Abstract
Aim:
This retrospective cohort study aimed to evaluate the prognostic implications of the distinct atrial fibrillation (AF) temporal patterns: first diagnosed, paroxysmal, and persistent or permanent AF.
Methods:
In this post hoc analysis of the MISOAC-AF trial (NCT02941978), a total of 1052 patients with AF (median age 76 years), discharged from the cardiology ward between 2015 and 2018, were analyzed. Kaplan-Meier and Cox-regression analyses were performed to compare the primary outcome of all-cause mortality, the secondary outcomes of stroke, major bleeding and the composite outcome of cardiovascular (CV) mortality or hospitalization among AF patterns.
Results:
Of patients, 121 (11.2%) had first diagnosed, 356 (33%) paroxysmal, and 575 (53.2%) persistent or permanent AF. During a median follow-up of 31 months (interquartile range 10 to 52 months), 37.3% of patients died. Compared with paroxysmal AF, patients with persistent or permanent AF had higher mortality rates (adjusted hazard ratio (aHR), 1.37; 95% confidence interval [CI], 1.08-1.74, P = .009), but similar CV mortality or hospitalization rates (aHR, 1.09; 95% CI, 0.91-1.31, P = .35). Compared with first diagnosed AF, patients with persistent or permanent AF had similar mortality (aHR, 1.26; 95% CI, 0.87-1.82, P = .24), but higher CV mortality or hospitalization rates (aHR, 1.35; 95% CI, 1.01-1.8, P = .04). Stroke and major bleeding events did not differ across AF patterns (all P > .05).
Conclusions:
In conclusion, in recently hospitalized patients with comorbid AF, the presence of persistent or permanent AF was associated with a higher incidence of mortality and morbidity compared with paroxysmal and first diagnosed AF.
Introduction
Atrial fibrillation (AF) represents the most common, clinically significant, cardiac arrhythmia worldwide. 1 Its prevalence is rising impressively in Europe 2 and in the United States of America, 3,4 reflecting the aging of the global population, the development of the current diagnostic methods, or the proliferation of the risk factors. 5 AF is a leading reason for all-cause mortality, 1 recurrent hospitalizations, and a major health and financial burden. 6,7 It is also consistently associated with morbidity, such as thromboembolic events and major bleeding. 5
The European Society of Cardiology AF Guideline Committee has classified AF into temporal patterns, determined by the occurrence and duration of the arrhythmia. 1 Whether the pattern of AF directly modifies the risk of mortality and morbidity, regardless of the co-existing comorbidities, is uncertain. 8,9 The prevailing notion is that persistent and permanent AF patterns are linked with worse survival outcomes. 9 -13 However, previous studies related to the association of specific AF subtypes with survival have been contradictory. 14,15 This study aims to investigate the frequency of AF patterns in a specific high-risk AF population and to assess their association with mortality and morbidity.
Methods
This is a retrospective cohort analysis of the MISOAC-AF (Motivational Interviewing to Support Oral AntiCoagulation Adherence in patients with non-valvular Atrial Fibrillation; ClinicalTrials.gov identifier: NCT02941978), a randomized trial, carried out in the cardiology department of a tertiary hospital (AHEPA University Hospital of Thessaloniki, Greece). The design and main results of this trial have already been described. 10,11 In brief, MISOAC-AF explored the role of patient–physician interviews and intentional scripted guidance in improving the conformity to OACs in AF patients. The study was approved by the institutional review board and ethics committee at the Aristotle University of Thessaloniki and conformed to the Declaration of Helsinki. 12 All patients that participated assigned written informed consent.
The MISOAC-AF randomized trial involved adult patients who were hospitalized in the Cardiology ward of a tertiary hospital and had comorbid AF, regardless of the discharge diagnosis. The current study source population consists of all patients randomized in the MISOAC-AF trial. Exclusion criteria were factors that would obstruct the follow up of the patients, such as survival expectancy less than 1 year and conditions likely to prevent patient participation in the interviews. Patients with unspecified-undefined AF pattern and patients diagnosed with atrial flutter were considered ineligible for the purpose of this study.
Prospective data from the MISOAC-AF patient dataset were used for patients discharged from the hospital between December 2015 and June 2018. Baseline clinical profiles, medical history, laboratory, and echocardiographic data, discharge diagnoses, and medications were available in the electronic database of the study. All these data were collected by trained independent physicians who interviewed patients while they were hospitalized and had access to their discharge notes and their full hospital record.
