Abstract
Limited data are available on atrial fibrillation (AF) and its clinical management and outcomes from an Australian perspective. This study was designed to examine the patient characteristics and antithrombotic treatment patterns among patients with AF in Tasmania, Australia. This retrospective observational study reviewed and followed patients with AF admitted to Tasmania’s 3 major hospitals between January 2011 and June 2012. Patients were excluded if they had only 1 episode of AF that reverted spontaneously or upon cardioversion without any documented recurrences. We reviewed the records of 2502 patients (≥18 years), and1469 were subsequently included in the study. The mean (±standard deviation [SD]) age of the patients was 76 (±12.3) years. The mean (± SD) CHADS2 score was 2.1 (±1.3), and 65.7% had a score ≥2. In total, only 55.6% of patients with CHADS2 score ≥2 were receiving anticoagulation and 9.9% were not receiving any antithrombotic treatment, whereas 85.4% of those at low risk (score 0) were on antithrombotic therapy. Hospitalization was associated with a significant increase in the rate of combination (antiplatelet plus anticoagulant) therapy (P < .001). Suboptimal use of antithrombotic therapy highlights the need to improve AF management in our jurisdiction.
Introduction
Atrial fibrillation (AF) is the most common sustained cardiac rhythm disorder and is increasing in incidence and prevalence in aging populations. 1 Studies indicate that 1.5% to 2% of the developed world population have AF. 2 Atrial fibrillation often coexists with hypertension (HTN), heart failure, coronary artery disease, and diabetes mellitus 3 and increases the risk of mortality and morbidity, particularly due to stroke or thromboembolism (TE). 4
Studies have provided details of the characteristics, risk profiles, management, and clinical outcomes of patients with AF. 5,6 Although all contemporary guidelines recommend prophylaxis with antithrombotic agents in people with AF and at least one other risk factor for stroke, 7,8 there have been consistent reports of underuse of anticoagulation as well as overuse in low-risk patients. 5,9 For instance, the EURObservational Research Programme on Atrial Fibrillation (EORP-AF) Pilot Survey revealed that antiplatelet therapy was still being overprescribed, with or without anticoagulation, while elderly patients were commonly undertreated with oral anticoagulation, despite their high risk of stroke and thromboembolic complications. 6
At present, there are limited data available on the characteristics, clinical management, and outcomes of patients with AF from an Australian perspective. The available literature suggests that there is underutilization of anticoagulant therapy, 10,11 although these data comes from relatively small observational trials in selected patient groups. 10,12 The Commonwealth Review of Anticoagulation Therapies in AF in Australia identified that stroke prevention in individuals with AF requires improvement and highlighted a range of issues to be addressed related to the assessment of patients for stroke and bleeding risk, appropriate choice of antithrombotic agent(s) in patients with multiple comorbidities, and the monitoring of patients. 13 The review stressed the need for local data on which to base recommendations regarding the treatment of AF. We designed this study, the Tasmanian AF (TAF) Study, as a starting point to providing comprehensive data describing the outcomes of current stroke prevention strategies in Tasmanian patients with AF. The aim of the current article is to describe the characteristics of patients with AF admitted to 3 different Tasmanian hospitals and the appropriateness of antithrombotic prescribing patterns according to existing guideline recommendations.
Materials and Methods
Study Design
Patients were identified by the Medical Record Departments at the Royal Hobart Hospital (RHH), Launceston General Hospital (LGH), and North West Regional Hospital (NWRH). The RHH is a 500-bed hospital servicing the southern region of Tasmania, Australia (population of 255 614). The LGH is a 300-bed hospital servicing residents of Launceston and the northern region of Tasmania (population of 143 544). Similarly, the NWRH is a 160-bed hospital providing services to North West Tasmania and King Island (population of 114 001). 14
We reviewed medical records (admissions between January 1, 2011, and June 30, 2012) of 2502 patients aged ≥18 years with a diagnosis of AF (both valvular and nonvalvular) at discharge (Australian Refined Diagnosis Related Group [AR-DRG] code I48: atrial fibrillation or flutter). Patients diagnosed with AF as their primary (ie, AF was the presenting complaint) or secondary condition (ie, AF was listed as a current illness in the medical history or discharge summary) were included. Patients were excluded if they had only 1 episode of AF that reverted spontaneously or upon cardioversion without any documented recurrences, as stroke prophylaxis may not be warranted in these patients. Contraindications (CIs) to anticoagulant therapy included a history of dementia, documented labile international normalized ratio (INR), bleeding disorders or allergies to anticoagulant therapy, and breast-feeding or pregnancy in women. We considered a patient’s index admission as their first admission within our data collection period, with a diagnosis of AF that met the study’s inclusion criteria.
