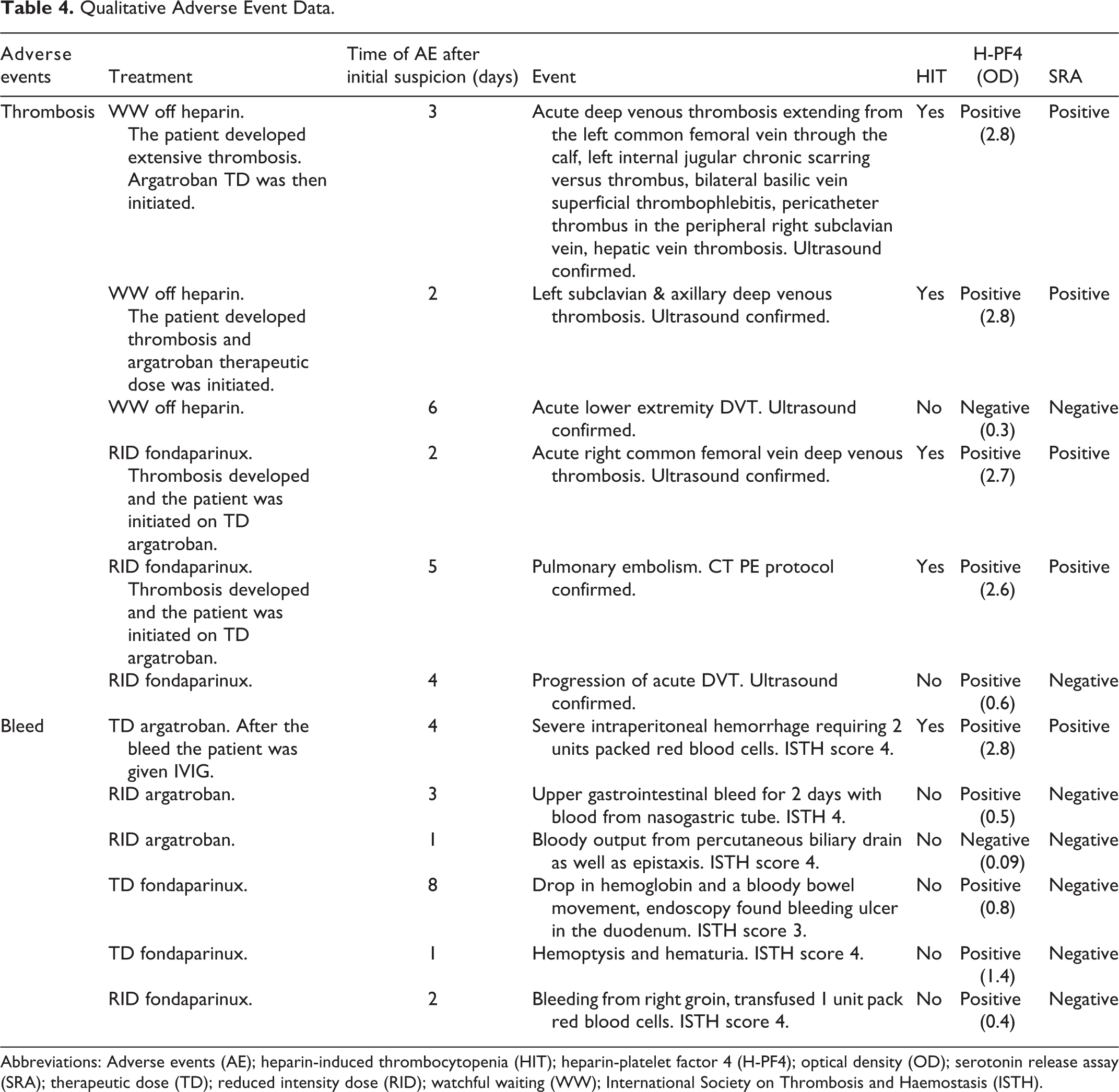
Abbreviations: Adverse events (AE); heparin-induced thrombocytopenia (HIT); heparin-platelet factor 4 (H-PF4); optical density (OD); serotonin release assay (SRA); therapeutic dose (TD); reduced intensity dose (RID); watchful waiting (WW); International Society on Thrombosis and Haemostasis (ISTH).
Discussion
Initial Empiric Management
Patients with higher 4Ts scores and who had positive H-PF4 results were more often treated with therapeutic non-heparin anticoagulation. The highest average 4Ts score was observed in the TD argatroban group, while the lowest average 4Ts scores occurred in the WW on heparin group (Table 1).
