Abstract
The novel oral anticoagulants (NOACs) apixaban, dabigatran, and rivaroxaban have been recently indicated for stroke prevention in patients with atrial fibrillation (AF) . Due to a lack of direct head-to-head trials comparing the NOACs, the current systematic review and network meta-analysis (NMA) were conducted to assess their relative efficacy and safety. Three phase III randomized controlled trials enrolling 50 578 patients were included. Results of the NMA show a clear trend favoring NOACs over warfarin with regard to the key outcomes of stroke/systemic embolism and all-cause mortality, with apixaban also showing a favorable response for major bleeding and total discontinuations. Although there were few significant differences among the NOACS with regard to efficacy outcomes, apixaban and dabigatran 110 mg were associated with significantly lower hazards of major bleeding compared with dabigatran 150 mg and rivaroxaban. The NOACs offer a therapeutic advance over standard warfarin treatment in stoke prevention in patients with nonvalvular AF.
Keywords
Introduction
Atrial fibrillation (AF) is the most common clinically significant cardiac arrhythmia. 1 The incidence of AF rises with age and is estimated to range from 0.7% in persons aged around 60 years to 18% in persons aged 85 years and older. 2 It is estimated that the extended survival of the elderly population will lead to a rapid increase in AF prevalence over the coming decades. 3 Although AF is known to increase the risk of overall mortality by as much as 60% compared with the general population, 4 its most serious manifestation is through the increased risk of stroke.
AF increases the risk of stroke by approximately 5-fold, 5 and more than 20% of all strokes are attributed to this arrhythmia. 6 The risk of stroke is dependent on a number of factors and ranges from an annual risk of 1% in patients aged 65 years and older with no risk factors to over 12% per year in patients with multiple risk factors. 7 Such risk factors include age, hypertension, diabetes, heart failure, or history of prior stroke. Furthermore, the risk of recurrent stroke within 5 years of the first stroke is up to 43%. 7 The treatment and management of stroke is associated with a significant burden on health care systems worldwide, 8,9 highlighting the need for an effective treatment algorithm.
Vitamin K antagonists (VKA, eg, warfarin) have been the standard of care and have shown to be effective when well managed in warfarin-suitable patients in clinical trials. 10 However, to achieve optimal clinical effectiveness, warfarin requires frequent monitoring and dose adjustments to ensure patients remain within a narrow therapeutic window as defined by the international normalized ratio (INR 2.0-3.0), and as a result a significant proportion of patients (up to 50%) will not achieve a stable INR. 11–13 In addition, INR levels above and below the target range are associated with substantial increases in bleeding and thromboembolic risk, respectively. 14–16 From a patient’s perspective, successful treatment with warfarin requires following numerous food and alcohol restrictions as well as being aware of likely interactions with other medications. 17 Several studies have indicated that up to 50% of all eligible patients do not receive warfarin and such underuse is a major barrier to effective stroke prevention leading to unnecessary strokes and disability. 18–21 Although antiplatelet agents do not require similar monitoring as VKAs, they are less effective in reducing the risk of stroke compared with VKAs, and recent European guidelines recommend only aspirin in patients who cannot tolerate oral anticoagulation therapy. 22
The unmet need for a preventative treatment that is both efficacious and safe led to the introduction of the novel oral anticoagulants (NOACs): the direct factor Xa inhibitors apixaban and rivaroxaban and the direct thrombin inhibitor dabigatran etexilate. Both dabigatran and rivaroxaban have been approved by the Food and Drug administration (FDA) and European Medicines Agency (EMA) for stroke prevention in patients with AF. 23–26 Apixaban has recently been approved by both the FDA and in Europe for this indication.
Several large, phase III randomized controlled trials (RCTs) enrolling over 50 000 patients have indicated the substantial benefit of the NOACs in patients with AF when compared with warfarin. 27–29 All studies reported NOACs to be at least as efficacious and to have a similar 28,29 or more favorable 27 bleeding profile compared with warfarin.
Although there are no published head-to-head studies comparing the NOACs in stroke prevention in patients with nonvalvular AF, several meta-analyses have recently been published reporting on the relative efficacy and safety of the NOACs in this population. 30–33 The current systematic review and meta-analysis extends the published data with regard to the number of outcomes reported and the data sets employed (intention to treat vs on treatment) using a Bayesian network meta-analysis (NMA) approach.
Methods
A systematic review protocol was written to define all aspects of the review prior to commencement. The predefined inclusion criteria are reported in Table 1.
Summary of Inclusion and Exclusion Criteria for the Systematic Review.

Abbreviations: AF, atrial fibrillation; GI, gastrointestinal; VKA, vitamin K antagonist.
Search Strategy
The data sources to identify published studies and ongoing (as yet unpublished) studies included:
Electronic databases accessed via OVID and the Cochrane library:
Medline in process and Medline
EMBASE
CINAHL
BIOSIS
The Cochrane Library (the Cochrane Database of Systematic Reviews, Database of Abstracts of Reviews of Effects, Cochrane Central Register of Controlled Trials, and Health Technology Assessment Database).
Hand searching of reference lists of included studies.
Hand searching of studies included in previously conducted systematic reviews/meta-analyses.
The following conference proceedings were searched (2007-2012):
European Congress of Cardiology and meetings of the Joint Working Groups of the European Society of Cardiology
Scientific sessions of the American Heart Association
Heart Rhythm Society
Annual meeting of the American College of Cardiology.
The original search was conducted in May 2011. The same search was rerun in February 2012 to identify any studies entered into the databases from May 2011.
One reviewer screened the title and abstract of studies identified against the eligibility criteria. Full-text articles were then assessed to ensure studies met the criteria or for those studies where eligibility could not be determined from the title/abstract. Any uncertainties as to eligibility were referred to a second reviewer and resolved by consensus. Data were extracted from eligible publications into a predefined, data extraction table by one reviewer and verified by a second reviewer.
Quality Assessment
The quality of RCTs was assessed according to the methodology checklist detailed in appendix D of the National Institute for Health and Care Excellence (NICE) Guidelines Manual 2009. 34 The likelihood of selection, attrition, and detection and performance bias was assessed by 2 reviewers working independently. Disagreements were resolved by discussion and/or additional referees.
