Abstract
The Multiplate, a whole blood (WB) platelet function test, has shown promising results identifying patients on antiplatelet therapy at increased risk of rethrombosis. In the present study, the influence of low platelet count on platelet aggregation was analyzed and compared with aggregation results in an artificial matrix, platelet-rich plasma (PRP). Heparinized and citrated blood was diluted with autologous plasma to platelet concentrations 200 to 25 × 109/L in WB samples (n = 10) and 200 to 100 × 109/L in PRP samples (n = 7). The platelet aggregation was investigated by the ADP-, ASPI-, COL-, and TRAP-test. The WB responses decreased at platelet concentration of ≤100 × 109/L (all P < .03), except for heparin-TRAP (50 × 109/L, P = .008) and citrate-ASPI (150 × 109/L, P = .03). In general, WB samples demonstrated higher aggregation than PRP samples at platelet concentrations 200 to 100 × 109/L (P < .05). In conclusion, platelet concentration of <150 × 109/L may influence Multiplate which should be considered in clinical settings. Furthermore, the findings emphasize the importance of evaluating haemostasis in its natural matrix, WB.
Introduction
The therapy of choice in preventing post-procedure thrombotic complications in patients undergoing percutaneous coronary intervention (PCI) is combination antiplatelet therapy with aspirin and clopidogrel. 1 Despite this combination therapy, more than 10% of the patients develop recurrent ischemic events.2–4 When evaluating platelet function by light transmission aggregometry (LTA), a subpopulation of patients display a reduced response to aspirin and clopidogrel, which is associated with increased risk of adverse cardiovascular events.2,5 Light transmission aggregometry, however, has important limitations for routine use, that is, it is time-consuming, requires skilled technicians, and is performed on platelet-rich plasma (PRP), and may hence not be suitable for routine clinical practice. 6 A new platelet function test, the multiple platelet function analyzer (Multiplate, Dynabyte Medical, Munich, Germany) has been introduced. This test is based on multiple electrode platelet aggregometry (MEA), which measures platelet aggregation in whole blood (WB) after stimulation with selective platelet agonists, and thereby allows analyzing the effect of antithrombotic drugs such as aspirin and clopidogrel on platelet aggregation. 6 The results obtained by the Multiplate have been reported to correlate with LTA. 7 Recently, a significant association between the degree of platelet inhibition, as evaluated by Multiplate, before PCI and the development of ischemic events 30 days post-PCI was found. This finding indicates that the assay may identify “high-risk patients” in this setting. 8
The influence of low-platelet concentration on the platelet aggregation result remains a matter of concern.9,10 A recent study reported reduced platelet aggregation at a platelet count of 100 × 109/L in hirudin-anticoagulated WB samples, when assessed by the Multiplate. 10 The clinical guidelines recommend standardized antiplatelet therapy no matter of the platelet count. 1 The consequences of a decreased aggregation response in patients with low platelet count could therefore be a misinterpretation of the result as sufficient antiplatelet therapy. These patients could therefore falsely be categorized as patients with no increased risk of a new thrombotic event measured by Multiplate. Given this, the main purpose of the present study was to assess the influence of a low platelet count on platelet aggregation against 4 different agonists (adenosine-diphosphate [ADP], arachidonic acid [ASPI], collagen [COL], and thrombin receptor-activating peptide 6 [TRAP]) in heparin- and citrate-anticoagulated WB evaluated by Multiplate. Furthermore, the purpose was to compare the platelet aggregation at different platelet concentrations in PRP and in WB.
Methods
After approval by Ethical Committee, 10 healthy volunteers, 5 men and 5 women, were enrolled in the WB study. In the PRP study, 7 of the 10 volunteers (4 women and 3 men) participated since the remaining 3 volunteers did not show up the day the blood samples were collected for the study. In the comparative study, the same 7 volunteers from the WB and PRP study were compared. Exclusions criteria were thrombocytopenia defined as platelet count below 150 × 109/L, daily use of any medication, and the use of antiplatelet medication within the last 7 days.
