Abstract
Anticoagulation using intravenous bolus administration of unfractionated heparin (UFH) aims to prevent thromboembolic complications in children undergoing cardiac catheterization (CC). Optimal UFH dosage is needed to reduce bleeding complications. We analyzed the effect of bolus UFH on activated clotting time (ACT) in children undergoing CC focusing on age-dependent, anesthesia-related, or disease-related influencing factors. This retrospective single-center study of 183 pediatric patients receiving UFH during CC analyzed ACT measured at the end of CC. After bolus administration of 100 IU UFH/kg body weight, ACT values between 105 and 488 seconds were reached. Seventy-two percent were within target level of 160 to 240 seconds. Age-dependent differences were not obtained (P = .407). The ACT values were lower due to hemodilution (total fluid and crystalloid administration during CC, both P < .001), with premedication of acetylsalicylic acid (P = .014) and low-molecular-weight heparin (P = .049). Arterial thrombosis (3.85%), venous thrombosis (0.55%), and bleeding (1.65%) following CC did not correlate with ACT values but occurred more frequently in children between 1 month and 1 year of age (91%). In conclusion, with a bolus of 100 IU UFH/kg, an ACT target level of 160 to 240 seconds can be achieved during CC in children in 72%, which is influenced by hemodilution and anticoagulant and antiplatelet premedication but not by age.
Introduction
Cardiac catheterization (CC) and cardiovascular surgery are needed for the diagnosis and treatment of congenital heart disease (CHD) in children. Not only cardiopulmonary bypass (CPB) but also vascular puncture and access by vascular sheath as well as endovascular and endocardial catheter manipulation activate blood coagulation. The use of foreign material, either temporary or permanent, including catheters and wires during diagnostic and interventional CC is a procoagulatory stimulus with the risk of endovascular or endocardial thrombosis and subsequent embolism. 1,2 Especially during and after interventional CC, the risk of intra- and extracardial thrombus formation and embolism remains one of the most severe complications using stents and other permanent implanted devices. 3 Therefore, anticoagulant treatment during CC is indicated to prevent thromboembolic complications. Nowadays, intravenous (IV) bolus administration of unfractionated heparin (UFH) is frequently used. 4 However, major side effects of heparin therapy include bleeding complications. 5,6 Due to an ongoing maturation process of hemostatic system, children are subject to higher bleeding risk compared to adults during and after CC. 7 –9 The dosage of heparin has to be chosen carefully, therefore, the blood coagulation system during CC can be monitored by measurement of activated clotting time (ACT). 10 –13 First described by Hattersley, ACT is the time, measured in seconds, until appearing of blood clots after incubating entire blood with an activator of the intrinsic coagulation pathway. 14 Multiple systems, using different activators and different methods for sensing blood clotting, are available.
In this study, we evaluated the hemostatic effect of bolus administration of UFH, consisting of 100 IU/kg body weight, on ACT during CC in children. The aim of the study was to search identify factors influencing ACT such as age-dependency, anesthesia-, disease-related factors, and laboratory parameters such as full blood cell count, liver enzymes, total fluid volume administration during CC, and anticoagulant or antiplatelet medication before CC.
Materials and Methods
Study Design
This is a retrospective single-center case series analyzing the effect of UFH on ACT during CC in children. We considered all consecutive patients undergoing elective CC between April 2012 and October 2013 at the University Children’s Hospital Zurich. Correspondent to our standard anticoagulation protocol, a single IV bolus of 100 IU/kg body weight (maximum 5000 IU) of UFH was administered after successful vascular puncture and vascular sheath access. The ACT was routinely measured at the end of CC to help determine further anticoagulant treatment. This can include another bolus administration of UFH or usually consists of a temporary continuation of UFH or low-molecular-weight heparin (LMWH) after CC for 24 to 36 hours, depending on ACT, duration of intervention, and individual estimated risk of thromboembolic events.
Only patients receiving 1 single dose of UFH during CC, no other than 100 IU/kg body weight, were included. Patients short time after cardiovascular surgery or with severe coagulation disorders were excluded. Patients were analyzed regarding possible influencing factors on ACT such as age, hemodilution (measuring hemoglobin [Hb], hematocrit [HCT], platelet count [PLT], and fluid infusion management during anesthesia), liver dysfunction, use of anticoagulant and antiplatelet medications before CC, and type of CHD (cyanotic and noncyanotic). Therefore, data were taken from medical records of all included patients.
Anesthesia and CC
Before CC laboratory values, including Hb, HCT, PLT, alanine aminotransferase (ALT), and aspartate aminotransferase (AST), assays measuring coagulation such as activated partial thromboplastin time, international normalized ratio (INR), prothrombin time and plasma fibrinogen concentration were determined routinely.