As in the MISOAC-AF trial, AF was electrocardiographically documented as a more than 30 seconds lasting episode of irregularly irregular ventricular rhythm (QRS complexes) accompanied by the absence of sinus P-waves. The AF pattern was classified according to the duration, incidence, and spontaneous or not termination of the episode. 5 First diagnosed AF was defined as AF that has never been diagnosed before. A self- or after intervention terminating AF within 7 days was defined as paroxysmal. Persistent AF was defined as AF that lasted more than 7 days and was restored to sinus rhythm after pharmacologic or electrical cardioversion. Permanent AF corresponded to the arrhythmia being accepted by both the patient and the physician, thus waiving the need for a rhythm control strategy. Persistent and long-standing persistent AF were not separated during data collection in MISOAC-AF. In this study, persistent, long-standing persistent, and permanent AF patterns were classified together, to attenuate classification bias and ease statistical analyses. AF pattern stratification was coded during baseline assessment of patients by trained personnel.
We calculated the CHA2DS2-VASc score (Congestive heart failure, Hypertension, Age ≥ 75, clinical history of Diabetes mellitus, prior Stroke/transient ischemic attack/thromboembolism, Vascular disease, Age 65-74, Sex [Female category]) for each patient to evaluate the thrombotic risk. For the bleeding risk assessment, we calculated the HAS-BLED Score (Hypertension, Abnormal liver or renal function, Stroke, Bleeding tendency or predisposition, Labile international normalized ratio (INR), Elderly: age >65 years old, Drugs: concomitant antiplatelet agents, non-steroidal anti-inflammatory drugs or alcohol).
The primary outcome in the current study was all-cause mortality. The composite of cardiovascular death or any hospitalizations, stroke or major bleeding events during the follow-up period constituted the secondary outcomes. Patients were followed annually from the index date of hospital discharge via telephone interviews about clinical outcome events. The follow-up period ended in February 2020 after a last telephonic contact with each patient. Using the insurance number of each patient, given after the written consent, we ascertained all deaths by the Greek electronic insurance system.
Patients’ data were grouped according to the AF pattern (first diagnosed, paroxysmal, and permanent or persistent). Continuous variables are expressed as means and standard deviation (SD), whereas categorical variables are presented as numbers and percentages. Baseline characteristics between patients with different AF patterns were compared using the Pearson chi-square or Fisher’s exact test for categorical variables and the one-way analysis of variance (ANOVA) for continuous variables. Kaplan-Meier plots were used to analyze time-to-event data, and the log-rank test was used to make comparisons between groups. Cox proportional-hazards models were performed to examine the impact of the AF pattern on mortality after adjustment for the following clinically relevant covariates: age, sex, the presence of heart failure, coronary artery disease, history of stroke/transient ischemic attack/systemic thromboembolic disease, levels of N-terminal pro-BNP, levels of creatinine, use of beta blockers and oral anticoagulation at discharge. Follow up was censored in the event of death. Multiple Imputations by Chained Equations (MICE) method was used to replace missing values (Table 1 in the Supplementary Appendix). Our analysis results are reported as hazard ratios (HR) with 95% confidence intervals (CI). A P-value of less than .05 using two-sided tests is considered to be statistically significant. R version 3.4.4 (R Foundation for Statistical Computing, Vienna, Austria) Stata statistical software, release 13 (StataCorp) and Stata statistical software, release 13 (StataCorp) were used to perform all analyses.
Results
Patient Characteristics
In the current analysis, a total of 1052 patients were included, after excluding 28 patients with unspecified AF pattern, 36 patients with atrial flutter, and 25 patients with unspecified type of arrhythmia (atrial fibrillation or flutter; Figure 1 in the Supplementary Appendix). The baseline characteristics of the included participants, according to AF pattern, are presented in Table 1. The median age was 73.8 ± 10.9, and 54.6% were men. Of patients, 121 (11.5%) had first diagnosed AF, 356 (33.8%) had paroxysmal AF, and 575 (54.7%) had persistent or permanent AF.
