Abstract
Several medicines are currently used to inhibit the platelet activity. We aim to monitor the residual platelet activity (RPA) despite antiplatelet therapy and assess its relationship with major adverse events. The impedance platelet aggregation was employed to determine RPA. Totally, 202 patients with acute coronary syndrome (ACS) were followed up for 10 months for major clinical events of myocardial infarction, cerebrovascular accident (CVA), and all cause mortality, and RPA after clopidogrel loading was assessed in 30 patients. The RPA at 2 hours after 300 mg clopidogrel loading was 1 Ω (± 2.3 Ω) induced by adenosine diphosphate. Residual platelet activity of patients who experienced death, MI, or CVA was significantly higher than those who did not experience (
Keywords
Introduction
Several classes of pharmacies are currently employed to inhibit platelet functionality. For instance, aspirin and clopidogrel are mostly used in combination in patients presenting with an acute coronary syndrome (ACS), in particular those undergoing percutaneous coronary intervention (PCI). In order to answer whether patients have achieved the desired intensity of inhibition, monitoring the residual platelet activity (RPA) despite antiplatelet drug treatment is increasingly requested. The measurement of platelet aggregation following stimulation of platelet by an agonist is the direct way to evaluate residual platelet reactivity. Although some studies indicated that assessment of platelet activity inhibition was highly test specific, 1,2 correlations between low responsiveness to antiplatelet therapy and high rates of ischemic events were shown in some trials. 3,4 In our study, we tried to monitor the RPA by impedance platelet aggregation in the setting of ACS and assess its relationship with major adverse events.
Methods
Study Population
A total of 164 healthy volunteers were recruited for reference interval calculations. Individuals without a history of hematology disease, cardiovascular disease, cerebrovascular disease and diabetes and not taking any drugs were qualified for inclusion. In all, 202 patients with ACS were consecutively enrolled. Patients who were more than 20 years old and taking either aspirin (100-300 mg/d) and/or clopidogrel (75 mg/d) for more than 7days were eligible for enrollment. Patients were excluded if ACS stabilization was not achieved within 7 days of ACS onset, if they had acute inflammatory illness, or if they were on chronic therapy with insulin or any nonsteroidal anti-inflammatory drugs (NSAIDs). The study was reviewed and approved by the hospital ethics committee and all participants provided informed consent.
Blood Samples
Whole blood was drawn in vacuum tubes (Becton Dickinson Medical Devices Co Ltd, Franklin Lakes, New Jersey) containing 0.3 mL of 3.2% sodium citrate to test platelet aggregation and fibrinogen. Blood anticoagulated with EDTA was collected for complete blood counts. Either impedance or optical platelet aggregation was performed on a whole blood lumi-ionized calcium aggregometer (CHRONO-LOG Model 560-CA, Chrono-Log Co, Havertown, Pennsylvania).
Impedance whole blood platelet aggregation
An electrode probe assembly was inserted into 450 μL of blood diluted with 450 μL of saline. Aggregation was performed using adenosine diphosphate (ADP; Chrono-Log Co) at 10 μmol/L, arachidonic acid (AA; Chrono-Log Co) at 0.5 mmol/L, and collagen (Chrono-Log Co) at 2μg/mL. The increased electrical resistance (Ω) was proportional to the amount of platelet aggregation.
Optical platelet-rich plasma platelet aggregation
The baseline optical density was set with 500 μL of platelet-poor plasma (PPP) prepared from platelet-rich plasma (PRP) at a higher centrifugation (2400
Precision evaluation
In all, 10 specimens from volunteers were tested 5 times by the impedance whole blood platelet aggregation procedures and by the optical PRP platelet aggregation procedures, respectively.
Responsiveness for loading dose of clopidogrel
In all, 30 specimens obtained at 2 hours after 300 mg clopidogrel loading were assayed by the impedance whole blood platelet aggregation procedures and by the optical PRP platelet aggregation procedures, respectively.