All CCs were performed under general anesthesia. Children aged ≥6 months received oral or rectal application of midazolam for premedication. General anesthesia was induced by inhalation of sevoflurane or via IV injection of etomidate. Endotracheal intubation was performed after muscle relaxation; anesthesia was then maintained using sevoflurane in oxygen/air or oxygen/nitrous oxide mixture, alfentanil, and atracurium. General IV fluid administration was by ringer acetate with 1% glucose (10 mL kg−1 h−1) but was adapted according to the individual patient’s requirements in case of hypovolemia or blood loss. If circulatory support during CC was needed, catecholamines such as phenylephrine or adrenaline were applied. Prophylactic antibiotics (cefazolin) were routinely given when foreign body material such as intracardiac devices was implanted during intervention. Depending on indication, medication for postoperative nausea and vomiting prophylaxis was also given to the patient (dexamethasone and ondansetron).
After skin disinfection, sterile coverage, and arterial or venous puncture, vascular sheaths were introduced using Seldinger technique. Immediately after vascular access, 100 IU/kg body weight of UFH was administrated IV. Iodinated contrast medium was given for angiography. Depending on the indication for CC diagnostic, investigations or interventions were carried out.
Activated Clotting Time Measurement
The ACT values were measured routinely at the end of CC using low-range cartridges of HemoTec ACT II (Medtronic Inc, Minneapolis, MN, USA). Analyses were obtained according to the manufacturer’s instructions. HemoTec is an automated ACT measurement system using whole blood samples. Exact time of heparin administration and time of ACT measurement were recorded in the CC and anesthesia protocol.
After filling a 2-cartridge system of the HemoTec ACT II with the patients’ blood sample, the patients’ blood is exposed to an activator substance (kaolin) and mixed by a plunger. Both chemical and mechanical exposures lead to the activation of the intrinsic coagulation pathway through contact activation. The fibrin clotting in the ACT device will have resultant plunger’s decrease. 13
Factors Influencing ACT Values
Laboratory data (Hb, HCT, PLT, ALT, and AST) were collected from medical records according to the correspondent CC date. Medications taken before CC including anticoagulant and antiplatelet drugs such as oral anticoagulant drugs (OACs), LMWH, and acetylsalicylic acid (ASA) were also extracted from medical records. According to our institutional protocol, OAC is routinely stopped 2 days before CC and antagonized with vitamin K analogs if INR before intervention is still over 2, LMWH is stopped 12 hours before CC, and ASA is not stopped before CC. Amount of total IV fluid intake during CC and consequent hemodilution were analyzed from anesthesia protocols. Exact amount of IV fluid administration, type of fluid such as colloid, crystalloid, red blood cell products (no other blood products were used), and the total volume, and the time between UFH administration and ACT measurement were evaluated.
Femoral Vessel Thrombosis and Bleeding Complications
Complications such as femoral vessel thrombosis (arterial and venous) and severe bleeding were analyzed within 24 hours after CC. Femoral vessel thrombosis were evaluated by vascular ultrasound and defined as a complete vascular closure of femoral arterial or venous vessels. Severe bleeding was defined as significant decrease in Hb concentration, hemodynamical relevance, and/or need of blood transfusion.
Statistics
Data are presented as mean ± standard deviation or median and range, as appropriate. For statistical analysis, SPSS (version 22.0, Chicago, Illinois) was used. A P value of ≤.05 was considered statistically significant. To detect influencing factors on ACT, nonparametric Kruskal-Wallis 1-way analyses of variance and Mann-Whitney U test (for age groups and treatment with ASA, LMWH, or OAC), as well as analysis of variance and regression analyses (for Hb, HCT, PLT, ALT/AST, and total fluid volume administration), were used. Correlation between ACT and factors possibly affecting ACT were calculated using Spearman rank correlation (ρ). Frequency analyses were used to evaluate correlation between ACT values and complications such as arterial or venous vessel thrombosis and bleeding complications after CC.
Ethics
The study was approved by the local ethical committee. The study was carried out as a data analysis.
Results
Patients
Between April 2012 and October 2013, 186 patients underwent CC at our institution. Twelve patients were not included in this study due to UFH administration different than 1 single bolus of 100 IU/kg body weight. A total of 174 patients (86 females and 88 males) were included into final analysis. Nine patients, who had CCs 2 times within this time period, appear twice in the study population, therefore, a total of 183 CCs performed within this period were included. The CC was performed at a median age of 2.8 years (range: 3 days-18.9 years), with a body weight of 12.6 kg (2.5-90 kg) and a body length of 90 cm (45-180 cm). The main cardiac diagnoses (at the time of CC) of all patients are summarized in Table 1. Of all, 49.4% of patients had noncyanotic CHD, 42% had cyanotic CHD, and 8.6% had other acquired cardiac diagnosis. However, some patients had combined cardiac diagnosis. Further associated diagnosis included different types of heterotaxy syndrome such as left isomerism (n = 2), situs inversus totalis with levocardia (n = 1) and dextrocardia (n = 2), cardiomyopathy (n = 1), and Scimitar syndrome (n = 1).