Characteristics of the Patients at Baseline.a

Abbreviations: ACE, angiotensin-converting–enzyme; AF, atrial fibrillation; ARB, angiotensin II–receptor blocker; BMI, body mass index; CHA2DS2-VASc, Congestive heart failure, Hypertension, Age ≥ 75 years, Diabetes mellitus, Stroke/transient ischemic attack, Vascular disease, Age 65 to 74 years, Sex category; COPD, chronic obstructive pulmonary disease; IQR, interquartile range; HAS-BLED, Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile INR, Elderly, Drugs/alcohol; HF, heart failure; LVEF, left ventricular ejection fraction; NT pro-BNP, N-terminal prohormone of brain natriuretic peptide; OAC, oral anticoagulant; SD, standard deviation; TIA, transient ischemic attack.
a Data were reported as absolute numbers (%), means ± SD, or medians (IQR). Statistically significant values are given in bold.
Patients with persistent or permanent AF were older, had more comorbidities (dyslipidemia, chronic obstructive pulmonary, heart failure) and higher rates of previous systematic thromboembolic and bleeding events. Median CHA2DS2-VASc, HAS-BLED scores and heart rate were also higher in persistent or permanent AF.
Overall, a rate control-strategy was preferred compared to a rhythm control-strategy at discharge. As expected, patients with persistent or permanent AF more commonly received rate control medication compared with patients with first diagnosed and paroxysmal AF. Rhythm control medication was more commonly selected in patients with paroxysmal AF compared with patients with first diagnosed and persistent AF. More electrical or pharmacological cardioversion attempts were performed in patients with paroxysmal AF.
Impact of AF Pattern on All-Cause Mortality
Mortality from any cause occurred in 414 (36.3%) patients over a median follow-up duration of 31 months (interquartile range 10 to 52 months). Mortality occurred in significantly higher rates in persistent or permanent AF (255 of 575 [44.3%]) as compared to first diagnosed (38 of 121 [31.7%]; unadjusted HR, 1.63; 95% [CI], 1.16 to 2.29, P = .005) and paroxysmal AF (109 of 356 [29.6%]; unadjusted HR, 1.586; 95% CI, 1.27 to 1.99, P < .001). After adjustment for baseline confounders (age, sex, presence of heart failure, coronary artery disease, history of stroke/transient ischemic attack/systemic thromboembolic disease, levels of N-terminal pro-BNP, renal function, use of beta blockers and oral anticoagulation at discharge), the risk of mortality remained significant only in persistent or permanent AF compared with paroxysmal AF (aHR, 1.37; 95% CI, 1.08 to 1.74, P = .009). This was not apparent in patients with first diagnosed AF (aHR, 1.26; 95% CI, 0.87 to 1.82, P = .22; Table 2 in the Supplementary Appendix). Kaplan-Meier curves for the primary outcome are presented in Figure 1.

Cumulative incidence of all-cause mortality according to AF pattern. aHR indicates adjusted hazard ratio; AF, atrial fibrillation; CI, confidence interval; CV, cardiovascular.
Impact of AF Pattern on CV Death or Hospitalization
The composite secondary outcome—CV death or hospitalization due to any cause—occurred more frequently in persistent or permanent AF (364 of 575 [63.3%]) than in first diagnosed (57 of 121 [47.1%]; unadjusted HR, 1.70; 95% CI, 1.28 to 2.25, P < .001 or paroxysmal AF (190 of 356 [53.4%]; unadjusted HR, 1.28; 95% CI, 1.08 to 1.53, P = .005; Figure 2). In multivariate analysis, persistent or permanent AF remained a significant predictor of CV death or all-cause hospitalization, compared with first diagnosed AF, which was not the case when compared with paroxysmal AF. The differences among AF patterns for each component of the secondary outcome (CV death and hospitalization) were not significant (Table 2 in the Supplementary Appendix).