Data collected at baseline (ie, at each patient’s index admission) included patient demographics, medications on admission, documented previous medical history, relevant laboratory data, discharge diagnosis, and discharge medications. The CHADS2 (1 point each for congestive heart failure (CHF), HTN, age ≥75 years, and diabetes and 2 points for previous stroke or transient ischemic attack (TIA) 15 ) and CHA2DS2-VASc (1 point each to CHF, HTN, diabetes, vascular disease (prior myocardial infarction, peripheral artery disease, or aortic plaque), age 65 to 74 years, and female gender and 2 points for age ≥75 years and previous stroke or TIA or TE. 16 ) risk assessment scores were used to estimate stroke risk. A CHADS2 score of 0 was considered low risk, 1 was considered intermediate risk, and ≥2 was considered high risk, while a CHA2DS2-VASc score of 0 represented low risk and ≥1 was considered intermediate–high risk. The HAS-BLED (1 point each for HTN, abnormal renal function, abnormal liver function, bleeding predisposition, age >65 years, labile INRs (if documented), the use of drugs predisposing patients to bleeding (NSAIDs), alcohol use (>8 drinks per week), and previous stroke. 17 ) score was used to estimate bleeding risk assessment. A HAS-BLED score of 0 indicated low risk, 1 to 2 indicated intermediate risk, and ≥3 indicated high risk. Although the CHA2DS2-VASc and HAS-BLED scores were not available to clinicians to guide treatment during the study time period, they were included in the analysis to assist in interpretation of our results. The Charlson Comorbidity Index (CCI) was used as a measure of comorbidity. 18
Our “antiplatelet” group consisted of patients receiving aspirin, clopidogrel, prasugrel, dipyridamole, or ticagrelor either alone or in combination with each other but not in combination with an anticoagulant. The “lone anticoagulant” group consisted of cases receiving warfarin, dabigatran, heparin, fondaparinux, or enoxaparin. Patients taking a combination of an anticoagulant with an antiplatelet agent constituted the “combination therapy” group. Finally, patients receiving any one of these drug groups were considered to be on antithrombotic therapy, that is, either on antiplatelet or anticoagulant or on combination therapy. We utilized the recommendations from the European Society of Cardiology (ESC) 2010 guidelines and American College of Chest Physicians (ACCP) 9th edition 7,8 to examine the appropriateness of antithrombotic prescribing at discharge of the index admission. We considered underutilization as nonprescribing of anticoagulation, with or without antiplatelet therapy, to patients without documented CIs to anticoagulant therapy and a CHADS2 score ≥2 as per these guidelines.
Statistical Analysis
Data were analyzed using SPSS version 21 (Prentice Hall, Upper Saddle River, New Jersey). Continuous variables were expressed as mean ± standard deviation (SD). Categorical variables were expressed as frequencies and percentages. Differences between cohorts were tested for statistical significance using Mann-Whitney U and chi-square tests, as appropriate. P < .05 was considered as statistically significant for all analyses.
Ethics
Ethics approval for the project was obtained from Tasmanian Health and Medical Human Research Ethics Committee.
Results
Baseline Characteristics
The medical records of 2502 patients were reviewed, of whom 1469 were included (RHH: 777, NWRH: 289, and LGH: 403) and 1033 were excluded (episode of AF that reverted spontaneously or upon cardioversion: 590, developed AF as a short-term complication: 288, no documented AF [coding error]: 155). The mean (±SD) age of the included patients was 76 (±12.3) years, and 55.6% were male. Valvular AF was observed in 11.3% (166) of patients. Hypertension was the most commonly associated comorbid condition (65.7%). The demographics and clinical characteristics of the patients are summarized in Table 1.