Network Meta-Analysis
In an NMA, treatment effects are calculated for all treatments using all available evidence in one simultaneous analysis. The NMA methods build on the principles of indirect comparisons and preserve the randomized comparisons within each trial. Both fixed- and random-effects models were fitted to the data for the NMA. The “goodness of fit” of each model was tested by calculating the residual deviance and the Deviance Information Criterion (DIC). 35 In the current analysis, the DIC values were similar for both the fixed- and random-effects models for each outcome, and so there was little difference in the model fit to the data. It is recommended that calculations investigating heterogeneity should be based on at least 10 studies, and in analyses where there are too few studies the random-effects model will produce poor estimates of the variation in between-study treatment effects. 36 Therefore, given that the current network only included 3 RCTs, only the fixed-effects model was considered to provide relevant estimates of treatment effects and uncertainty and is reported in the current publication.
WinBUGS software (MRC Biostatistics Unit, Cambridge, UK) was used to conduct Bayesian NMA. A Poisson likelihood with a log link was used to calculate the hazard ratio (HR) for all treatments compared with other treatments 37
Point estimates and 95% credible intervals for HRs using Markov Chain Monte Carlo methods were modeled. Vague or flat priors, such as N (0, 100), were assigned for basic parameters throughout. 37 To ensure convergence was reached, trace plots were assessed. 38
After running the WinBUGS models for 1 00 000 iterations to ensure the model had converged, the HR (for event rates) for each of the next 20 000 simulations was estimated, and the point estimate of the calculated data was taken to be the median of the 20 000 simulations and the 95% credible intervals for the calculated data were taken from between the 2.5th and 97.5th percentiles from the distribution of the calculated data. The assumption of consistency between the direct and the indirect evidence could not be assessed formally in the full NMA network, as there were no independent loops of evidence in the network.
Data Source
The analysis of event rates considered the total number of events that occurred during the patients’ exposure to the risk (total events across all patients divided by total patient-years exposed). This allowed for cases where the patient may have experienced the event more than once and for differences in trial follow-up. If event rates were not reported in the publication, the probability of experiencing an outcome at the median follow-up point was calculated by dividing the number of first events by the number of patients randomized and converting this probability into an annual rate as:
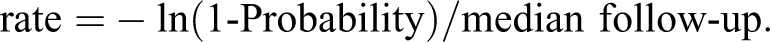
This approximation accurately predicted the event rate for studies, where both the rate and the number of patients with events were reported.
The following data were used for the base case analyses:
RE-LY 2010 data from the updated publication reporting additional outcome events identified during routine clinical site closure visits. 39 This was deemed to be a more appropriate data source compared with the original 2009 publication 28 in which several efficacy and safety outcomes may have been underreported. Additional events were reported for the following outcomes: stroke or systemic embolism (SE), total stroke, myocardial infarction (MI), intracerebral hemorrhage (ICH), major bleeding, and any bleed (Table 2).
ROCKET intention to treat (ITT) efficacy data—Analyses of efficacy in the primary ROCKET-AF trial publication were conducted on a per protocol (PP) population to demonstrate noninferiority, with superiority and safety analyses were run on the on treatment (OT) population. 29 However, in the base case for the current NMA, the ITT event-rate data were sourced for hemorrhagic stroke, ischemic stroke, non–central nervous system embolism, MI, and all-cause mortality (Table 2). This has been accepted by the FDA and the EMA as the most appropriate efficacy analysis. 43 The ITT event-rate data were not available for fatal stroke, disabling stroke, and nondisabling stroke, and OT data were used for these outcomes.
Outcomes Analyzed in NMA and Additional Data Sources.a

Abbreviations: CRNM, clinically relevant non-major; FDA, Food and Drug Administration; NMA, network meta-analysis; ITT, intention to treat.
a ✓ Shows data identified for that outcome.
c Calculated by adding adjudicated minimal bleeding event rates reported in the FDA briefing document for rivaroxaban 42 to other bleeding event rates.
d RE-LY update publication (Connolly et al 2010) reported additional primary efficacy outcome events recorded during routine clinical site closure visits after the database was locked. 39
e ITT data reported in online appendix of publication.
Table 2 shows the outcomes for which additional data not available in the primary publication were identified (represented by ✓ ) and the source of this data (in the table note). The data used in the base case analysis for each outcome are presented in Supplementary Table 1, together with the dataset used for each analysis.
Data Assumptions for NMA
For several outcomes, assumptions were made in order to calculate the relevant event-rate data. These are detailed below:
Any bleed: This outcome is an important composite end point and is presented for apixaban and dabigatran. Given the omission of the any bleed outcome reported from the ROCKET-AF publication, 29 the authors calculated this by adding adjudicated minimal bleeding event rates reported in the FDA briefing document for rivaroxaban 42 to other bleeding event rates. The authors are aware that this may overestimate the total number of “any bleeding” events in the rivaroxaban study; however, it is the best available estimate.
Disabling stroke: To obtain data for the RE-LY and ARISTOTLE studies, the incidence of “fatal stroke” was subtracted from the incidence of “fatal or disabling stroke.”
Nondisabling stroke: To obtain data for the ARISTOTLE study, the incidence of “disabling or fatal stroke” was subtracted from the incidence of “total stroke.” Since this trial investigated first stroke events, the likelihood of double counting was very small.
“Other” major bleed: this outcome was calculated for the included studies by subtracting the incidence of ICH from the incidence of major bleeding.
Sensitivity Analyses
The following sensitivity analyses were considered for each outcome (where data were available):
Substitution of the RE-LY 2010 efficacy data (from the update 39 ) with the RE-LY 2009 data from the primary publication 28 for applicable outcomes.
Substitution of the ROCKET-AF efficacy ITT data with ROCKET-AF efficacy OT data (from the primary publication 29 ).
The data used in the sensitivity analyses are presented in Supplementary Table 2.
Results
Systematic Review Results
A total of 4596 potentially relevant publications were identified for inclusion in the systematic review, of which 4370 were excluded on the basis of title and abstract. Upon reapplication of the inclusion criteria to the full text of the 226 remaining articles, a further 184 were excluded. Finally, 56 publications, detailing 41 RCTs, met the inclusion criteria (Figure 1).