Follow-Up
Enrolled patients with ACS were followed by telephone interviews and review of medical records. End points were myocardial infarction (MI), cerebrovascular accident (CVA), and all-cause mortality. Myocardial infarction was defined as the detection of rise in troponin or creatine kinase MB mass method above the 99th percentile of the upper reference limit in addition to evidences of myocardial ischemia. The CVA was defined as an acute neurologic vascular event with neurological signs for more than 24 hours.
Statistical Analysis
Coefficient variation (CV) was calculated from the ratio of the standard deviation (SD) to the mean. Reference intervals for impedance platelet aggregation are estimated as the 2.5th and 97.5th percentile of the distribution. Mean ± SD and range (maximum to minimum) are used for clopidogrel loading dose assessment. The Mann-Whitney test was performed for RPA comparison between patients with different outcomes. Receiver–operator characteristic (ROC) curves were used to generate clinical cutoff values for predicting patients’ outcomes.
Results
Precision and Reference Intervals for Impedance Platelet Aggregation
The mean values of within-day CV for the impedance procedure and optical procedure are 4.81% and 7.67%, respectively.
In all, 162 healthy volunteers (male 68 and female 84) of Chinese Han ethnicity had a mean age of 59, ranging from 38 to 83 years of age. The reference intervals of different agonists are presented in Table 1. Platelet activities induced by ADP, AA, and collagen showed mutual correlations (
Reference Intervals for Impedance of Whole Blood Platelet Aggregation.

Abbreviations: AA, arachidonic acid; ADP, adenosine diphosphate.
Linear Regression Among Platelet Activity Stirred by Different Agonists.
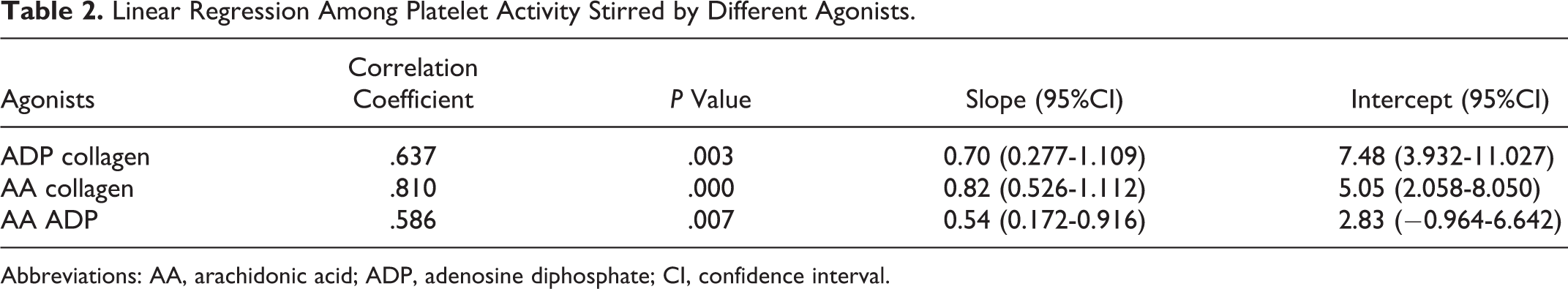
Abbreviations: AA, arachidonic acid; ADP, adenosine diphosphate; CI, confidence interval.