Baseline Characteristics of Patients Enrolled in TAF Study.

Abbreviations: AP/AC, antiplatelet and anticoagulant; AF, atrial fibrillation; CCI, Charlson comorbidity index; DVT, deep vein thrombosis; H2Bs, histamine-2 receptor blockers; PPIs, proton pump inhibitors; NSAIDs, non-steroidal anti-inflammatory drugs; SD, standard deviation; TAF, Tasmanian atrial fibrillation.
The mean ± (SD) CHADS2 score was 2.1 ± (1.3), and 65.7% had a score ≥2. The mean ± (SD) CHA2DS2-VASc score was 3.7 ± (1.7), and 8.2% had a score of 1 while 88.6% had a score ≥2. The majority of the patients (73.5%) had an intermediate HAS-BLED score of 1 to 2, as shown in Table 1. A contraindication (CI) to antithrombotic treatment was documented in 9.9% of patients.
Thromboprophylactic Treatment
Based on ESC 2010 guidelines and ACCP 9th edition, 7,8 64.1% (n = 808) of the 1261 patients with no documented CIs to antithrombotic therapy had a CHADS2 score ≥2 and were therefore eligible for anticoagulant therapy (Table 2). An anticoagulant was prescribed for 55.6% (n = 449) of these patients at discharge. These percentages were broadly similar for the intermediate risk (CHADS2 = 1) group. Antiplatelet agents were prescribed in an additional 34.5% (n = 279) of patients. Eighty (9.9%) patients with a CHADS2 score ≥2 were not receiving any antithrombotic therapy. “Physician decision” was the only documented reason for not prescribing antithrombotic therapy (n = 34, 3.0%), despite patients having intermediate to high risk of stroke (CHADS2 score ≥1) without any CIs. In contrast, 85.4% (n = 123 of 144) of patients in whom no treatment may have been indicated due to their low risk of stroke (CHADS2 = 0) were receiving antithrombotic therapy. Only 5.6% (n = 8 of 144) of these patients had an history of embolic disease other than AF and therefore alternative indications for anticoagulation. Furthermore, among those receiving lone antiplatelet agents and having a CHADS2 = 0, 16.3% (8 of 49) had a history of ischemic heart disease (IHD).
Use of Antithrombotic Therapies, Overall and According to CHADS2 and CHA2DS2-VASc Scores on Discharge.a
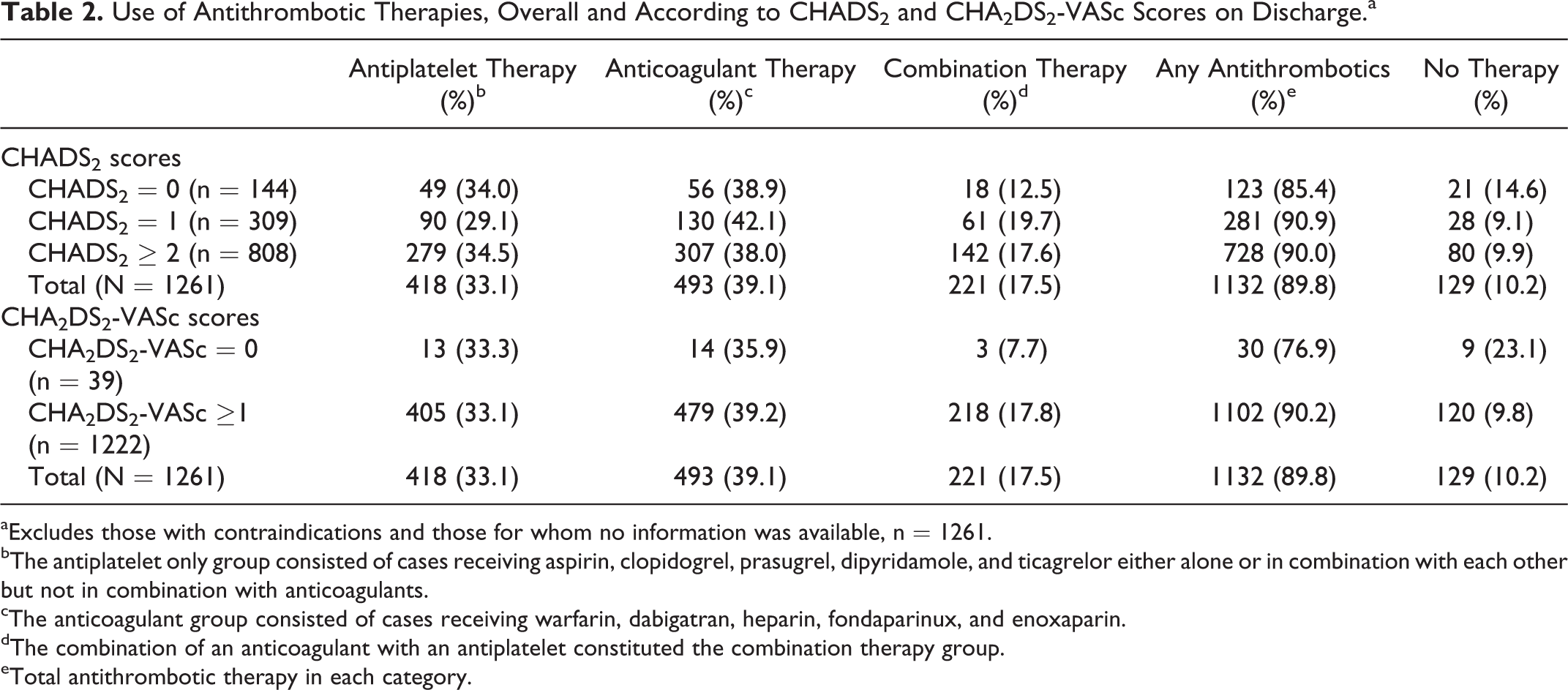
aExcludes those with contraindications and those for whom no information was available, n = 1261.
bThe antiplatelet only group consisted of cases receiving aspirin, clopidogrel, prasugrel, dipyridamole, and ticagrelor either alone or in combination with each other but not in combination with anticoagulants.