Distribution and Variety of Cardiac Diagnosis of All 174 Patients Undergoing Diagnostic and Interventional Cardiac Catheterization.

Abbreviations: D-TGA, D-Transposition of the great arteries; VSD, ventricular septal defect.
*Other heart disease are coronary artery anomaly (n = 2), infective endocarditis (n = 1), cardiomyopathy (n = 2), Kawasaki syndrome (n = 1), arterial tortuosity syndrome (n = 1), heart transplantation (n = 5), normal anatomy (n = 3)
Procedures
In 39 (21%) cases, CCs were diagnostic, whereas 144 (79%) were interventional. Interventional catheterizations included the whole spectrum of procedures such as balloon angioplasty and valvuloplasty, stenting, and device closure of atrial and ventricular septal defect.
Activated Clotting Time Values
The ACT was measured at the end of CC (median: 57 minutes, range: 17-200 minutes) after UFH bolus administration. We analyzed 183 ACT values according to time of measurement after heparin administration. Mean values and 95% confidence interval (dotted line) are shown in Figure 1. Median ACT was 204 seconds (range: 105–480 sec) with an interquartile range (IQR) of 182 to 229 sec, mean ACT value was 213 ± 55.3 sec, respectively. HemoTec low-range cartridge ACT values within 160 to 240 seconds are regarded as target level during CC at our institution. In this study within 132 (72%) CCs, ACT was within target levels. Only in 16 (9%) CCs, ACT was measured below 160 seconds, whereas in 35 (19%) measurements, ACT values were greater than 240 seconds. Nine patients underwent 2 CCs within the given study period (Figure 2). Besides 2 of these patients (patients 3 and 4 in Figure 2), the ACT values of the 2 measurements correlated according to the 2 time points of measurement.
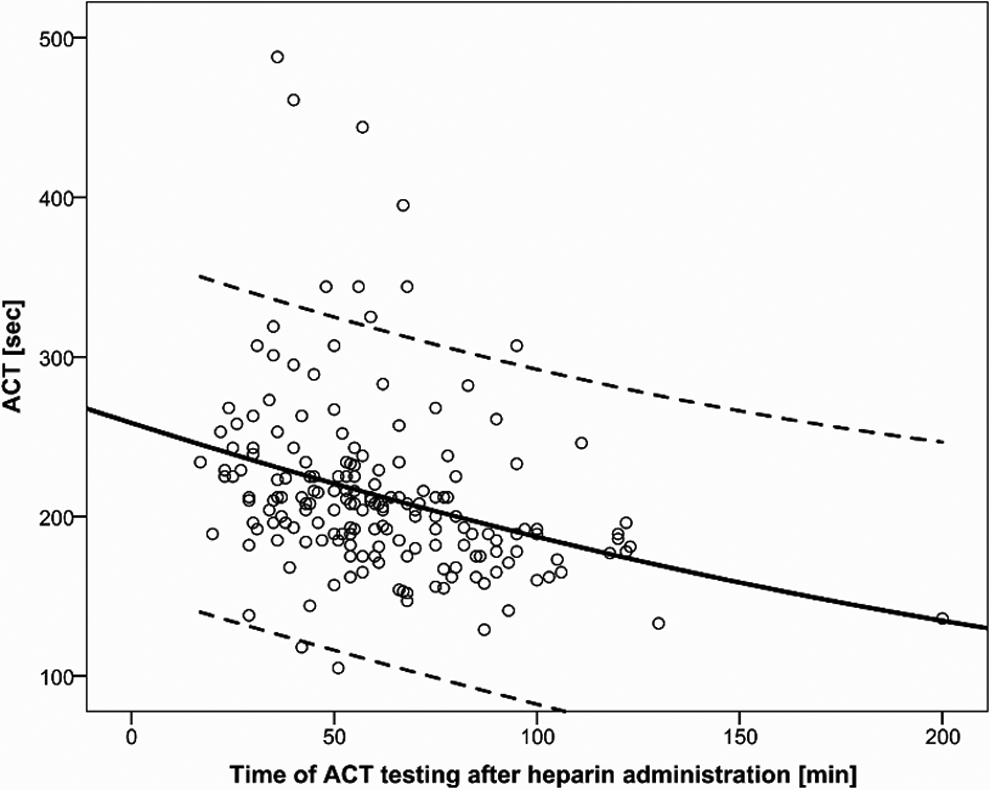
Effect on activated clotting time (ACT) of single intravenous bolus administration of 100 IU/kg body weight of unfractionated heparin during 183 cardiac catheterization in childhood. Mean (solid) and 95% confidence interval (dotted line) are given.