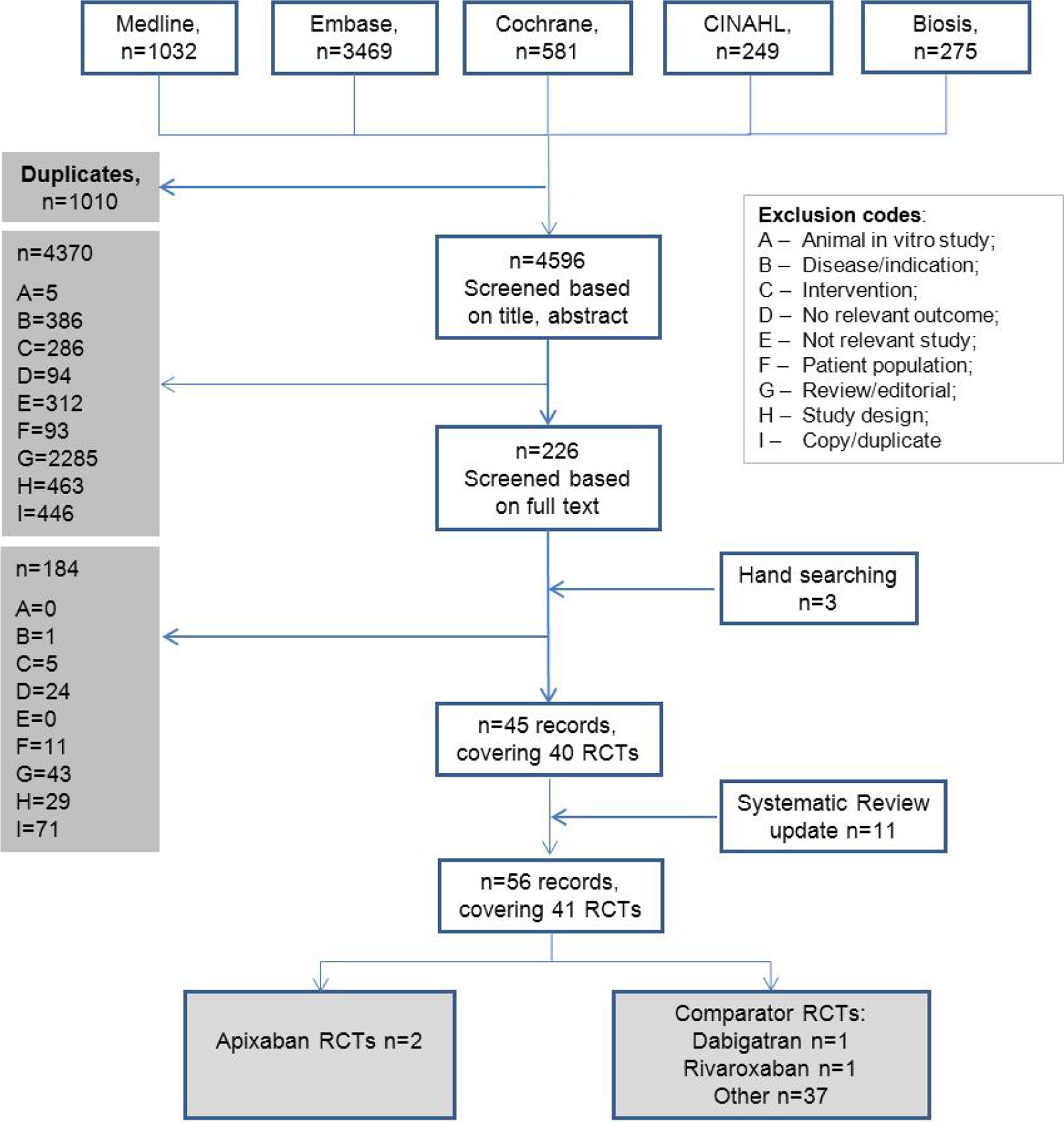
Schematic for the systematic review of clinical evidence.
Description of Studies/Key Findings of the Review
Table 3 indicates that there were several potential sources of heterogeneity among the 41 studies included in the review. These included:
Summary of Studies Meeting Inclusion Criteria for the Systematic Review.
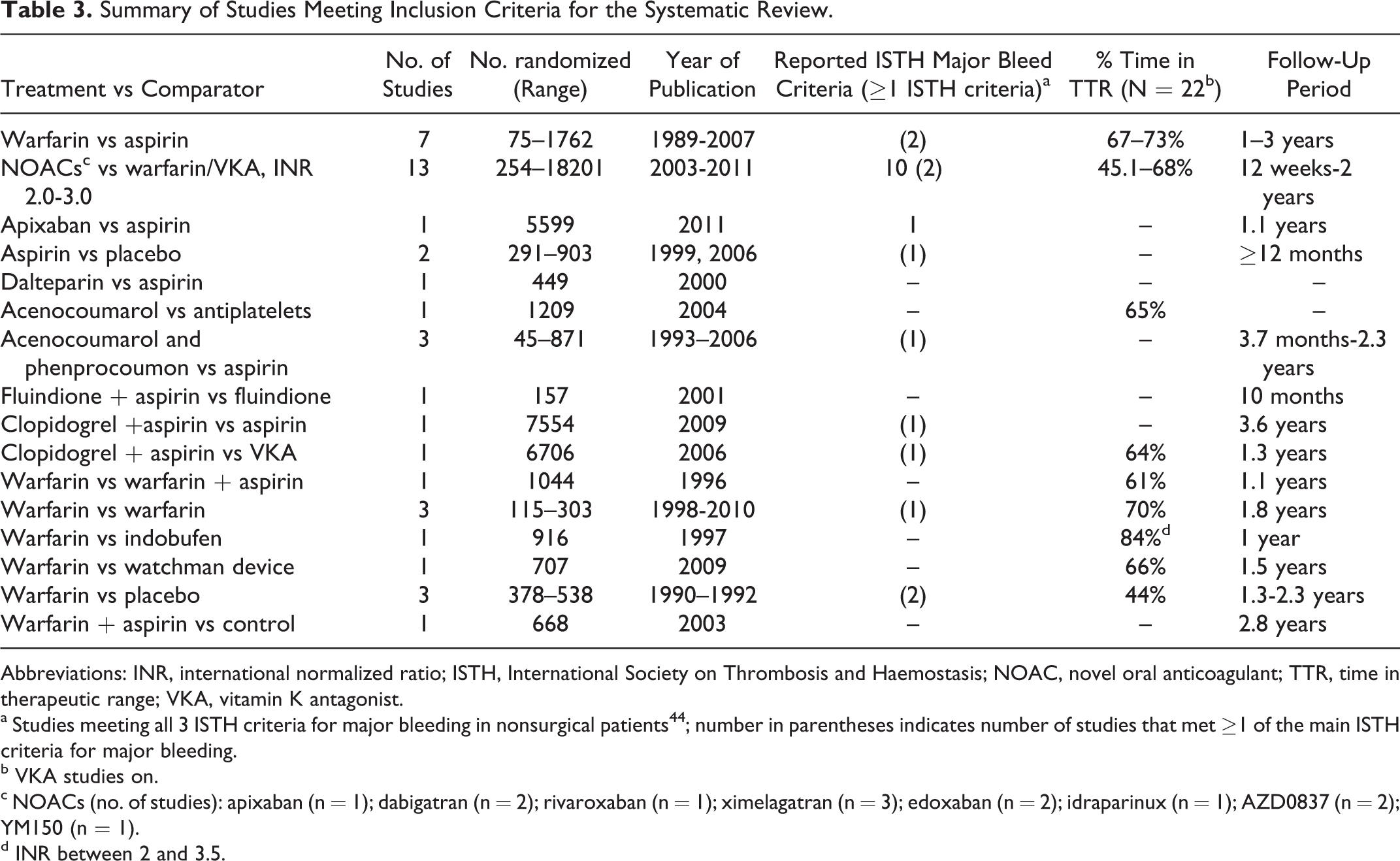
Abbreviations: INR, international normalized ratio; ISTH, International Society on Thrombosis and Haemostasis; NOAC, novel oral anticoagulant; TTR, time in therapeutic range; VKA, vitamin K antagonist.
a Studies meeting all 3 ISTH criteria for major bleeding in nonsurgical patients 44 ; number in parentheses indicates number of studies that met ≥1 of the main ISTH criteria for major bleeding.
b VKA studies on.
c NOACs (no. of studies): apixaban (n = 1); dabigatran (n = 2); rivaroxaban (n = 1); ximelagatran (n = 3); edoxaban (n = 2); idraparinux (n = 1); AZD0837 (n = 2); YM150 (n = 1).
d INR between 2 and 3.5.
Year of publication: Although the majority of the studies (including all NOAC studies) were published post-2000 (n = 28), 12 studies were published between 1990 and 1999, with a single study published prior to 1990.
Follow-up period that ranged from 12 weeks to 3.6 years.
Number of patients randomized that ranged from 45 to 18 201 patients.
Warfarin eligibility: Of the 41 studies, 34 (83%) were conducted in patients who were eligible for warfarin use, compared with 4 studies that enrolled patients considered unsuitable for warfarin use (eg, INR not maintained in therapeutic range or intolerance).
Study design: In all, 21 studies employed either a double- (n = 19) or single-blind design (n = 2), compared with a further 19 that had an open-label design.
Time in therapeutic range (TTR) ranged from 43.7% (INR 2.0-3.0) to 83.5% (INR 2.0-3.5).