Whole Blood Preparation
Before blood collection, the volunteers were resting in a chair for 5 minutes. The blood was drawn by clean venipuncture from an anticubital vein using a 21-gauge butterfly (Vacuette, Breiner Bio-One GmbH, Kremsmünster, Austria). From each donor, six 4-mL WB samples, 3 containing 1/10 mL of 3.2% sodium citrate (0.109 mol/mL, Greiner Bio-One GmbH, Kremsmünster, Austria) and 3 containing lithium heparin (18 IU/mL, Greiner Bio-One GmbH, Kremsmünster, Austria) were collected. Immediately after blood collection, processing of the blood started with the measurements of the platelet count (Automated Haematology Analyser, SF-3000, Sysmex Corporation, Kobe, Japan). To generate autologous plasma, 8 mL of citrated and heparinized WB were centrifuged at 3000g for 10 minutes. The remaining 4 mL WB was resting for 20 minutes at room temperature before dilution with autologous plasma to achieve platelet concentrations of 200 × 109/L, 150 × 109/L, 100 × 109/L, 50 × 109/L, and 25 × 109/L.
Platelet-Rich Plasma Preparation
Sample collection and measurement of platelet count were performed as described above. Whole blood was centrifuged at 114g for 15 minutes to recover PRP in the supernatant and further centrifuged at 3500g for 10 minutes to recover platelet-poor plasma (PPP). The PRP samples were diluted with autologous PPP to achieve platelet concentrations of 200 × 109/L, 150 × 109/L, and 100 × 109/L. Because of the recognized difficulties in assessing platelet aggregation in very low platelet concentrations (<100 × 109/L), 9 the concentrations below this were not analyzed in the PRP study.
Multiplate Assay
The Multiplate is a platelet function analyzer based on WB impedance aggregometry. The principle of the method is that the attachment of the platelets aggregates onto metal electrodes is leading to an increase of the electrical impedance determined by each sensor (2 electrodes). The Multiplate applies 4 electrodes per test cell and this duplicate of measurements serves as an internal control. The results of the test are 2 curves with arbitrary aggregation units (AU) plotted against time. The mean value of the 2 curves is automatically expressed in AU. For each applied platelet agonist, the area under the aggregation curve (AUC, AU*min or U [1 U = 10 AU*min]) after 6-minute analysis is recorded by the Multiplate analyzer. In the clinical setting, the value U is used.11,12
The platelets were stimulated with 4 platelet agonists. The final concentration of each agonist was according to the manufacture’s instructions (Instrumentation Laboratory, Munich, Germany): Adenosine-diphosphate (ADP-test: 0.2 mmol/L per mL; 20 μL, final concentration: 6.5 μmol/L ADP), COL-test (100 μg/mL; 20 μL, final concentration: 3.2 μg/mL COL), ASPI-test (15 mM/mL; 20 μL, final concentration 0.5 mmol/L ASPI), and (TRAP-test: 1 mmol/L per mL; 20 μL, final concentration: 32 μmol/L per mL). Multiplate aggregation measurements in WB and PRP preparations were performed within 120 minutes after collection of the blood samples.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism Version 5.01. The effect of platelet concentration on platelet aggregation was investigated by Friedman test with the platelet concentration as grouping variables: 200 × 109/L (control), 150 × 109/L, 100 × 109/L, 50 × 109/L, and 25 × 109/L. Significant effect of concentration by Friedman test was evaluated by comparing platelet concentration of 200 × 109/L with each of the lower platelet concentrations by Bonferroni adjusted (×4) post hoc Wilcoxon signed rank test. Whole blood vs PRP at platelet concentrations 200 × 109/L, 150 × 109/L, and 100 × 109/L were compared by Wilcoxon signed rank test. P Values of <.05 were considered significant.