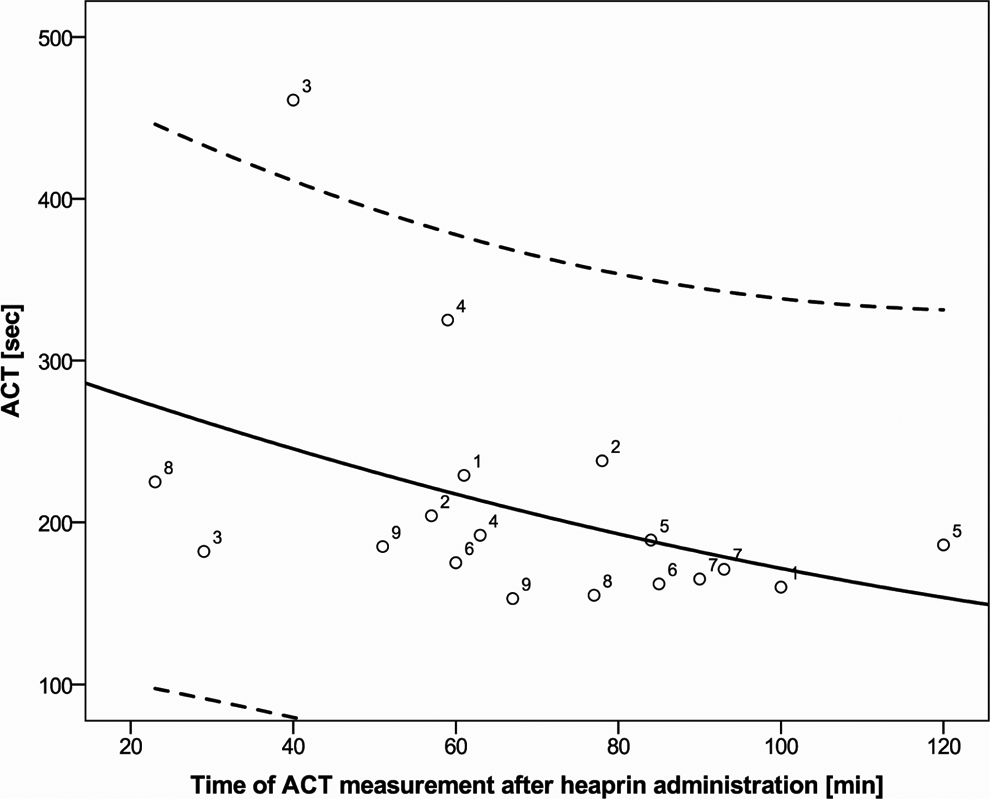
Activated clotting time (ACT) in 9 individual patients with 2 subsequent cardiac catheterizations. Mean (solid) and 95% confidence interval (dotted line) are given. Patient numbers (1 to 9) show the different ACT values (seconds) according to time of measurement after heparin administration (minutes) in 9 individual patients undergoing 2 subsequent cardiac catheterizations.
Mean (±SD) ACT values in interventional CCs were 208 ± 47 sec, respectively median ACT values 200 sec (range: 105–444 sec, IQR 178–225 sec), comparable to diagnostic CC with Mean (±SD) ACT values of 231.5 ± 77 sec, respectively median ACT values 208 (range: 136–488 sec, IQR 190.5–236 sec).
Age at Intervention
Figure 3 shows distribution of ACT values in different age groups. The number of patients per age group differed (age group 0-1 month: n = 13; age group 1-12 month: n = 47; age group 1-6 years: n = 72; age group 6-12 years: n = 27; age group >12 years: n = 24). Median (IQR) ACT levels according to age group were at age group 0–1 month 185 sec (IQR 178–216 sec), at age group 1–12 month 204 sec (IQR 180–226 sec), at age group 1–6 years 206 sec (IQR 183–225 sec), at age group 6–12 years 223 sec (IQR 192–258 sec), and at age group > 12 years 200 sec (IQR 184–229 sec). Age-dependent differences in ACT values between different age groups from neonates to adolescents were not obtained (P = .407).