Primary analysis: Noninferiority or superiority and definitions used for reporting noninferiority or superiority.
Definitions used for efficacy/bleeding outcomes: The diversity in the reporting of outcomes adds to the difficulty in comparing data between trials. In particular, several different bleeding scores were reported across the included studies, each employing a different definition and resulting in unique separation into low-, moderate-, and high-risk strata. In 2005, the International Society on Thrombosis and Haemostasis (ISTH) introduced a uniform definition of major bleeding consisting of 3 criteria.
44
The 41 eligible studies conformed as follows: Meeting all 3 ISTH criteria (n = 11) Meeting ≥1 ISTH criteria (n = 11) Other definition (n = 10) Definition not reported (n = 9)
Patient characteristics: There were several additional factors that could influence patient outcome and that varied across trials: stroke severity (assessed on CHADS2 scale) types of AF (paroxysmal, persistent, or permanent) age presence of comorbidities use of concomitant antithrombotic medication (eg, antiplatelet therapy).
A consequence of these between-study differences is that a broad meta-analysis including all potential studies reporting data may introduce additional statistical heterogeneity into the NMA for a particular outcome. To avoid this, we restricted the network to phase III studies with a NOAC treatment arm conducted in a warfarin-eligible population. Therefore, of the 41 eligible RCTs, only 3 phase III studies compared a NOAC of interest with warfarin: the ARISTOTLE trial evaluated apixaban, 27 ROCKET-AF investigated rivaroxaban, 29 and dabigatran was evaluated in RE-LY. 28 The network diagram is shown in Figure 2.
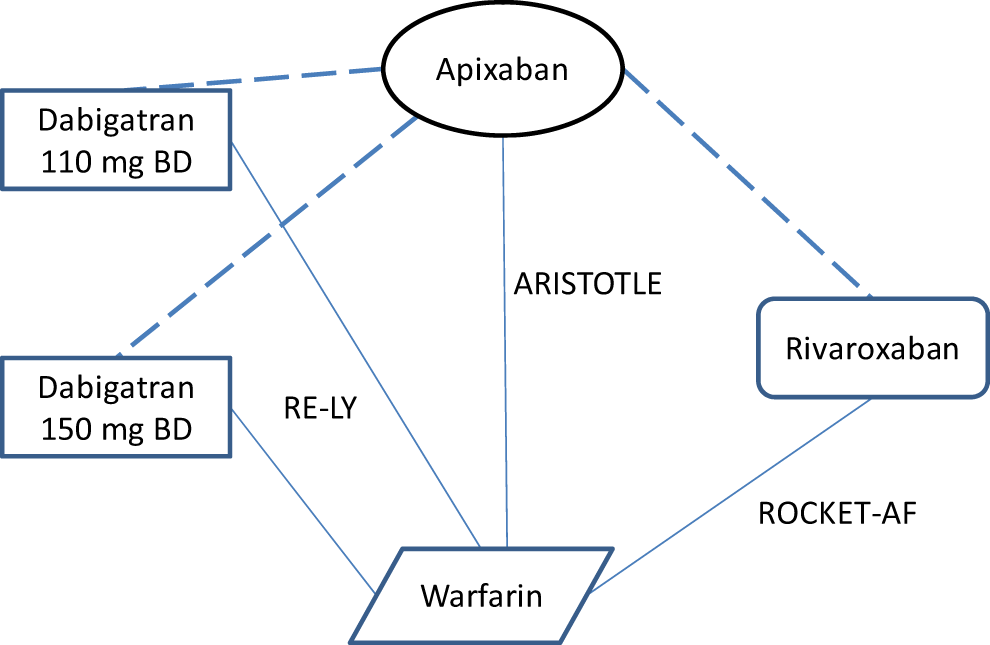
Network diagram for the warfarin-eligible population.