cThe anticoagulant group consisted of cases receiving warfarin, dabigatran, heparin, fondaparinux, and enoxaparin.
dThe combination of an anticoagulant with an antiplatelet constituted the combination therapy group.
eTotal antithrombotic therapy in each category.
Similar findings arose when prescribing was evaluated against patients’ CHA2DS2-VASc scores. In this case, 96.9% (n = 1222 of 1261) of patients with no CIs to antithrombotic therapy had at least 1 additional risk factor for stroke and were therefore eligible for anticoagulant therapy (Table 2). An anticoagulant was prescribed for 57.0% (n = 697) of these patients. Antiplatelet agents were prescribed in 33.1% (n = 405).
In assessing whether anticoagulant underuse was associated with a high HAS-BLED score, we observed that 22.0% (n = 278 of 1261) of patients without any CIs had an HAS-BLED score, which exceeded their CHADS2 score. Of these patients, 56.5% were on anticoagulant therapy, 34.9% were on antiplatelet therapy, and 8.6% received no therapy, which was largely consistent with the overall population.
Of the patients without CIs, 11.4% (144 of 1261) had valvular AF. Among these patients, 54.9% were prescribed lone anticoagulant therapy, followed by combination therapy (23.6%), and 15.3% were taking antiplatelet monotherapy. The prescribing pattern of antithrombotic therapy differed significantly between the patients with valvular and nonvalvular AF, with higher rates of prescribing of lone anticoagulant therapy (54.9% vs 37.1%, P < .001) and combination therapy (23.6% vs 16.7%, P < .001) and a correspondingly lower rate of lone antiplatelet therapy (15.3% vs 35.6%, P < .001).
We also observed a significant difference in the prescribing pattern of combination antithrombotic regimens between patients who were newly initiated (ie, started antithrombotic therapy during the index admission) and continuing therapy (ie, those on antithrombotic treatment prior to their index admission). A larger proportion of newly initiated patients were prescribed combination antithrombotic therapy (25.8% vs 18.0% for the continuing group; P < .001). Recent diagnosis of acute coronary syndrome (ACS), as a potential indication for combination therapy, did not seem to have increased its prescribing between these groups, with 4.0% of those newly initiated and 4.5% of those continuing on antithrombotic therapy diagnosed with ACS during their index admission (P = .71).