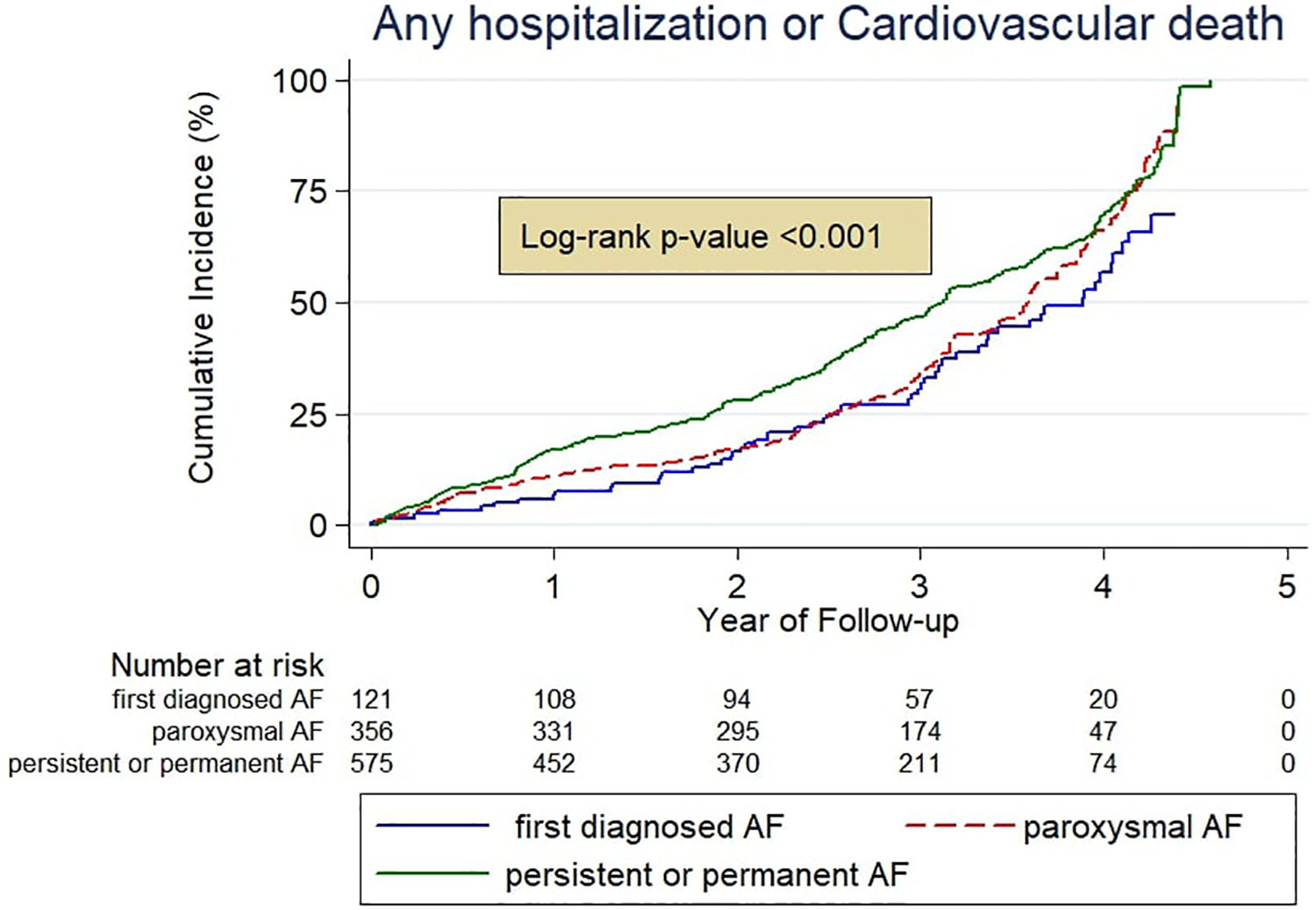
Cumulative incidence of any hospitalization or cardiovascular death according to AF pattern. aHR indicates adjusted hazard ratio; AF, atrial fibrillation; CI, confidence interval.
Associations of AF Patterns With Risk of Stroke and Major Bleeding
Regarding the secondary outcome of stroke, no significant differences were observed between persistent or permanent AF and first diagnosed AF (unadjusted HR, 0.80; 95% CI, 0.35 to 1.86, P = .60) and between persistent or permanent AF and paroxysmal AF (unadjusted HR, 0.86; 95% CI, 0.47 to 1.57, P = .62). Adjusting for covariates did not change the results (persistent or permanent AF versus first diagnosed AF: aHR, 1.18; 95% CI, 0.34 to 4.12, P = .79, and persistent or permanent AF versus paroxysmal AF: aHR, 0.73; 95% CI, 0.38-1.37, P = .32; Figures 3 and 4).

Cumulative incidence of stroke and major bleeding according to AF pattern. aHR indicates adjusted hazard ratio; AF, atrial fibrillation; CI, confidence interval.
