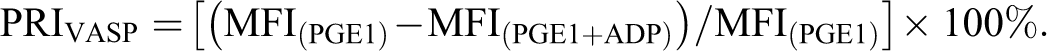Abstract
Antiplatelet medicines have been one of the cornerstones in the treatment of patients with acute coronary syndrome (ACS). However, adverse cardiovascular events still occur in some patients on standard antiplatelet therapy. Therefore, a reliable laboratory test to monitor the residual platelet activity (RPA) is urgent. We aim to modify the impedance whole blood platelet aggregation (WBA) assay associated with release assay to monitor RPA, despite antiplatelet therapy and assess their relationship with clinical ischemic events. In this study, RPA was tested in 133 patients with ACS postpercutaneous coronary intervention between 24 and 36 hours after a 300-mg clopidogrel loading dose by modified assay. Then, these patients were followed up for 3 months for clinical ischemic events. Meanwhile, platelet activity of 58 healthy volunteers was also tested by modified assay. Results showed that in modified assay the point of platelet magnified activation time (MAT) and maximal platelet adenosine triphosphate release values (RV) have significant differences between healthy volunteers and patients ([90.86 ± 27.60 seconds] vs [206.44 ± 58.97 seconds] and [2.07 ± 0.64 nmol] vs [0.98 ± 0.49 nmol]; P < .001 and P < .001, respectively). During follow-up, 5 patients present ischemic events. Receiver-operator characteristic curve showed that the cutoff values for MAT and RV were 156.5 seconds and 1.05 nmol, respectively, with the sensitivity and specificity of 60.00% and 83.30% and 80.00% and 67.50%, respectively; when MAT combined with RV, the sensitivity can be increased to 100%. Therefore, modified impedance WBA and release assay may be a potentially recommended reliable laboratory assays for monitoring the RPA.
Keywords
Introduction
Dual antiplatelet treatment (DAPT) is the cornerstone in the management of atherothrombotic diseases, especially for patients with acute coronary syndromes (ACSs) and/or undergoing percutaneous coronary interventions (PCIs). 1,2 Among the antiplatelet drugs, clopidogrel which is an adenosine diphosphate (ADP) receptor inhibitor, alone or in combination with aspirin, has proved significant clinical benefits. However, recently several studies have described that many patients experience recurrent ischemic events, despite recommended antiplatelet therapy and good responsiveness to antiplatelet agents by laboratory assessments. 3,4 Might laboratory tests of platelet function have limitations and can’t detect all high on-treatment platelet reactivity? Nowadays several laboratory methods (such as impedance whole blood platelet aggregation [WBA], turbidimetric light transmittance aggregometry) have been used to assess clopidogrel-induced antiplatelet effects. These methods are all performed on citrate-anticoagulated blood. Citrate can bind all extracellular calcium (Ca2+) that plays an important role in the platelet activation. Therefore, these methods may provide misleading information on platelet reactivity and antiplatelet effects in vivo. 5,6 Due to the absence of calcium in citrate-anticoagulated blood, these methods can’t detect a strong effect of thrombin on platelet activation. Meanwhile, these methods are unable to reflect platelet-amplified activation and release function actually. Therefore, it puts forward the urgent request to improve laboratory testing method of platelet function. In our study, in order to overcome the problems created by citrate, we adjust in vitro citrate-anticoagulated blood Ca2+ concentration to physiological levels before testing platelet function by modified impedance WBA and release method.
Methods
Study Population
Fifty-eight healthy volunteers of Chinese Han ethnicity were recruited. Individuals without a history of cardiovascular disease, hematology disease, cerebrovascular disease, cancers, autoimmune disease, and not taking any drugs were qualified for inclusion. A total of 157 patients with ACS were enrolled, while 133 patients with PCI met the inclusion criteria. Some patients, who didn’t take clopidogrel for at least 7 days before PCI, received a 300-mg loading dose of clopidogrel followed by a maintenance dose of 75 mg daily. Other patients, who had taken clopidogrel for over 7 days before PCI, continued to receive a maintenance dose of 75 mg daily. All patients received aspirin at a maintenance dose of 100 mg daily. Patients were tested for platelet function between 24 and 36 hours after a 300-mg clopidogrel loading dose. Patients were excluded mostly due to ACS without PCI, patients with acute inflammation, and patients’ treatment with any platelet inhibitor expect aspirin and clopidogrel. The study was reviewed and approved by the Hospital Research and Ethical Committee. All study participants voluntarily provided informed consent personally before any study procedures were undertaken.