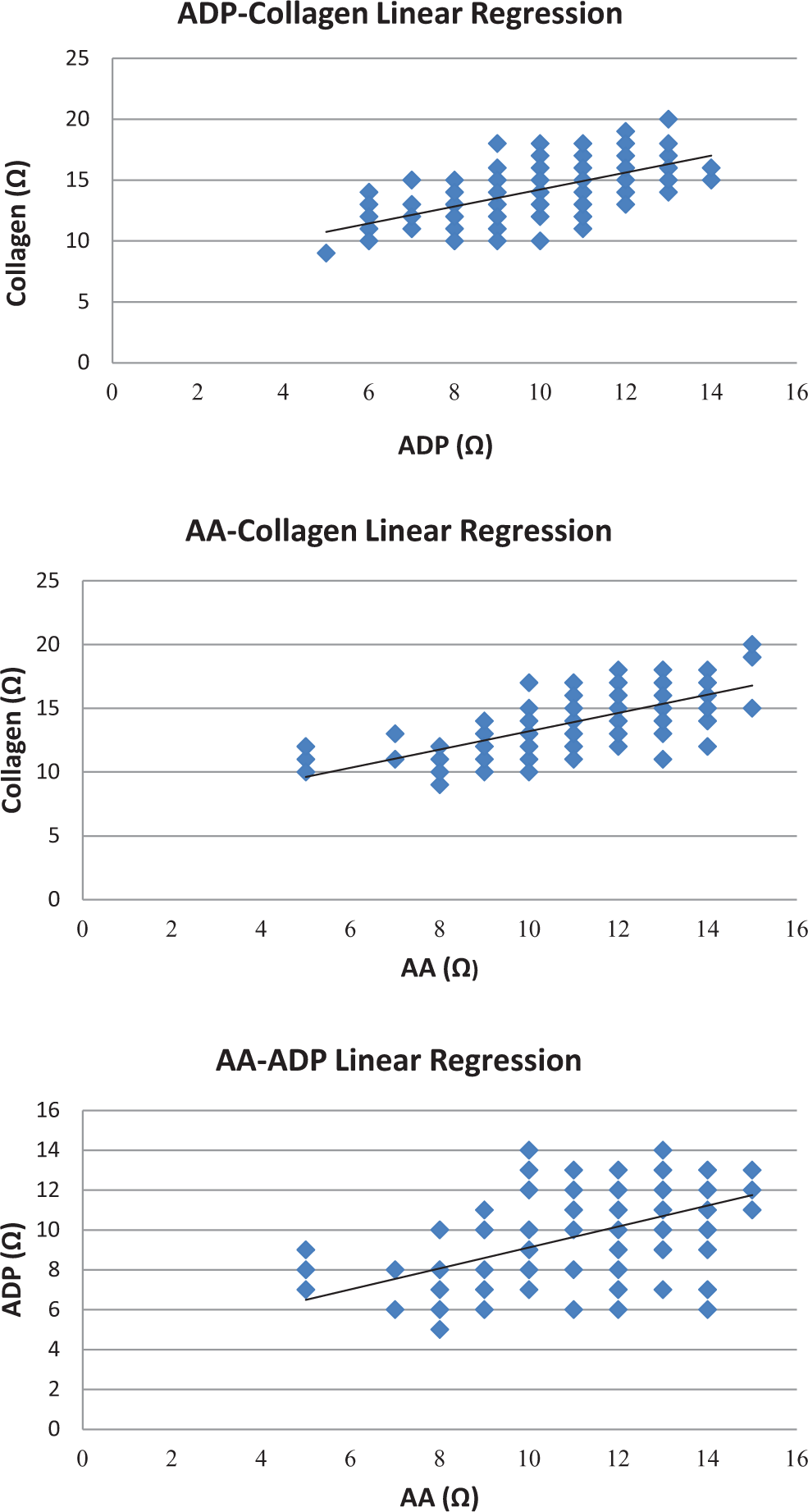
Linear regression among platelet activity stirred by different agonists.
Responsiveness for Loading Dose of Clopidogrel
A total of 30 patients’ RPAs at 2 hours after 300 mg clopidogrel loading are presented in Table 3. Employing ADP as the agonist, the mean RPAs of the impedance method was 1 Ω (± 2.3 Ω), corresponding to 54.9% (± 14.0%) of the optical method. Meanwhile, employing collagen as the agonist, the mean RPAs of the impedance method was 6.7 Ω (± 2.8 Ω), corresponding to 54% (± 19%) of the optical method.
Residual Platelet Activities 2 Hours After Loading 300 mg Clopidogrel.
