Abstract
Selective intensification of platelet inhibition may improve high on treatment platelet reactivity (HPR). We evaluated the efficacy of dual-antiplatelet therapy, including clopidogrel (CPG), compared to new P2Y12-receptor antagonists in patients with HPR undergoing percutaneous coronary intervention, regarding the outcome of composite major adverse cardiac events (MACEs, including death, acute coronary syndrome [ACS], and stent restenosis). The presence of HPR (71 of 181 patients) almost doubled the risk of MACEs. The new antiplatelet agent reduced MACEs (45.8%, 26%, and 16.7% for CPG, prasugrel, and ticagrelor [TGL]; RR 0.36; 0.13-0.98, P = .03, TGL), specifically in patients with ACS. Failure to reduce HPR after the antiplatelet change and diabetes were independent predictors for MACEs. The HPR was early and effectively reduced after changing the antiplatelet therapy, but the intensity of this reduction did not significantly decrease the risk of MACEs. These findings support the benefit of HPR-guided intensification of platelet inhibition. Whether the intensity of this reduction improves the patient’s clinical outcomes deserves further investigation.
Keywords
Introduction
Clopidogrel (CPG) is widely used in the management of coronary artery disease, as part of a dual antiplatelet therapy either in the setting of acute coronary syndrome (ACS) or for the treatment of stable angina. Additionally, CPG is frequently used in the settings of percutaneous coronary intervention (PCI) to prevent stent thrombosis and ACS. However, many patients still experience recurrent ischemic events, which may be due in part to the variability of the response to dual antiplatelet therapy. The so-called high on treatment platelet reactivity (HPR) refers to a poor response to antiplatelet agents, which is consistently associated with the recurrence of ischemic events. The identification of HPR may predict a higher risk of ischemic complications in patients with stable angina who are submitted to PCI; furthermore, therapy with novel P2Y12-receptor antagonists may reduce HPR and provide a greater antiplatelet effect than CPG in patients with ACS, both during the first hours of treatment and during the maintenance therapy. 1,2
To overcome HPR, changing from the standard CPG treatment to other P2Y12-receptor antagonists with higher antiplatelet effects has been investigated. Most of these studies 3,4 have demonstrated a decreased ischemic risk associated with a change in antiplatelet therapy and a consequent reduction in HPR. Whether the intensity of platelet inhibition is related to a lower risk is not yet clear, because the results from these studies are not consistent, and these studies are heterogeneous regarding the study design, the trial drug, and/or the cardiovascular risk of the study population. 3 –5
The present study was designed to explore the effect of HPR in a cohort of patients treated with CPG who were submitted to urgent and elective PCI and to estimate the impact of changing to new P2Y12-receptor antagonists on the clinical outcome of major adverse cardiac events (MACEs) during a 6-month follow-up.
Methods
Patients
A total of 181 patients with risk factors and/or a previous history of ischemic heart disease were treated for current coronary artery disease (chronic stable angina and ACS, including unstable angina, ST-segment elevation myocardial infarction [STEMI], and non-ST-segment elevation myocardial infarction [NSTEMI]) through coronary stenting with drug-eluting stents according to standard techniques. These patients were followed from March 2012 to February 2013. Patients with active bleeding, hemorrhagic diathesis, aspirin or thienopyridine allergy, history of stroke, weight lower than 60 kg, recent use of prasugrel (PSG) or ticagrelor (TGL), recent use of periprocedural glycoprotein IIb/IIIa inhibitors, or any anticoagulant were excluded from the study. Dual antiplatelet therapy previous to PCI was aspirin 81 to 300 mg/d and CPG 75 mg/d in patients with chronic stable angina. Patients with ACS received an aspirin loading dose (LD) of 300 mg and a CPG LD of 600 mg immediately before PCI. This study was designed and conducted according to the Declaration of Helsinki and was approved by the local ethics committees. All patients signed the written informed consent.
Platelet Function Test and Definition of HPR
The platelet reactivity to CPG was assessed at the beginning of the study. The platelet reactivity test was performed with a commercially available “VerifyNow P2Y12 Test” (Accumetrics, San Diego, California) by following the supplier’s recommendations. Briefly, 3 mL of arterial blood was obtained during the PCI procedure, was placed in a tube containing a 3.2% sodium citrate solution (GreinerBio-OneVacuette; North America, Inc, Monroe, North Carolina), and was analyzed within the next 15 minutes. The assay is based on adenosine diphosphate (ADP)-stimulated platelet activation and prostaglandin E1-induced increases of intraplatelet cyclic adenosine monophosphate to achieve more specific effects of ADP on the P2Y12 receptor. Platelet function was measured based upon the ability of activated platelets to bind to fibrinogen-coated microparticles and consequent aggregation with an expected reduction in binding after exposure to the P2Y12 inhibitor. The assay reports the extent of platelet aggregation in P2Y12 reaction units (PRUs), which depicts the amount of ADP-mediated aggregation specific to the platelet P2Y12 receptor and is calculated as a function of the rate and extent of platelet aggregation in the ADP channel. In this study, HPR was defined as higher than 208 PRU during CPG treatment as described previously.