Blood Samples
Whole blood samples for analysis were drawn by venipuncture of an antecubital vein and collected into vacuum tubes (Becton Dickinson Medical Devices Co Ltd, Franklin Lakes, New Jersey) containing 0.3 mL of 3.2% sodium citrate. The initial 3 mL of blood drawn was discarded to avoid inclusion of activated platelets. Either impedance WBA or platelet release function was performed on Choron-Log lumi-aggregometer instruments (CHRONO-LOG Model 560-CA, Chrono-Log Co, Havertown, Pennsylvania). The vasodilator stimulated phosphoprotein (VASP) phosphorylation analysis was performed on a flow cytometer (Beckman Coulter, Margency, France).
Impedance WBA and release assay
An electrode probe assembly was inserted into 450 μL of blood diluted with 450 μL of saline which was incubated at 37ºC for 5 minutes before; then, 100 μL Chronolume (Chrono-Log Co, Havertown, Pennsylvania), which was used to measure the adenosine triphosphate (ATP) released from platelets, 7,8 and 10 μL ADP (Chrono-Log Co) at 10 μmol/L were added. The maximal increased electrical resistance (Ω) values and the maximal platelet ATP release values (RV) were recorded.
Modified impedance WBA and release assay
An electrode probe assembly was inserted into 450 μL of blood diluted with 450 μL of saline which was incubated at 37ºC for 5 minutes before; 100 μL Chronolume (Chrono-Log Co, Havertown, Pennsylvania) was added and incubated at 37ºC for 2 minutes; and then, CaCl2 (at a final concentration of 2.5 mmol/L 9 ) and ADP (at 10 μmol/L) were added. The point of platelet MAT and the maximal platelet ATP RVs were recorded.
Flow cytometric VASP phosphorylation assay
Blood samples were incubated with prostaglandin E1 (PGE1) or with PGE1 and ADP 10 μmol/L at room temperature for 10 minutes and then fixed with paraformaldehyde, after which the platelets were permeabilized with nonionic detergent. Subsequently, cells were labeled with a monoclonal antibody against serine 239-phosphorylated VASP (16C2) or a negative isotypic control antibody using fluorescein isothiocyanate (FITC) staining and a CD61 phycoerythrin-labeled platelet-specific antibody. Analyses were performed on a flow cytometer (Beckman Coulter, Margency, France). A platelet reactivity index (PRI) VASP was calculated from the mean fluorescence intensity (MFI) of samples incubated with PGE1 or PGE1 and ADP according to the following formula:
Precision evaluation
Ten specimens from healthy volunteers were tested 5 times by modified impedance WBA and release method.
Genotyping
Blood samples of patients with recurrent ischemic events were collected in tubes containing EDTA for genetic testing. The genomic DNA was extracted from whole blood using a commercially available DNA isolation kit (Tiangen Biotech Co, Ltd, Beijing, China) according to manufacturer’s instruction. CYP2C19 polymorphisms were analyzed for the loss of functional alleles *2 and *3 by identifying 2 polymorphic positions, 681G > A in exon 5 and 636G > A in exon 4, respectively. The region including the polymorphisms was amplified by polymerase chain reaction (PCR), and genotyping was performed using a commercially available validated drug metabolism genotyping assay (Sky Molecular Biotechnology, Suzhou, China).
Follow-Up
Patients were followed by telephone interviews and review of medical records after 3 months of PCI (96 ± 11 days). End points were clinical ischemic events. Clinical ischemic events were defined as myocardial infarction (MI; excluding in-stent thrombosis), in-stent thrombosis, cardiac death, and stroke. 10
Statistical Analysis
Data were checked for normality. Normally distributed continuous data are expressed as mean and standard deviation (SD); otherwise, median (M) and quartile ranges from the lower quartile Q1 to the upper quartile Q3 (Q1-Q3) were used. Coefficient of variation (CV) was calculated from the ratio of SD to the mean. Reference intervals were estimated as the 2.5th and 97.5th percentile of the distribution. Statistical comparisons were tested with t test in the case of normally distributed data or with Mann-Whitney U test when data distribution was asymmetrical. Receiver–operator characteristic (ROC) curves were used to generate clinical cutoff values for predicting patients’ outcome. A 2-tailed P value <.05 was considered significant. All statistical analyses were performed with SPSS Version 16.0.