Impact of Hospitalization on Antithrombotic Prescribing
When the antithrombotic prescribing patterns on admission and discharge were compared among the 693 patients who had a preexisting AF continuing on antithrombotic treatment, significant changes were observed, with a reduction in the prescribing of lone antiplatelet (40.8% vs 33.5%, P < .001) and anticoagulant therapy (49.2% vs 47.5%, P < .001), and an increased rate of use of combination therapy (10.0% vs 18.9%, P < .001; Table 3). Only 4.0% of those with preexisting AF continuing on antithrombotic therapy were diagnosed with ACS during their index admission.
Antithrombotic Therapy Change Associated With Hospitalization.a
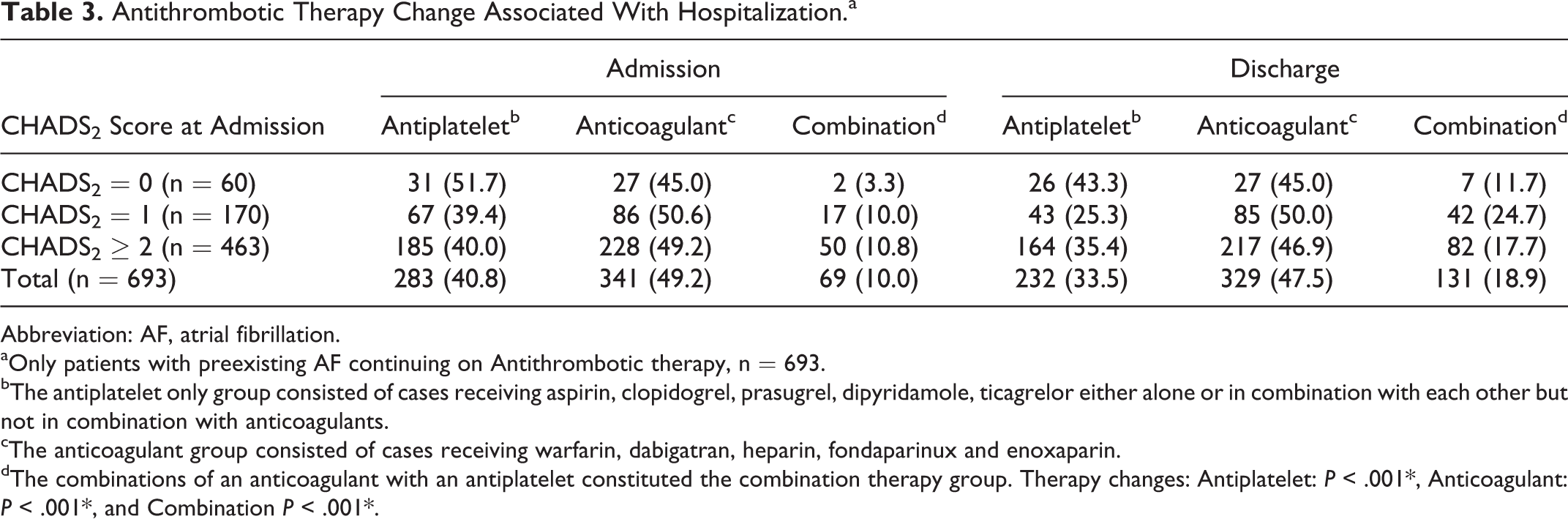
Abbreviation: AF, atrial fibrillation.
aOnly patients with preexisting AF continuing on Antithrombotic therapy, n = 693.
bThe antiplatelet only group consisted of cases receiving aspirin, clopidogrel, prasugrel, dipyridamole, ticagrelor either alone or in combination with each other but not in combination with anticoagulants.
cThe anticoagulant group consisted of cases receiving warfarin, dabigatran, heparin, fondaparinux and enoxaparin.
dThe combinations of an anticoagulant with an antiplatelet constituted the combination therapy group. Therapy changes: Antiplatelet: P < .001*, Anticoagulant: P < .001*, and Combination P < .001*.