Change in Antiplatelet Therapy
Patients under dual antiplatelet therapy, including CPG, who were also identified with HPR, were randomly assigned to a newer P2Y12-receptor antagonist, including PSG LD of 60 mg and 10 mg/d or TGL LD 180 mg and 90 mg twice a day. For comparison, a group of patients continued CPG LD of 300 mg and 75 mg/d. The platelet reactivity was measured again after 24 hours from the first determination as well as after 6 months; the persistence of a PRU value higher than 208 at these time points was considered to be an additional variable. The percentage of reduction in platelet reactivity was calculated to perform the quartile-grouped analysis. Patients, attending physicians, and laboratory personnel assessing the platelet reactivity were blinded to the study treatments assigned after detection of HPR, including change to PSG, TGL, or maintenance under CPG therapy.
Statistical Analysis
For descriptive statistics, continuous variables are reported as the mean ± standard deviation, and categorical variables are reported as frequencies and percentages. The normal distribution of the variables was estimated by the Kolmogorov-Smirnov test. The comparative analysis of the continuous variables was performed with an unpaired t test, Mann-Whitney U test, or analysis of variance; categorical variables were compared with a chi-square or Fisher exact test (applied as appropriate). The association between antiplatelet treatment and primary end points was calculated by the relative risk and 95% confidence intervals (95%CIs) or by a comparison through quartiles based on the percentage of reduction in the PRU value. An independent association of the baseline characteristics through a multivariate analysis was also performed using logistic regression. The statistical analyses were performed using SPSS v19.0 for Windows (SPSS Inc, Chicago, Illinois); P values <.05 (2 tailed) were considered to be significant.
End Points and Definitions
The primary end points were determined during the 6-month follow-up period and consisted of composite MACEs (death, ACS, and stent restenosis) in a study population with HPR treated with a standard CPG dose. Acute coronary syndrome was defined as the presence of unstable angina, NSTEMI, or STEMI. Unstable angina was defined as ischemic discomfort longer than 10 minutes occurring at rest or in an accelerating pattern with frequent episodes prompting an unscheduled visit to an emergency department within 24 hours of the most recent symptoms with a hospital stay of 24 hours and associated with new or worsening ST- or T-wave changes and negative myocardial necrosis markers. Myocardial infarction after PCI was defined as the presence of clinical signs with concomitant increases in creatine kinase MB (CK-MB) and troponin higher than 3 times the upper reference values with a previous determination of myocardial necrosis markers within the reference values and a subsequent increment of more than 50% the baseline value in patients with already elevated baseline levels of CK-MB (similar to the recommendation from the Joint European Society of Cardiology/American College of Cardiology Foundation/American Heart Association/World Heart Federation task force consensus statement on the redefinition of Myocardial Infarction for clinical trials on coronary intervention). Spontaneous ischemic events with similar characteristics associated with electrocardiographic changes indicative of new ischemia (new ST-T changes or new left bundle branch block), which occurred at any time within the 6-month follow-up post-PCI, were also recorded. The NSTEMI or STEMI was classified according to ST-elevation, which is defined as a new ST-segment elevation at the J point in 2 contiguous leads with the cut-points: higher than 0.1 mV in all leads other than leads V2 to V3, where applicable cut-point is higher than 0.2 mV. Finally, stent restenosis was defined as the angiographic evidence of a filling defect or lucency surrounded by contrast material found in multiple projections that originated in the stent in the segment 5 mm proximal or distal to the stent and the presence of at least 1 of the following criteria within a 48-hour time window: acute onset of ischemic symptoms at rest, new ischemic electrocardiogram changes that suggested acute ischemia, or typical dynamic curve of myocardial necrosis markers. The other end point was the composite MACEs and platelet reactivity in a study population with HPR that changed their antiplatelet agent. The secondary end points included each of the MACEs components and any adverse drug reactions and drug discontinuation during the study time period.
Results
Baseline Characteristics and Prevalence of HPR
A total of 181 patients with current coronary artery disease, 96 with chronic stable angina and 85 with ACS (including 23 with unstable angina, 38 with STEMI, and 24 with NSTEMI) submitted to coronary stenting, were enrolled. After platelet function screening, patients with HPR were randomly changed to either PSG LD of 60 mg followed by 10 mg once daily (n = 23) or TGL LD of 180 mg followed by 90 mg twice a day (n = 24). A control group of patients with HPR was maintained on CPG LD of 300 mg followed by 75 mg once daily (n = 24). The clinical demographic characteristics of the study population are shown in Table 1.