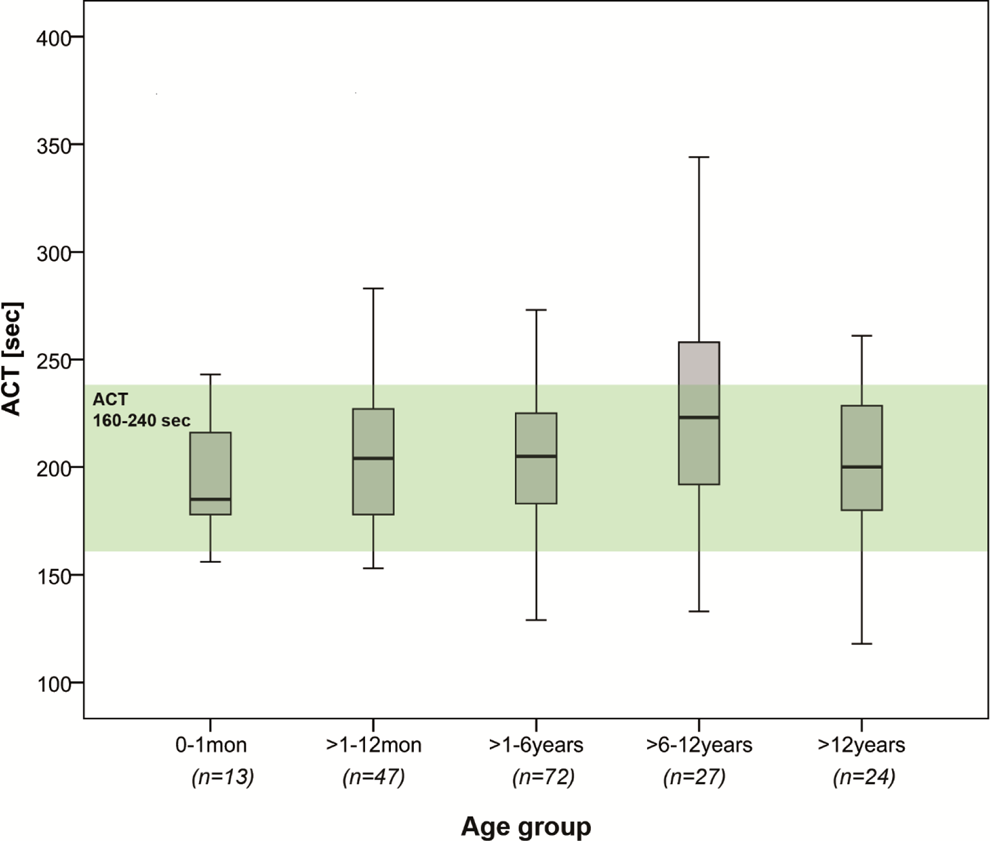
Activated clotting time (ACT) values according to age groups. Box plot diagram of ACT values according to age groups (neonates, infants, preschool children, school-age children, and adolescents). Target level within our clinic for HemoTec low-range cartridge ACT during CC: 160 to 240 seconds (green beam).
Fluid Volume During CC
The amount of total fluid volume administration during CC (crystalloid, colloid, and blood products) was median 17.7 mL/kg (range: 13.1-26.5 mL/kg) and negatively correlated with ACT values (Spearman ρ = −0.241, P < .001). Calculation of total fluid volume intake consisted of all crystalloid, colloid, and red blood cell administrated during intervention. Only crystalloid administration showed significant negative correlation with ACT (Spearman ρ = −0.254, P < .001), whereas correlation between colloid (P = .277) and red blood cell administration (P = .469) and ACT values were not obtained.
Anticoagulant and Antiplatelet Medication Before CC
Sixty-four patients were treated with anticoagulant or antiplatelet drugs before CC, with ASA (n = 48), LMWH (enoxaparin; n = 8), and/or OAC (phenprocoumon; n = 8). Medications before CC such as ASA (P = .014), LMWH (P = .049), but not phenprocoumon (P = .863), interfered with ACT values during CC. Patients receiving ASA and LMWH showed lower ACT values than patients without ASA and LMWH before CC. However, 38 (79%) of the 48 patients receiving ASA before CC reached target ACT levels (between 160 and 240 seconds), 5 (10.5%) were above target level and 5 (10.5%) had ACT values lower than 160 seconds. Of 8, 6 (75%) patients treated with LMWH before CC reached target (as mentioned above), whereas only 2 (25%) patients showed values below target level and none of these patients reached ACT values higher than 240 seconds. Of all patients treated with OAC before CC (n = 8), 4 (50%) had ACTs above 240 seconds, others were within target level.
Factors Influencing ACT Level: Hb, HCT, PLT, and Transaminases
Laboratory parameters such as Hb (P = .064), HCT (P = .087), and PLT (P = .576) did not correlate significantly with ACT values, neither did elevated transaminases (ALT, AST; P = .291).
Femoral Vessel Thrombosis and Bleeding
In 8 (4.4%) of the 183 CCs, arterial (n = 7) or venous (n = 1) femoral vessel thrombosis occurred, whereas bleeding complications occurred in 3 (1.65%) of the 183 CCs. All vascular complications after CC (thrombotic and bleeding) did not correlate with ACT values (P = .614). An overview of all patients with complications after CC and ACT values is shown in Table 2.