Trial design and baseline characteristics are shown in Table 4. Although the trials were all multinational RCTs and were broadly similar in terms of several patient characteristics and reported outcomes (eg, major bleeding definition), there were a number of differences in the design of the included studies:
Summary of the Trials Used to Conduct the NMAs.
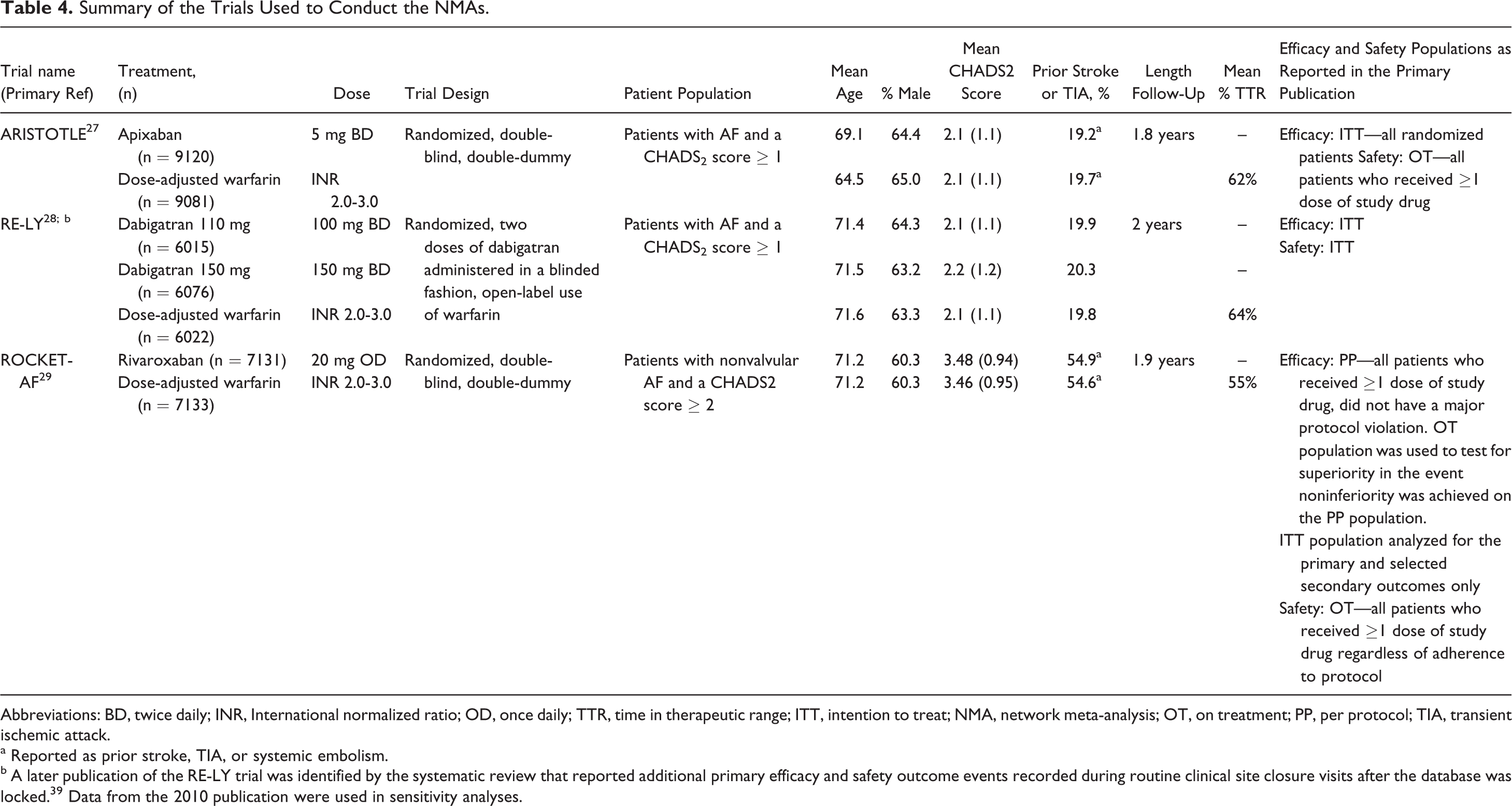
Abbreviations: BD, twice daily; INR, International normalized ratio; OD, once daily; TTR, time in therapeutic range; ITT, intention to treat; NMA, network meta-analysis; OT, on treatment; PP, per protocol; TIA, transient ischemic attack.
a Reported as prior stroke, TIA, or systemic embolism.
b A later publication of the RE-LY trial was identified by the systematic review that reported additional primary efficacy and safety outcome events recorded during routine clinical site closure visits after the database was locked. 39 Data from the 2010 publication were used in sensitivity analyses.
The ARISTOTLE and ROCKET-AF trials were double-blind, double-dummy studies. In contrast in the RE-LY study, the assignments to dabigatran or warfarin were open label.
Patients were enrolled in the ARISTOTLE and RE-LY studies if they had a CHADS2 score of ≥1, whereas ROCKET-AF enrolled a higher risk population (CHADS2 ≥ 2, with >90% CHADS2 ≥ 3). At baseline, the mean CHADS2 score for the ROCKET-AF study was 3.58 compared with 2.1 for both RE-LY and ARISTOTLE.