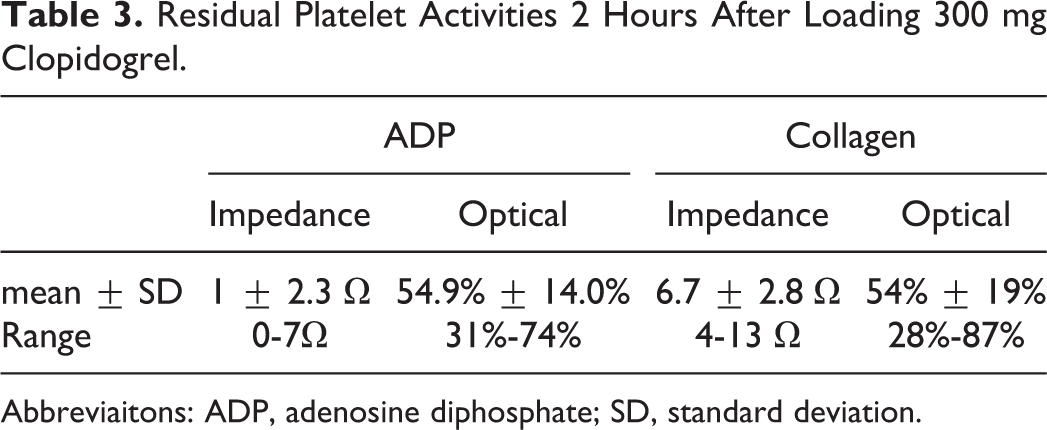
Abbreviaitons: ADP, adenosine diphosphate; SD, standard deviation.
Residual Platelet Activity Associated With Major Clinical Events
An average of 10 months of follow-up was achieved in patients with ACS enrolled in this study. Pertinent demographic information of followed patients with ACS is provided in Table 4. Major events occurred in 18 (9%) of the 202 patients.
Pertinent Demographic Information of Followed Patients With ACS.
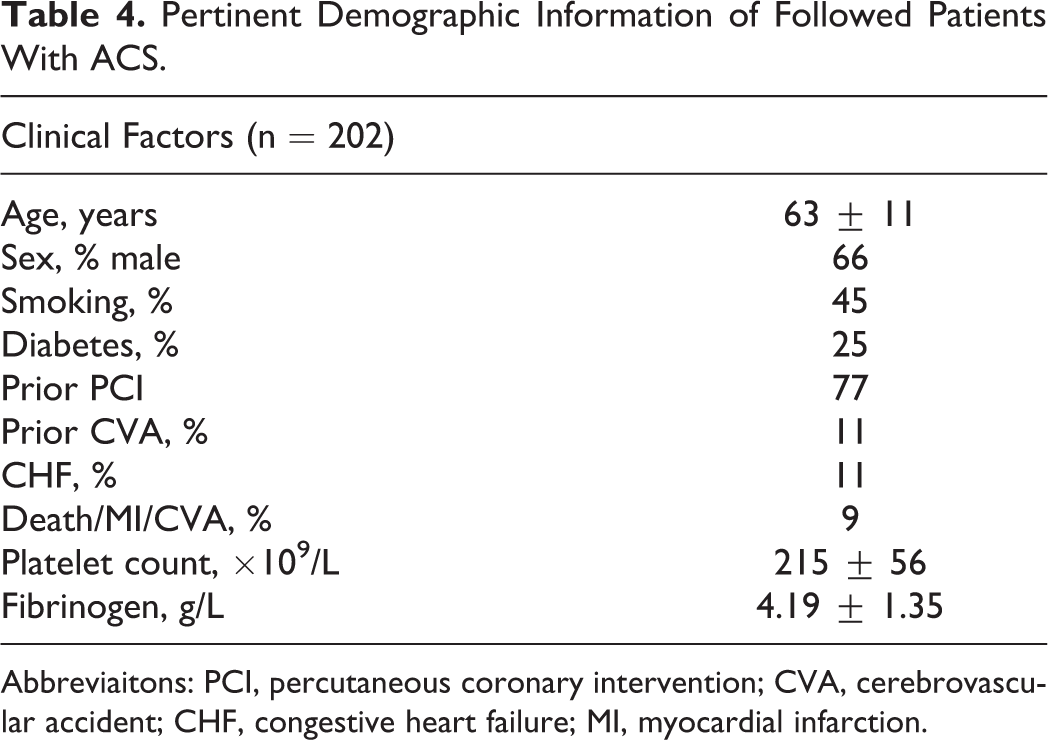
Abbreviaitons: PCI, percutaneous coronary intervention; CVA, cerebrovascular accident; CHF, congestive heart failure; MI, myocardial infarction.