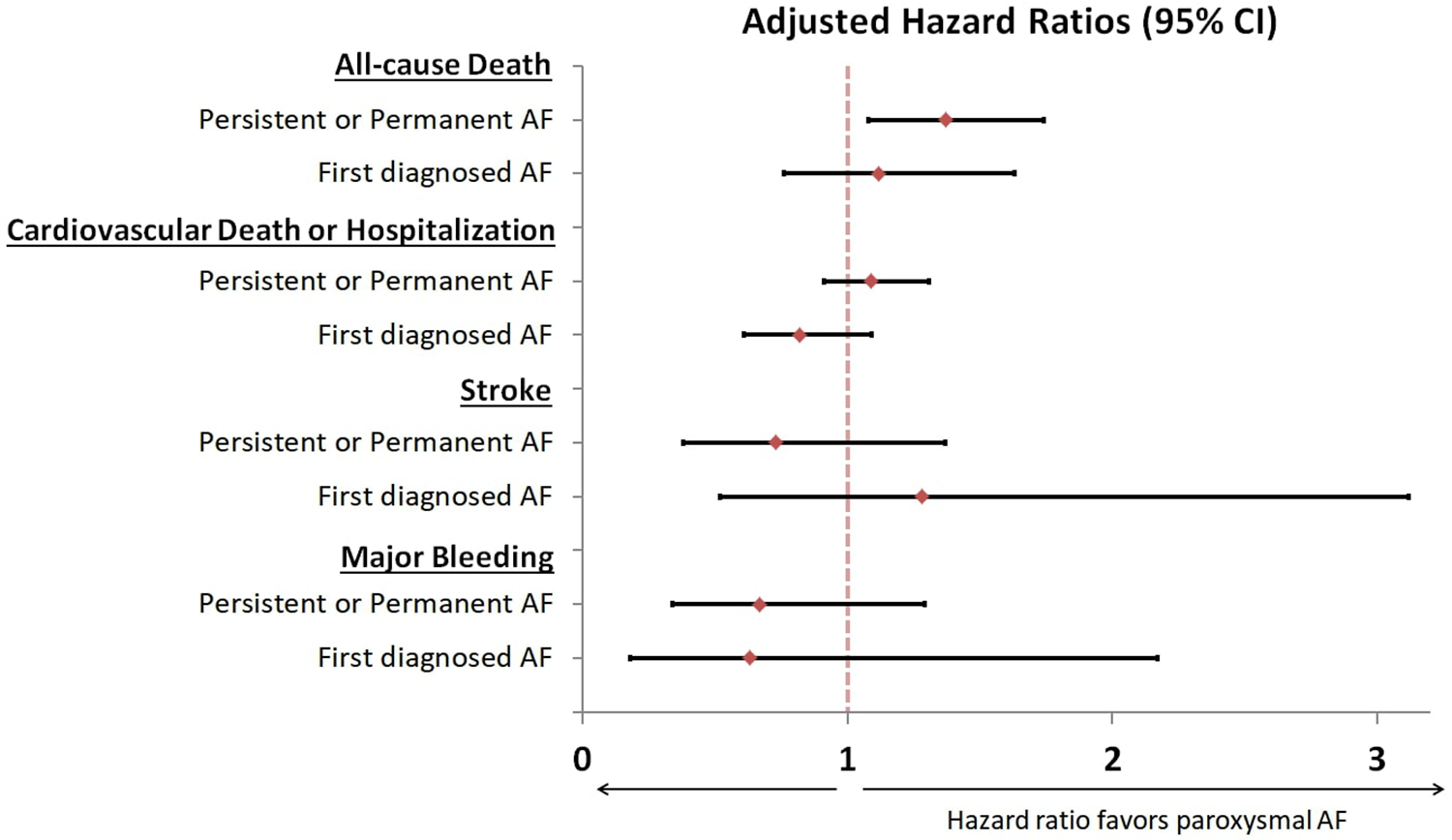
Risk comparisons for outcomes among AF patterns. Hazard ratios of outcomes are presented by AF pattern, using paroxysmal AF as reference. HRs with 95% confidence intervals were calculated using Cox models, adjusted for age, sex, the presence of heart failure, coronary artery disease, history of stroke/transient ischemic attack/systemic thromboembolic disease, levels of N-terminal pro-BNP, levels of creatinine, use of beta blockers and oral anticoagulation at discharge. AF indicates atrial fibrillation; CI, confidence interval.