Discussion
Observational studies can be valuable in evaluating disease state management and its outcomes in a population, especially in countries such as Australia that lack effective large-scale health care data linkage systems. The TAF study, an ongoing observational study of adults with AF, aims to comprehensively evaluate the management and outcomes of patients with AF. This article provides a salient snapshot of AF management patterns among patients with AF who experienced a hospital admission in 3 Tasmanian hospitals during 2011 and 2012. In this large study of patients with AF, we noted that, despite the high risk of stroke and thromboembolic complications, anticoagulant therapy was suboptimal.
As our patient population was identified during hospital admission, it is perhaps unsurprising that they demonstrated high rates of comorbid conditions, especially cardiovascular diseases. Nevertheless, their characteristics proved to be largely similar to those of the populations studied in the Stockholm AF database, Fushimi AF Registry, and ORBIT-AF Registry. 19 –21 They were noted to be older and at higher risk of stroke compared to the Global Anticoagulant Registry in the FIELD (GARFIELD) study population, 5 but as GARFIELD is enrolling only patients with new-onset AF, it is expected that this will have a younger, lower risk cohort. The retrospective cohort of the TAF Study also compares well to the patient cohorts studied in clinical trials comparing novel oral anticoagulants (NOACs) to warfarin, in terms of mean age (76 vs 70-73 years) and stroke risk (mean CHADS2 2.1 vs 2.1-3.5). 22 –25 This suggests that our patient population is broadly comparable to that of other countries and to those studied in recent pivotal trials, indicating the likely transferability of international guidelines and the benefits indicated by these trials’ results to our population.
Antiplatelet and anticoagulant therapies are available for stroke prevention in AF. Use of anticoagulation therapy for stroke prevention in AF is well accepted, established, and recommended widely. 26 All contemporary guidelines recommend prophylaxis with antithrombotic agents in people with AF and at least 1 other risk factor for stroke, 7,8 however, discordance between AF guideline recommendations and anticoagulant prescribing patterns has been reported in various international studies. 5,27
In our study, despite the high risk of stroke observed (64.1% of patients had a CHADS2 ≥2, and 96.9% had a CHA2DS2-VASc score of 1 or more), anticoagulant therapy was found to be underutilized according to guideline recommendations. 7,8 At discharge from hospital, almost 10% of patients with a CHADS2 ≥2 and CHA2DS2VASc score ≥1 were not taking any antithrombotic medication, and approximately one-third were prescribed antiplatelet monotherapy. Given the time frame of our study and the guideline recommendations 8 during that time, CHADS2 was the more realistic tool for assessing appropriateness of therapy in relation to estimated stroke risk; however, we have presented the results using both CHADS2 and CHA2DS2-VASc scores believing that physicians might have also considered the additional risk factors subsequently included in CHA2DS2-VASc score.
This underutilization of anticoagulant therapy in patients with AF at relatively high risk of stroke is consistent with previous findings, 11,28 with many clinicians considering the risk of anticoagulant treatment to exceed the benefits, when in fact those at higher risk of stroke often receive the greatest benefit from anticoagulation. 29 The potential risk of bleeding is one of the most frequent reasons of anticoagulant underuse among treating physicians. 30 Since 22.0% of our patients without any CIs had an HAS-BLED score that exceeded their CHADS2 score, this might have contributed to the observed underutilization of anticoagulant therapy. Given that the majority of our patient population were elderly and had multiple comorbidities, this might have led to physicians being more reluctant to prescribe anticoagulant therapy. A history of anemia, history of hospitalization/emergency department visits, elderly age, comorbidities, risk of falls, and previous bleeding have all been identified as reasons for nonprescribing of anticoagulants. 5,31 –33 The belief that antiplatelet therapy alone is sufficient for stroke prevention and/or lack of awareness of guideline recommendations 11 may also have contributed to the underutilization of anticoagulant therapy. Nevertheless, the overall use of antithrombotic treatment seems to have improved since 1997 to 1999, when a study conducted among 505 patients diagnosed with AF at the RHH found that 18.2% of patients at high risk were discharged without any antithrombotic therapy. 10
Over 80% of the patients with a CHADS2 score of 0, who are considered to be “low-risk” patients, were receiving antithrombotic drugs at discharge. Nearly 40% of these patients were on anticoagulants, and this overuse pattern is very similar to that found in the GARFIELD study (38.7%). 5 Other indications apart from AF might have justified the use of anticoagulants in some of these patients; however, only 5.6% had a history of embolic disease, and data regarding other possible indications was not collected within the confines of this study. Similarly, a history of IHD among those with a CHADS2 score of 0 (16.3%) might have made some patients eligible for antiplatelet therapy, despite being at a low risk of stroke.