Residual platelet activities of patients who experienced death, MI, or CVA were significantly higher than those who did not experience (3 agonists resulting in
Differences of Residual Platelet Activity Between Different Outcomes.
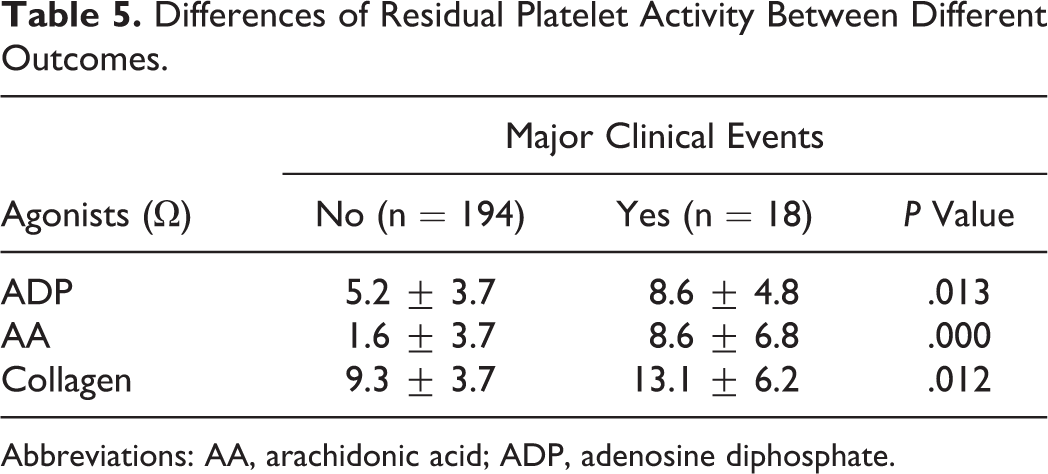
Abbreviations: AA, arachidonic acid; ADP, adenosine diphosphate.
Clinical Performance of Impedance Method for Major Clinical Events.a
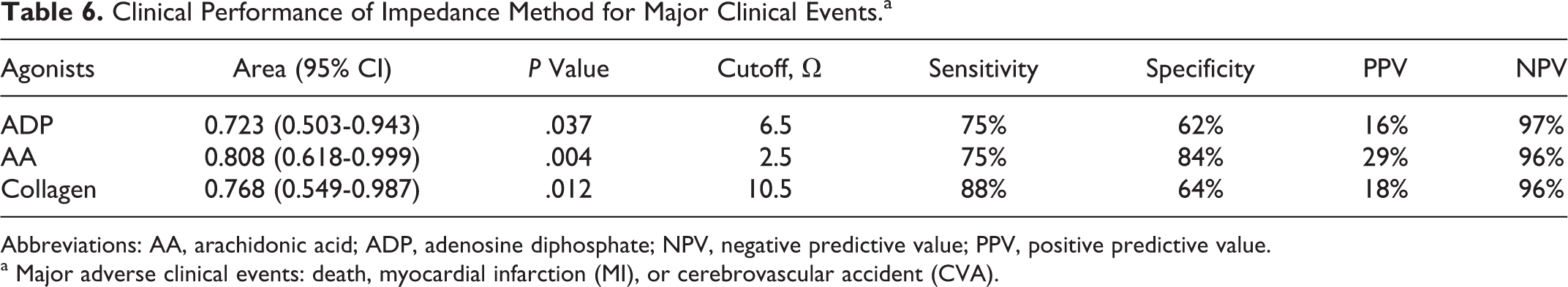
Abbreviations: AA, arachidonic acid; ADP, adenosine diphosphate; NPV, negative predictive value; PPV, positive predictive value.
a Major adverse clinical events: death, myocardial infarction (MI), or cerebrovascular accident (CVA).