Basal Characteristics in the Study Population.a
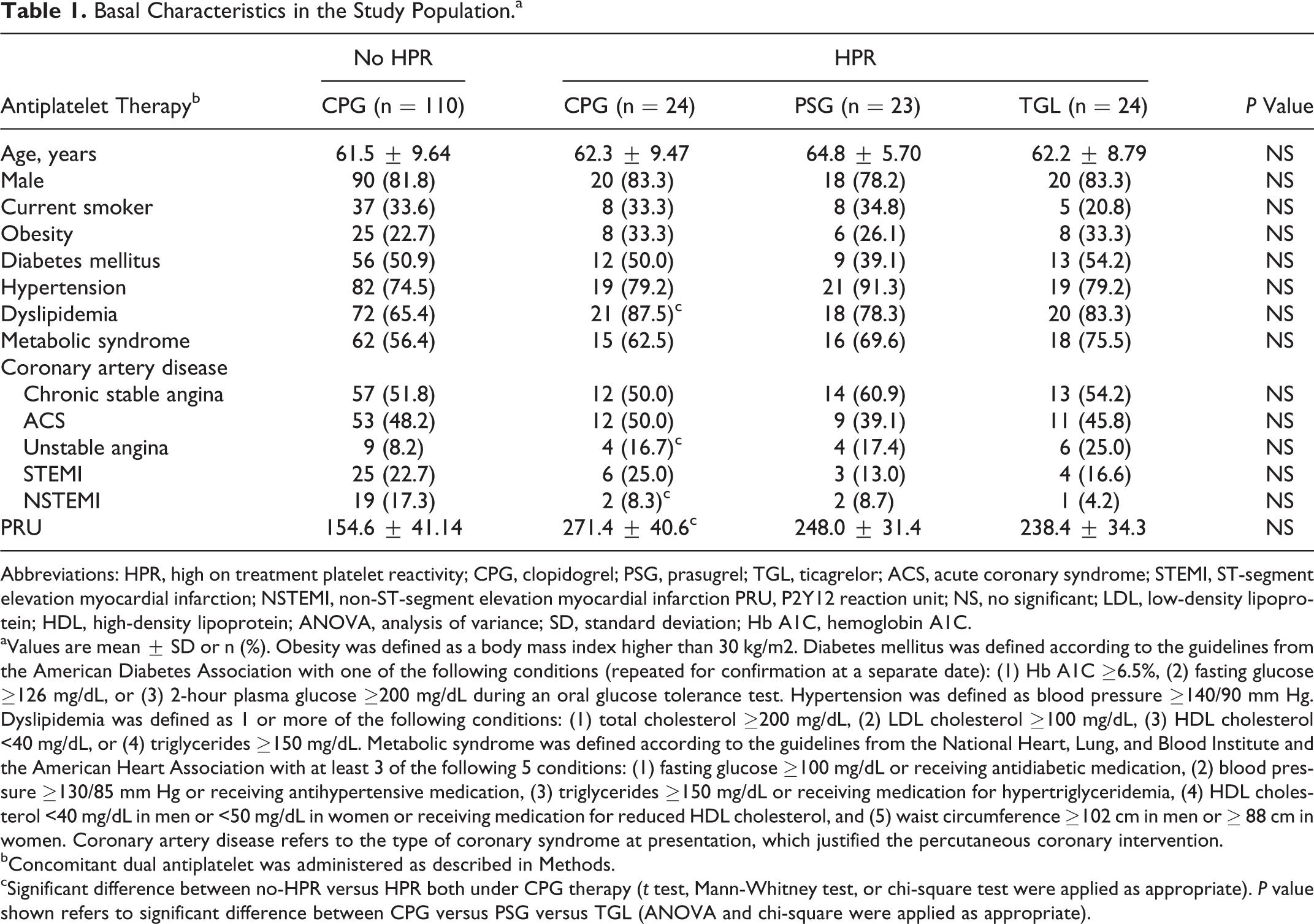
Abbreviations: HPR, high on treatment platelet reactivity; CPG, clopidogrel; PSG, prasugrel; TGL, ticagrelor; ACS, acute coronary syndrome; STEMI, ST-segment elevation myocardial infarction; NSTEMI, non-ST-segment elevation myocardial infarction PRU, P2Y12 reaction unit; NS, no significant; LDL, low-density lipoprotein; HDL, high-density lipoprotein; ANOVA, analysis of variance; SD, standard deviation; Hb A1C, hemoglobin A1C.