Results
Precision and Reference Intervals for Modified Assay
The modified impedance whole blood platelet assay obviously shows the point of platelet MAT in the aggregation curve (Figure 1). The mean within-day CVs for MAT and RV are 9.31% and 6.13%, respectively (Table 1).
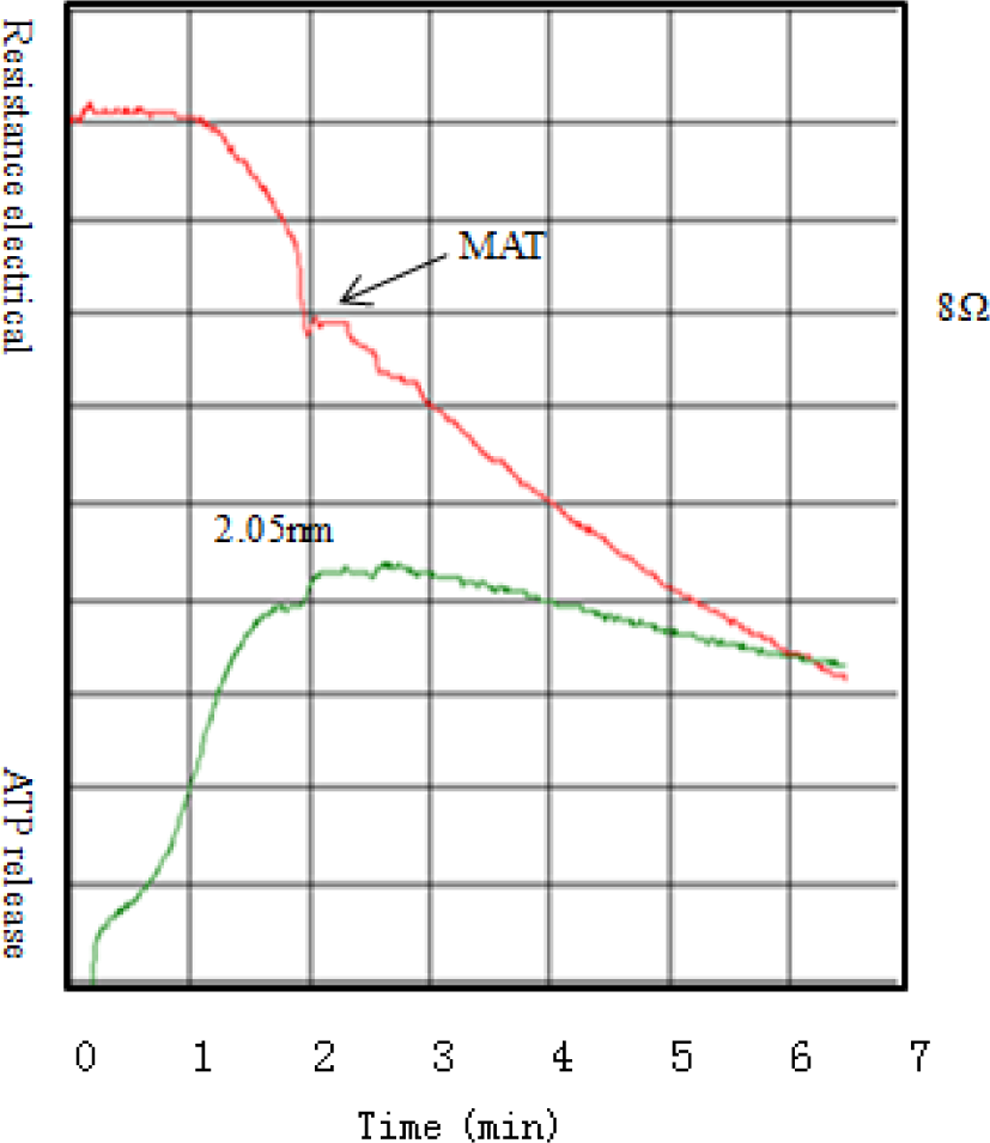
Modified impedance whole blood platelet aggregation (WBA) and release curves. The upper curve: impedance WBA curve; the lower curve: platelet ATP release curve. MAT indicates magnified activation time; ATP, adenosine triphosphate.