Discussions
“Platelet function assays are inherently variable because they measure cell function rather than a single analyte.” 5 Platelet function results, therefore, have poor comparability within patients and between patients. Optical platelet aggregation is usually regarded as the gold standard of platelet function testing. However, it is relatively nonphysiological as platelets are induced after being separated from other blood cells by centrifugation, which may contribute to certain part of its results variation. In response to this disadvantage, impedance platelet aggregation activating platelets in whole blood is more physiological and less sample volume is required. 6 According to the results of our study, the impedance platelet aggregation has a better precision than the optical platelet aggregation (CV 4.81% vs 7.67%), which mostly attributes to no centrifugation involved in impedance aggregation procedures.
Each laboratory must establish its particular reference intervals based on its instrumentation and demographics of the population it serves, Conforming to the CLSI document CA28-A3c, 7 we established the reference intervals based on Han ethnicity population for local laboratory reference. In light of the statistics listed in Table 2, the results of platelet activity induced by ADP, AA, and collagen presented good mutual correlations that are consistent with the synergistic effects among antiplatelet drugs.
The recommended loading dose of clopidogrel is 300 to 600 mg in the setting of PCI. 8 We assessed 30 patients’ RPAs at 2 hours after 300 mg loading by both the impedance method and the optical method. Comparing the reference intervals established, platelet activities induced by ADP and collagen were both dramatically inhibited within 2 hours, though the ranges of minimum to maximum are quite wide (Table 3), implying that the variations in loading dose inhibition effect among the patients are large. Optimum RPAs that resulted from clopidogrel loading have not been well defined, 9,10 but ADP-induced aggregation (by optical method) >40% may experience lower risk of perioperative bleeding according to the study of Chen et al. 11 Consequently, the mean ADP aggregation of 1 Ω by the impedance method, corresponding to 54.9% by the optical method, obtained in our study may indicate that 300 mg loading dose generally do not expose patients undergoing emergent PCI to the risk of perioperative bleeding. Besides, collagen is highly thrombogenic. There have been studies 12,13 demonstrating that surface expression of platelet collagen receptor glycoprotein VI (GPVI) is elevated in patients with ACS and associated with acute coronary events. In our study, collagen-induced platelet aggregation, mainly mediated by GPVI, declined after loading 300 mg clopidogrel. This is consistent with the fact that clopidogrel may limit ADP-induced GPVI dimerization, resulting in the reduction of collagen-induced platelet activation. 14
Residual platelet activity may be associated with adverse cardiovascular events in patients with ACS.
5,15
–17
In our study, patients who experienced death, MI, or CVA had significantly higher RPA than patients who did not experience (3 agonists resulting in
There are a few limitations to this study. Residual platelet activities of followed patients with ACS were tested only once and merely 18 patients who had major clinical events during the follow-up may compromise the efficacy of statistics. In addition, we focused on the association between RPAs and clinical outcomes without unifying the regimen of antiplatelet drugs.
Conclusions
Impedance whole blood aggregation may be a potentially reliable method for monitoring RPA despite antiplatelet drug therapy whose optimal clinical cutoff values have high exclusionary predictive value for the occurrence of major clinical events in patients with ACS.
Footnotes
Acknowledgments
The authors are very grateful to Dr Joyce Rogers (Pathology and Laboratory Medicine Institution, Cleveland Clinic) for her kind review of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by The Seeds Fund of Peking University Third Hospital.