aValues are mean ± SD or n (%). Obesity was defined as a body mass index higher than 30 kg/m2. Diabetes mellitus was defined according to the guidelines from the American Diabetes Association with one of the following conditions (repeated for confirmation at a separate date): (1) Hb A1C ≥6.5%, (2) fasting glucose ≥126 mg/dL, or (3) 2-hour plasma glucose ≥200 mg/dL during an oral glucose tolerance test. Hypertension was defined as blood pressure ≥140/90 mm Hg. Dyslipidemia was defined as 1 or more of the following conditions: (1) total cholesterol ≥200 mg/dL, (2) LDL cholesterol ≥100 mg/dL, (3) HDL cholesterol <40 mg/dL, or (4) triglycerides ≥150 mg/dL. Metabolic syndrome was defined according to the guidelines from the National Heart, Lung, and Blood Institute and the American Heart Association with at least 3 of the following 5 conditions: (1) fasting glucose ≥100 mg/dL or receiving antidiabetic medication, (2) blood pressure ≥130/85 mm Hg or receiving antihypertensive medication, (3) triglycerides ≥150 mg/dL or receiving medication for hypertriglyceridemia, (4) HDL cholesterol <40 mg/dL in men or <50 mg/dL in women or receiving medication for reduced HDL cholesterol, and (5) waist circumference ≥102 cm in men or ≥ 88 cm in women. Coronary artery disease refers to the type of coronary syndrome at presentation, which justified the percutaneous coronary intervention.
bConcomitant dual antiplatelet was administered as described in Methods.
cSignificant difference between no-HPR versus HPR both under CPG therapy (t test, Mann-Whitney test, or chi-square test were applied as appropriate). P value shown refers to significant difference between CPG versus PSG versus TGL (ANOVA and chi-square were applied as appropriate).
High on Treatment Platelet Reactivity-Associated Risk