The final issue of concern raised by our data was the relatively high rate of prescribing of combination anticoagulant/antiplatelet therapy; this rate was especially high in patients newly initiated on therapy and also increased from admission to discharge in patients with existing AF. One explanation for this prescribing pattern may be the diagnosis of a compelling guideline-recommended indication for antiplatelet therapy (eg, ACS) in patients requiring anticoagulation for AF thromboprophylaxis. 8 The ACS was only diagnosed among 4.0% of those with old AF continuing on antithrombotic therapy during the index admission in our study; however, this is unlikely to have been a major influencing factor. Other potential reasons for the prescribing of a combination therapy may have been the use of antiplatelet agents as “bridging therapy” during anticoagulant initiation or inappropriate failure to cease antiplatelet therapy upon initiation of anticoagulation. This issue warrants further investigation as combination therapy is associated with a significantly higher risk of bleeding. 8
There is clearly an evidence to practice gap in guideline adherence to rational prescribing of antithrombotic regimens in patients with AF in our population. Local guidelines and other contextual factors may influence anticoagulant prescribing. 11 At present, there are no up-to-date Australian guidelines for the management of AF; thus, international guidelines are the basis of its management, and data regarding predictors of antithrombotic prescribing among patients with AF are as yet limited in Australia. The absence of robust national AF management guidelines might have influenced underutilization or overutilization of anticoagulant therapy in our population. Intervention programs, such as described previously, 34,35 may be required to assist with optimizing guideline concordance and therefore the proportion of Australian patients receiving appropriate thromboprophylaxis for stroke prevention in AF.
Limitations
As an observational study, this study is subject to certain limitations such as the collection of nonrandomized data. Data were collected retrospectively from 3 different hospitals, and the presence of missing or incomplete data was unavoidable. Recent hospitalization for surgery may have constituted a reasonable indication for temporary cessation of anticoagulation, which may have influenced the findings regarding suboptimal anticoagulation. Our study only included patients with AF who experienced a hospitalization meaning that they were potentially already at higher risk than “average” Tasmanian patients with AF. This could limit the generalizability of our results to the broader Australian population.
Conclusion
Our study highlights a gap between the evidence-based risk stratification and antithrombotic management pattern among patients with AF in Tasmania. In the absence of contemporary local guidelines, there appears to be a need to better support prescribers to assist in the identification and quantification of patient risk according to accepted international guidelines to optimize thromboprophylaxis and reduce the risk of thromboembolic and bleeding complications in this vulnerable patient group.
Footnotes
Acknowledgments
We would like to acknowledge the Royal Hobart Hospital Research Foundation (RHHRF) for providing funding to conduct our study. We would also like to thank Mr Peter Gee for his support in designing the electronic TAF database and in the data extraction process. We would also like to thank Mr Chris Ballard, Dr Carley Bagley, Dr Georgia McGuinness, and Mr Cameron Wright for their help in data collection.
Authors’ Notes
DB contributed to design of the work; acquisition, analysis, or interpretation of data; literature search; and manuscript writing. LC contributed to the conception of the work, manuscript drafting, critical revising, and final approval of the version to be published. GP contributed to the conception of the work, critical manuscript review, and final approval of the version to be published. LB contributed to the conception of the work critical manuscript revising and final approval of the version to be published. This study was delivered as an oral presentation at the Australasian Pharmaceutical Science Association (APSA), Dunedin, New Zealand (December, 2013).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Leanne Chalmers, Luke R. E. Bereznicki and Gregory M. Peterson have received consultancy funding from Aspen Pharmacare Australia and Boehringer Ingelheim. Luke R. E. Bereznicki has also received consultancy funding from Roche Diagnostics Australia and Sanofi Aventis and speaker honorarium payments from Roche Diagnostics Australia and Boehringer Ingelheim.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: received financial support from the Royal Hobart Hospital Research Foundation for the research.