Precision and Reference Intervals for Modified Impedance Whole Blood Platelet Aggregation (WBA) and Release Assay.
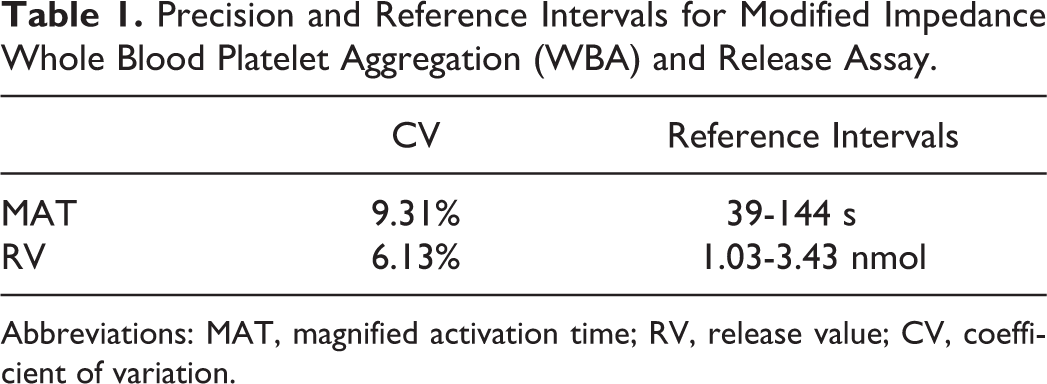
Abbreviations: MAT, magnified activation time; RV, release value; CV, coefficient of variation.
In all, 58 healthy volunteers (male 24 and female 34) had a mean age of 41 years, ranging from 23 to 68 years of age. The reference intervals of modified impedance whole blood platelet MAT and RV are presented in Table 1. The RV of healthy volunteers in the modified assay was significantly higher than that in unmodified assay ([2.07 ± 0.64]) vs [1.32 ± 0.432], P < .001; Figure 2). The MAT and RV between healthy volunteers and patients have significant differences (P < .001 and P < .001), respectively (Table 2; Figure 3).
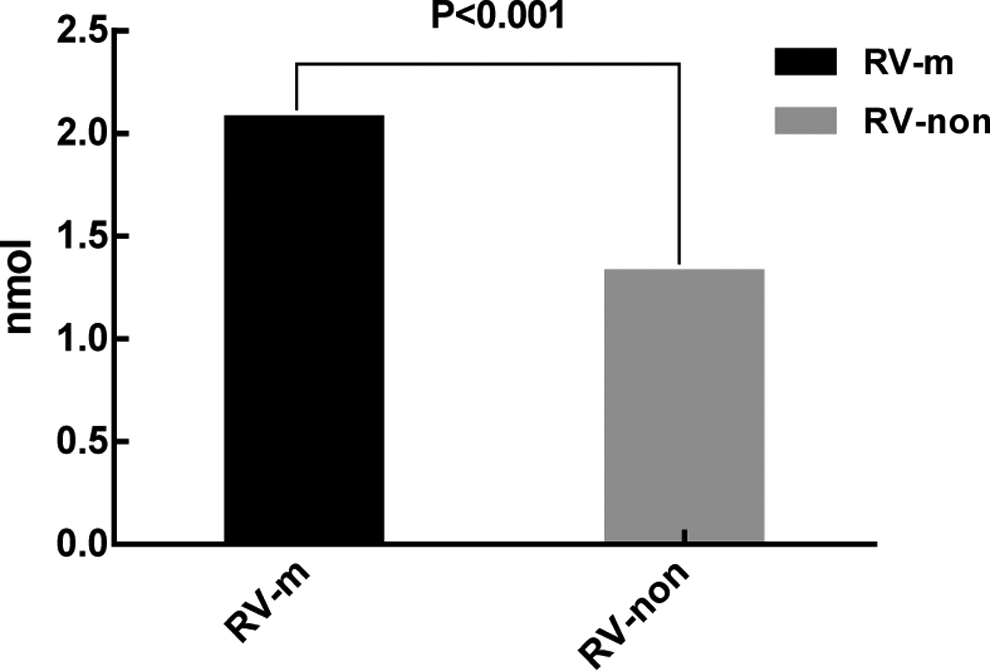
Differences in healthy volunteer maximal platelet ATP release values (RV) between the modified assay and the unmodified assay. RV-m were significantly higher than RV-non ([2.07 ± 0.64] vs [1.32 ± 0.432], P < .001). RV-m indicates maximal platelet ATP RV in modified assay; RV-non indicates maximal platelet ATP RV in unmodified assay; ATP, adenosine triphosphate; RV, release value.