The prevalence of HPR in the study population was 39.2% (71 of 181) and nearly doubled the risk of the composite end point (Table 2, upper row). After the stratification analysis for risk adjustment by acute or stable settings and the heterogeneously distributed characteristics (Table 2, lower row), the presence of ACS, dyslipidemia, and previous myocardial infarction were significant positive interacting factors. The HPR did not significantly affect the risk of the secondary end point according to the analysis for each particular event: stent restenosis RR 1.97 (95% CI = 0.71-5.41; P = .14); ACS, RR 3.05 (95% CI = 0.54-17.3; P = .22), and both RR 4.58 (95% CI = 0.68-30.9; P = .15).
Effect of HPR on Prevalence and Risk of Primary Composite End Point.a
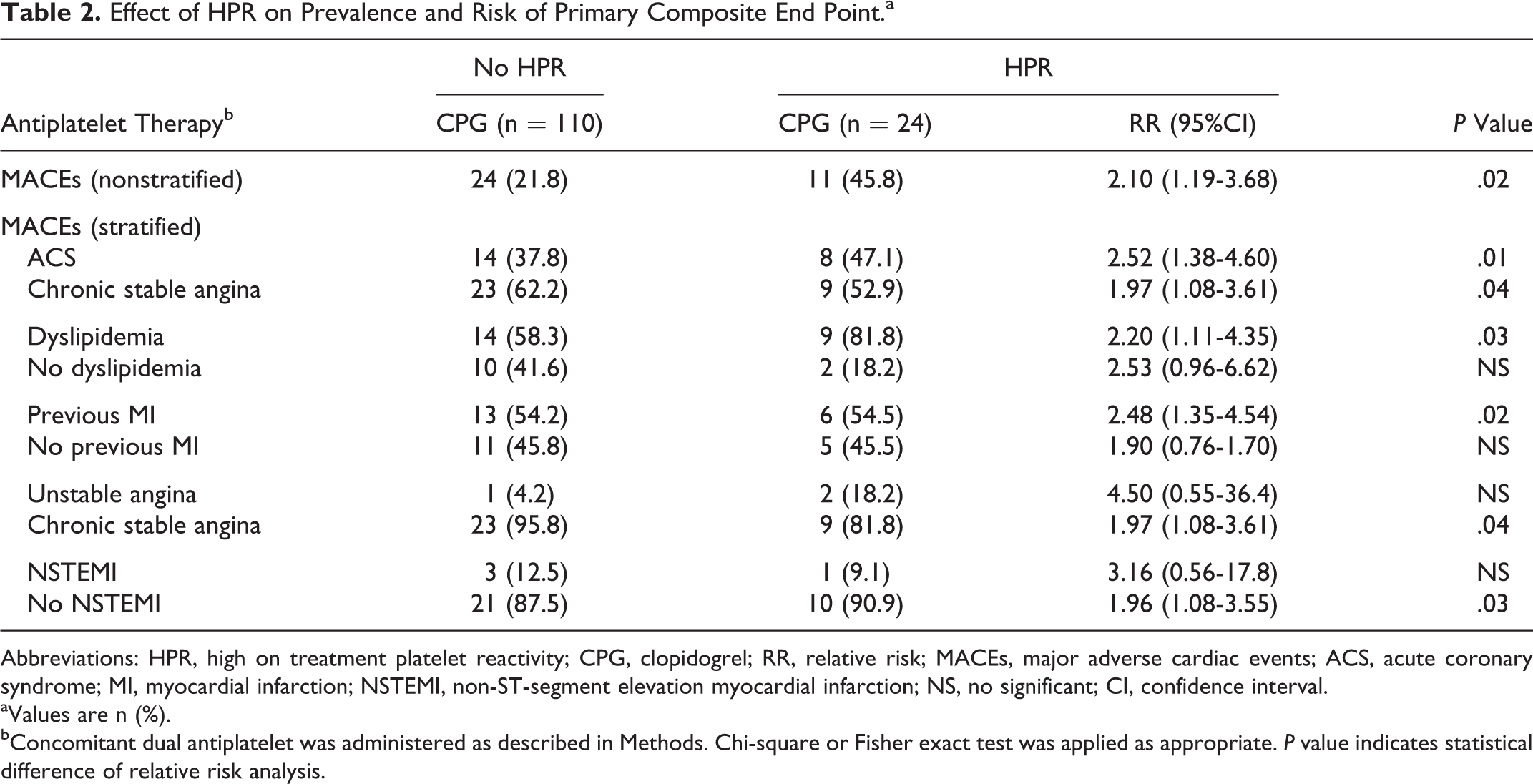
Abbreviations: HPR, high on treatment platelet reactivity; CPG, clopidogrel; RR, relative risk; MACEs, major adverse cardiac events; ACS, acute coronary syndrome; MI, myocardial infarction; NSTEMI, non-ST-segment elevation myocardial infarction; NS, no significant; CI, confidence interval.
aValues are n (%).
bConcomitant dual antiplatelet was administered as described in Methods. Chi-square or Fisher exact test was applied as appropriate. P value indicates statistical difference of relative risk analysis.
Effect of Antiplatelet Change
Patients with HPR were subdivided and randomly assigned to an antiplatelet change group or control group that remained on CPG therapy. The resulting subgroups showed similar clinical demographic characteristics (Table 1), and all of them completed the 6-month follow-up. No adverse drug reactions, side effects, or drug discontinuations were recorded during this period.
The prevalence and relative risk of the primary composite MACEs are shown in Table 3. The percentage of MACEs decreased in the antiplatelet change group, which resulted mainly from TGL when comparing each antiplatelet. A positive interaction was observed with the ACS, and no significant risk was attributed to the individual components of the primary MACE end point.
Prevalence and Risk of MACEs After Antiplatelet Change.a
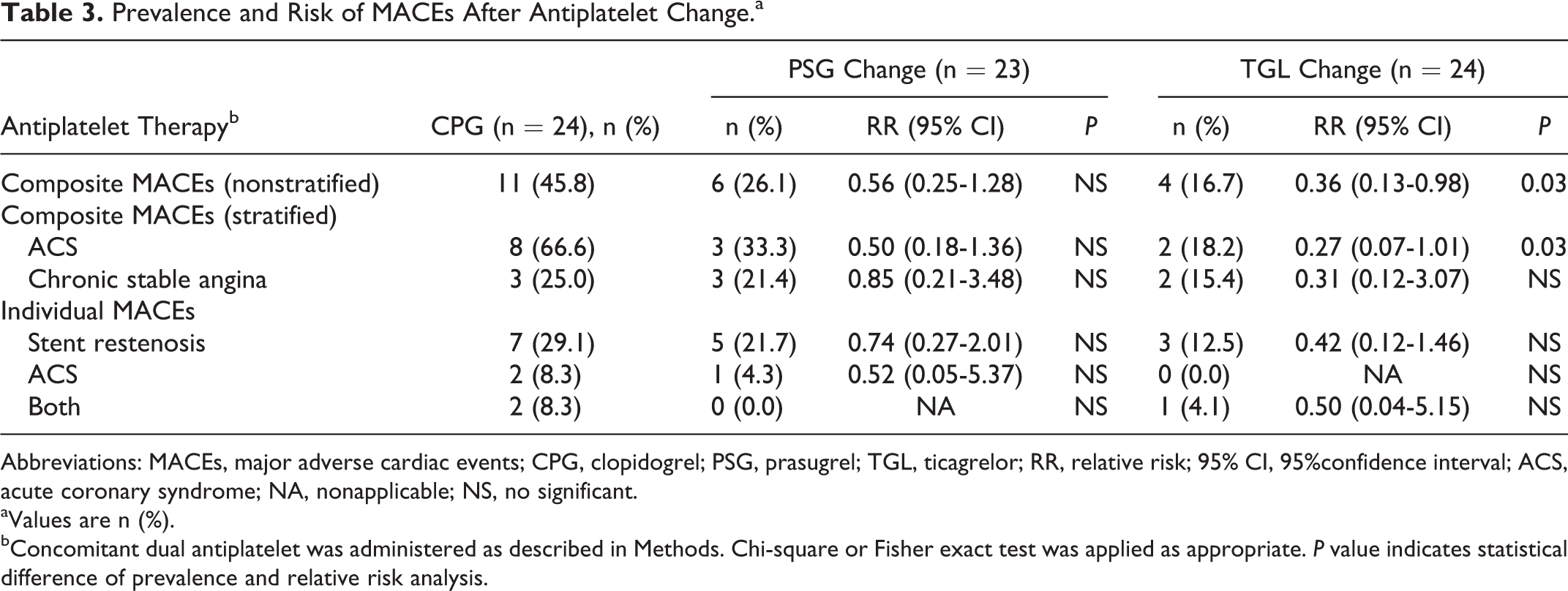
Abbreviations: MACEs, major adverse cardiac events; CPG, clopidogrel; PSG, prasugrel; TGL, ticagrelor; RR, relative risk; 95% CI, 95%confidence interval; ACS, acute coronary syndrome; NA, nonapplicable; NS, no significant.