Results
There were no differences between males and females with regard to the platelet aggregation results and the data from both genders were consequently pooled (Figure 1A and B, data from WB study not shown). The correlation data for the heparinized samples showed a higher degree of variation (Figure 1A) as compared to citrated samples (Figure 1B). An explanation for this could be heparin-induced platelet activation and accompanying spontaneous platelet aggregation during the preparation of PRP in heparinized blood, leading to a desensitization of the platelets before stimulation. 13 However, only 2 out of 7 heparinized samples displayed a low-aggregation result as compared to 0 of the citrated samples (n = 7), emphasizing that the small sample size may have introduced a type II error.
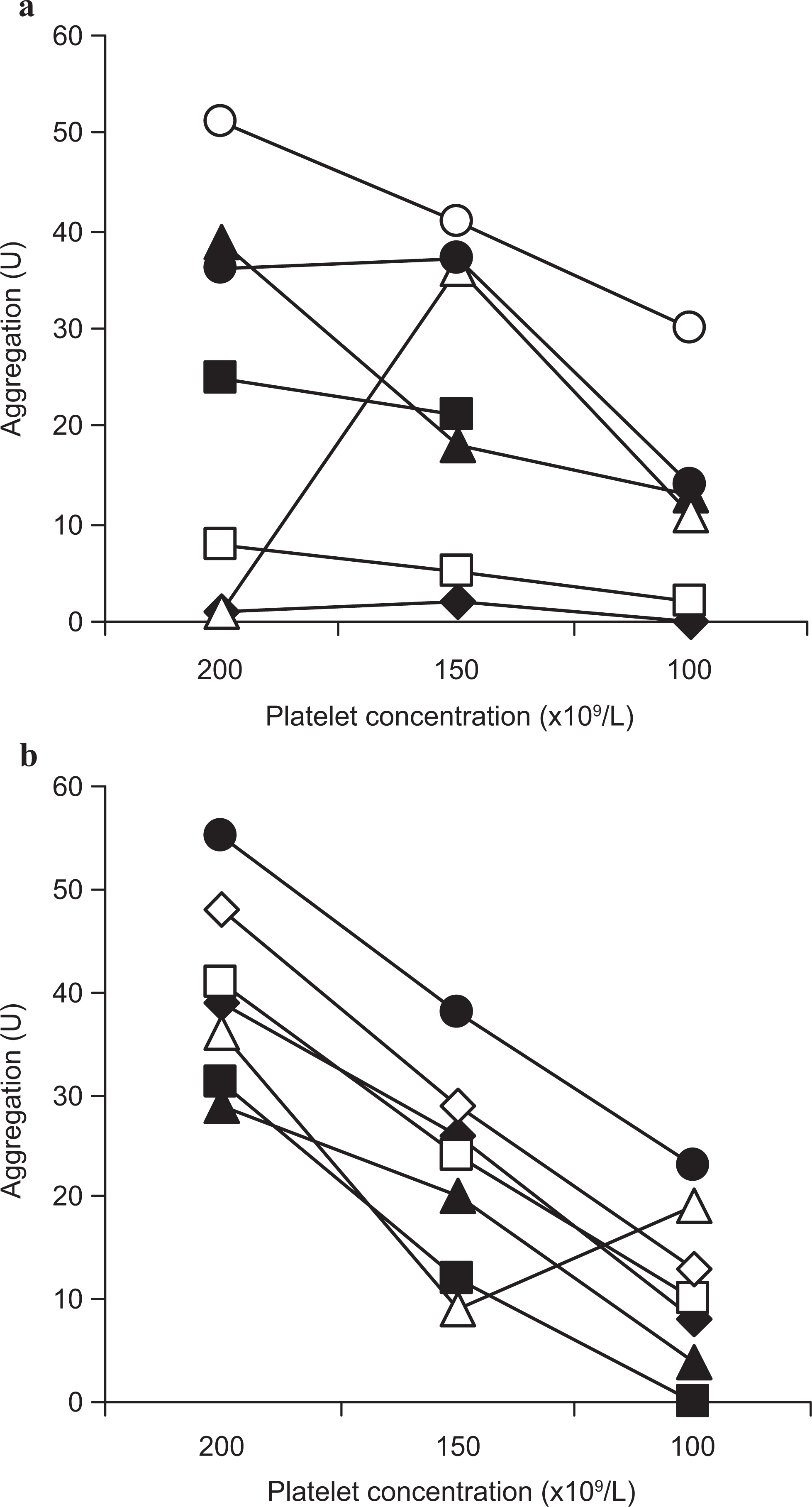
Platelet aggregation in platelet-rich plasma (PRP). Individual adenosine-diphosphate (ADP)-induced aggregation in 7 volunteers. The aggregation response was analyzed in heparinized (A) and citrated (B) PRP with platelet concentration of 200, 150, and 100 × 109/L. U indicates area under curve. Dark symbols (●), women; white symbols (○), men.