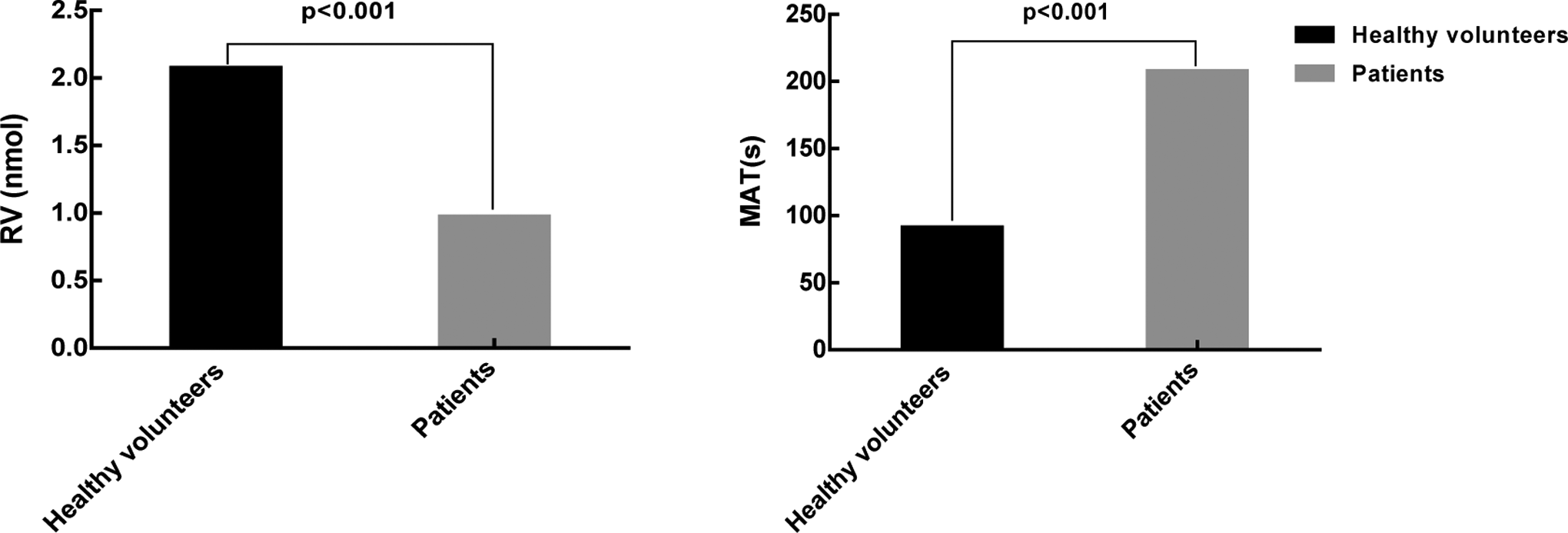
Comparison of the modified assay in healthy volunteers and patients. MAT in healthy volunteers is significantly shorter than that in patients (90.86 ± 27.60 seconds vs 206.44 ± 58.97 seconds, P < 0.001). RV in healthy volunteers is significantly higher than that in patients (2.07 ± 0.64 nmol vs 0.98 ± 0.49 nmol, P < 0.001). MAT indicates magnified activation time; RV, release value.
Differences in Modified Impedance Whole Blood Platelet Aggregation and Release Assay Between Healthy Volunteers and Patients.