aValues are n (%).
bConcomitant dual antiplatelet was administered as described in Methods. Chi-square or Fisher exact test was applied as appropriate. P value indicates statistical difference of prevalence and relative risk analysis.
The change in antiplatelet treatment effectively reduced platelet reactivity in the patients with HPR. As early as 24 hours from antiplatelet change (Figure 1, left), there was a 31% and 45% reduction in PRU after change to PSG and TGL, respectively; and after 6 months (Figure 1, right), there was a 47% and 62% reduction, respectively. Notably, a higher reduction was observed in the group treated with TGL at both time points.
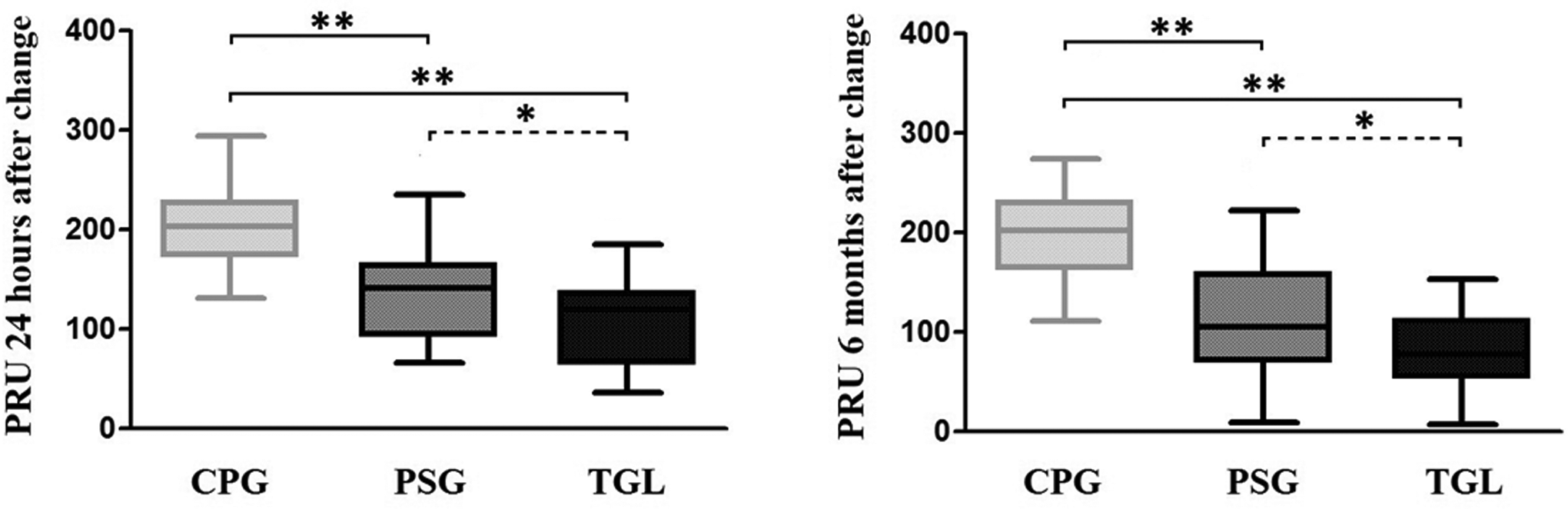
Reduction in high on treatment platelet reactivity (HPR) after antiplatelet change. t Test or Mann-Whitney test were used as appropriate. Statistical difference in PRU values (CPG, PSG, and TGL: 203.7 ± 40.98, 140.0 ± 47.72, and 106.1 ± 45.87, respectively, at 24 hours, while 199.8 ± 42.49 vs 109.6 ± 55.85 vs 75.7 ± 39.96, respectively, at 6 months) is indicated as (*) P < .01 and (**) P < .0001. PRU indicates P2Y12 reaction unit; CPG, clopidogrel; PSG, prasugrel; TGL, ticagrelor.