Major bleeding events appeared to be similar in persistent or permanent AF when compared with first diagnosed AF (unadjusted HR, 1.68; 95% CI, 0.50 to 5.61, P = .40) and when compared with paroxysmal AF (unadjusted ηR, 0.82; 95% CI, 0.44 to 1.53, P = .52). The same was true after adjustment for confounding factors (persistent or permanent AF versus first diagnosed AF: aHR, 1.18; 95% CI, 0.34 to 4.12, P = .79, and persistent or permanent AF versus paroxysmal AF: aHR, 0.67; 95% CI, 0.34-1.29, P = .22; Figures 3 and 4). Further data are shown in Table 2 in the Supplementary Appendix.
Discussion
The current post hoc analysis of a randomized clinical trial of patients discharged from hospital with comorbid AF revealed differences in prognosis according to AF pattern. Persistent or permanent AF was prevalent in more than half of the patients with AF, and was associated with higher mortality risk, as compared with paroxysmal AF. Furthermore, persistent or permanent AF was associated with an increased risk of the composite outcome of cardiovascular death or all-cause hospitalization, as compared with patients with first diagnosed AF. The incidence of stroke and major bleeding did not differ when stratified by AF pattern.
Only few prior studies have been focused on examining the AF pattern as an independent predictor for all-cause mortality. Most are consistent with the results of our study. 13 -17 ENGAGE AF TIMI-48 found significantly higher annual mortality rates in persistent (4.41%) and permanent AF (4.41%) compared with paroxysmal AF (2.99%). 14 More evidence for the role of AF pattern per se in mortality was provided by GARFIELD-AF, 17 where aHRs for mortality between persistent and paroxysmal AF and between permanent and paroxysmal AF were 1.38 (95% CI 1.23 to 1.54) and 1.63 (95% CI 1.46 to 1.81) respectively. However, a smaller study by Keating et al 9 presented a higher mortality risk in patients with paroxysmal AF compared with persistent AF. Permanent AF was found to have the poorest prognosis of all AF patterns in the latter study. In the SCAF trial, also, paroxysmal AF was associated with worse survival, but in multivariate analysis, this finding was no longer significant. 8
An electrophysiologic approach could explain the differences in all-cause mortality among AF patterns. A more discrete atrial electrical remodeling is presented in persistent AF, leading to the AF prolongation, 18 while the AF circle lasts less. 19 In addition, high LA pressures and structural heart disease are often encountered in more progressive AF patterns. The identification and focus on the distinct mechanisms, which are associated with increased mortality in AF patterns with an increased AF burden could contribute to the development of new more targeted treatments, and merits further research.
The relationship of AF burden to mortality and cardiovascular outcomes is little investigated by previous studies over the years. 20 Subjacent risk factors and comorbidities interrelate and are responsible for the atrial remodeling, resulting in AF development. They complicate the therapeutic strategies in AF, increase the risk of cardiovascular events and therefore deteriorate the prognosis. Consequently, the identification and intensive management of the cardiovascular comorbidities (arterial hypertension, diabetes mellitus, heart failure, coronary artery disease, sleep apnoea) as well as the targeted lifestyle modification (weight loss, abstinence from alcohol and physical exercise) could contribute to the reduction of AF burden and eliminate the progression of persistent AF to permanent AF. Progression to more permanent forms of AF could be halted by intensive rhythm control management or catheter ablation in selected patients, as outlined by recent guidelines. 1
Compared with first diagnosed AF, persistent or permanent AF exhibited a significant increase in the composite of CV mortality or hospitalizations in our cohort. Compared with paroxysmal AF, persistent or permanent AF did not yield significant differences. Data from previous studies are conflicting. The EORP-AF study did not detect any differences regarding the causes of death among AF patterns. 21 In contrast, GARFIELD-AF 17 and ENGAGE AF-TIMI 48 14 studies have correlated paroxysmal AF with a lower risk of CV mortality. Regarding hospitalizations, a recent small study suggested that persistent AF be associated with higher future AF-hospitalizations. 22 We consider our findings important from an epidemiological perspective, given the association of multiple hospitalizations with an impaired quality of life and an increased financial burden worldwide. 7,23 -25