Abbreviations: MAT, magnified activation time; RV, release value.
Residual Platelet Activity Associated With Clinical Ischemic Events
An average of 3 months of follow-up was achieved in patients having ACS with PCI enrolled in this study. The baseline characteristics of patients are included in Table 3. Clinical ischemic events occurred in 5 (3.76%) of the 133 patients. In all, 2 patients had MI, 1 patient had early in-stent thrombosis (presented STEMI), 1 patient had late in-stent thrombosis (presented unstable angina), and 1 patient had stroke (caused by cerebral vascular thrombosis).
Baseline Characteristics of Patients With ACS.
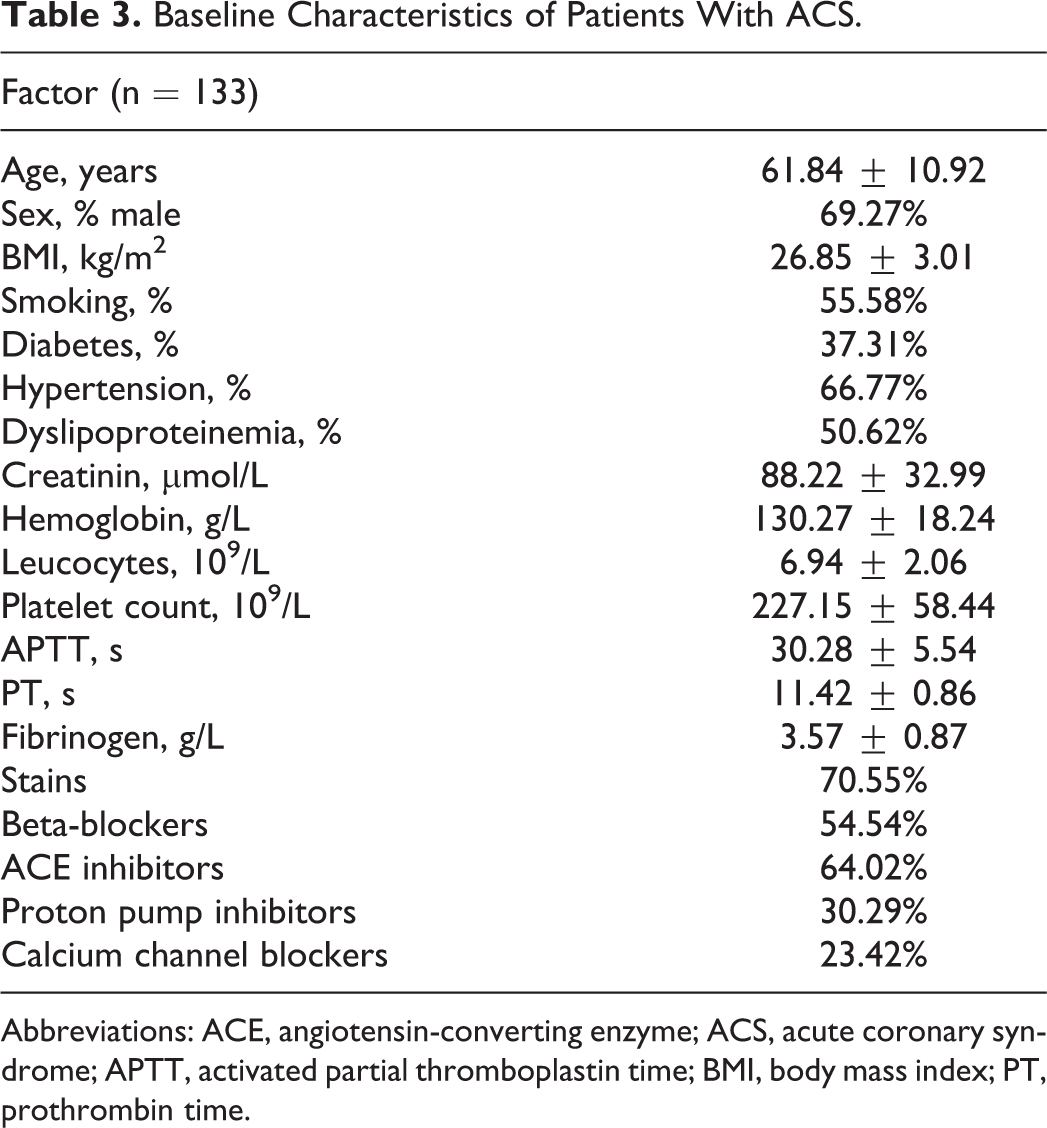
Abbreviations: ACE, angiotensin-converting enzyme; ACS, acute coronary syndrome; APTT, activated partial thromboplastin time; BMI, body mass index; PT, prothrombin time.