When grouped by quartiles of percentage of reduction in platelet reactivity (PRU value), a nonsignificant risk reduction for MACEs was observed when analyzed as the combined results of PSG and TGL (0.35, 95% CI 0.11-0.98, P = .06). Likewise, low association was observed between quartiles and the prevalence of MACEs within the different antiplatelet groups (Figure 2). A correlation was also calculated for comparison.
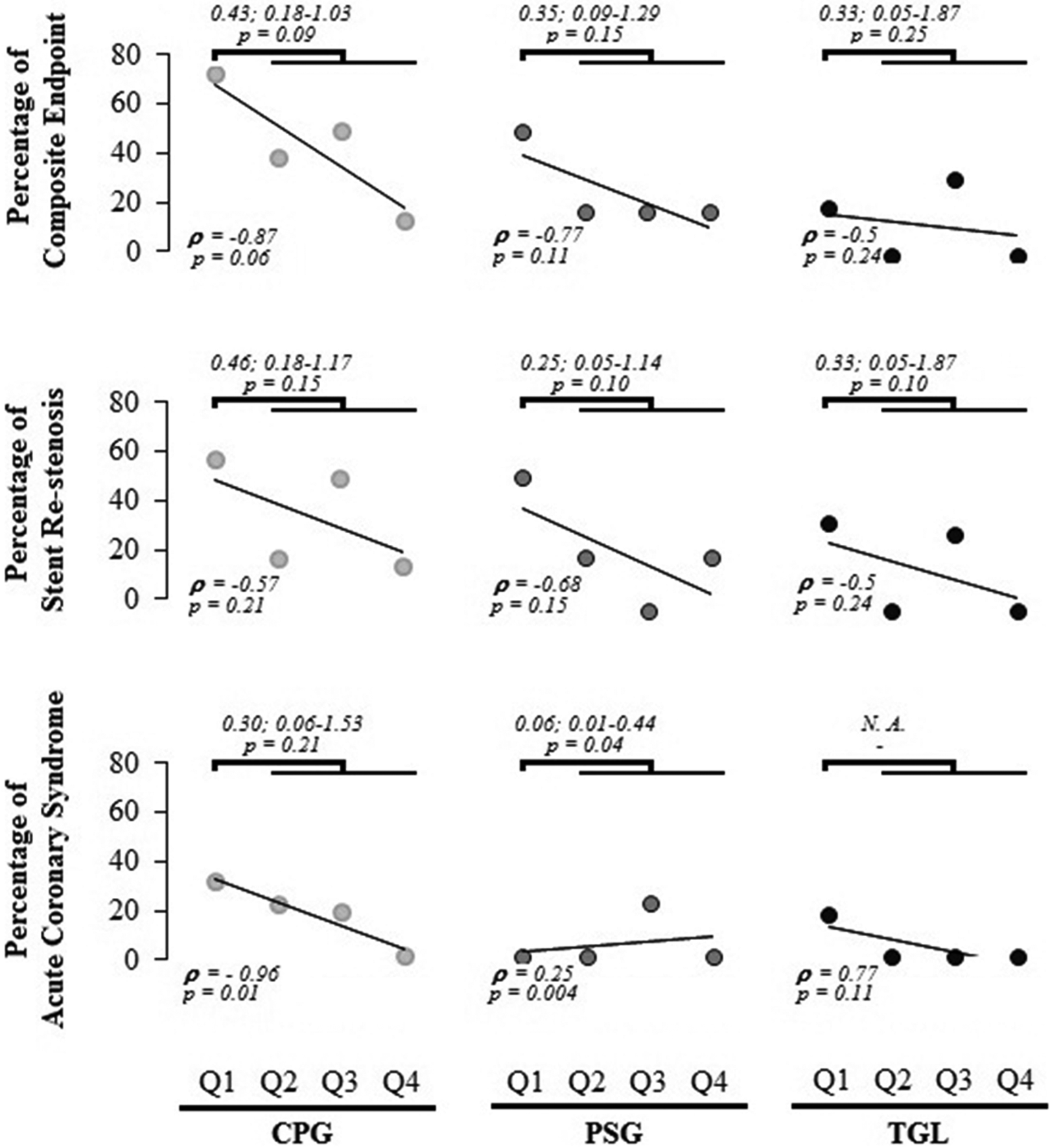
Comparison of major adverse cardiac events (MACEs) between quartiles (Q) of reduction in platelet reactivity. Quartiles according to percentage of reduction in PRU value were formed. CPG: Q1 (0-12.16), Q2 (12.16-25.02), Q3 (25.02-35.33), and Q4 (>35.33); PSG: Q1 (0-35.29), Q2 (35.29-40.25), and Q3 (40.25-60.09), Q4 (>60.09); and TGL: Q1 (0-41.88), Q2 (41.88-50), Q3 (50-74.77), and Q4 (>74.77). Relative risk (indicated on top, expressed as mean; 95% confidence interval) and correlation (ρ, left-bottom) were calculated. CPG indicates clopidogrel; PSG, prasugrel; TGL, ticagrelor; PRU, P2Y12 reaction unit.