Patient and Clinical Characteristics Including ACT Values of 11 Children With Femoral Vessel Thrombosis and Bleeding Complications After Cardiac Catheterization.
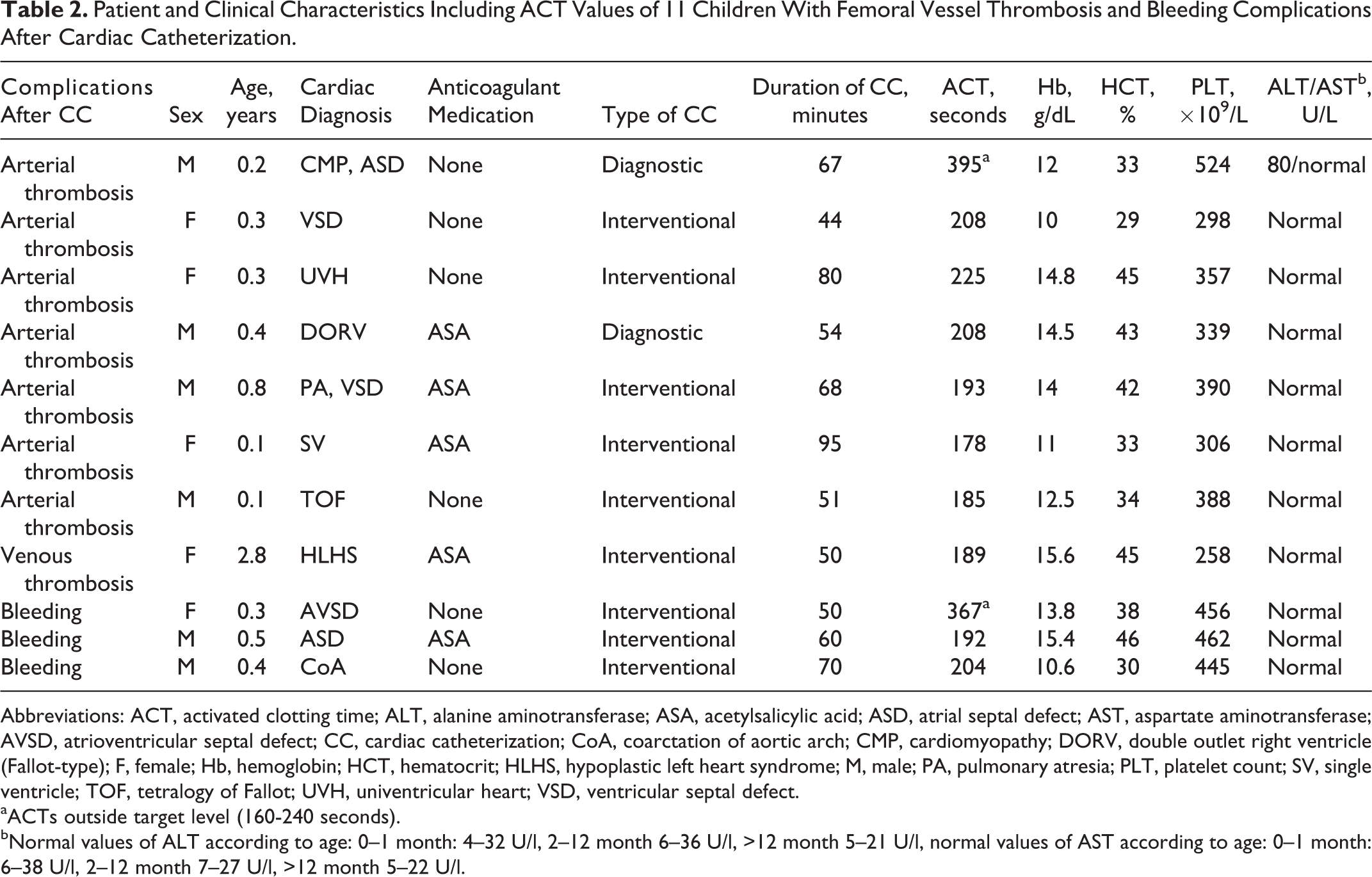
Abbreviations: ACT, activated clotting time; ALT, alanine aminotransferase; ASA, acetylsalicylic acid; ASD, atrial septal defect; AST, aspartate aminotransferase; AVSD, atrioventricular septal defect; CC, cardiac catheterization; CoA, coarctation of aortic arch; CMP, cardiomyopathy; DORV, double outlet right ventricle (Fallot-type); F, female; Hb, hemoglobin; HCT, hematocrit; HLHS, hypoplastic left heart syndrome; M, male; PA, pulmonary atresia; PLT, platelet count; SV, single ventricle; TOF, tetralogy of Fallot; UVH, univentricular heart; VSD, ventricular septal defect.
aACTs outside target level (160-240 seconds).
bNormal values of ALT according to age: 0–1 month: 4–32 U/l, 2–12 month 6–36 U/l, >12 month 5–21 U/l, normal values of AST according to age: 0–1 month: 6–38 U/l, 2–12 month 7–27 U/l, >12 month 5–22 U/l.