The response results of different tests and genotyping in 5 patients with clinical ischemic events are shown in Table 4. The cutoff values for modified impedance WBA MAT and RV to discriminate patients who are likely to have clinical ischemic events are 156.5 seconds and 1.05 nmol, respectively, and their sensitivity and specificity are presented in Table 5. When RV combined with MAT, the sensitivity can increase to 100%. The cutoff values for VASP-PRI to discriminate clinical ischemic events is 58.44%, and the sensitivity and specificity are presented in Table 5.
The Response Results of Different Tests and Genotyping in 5 Patients With Clinical Ischemic Events.

Abbreviations: MAT, magnified activation time; RV, release values; PRI, platelet reactivity index; WBA, impedance whole blood platelet aggregation; No. I-V, patients with clinical ischemic events; G, clopidogrel good response according to the different tests; P, clopidogrel poor response according to the different tests.
Clinical Performance of Impedance Assay and VASP for Clinical Ischemic Events.
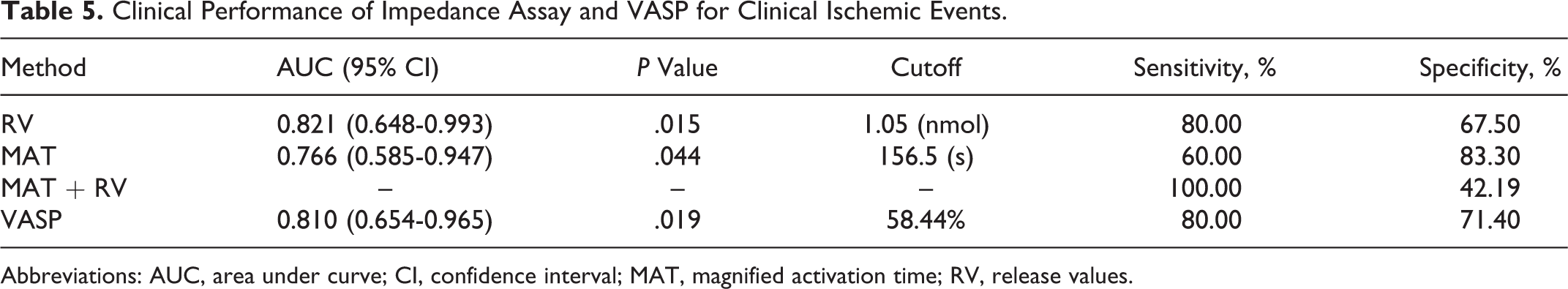
Abbreviations: AUC, area under curve; CI, confidence interval; MAT, magnified activation time; RV, release values.
Discussion
Many patients experience recurrent ischemic events, despite optimal antiplatelet therapy. Nowadays, laboratory tests of platelet function have identified “resistance” in 4% to 30% of patients taking clopidogrel. 11 –14 However, these laboratory tests of resistance have not correlated closely with subsequent recurrent ischemic events and have not reliably identified nonresponders to antiplatelet therapy. 15,16 So far, the mechanisms leading to resistance are not fully clarified. Some research show that genetic factors, underdosing and drug–drug interactions, upregulation of the P2Y(12) pathways, accelerated platelet turnover, high baseline platelet reactivity, and poor compliance all likely lead to resistance. 14 In our study, another factor that is likely causing resistance is found. We found that many laboratory tests were almost collected into citrate-anticoagulated tubes that bind all extracellular calcium. 6 Therefore, these tests, such as impedance WBA, are relatively nonphysiological because of lacking Ca2+ which plays an important role in platelet activation, aggregation, and release in reaction system. In response to this disadvantage, we adjust in vitro citrate-anticoagulated blood Ca2+ concentration to physiological levels to modified impedance WBA and release method. Meanwhile, this modified method can also reflect the strong effect of thrombin on platelet magnified activation (Figure 1).