The mean percentage of time in which the INR was in the TTR of 2.0 to 3.0 for warfarin was 64% in the RE-LY trial, 62% in the ARISTOTLE trial, and 55% in the ROCKET-AF trial.
As discussed earlier, the RE-LY study publication based all efficacy and safety analyses on the ITT principle. In the ARISTOTLE study publication, efficacy analyses were conducted on the ITT population and safety analyses on the OT, safety population. Analyses of efficacy in the ROCKET-AF trial publication were conducted on a PP population to demonstrate noninferiority, with superiority and safety analyses run on the OT population. Efficacy analyses were also conducted on the ITT population.
The NMA Comparison of NOACs With Warfarin
Results are reported in Table 5 and for the key outcomes of stroke/SE, major bleeding, all-cause mortality, and discontinuations results are displayed in Figure 3. (For completeness, results for the opposite treatment comparisons to those reported in Table 5 are reported in Supplementary Tables 3 and 4.) Comparing the efficacy of NOACs with warfarin, both apixaban and dabigatran 150 mg showed significantly reduced HRs for the primary outcome of stroke/SE, in addition to any stroke and fatal stroke. Although the hazards were lower for rivaroxaban and dabigatran 110 mg compared with warfarin for these outcomes, they did not reach statistical significance. The reduction in stroke was primarily driven by a significant reduction in hemorrhagic stroke for all the NOACs, with dabigatran 150 mg the only NOAC to also report a significant reduction in ischemic stroke. All-cause mortality was lower for all NOACs compared with warfarin, and this approached statistical significance for apixaban.
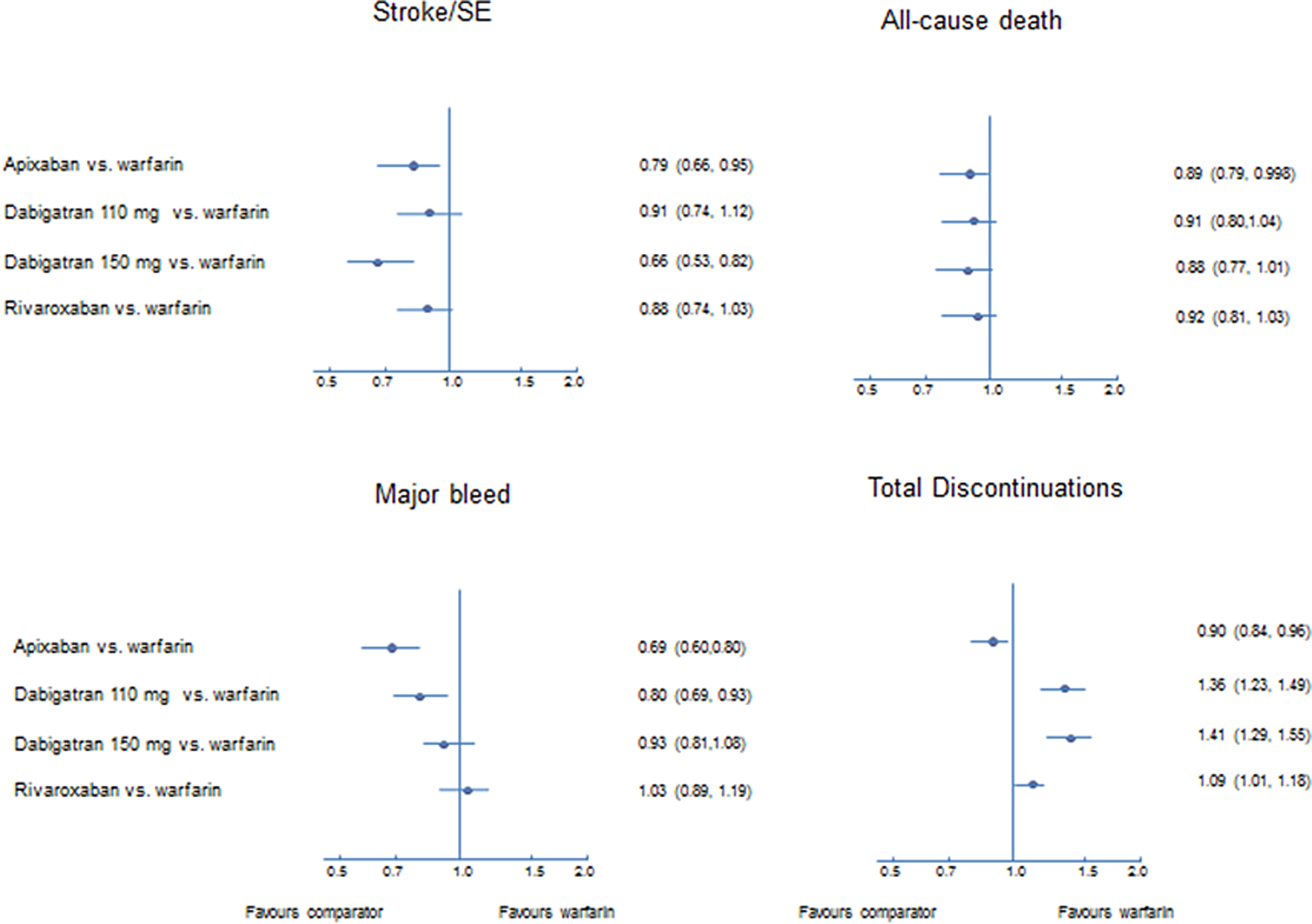
Direct comparison results versus warfarin for 4 key outcomes.