Discussion
Vascular interventions such as CC are a well-known risk factor for thrombus formation caused by intimal vascular damage. 15 Intravenous heparin administration during pediatric CC as primary prevention of thromboembolic events is standard of care. 4 But the most feared side effects of heparin include bleeding complications. 5,6 Especially small children, with their immature hemostatic system, are at risk for both thrombotic and bleeding complications after CC. 7 –9 To find accurate UFH dose for every individual patient, monitoring of UFH effect has been routinely established using ACT as a bedside tool. 10 –13 But ACT can be influenced by multiple factors apart from the anticoagulant effect of UFH. 13 Also significant variability of ACT values exists between different analyzers. 11 This makes interpretation of ACT, defining target levels for ACT and UFH dosage, a challenging task until today. This study was planned to establish a safe anticoagulation regimen and to detect influencing factors on ACT by showing the effect of UFH bolus on ACT during CC in pediatric patients from the neonate to the young adult.
We could not show age dependency for ACT levels in this study (Figure 3). Although median ACT levels according to age groups showed the lowest levels for neonates (0-1 month), in 7 (91%) of the 8 patients, thrombosis occurred after CC in children between 1 month and 1 year of age. This confirms the fact that femoral vessel thrombosis is more frequent in younger children. 16 This could imply that UFH dose in this age group is often underestimated. But further risk factors for femoral vessel thrombosis are based on the circumstance that those neonates undergoing CC often have severe type CHD, mostly cyanotic CHD, which has been also described as hemostatic risk factor. 16 But also bleeding complications in 3 (100%) of 3 patients in our study population occurred in children < 1 year. These findings are supported by Monagle and Newall 16 who pointed out that anticoagulation management particularly in neonates can be challenging and implies therapeutic uncertainty of a safe anticoagulant therapy in this specific young age group, especially in children with CHD. Multiple factors have been described for pediatric patients including immaturity of the hemostatic system and physiological changes in the blood coagulation system during childhood, extreme hemodilution in small children, and faster heparin metabolism in children compared to adults. 7,8,17 Therefore, weight-adapted heparin protocols transferred from adult data contain the risk of imprecise dosage in children, especially in neonates and children with CHD. Bembea et al 18 demonstrated that ACT was higher in neonates compared to infants and children, whereas in contrast, Kim et al 12 revealed age differences depending on whether children were healthy (ACT lower in children <3 years) or had CHD (higher values in younger children).
Apart from age, ACT may also be influenced by further factors. Dalbert et al 19 showed hemodilution being a significant influencing factor on ACT monitoring. In this study, colloid fluid administration and total fluid volume applied during CC were fairly low, and thus, no positive correlation with ACT levels was observed. Dilution-dependent increase in ACT was demonstrated in a study of adult patient with cardiac, while the prolongation was described to be independent of the dose of UFH administered. 19 Other influencing factors on ACT such as hypothermia were described. 13,20 Hypothermia, which occurs during CPB, prolongs ACT, because of slowing down enzymatic pathways. The CC procedures are shorter and hypothermia is strictly avoided, still other influencing factors remain. Prolongation of ACT was not observed following administration of therapeutic medication of LMWH. This finding was not supported by our results. Likewise, the linear increase in ACT due to ASA and OAC was also not observed in our study cohort. To summarize, ACT measurement may not be an optimal reliable tool to monitor anticoagulation therapy, because it is influenced by the sum of factors such as CHD and other comorbidities, intimal injury, interventional procedure, French-size catheters, and duration of CC. In addition, we could not find any other influencing factors of laboratory parameters such as Hb, HCT, PLT, ALT, and AST.
In this study, only patients receiving 100 IU/kg body weight of UFH during CC were included. According to the evidence-based clinical practice guidelines of the American College of Chest Physicians, 4 this dosage is recommended today. Reasons for this dosage other than 100 IU/kg are short duration of CC, heparin resistance, or other known coagulation disorders. Grady et al 21 showed that with Hemochron 801 ACT monitor (International Technidyne Corporation, Edison, NJ, USA), ACT values prolonged to 209 ± 52 seconds after 50 IU/kg versus 270 ± 57 seconds after 100 IU/kg bolus UFH administration. Hanslik et al 2 compared low- versus high-dose heparin protocols during CC (50 vs 100 IU/kg bolus, followed by 20 IU kg−1 h−1 continuous infusion) and were able to show no differences regarding femoral vessel thrombosis and bleeding complications.