No significant differences in stroke incidence were detected among all AF patterns. Our findings are in line with the so far established theory that the AF pattern is irrelevant to the thromboembolic events in patients with comorbid AF. Similar incidences of stroke between paroxysmal and permanent AF were found in SCAF (26 vs 29 events/1000 patient-years) 26 and SPAF (32 and 33 events/1000 patient-years) 27 studies. A substudy of ACTIVE-W 28 also concurred with our outcomes. In the GARFIELD-AF study, among non-anticoagulated patients, those with non-paroxysmal AF were associated with higher stroke risk, but among coagulated patients, the stroke risk was similar between paroxysmal and non-paroxysmal AF. 17 The increased stroke risk rates in paroxysmal AF found by older studies could be explained by the inadequate anticoagulation therapy in this particular AF pattern. AF burden, clinical risk factors, underlying comorbidities, and atrial pathology may be more important modifiers of quantification of thromboembolic risk compared to AF pattern. 29
In our cohort, no differences in anticoagulated patients between temporal AF patterns were found regarding the incidence of major bleeding events. Interesting but still unexplained is the fact that the paroxysmal AF pattern presented the highest rates of major bleeding (5.7%) during a median follow-up of 31 months. In ENGAGE AF-TIMI 48 Trial major bleeding rates were 2.86% in paroxysmal AF versus 2.65% in persistent and 2.73% in permanent AF annually. 14 Almost none of the previous studies has concluded in a different assumption. 14,17,30
Limitations
Firstly, the current study was not designed at the commencement of the MISOAC-AF trial and is subsequently retrospective and nonrandomized, restricted to a single center dataset. Additionally, the AF pattern classification was based on electrocardiographic data and physician-assigned diagnosis, which might have caused a classification bias. The AF pattern of each patient was, also, determined only once, on admission and progressions from one pattern to another were not detected and therefore not included in our study. Last, multivariate models were applied based on baseline characteristics, whereas variations in clinical features near to the time of death were not included. Although all outcomes were adjusted for confounding factors, we cannot exclude other confounders’ potential existence, not measured in our cohort.
Conclusion
In patients discharged from hospital with a primary or secondary diagnosis of AF, persistent or permanent AF was independently associated with a significant increase in all-cause mortality compared with paroxysmal AF. As compared with first diagnosed AF, persistent or permanent AF was also independently associated with higher risk of the composite of CV mortality or all-cause hospitalization. Stroke and major bleeding events appeared to be similar across AF patterns.
Supplemental Material
Supplemental Material, sj-docx-1-cpt-10.1177_10742484211069422 - Associations of Atrial Fibrillation Patterns With Mortality and Cardiovascular Events: Implications of the MISOAC-AF Trial
Supplemental Material, sj-docx-1-cpt-10.1177_10742484211069422 for Associations of Atrial Fibrillation Patterns With Mortality and Cardiovascular Events: Implications of the MISOAC-AF Trial by Amalia Baroutidou, Anastasios Kartas, Athanasios Samaras, Andreas S. Papazoglou, Eleni Vrana, Dimitrios V. Moysidis, Evangelos Akrivos, Anastasios Papanastasiou, Ioannis Vouloagkas, Michail Botis, Evangelos Liampas, Artemios G. Karagiannidis, Efstratios Karagiannidis, Georgios Efthimiadis, Haralambos Karvounis, Apostolos Tzikas and George Giannakoulas in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgments
Authors acknowledge Dimitra Vasdeki, George Dividis, George Fotos, Eleni Paschou, Evropi Forozidou, Paraskevi Tsoukra, Eleni Kotsi, and Ioannis Goulas, who contributed to data collection during the conduction of the MISOAC-AF trial.
Author Contributions
AB: Writing—Original draft; AK: Conceptualization, Writing—Review & Editing; AS: Software, Supervision; ASP: Writing—Review & Editing; EV: Writing—Review & Editing; DVM: Writing—Review & Editing; EA: Software, Formal analysis; IV: Resources; AP: Resources; MB: Resources; EL: Resources; AGK: Writing—Review & Editing; EK: Resources; GE: Conceptualization; HK: Conceptualization; AT: Conceptualization, Supervision; GG: Conceptualization, Project administration, Supervision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