Results of the Base Case Analysis.a
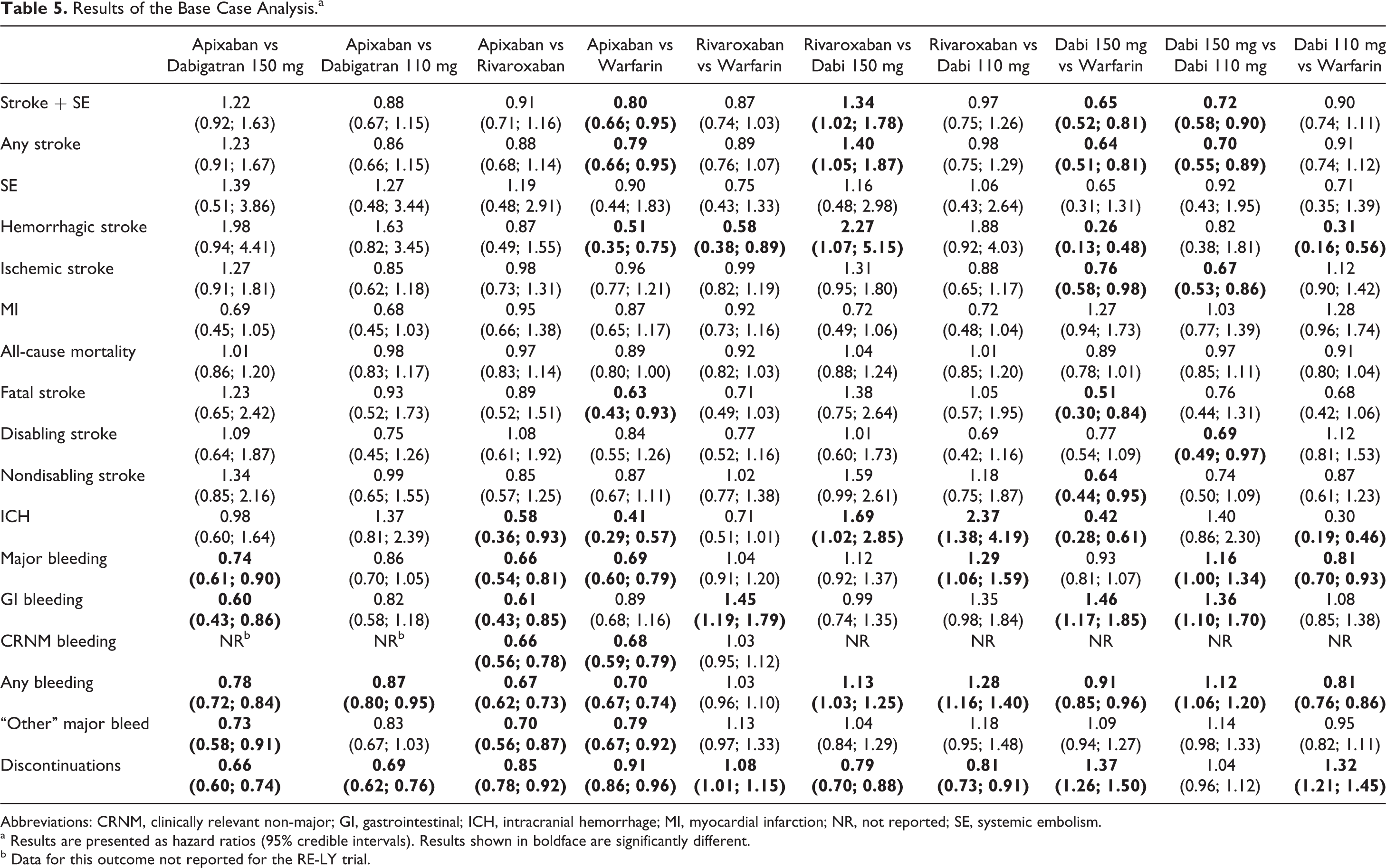
Abbreviations: CRNM, clinically relevant non-major; GI, gastrointestinal; ICH, intracranial hemorrhage; MI, myocardial infarction; NR, not reported; SE, systemic embolism.
a Results are presented as hazard ratios (95% credible intervals). Results shown in boldface are significantly different.
b Data for this outcome not reported for the RE-LY trial.
Regarding safety outcomes, the hazard of major bleeding or any bleed was significantly lower for both apixaban and dabigatran 110 mg compared with warfarin (dabigatran 150 mg reported significantly lower hazards for any bleed only). Rivaroxaban and dabigatran 150 mg were associated with significantly higher hazards of gastrointestinal (GI) bleeding compared with warfarin. With the exception of rivaroxaban, the remaining NOACs were associated with significantly lower hazards of ICH. Finally, apixaban was the only NOAC associated with significantly lower hazards of discontinuation compared with warfarin (significantly higher hazards reported for rivaroxaban and both doses of dabigatran).
NOAC Versus NOAC
Results from the NMA comparing the 3 different NOACs are reported in Table 5. The hazards of a stroke/SE, any stroke, or hemorrhagic stroke were significantly higher for rivaroxaban compared with dabigatran 150 mg. Treatment with dabigatran 110 mg was associated with a significantly higher hazard of a stroke/SE, any stroke, disabling stroke, or ischemic stroke compared with dabigatran 150 mg. There were no other statistically significant differences among the NOACs for the efficacy outcomes analyzed; and for the outcome of all-cause mortality, there was little difference observed between the treatments.
Considering bleeding events, apixaban and dabigatran 110 mg were associated with significantly lower hazards of any bleed compared with rivaroxaban. In addition, the hazards of major bleed, GI bleed, and “other” major bleed were significantly reduced following treatment with apixaban when compared with dabigatran 150 mg twice daily or rivaroxaban and nonsignificantly reduced compared with dabigatran 110 mg (significantly reduced for any bleed). Rivaroxaban and dabigatran 150 mg were also associated with significantly higher odds of major bleeding compared with dabigatran 110 mg. The hazards of ICH were significantly lower for apixaban and both doses of dabigatran compared with rivaroxaban. Apixaban also reported a statistically significant lower incidence of clinically relevant non-major bleeding compared with rivaroxaban (comparison with dabigatran not possible due to lack of data). Apixaban was significantly better than all the comparators for the outcome of total discontinuations (confirming the findings from direct analysis vs warfarin). Rivaroxaban treatment was associated with significantly lower hazards of discontinuation compared with both doses of dabigatran.
The results of the sensitivity analyses support the base case conclusions, except for the MI outcome when the RE-LY 2009 data 28 were substituted for the RE-LY 2010 data. 39 In this sensitivity analysis, the incidence of MI was statistically significantly lower for apixaban compared with both doses of dabigatran (data reported in Supplementary Table 5).