When the Ca2+ (a final concentration of 2.5 mmol/L 9 ) is added into the reaction system, the point of platelet MAT appears in the impedance curve (Figure 1). From Figure 1, we can observe that the trend of platelet aggregation curves show almost a straight line down after the point of MAT. Furthermore, after finishing the procedure, we can also observe big coagulation clots in between 2 electrodes and some coagulation clots floating in the blood, which lead to electrical resistance instability. This phenomenon had also been described in the study of Li et al. 17 In addition, after the point of MAT, the aggregation curves include 2 components—coagulation and platelet aggregation—which can’t reflect the function of platelet aggregation, alone. So, in our study, the point of MAT is recorded instead of electrical resistance in modified method.
In this study, the precision of MAT and RV in modified method was evaluated (9.31% and 6.13%). All values of the CV were less than 10%, thus, this modified method can detect the samples stability. Furthermore, conforming to the CLSI document CA28-A3c, 18 we established the reference intervals of this modified method based on Han ethnicity population for local laboratory reference (Table 1). Analysis of modified method parameters in the groups of healthy volunteers and patients showed a significant difference (MAT: P < .001, RV: P < .001; Table 2). It showed that this modified method can separate healthy participants and patients receiving clopidogrel.
Residual platelet activity may be associated with adverse cardiovascular events in patients with ACS. 19,20 In our study, 5 patients had clinical ischemic events during the follow-up. Analysis of the ROC curve of RV showed that when the cutoff value was 1.05 nmol, the clinical effectiveness was good (sensitivity 80.00% and specificity 67.50%), and the analysis of the ROC curve of MAT showed that when the cutoff value was 156.5 seconds, the clinical effectiveness was good (sensitivity 60.00% and specificity 83.30%). When RV combined with MAT, the sensitivity could be increased to 100%. In the previous study of Qiao et al, 21 the clinical effectiveness of impedance WBA showed that sensitivity and specificity were 75% and 62%, respectively. It showed that the clinical effectiveness of modified assay could be better. Analysis of the ROC curve of VASP showed that when the cutoff value was 58.44%, the clinical effectiveness was good (sensitivity 80.00%, specificity 71.40%). In this study, the cutoff value 58.44% of VASP is different from some literature that described the cutoff value of VASP was 50%. 22,23 Recently, some study showed that 60% cutoff value of VASP was associated with cardiovascular death in patients treated with PCI. 24 In this present study, the cutoff value of 58.44% of VASP was close to 60%. In our study, of 5 patients with clinical ischemic events (Table 4), 1 patient was “clopidogrel poor responder” by WAB; 4 patients were “clopidogrel poor responders” by VASP; 3 patients were “clopidogrel poor responders” by MAT; and 4 patients were “clopidogrel poor responders” by RV. The MAT combined with RV can increase the detection of clopidogrel poor responders to 5 patients. In addition, Table 5 suggests that the sensitivity of MAT 60% is lower than that of VASP; the sensitivity of RV (80%) is almost equivalent to VASP; and when MAT combined with RV, the sensitivity is much better than VASP (100% vs 80%). However, compared with modified impedance WBA and release assay, VASP detection methods are expensive, time consuming, and have complex procedure. The current study suggests that modified impedance WBA and release test may be a good laboratory method to detect the responsiveness to antiplatelet agents.
Several trials have found an association between CYP2C19 and poor clinical prognosis. 25,26 In this study, the results of CYP2C19 analysis showed that only 3 of the 5 patients with clinical ischemic events had CYP2C19 (*2 and *3) loss of function mutation. It indicated that genetic factors might only explain part of clopidogrel poor responders.
There are a few limitations to this study. First, the number of patients is not enough and only 5 followed patients had clinical ischemic events. Second, we didn’t focus on the relationship between other platelet agonists (such as arachidonic acid and collagen) and modified method.
Conclusion
Modified impedance WBA and release assay may be a potentially reliable laboratory method for monitoring the responsiveness to antiplatelet agent.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Key Technology R&D Program (2012BAI37B01) and the Application Research of Capital Clinical Characteristic (Z141107006614003).