With a multivariable analysis of the basal characteristics of the HPR group, the independent predictors of MACEs were the persistently high values of platelet reactivity after 24 hours of CPG change Exp(B) 22.8 (3.73-139.81; P < .001) and diabetes mellitus Exp(B) 6.1 (1.10-34.01; P < .056).
Discussion
Almost 40% of the study population demonstrated HPR with CPG. This prevalence is moderately high when compared with other studies that demonstrate HPR ranging from 5% and 21% up to 55% depending on the race, type of intervention, and assay for platelet reactivity. 6 –8
To our knowledge, this the first study in a Latin American population submitted to PCI analyzing the HPR-related risk of MACEs and assessing the impact of new P2Y12-receptor antagonists. Our data analysis indicates that HPR almost doubled the risk of the composite end point of MACEs during the 6-month follow-up, which was nonsignificant for each one of the components. Our results are consistent with reports of composite MACEs involving death, myocardial infarction, and stent thrombosis from previous studies, either in the HPR-associated risk (10.9%-15.8% vs 6.9%-5.8% of composite MACEs in population with HPR vs no HPR, respectively) or in the risk attributed to the change in antiplatelet (1%-1.2% up to 9.8% vs 2.9%-3.7% up to 22.6% in the group changed to P2Y12 antagonists vs the group maintained under CPG treatment, respectively). 4,9 –13 Although most of these studies used larger samples than the present study, the sample size calculated from previous reports at fixed errors α of .05 and β of .2 results in approximately 20 and 44 patients for the study of HPR-associated risk and the effect of antiplatelet change, respectively. A similar number of patients were included in this study (24 patients with HPR under CPG treatment; 47 patients in the group changed to PSG or TGL). Finally, the ultimate risk observed here deserves careful interpretation due to potential interactions with situations such as the acute clinical setting and the antecedents of ischemic heart disease or dyslipidemia as suggested from a standardization analysis.
The secondary safety end point confirmed that changing to a new P2Y12-receptor antagonist is a safe procedure. There were no adverse events or drug discontinuations. Similarly, an HPR-guided change in antiplatelet drug elicited a reduction of more than 50% in the risk of composite MACEs during the 6-month follow-up. This effect was almost completely attributable to TGL and was specific to patients with ACS. Only a trend could be observed in the PSG groups or within a stable setting. A smaller effect was observed with each particular component. Such discrepancies with the composite end point may be due to the heterogeneity of the size of the comparative groups and/or an underestimation of the effect due to the short follow-up duration.
The efficacy of HPR-guided antiplatelet change has been demonstrated in different studies. 4 However, this benefit has been questioned by the results obtained in a series of trials. 11 –13 Although these trials have demonstrated no statistical risk modification, some claims have been raised regarding the trial drug’s antiplatelet effect and the low cardiovascular risk in the study population. 5,14 Hence, a definitive position has not been clearly established. In the present study, we performed an HPR-based change to a newer P2Y12-receptor antagonist with higher antiplatelet effects with PSG or TGL in a population with high cardiovascular risk. Our results support the advantage of changing to a new P2Y12-receptor antagonist for the prevention of MACEs in patients submitted to PCI.
As expected, antiplatelet change effectively reduced platelet reactivity in the population with HPR (up to 38% and 54% reduction after 24 hours and 6 months, respectively) compared to the control CPG-treated group. The highest reduction was observed in the group treated with TGL, which also demonstrated the highest reduction in risk of the composite end point. Moreover, a reduction in platelet reactivity and DM were both strong predictors of the composite end point in the multivariable analysis. Similarly, studies such as Platelet Inhibition and Patient Outcomes (PLATO) and Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with Prasugrel (TRITON) have demonstrated that the use of new P2Y12-receptor antagonists significantly reduces HPR. 15,16 However, whether the strength of platelet inhibition is associated with a risk modification of the clinical outcomes was not conclusive in the present study. There was a nonsignificant trend obtained when the population was grouped by the quartiles of percentage reduction in platelet reactivity.
In conclusion, the prevalence of HPR and the related risks in a Latin American population submitted to PCI is similar to that reported worldwide. The change to new P2Y12-receptor antagonists, but not the amount of platelet reactivity reduction, decreased the risk of MACEs in the group of poor CPG responders.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