The target range of ACT values between 160 and 240 seconds is used at our institution corresponding to the empirical ACT target value for HemoTec low-range cartridge. The average baseline ACT without UFH administration using HemoTec low range is reported to be 90 to 120 seconds. 22 A safe minimal ACT level during CPB is evidently not determined yet, but in cardiac surgery, most institutions aim for values greater than 350 seconds. 23 Glatz et al 24 found ACT levels below 250 seconds associated with a higher risk for acute occlusive arterial injury following CC. In this study, only in 9% of all CCs, ACT values did not reach target level of 160 seconds, but in 19%, ACT values were greater than 240 seconds. Thromboembolic and bleeding complications did not correlate with low or high ACT values. Only in 1 of 3 cases with bleeding complications, ACT was above target levels over 367 seconds (Table 2). Of the 35 patients, 34 with ACT values higher than 240 seconds were not affected by bleeding complications, in 1 case yet thrombosis occurred. On the other hand, all 16 patients with ACT below target level did not have thromboembolic complications after intervention. Only in 1 of 8 cases of thrombosis ACT was outside target level over 395 seconds (Table 2), where we rather would expect bleeding complications instead of thrombosis. However, incidence of arterial and venous vessel thrombosis (4.4%) and bleeding (1.65%) as a complication following CC in our population (Table 2) was comparable to other data. 2,3 Monagle et al 4,16 mentioned a reduction in the incidence of thrombosis from 40% to 8%, introducing a standardized UFH regime of 100 to 150 IU/kg. Hanslik et al 2 counted 3% thromboembolism with administration of 100 IU/kg UFH, versus 4% with 50 IU/kg UFH, whereas bleeding occurred in 12% versus 7%. Although according to Yamada et al, 25 ACT may not predict blood loss, because it does not primarily measure clot formation.
Measurement of ACT values at different time points after UFH administration was unavoidable in this retrospective study due to practical reasons. Activated clotting time is routinely measured at the end of CC for decision-making of further anticoagulant treatment. Due to a fairly high number of CCs executed at a central institution for pediatric CC, wide range and high complexity of cardiac diagnosis and interventions had to be taken into account. This led to a broad range of duration of CC. Netz et al 26 also reported time dependency of ACT after UFH bolus administration. This of course could be relevant for the observed differences in ACT measurements in this study.
For the inconsistency of ACT measurements (intraindividual variability), which was shown by these 9 patients, that incidentally appear twice in our study population, because they underwent 2 CCs in the time period of this study (Figure 2), we did not find an obvious explanation. This leads to the assumption that ACT may also be affected by individual patient- and/or procedure-related factors, which have not been measured in this study. Significant differences in ACT according to different CC (diagnostic vs interventional) have not been proven, but foreign body material as it is more often used in interventional CCs could possibly activate coagulation before ACT measurement and therefore might have an influence on ACT. Because of this known inconsistency of ACT values, other bedside systems for monitoring coagulation in children undergoing CPB have been established. In children younger than 6 months, the entire blood heparin concentration can be measured with Hepcon (Hepcon Hemostasis Management System Plus; Medtronic, Minneapolis, Minnesota). This method has shown better correlation with actual plasma heparin levels (measured by anti-Xa assays) than ACT. 27 But there are no recommendations for the use of Hepcon in CC.
Our study has some limitations such as the retrospective study design, which limited the number of ACT measurements to a single analysis at the end of CC. Future trials should include a prospective study design with at least 2 or more analysis of ACT measurements. Patients in this study were mostly small children at a median age of 2.8 years, so age-dependent differences may be missed due to the age distribution and total number of patients in the other age groups. Further studies could compare neonates (<28 days), infants (>28 days to <1 year), and older children (>1 year) in a prospective clinical study.
Conclusions
Intravenous bolus administration of 100 IU/kg UFH during CC in children resulted in ACT values of median 204 seconds (range: 105-488 seconds), which corresponded in 72% (n = 132) to be within the empirical ACT target value for HemoTec low-range cartridge as it is used in our institution (160-240 seconds).
Both ideal UFH dose and safe ACT target levels to avoid thrombosis as well as bleeding following CC are not universally established. Especially limited data exist for children. Intraindividual variability of ACT measurements makes the design of a standard protocol even more difficult.
In our study, there were no significant differences in ACT according to age, but whether measurement of ACT is the right diagnostic tool analyzing age-dependent differences concerning coagulation remains unclear. In summary, with single-shot administration of 100 IU/kg body weight of UFH reaching ACT values between 160 and 240 seconds, the incidence of complications in our study was comparable to other data. Further studies are required to prove safety of this anticoagulant regimen during CC.
Footnotes
Author Contributions
Oliver Kretschmar and Walter Knirsch contributed as senior authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