The IMPROVE bleeding RAM score differed significantly (p = 0.002) among empiric management groups, suggesting that bleeding risk influenced management decisions (Table 1). Clinicians were not prompted to calculate a bleeding risk score prior to selecting empiric HIT management, however our findings support that in clinical practice providers were using a gestalt comparison between bleeding risk and suspicion for HIT when selecting empiric treatment and favoring more conservative management when bleeding risk was estimated to be high.
Adverse Events
AE occurred in 7.1% of patients (Table 2). 50% of all events were thrombotic and 50% were clinically significant bleeding. AE were more common among patients who were ultimately diagnosed with HIT.
It should be noted that regardless of the empiric choice of management initially made, clinicians actively altered empiric management based on incoming clinical information. For example, if new or progressive thrombosis occurred clinicians initiated more aggressive anticoagulation and, conversely, if bleeding occurred anticoagulation was routinely stopped (Table 4). This dynamic approach to HIT management currently ongoing in clinical practice is challenging to objectively quantify, however it is conceptually intuitive.
This study supported the importance of calculating pre-test probability of HIT before subjecting patients to empiric HIT management and the utility of a cautious WW approach in patients without thrombosis and with high bleeding risk. Patients ultimately without HIT frequently experienced clinically significant bleeding events while on non-heparin anticoagulation and thrombotic events when anticoagulation was held or dose reduced.
Impact of Empiric Management Choice
Half of all AE occurred in patients treated with fondaparinux. Logistic regression analyses revealed fondaparinux was associated with more overall AE than watchful waiting. However, argatroban was not associated with more overall AE compared to WW, when considering that most patients were negative by SRA HIT functional assay. This finding suggests that if selecting a non-heparin anticoagulant, argatroban may be a preferred agent, but this likely warrants additional study considering potential cost and convenience of subcutaneous fondaparinux dosing. This finding counters recent retrospective studies that support off-label fondaparinux use in patients with confirmed HIT.
The decisions to observe off anticoagulation or prescribe RID anticoagulation, although largely not supported by evidence, is often employed by clinicians who perceive significant bleeding risks associated with full-dose anticoagulation despite clinical suspicion for HIT. These treatment options were examined in this study. The significantly higher retrospectively-assigned IMPROVE RAM score among those treated with RID anticoagulation or WW suggested clinicians used estimated bleeding risk to guide dosing (Table 1). Interestingly, results showed the intensity of anticoagulation, with and without controlling for bleeding risk, was not associated with a significant difference in overall thrombotic and bleeding events. Our study found no significant difference in creatinine or PT/INR between treatment groups, however activated partial thromboplastin time (aPTT) during anticoagulation was not collected in this study to better evaluate the possibility of equivalent anticoagulation despite RID in the setting of patient-specific organ dysfunction (Table 1).
Our study included all patients with suspected HIT, not only patients with confirmed HIT. It should be noted that our study did not compare argatroban to fondaparinux directly. Furthermore, our study found that time to treatment initiation was not associated with a significant difference in overall thrombotic and bleeding events. This finding was likely multifactorial and due in part to the fact that empiric management was initiated on average < 24 hours within initial suspicion. This was paired with rapid testing turn-around time, despite the fact that the SRA was a send-out test (Table 1). The impact of timing may be worthy of additional examination in clinical settings where results are more delayed.
Study Limitations
Limitations of this study include its retrospective nature, which poses risk for selection bias, potential for unaccounted for confounding variables and inability to calculate causation or relative risk. Low HIT prevalence and limited sample size inhibited a priori power calculation and overall robustness of findings. The dynamic management of empiric HIT treatment, with numerous approaches taken as well as adjustment of approach during individual cases made findings difficult to quantify and interpret. Despite these limitations, findings from this study challenge current paradigms of suspect HIT management and call for larger, prospective studies that examine empiric HIT treatment.
Conclusion
Empiric HIT management varies widely, with multiple clinical factors likely impacting management decisions and AE. Bleeding and thrombotic AE occurred at higher proportions in patients ultimately diagnosed with HIT. Fondaparinux was significantly associated with more overall AE than WW, both with and without heparin. Intensity of anticoagulation was not associated with a significant difference in AE in this limited study. Larger studies are needed to further investigate and validate these findings.
Footnotes
Authors’ Note
K. Dykes contributed to study design and performed data collection, data analysis and manuscript writing. C. Johnson contributed statistical analysis and data interpretation. J. Gong and S. McKenzie contributed to study design, data interpretation and manuscript editing. H. Husseinzadeh contributed to study design, data analysis and manuscript writing. All authors agreed on the final manuscript. Our institution did not require ethical approval for this retrospective chart review and informed consent was waived given the anonymous and retrospective nature of the study. Fresenius Kabi employed C. Johnson in August 2020, after her contribution to the manuscript had been completed.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