Discussion
The aim of the current systematic review and meta-analysis was to compare the efficacy and safety of treatments for the management of patients with mild-to-moderate AF at increased risk of developing a stroke, with a particular focus on the NOACs. The NMA is required to compare the NOACs, as there are no head-to-head studies, and such a trial is unlikely due to the large number of patients that would be required and the resulting expense. An extensive systematic review was conducted to ensure identification of all relevant RCTs. Although 41 studies met the eligibility criteria for inclusion in the systematic review, there were considerable differences between studies in terms of trial design, patient characteristics (eg, stroke severity, age, presence of comorbidities, and use of concomitant antithrombotic medication), and definition of reported outcomes. Eligible studies were published over a 2-decade period, and the treatment pathway for management of patients with AF has evolved considerably over this timescale. 45 In addition, the TTR varied across the studies ranging from 43.7% (INR 2.0-3.0) to 83.5% (INR 2.0-3.5). A relationship between poor therapeutic range and an increase in the incidence of vascular events and major bleeding in patients receiving anticoagulation has been reported in several studies. 46,47 Finally, the diversity in the reporting of outcomes adds to the difficulty in comparing data between trials that have not defined outcomes using the same criteria. In particular, several different bleeding scores were reported across the included studies, each employing a different definition and resulting in unique separation into low-, moderate-, and high-risk strata. 48,49
Any NMA has the underlying assumption that trials and outcomes are sufficiently similar to allow for data to be pooled, and the consistency assumption relies on there being no imbalance in modifiers of relative treatment effects across the studies. The presence of significant methodological or clinical heterogeneity between the studies included in any meta-analysis may limit the ability to make a robust decision about the relative efficacy of treatments. Indeed in the recent NICE submission for rivaroxaban, the Evidence Review Group raised concerns about the validity of the results from the NMA, due to the high levels of statistical heterogeneity and recommended the use of a restricted network limited to comparators of interest only in order to minimize heterogeneity. 50 Therefore, in the current analysis, the decision was taken to restrict the NMA network to the comparators of interest in a warfarin-eligible population. This led to the inclusion of 3 RCTs in the network, 27–29 and use of this restricted analysis may reduce the degree of heterogeneity observed in the NMA.
In the current NMA, the similarity assumption was supported by the eligibility criteria applied for the selection of studies. There remained, however, study differences between the included RCTs, which highlight the challenges associated with cross-trial indirect comparisons in this indication. However, although these differences might present a challenge to the similarity assumption, it does, however, better support the external validity of these results as this variation in patient populations is more likely to reflect real-world practice. One of these issues related to the open-label design of RE-LY compared with both ARISTOTLE and ROCKET-AF, which were double-blind, double-dummy studies. Studies employing an open-label design may be subject to bias and overestimate the effect of the treatment being studied. 51,52 A recently published analysis has attempted to correct for the potential bias of the open-label design in RE-LY 53 and reported that following correction the magnitude of difference between apixaban and dabigatran 150 mg for the outcomes of stroke/SE and ischemic stroke was reduced and closer to parity.
It is generally accepted that in stroke prevention in patients with AF, higher efficacy is associated with a higher incidence of major bleeding events. 54 However, major bleeds are not simply benign events with no clinical consequences. Indeed, a reduction in bleeding is of particular clinical significance as there is increasing evidence to support a significant association between an in-hospital major/GI bleeding event and short- and long-term (up to 4 years) adverse prognosis as indicated by an increased risk of ischemic complications (eg, MI and stroke) as well as death in patients with cardiovascular disease 55–57 including those with AF. 58 Therefore, when assessing new interventions in this indication, it is prudent to consider their effect on both stroke prevention and bleeding outcomes simultaneously and not in isolation to achieve a more accurate assessment of their net clinical benefit. In this case, apixaban may be considered to have the efficacy advantage of dabigatran 150 mg combined with the bleeding outcomes of the 110 mg dose.
Results from the NMA indicate that compared with warfarin, only apixaban and dabigatran 150 mg have demonstrated significantly better results for the key outcome of stroke/SE, suggesting that these treatments hold promise in patients who are at high risk of stroke/SE. Similarly, apixaban and dabigatran 110 mg were reported to be the interventions with the lowest incidence of major bleeding, thereby providing a better alternative to the prevailing interventions in patients with AF, with a high risk of major bleeding. 59 Discontinuation rates were significantly in favor of apixaban compared with both warfarin and the other NOACs (discontinuation rates were statistically higher for rivaroxaban and both dabigatran doses compared with warfarin), which may imply apixaban may be better tolerated compared with other NOACs in dealing with chronic conditions like stroke prevention in AF, where long-term patient compliance with treatment is of critical importance. Results from the current NMA indirect comparison of the 3 NOACs indicate that treatment with apixaban was associated with significantly less major bleeding and GI bleeds compared with dabigatran 150 mg twice daily or rivaroxaban and nonsignificantly less compared with dabigatran 110 mg.
Data from the currently published studies indicate that use of the NOACs could represent a major advance in stroke prevention in patients with AF, as results from the present analysis indicate a trend toward both increased efficacy and safety (ie, reduction in bleeding events compared with warfarin). These results confirm the findings from other recently published meta-analyses 30–33 , assessing the relative efficacy and safety of the NOACs and extended the findings to additional efficacy and safety outcomes and the use of additional data sets (eg, ITT efficacy data for ROCKET-AF). Among the 3 NOACs included in the NMA, apixaban appears to be the most promising as a result of the favorable outcome with regard to preventing stroke and reducing bleeding events in warfarin-eligible patients with AF. In addition, although not considered as part of the current network, results from the AVERROES trial indicate that apixaban is also associated with a significantly reduced incidence of stroke/SE (and no difference in incidence of major bleed) in comparison with aspirin in patients who are judged to be unsuitable for oral anticoagulation with warfarin. 60 Long-term follow-up data from real-world studies and ongoing surveillance is required to confirm the current published evidence and to help define the place of the NOACs in clinical practice.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IL is an employee of Pfizer Ltd. SM, SR, and MO were paid consultants to Bristol Myers Squibb in connection with conducting the meta-analysis and development of the manuscript. PD, DJ, and TS are employees of Bristol Myers Squibb.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Bristol Myers Squibb.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