Values of red blood cells (RBCs), hematocrit, and white blood cells (WBCs) after adjustment of the platelet concentrations are summarized in Table 1 .
Dilution of Red Blood Cells (RBCs), Hemotocrit (Hct), and White Blood Cells (WBCs) a
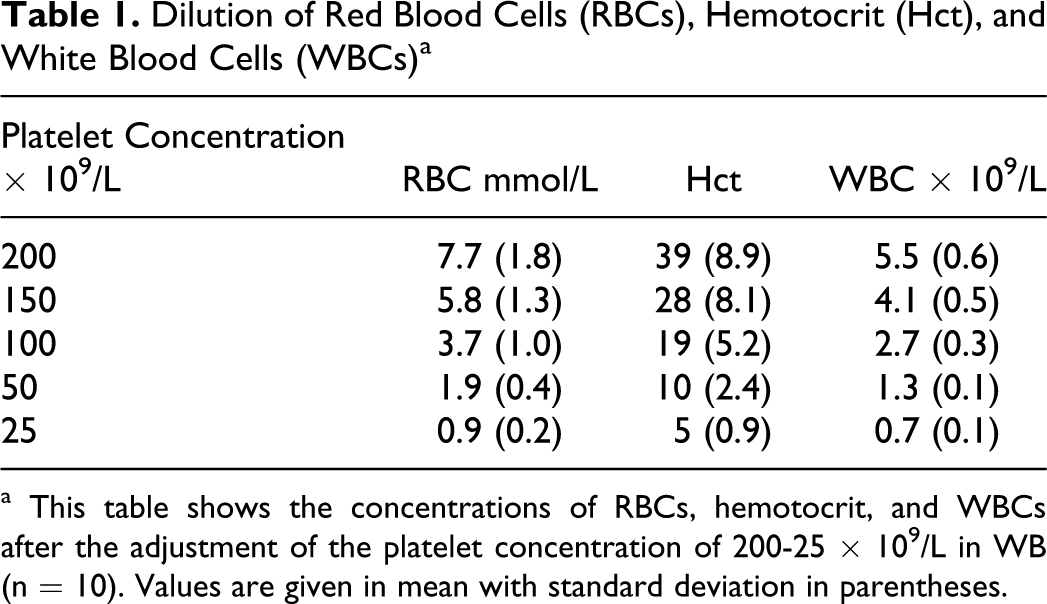
a This table shows the concentrations of RBCs, hemotocrit, and WBCs after the adjustment of the platelet concentration of 200-25 × 109/L in WB (n = 10). Values are given in mean with standard deviation in parentheses.
Whole Blood Aggregometry
In the heparinized and citrated WB samples, significant effects of the platelet concentrations on platelet aggregation were observed against all agonists (all P < .0001). In the heparinized WB samples, this was due to significantly reduced platelet aggregation in the ADP-, ASPI-, and COL-test (all P < .03) or borderline-reduced TRAP-test (P = .07) for platelet concentration of ≤100 × 109/L when compared to the 200 × 109/L control. In the TRAP-test, the aggregation only decreased significantly at platelet concentration of ≤50 × 109/L compared to the control (P = .008). No differences were found between the 200 and 150 × 109/L concentrations in the heparinized WB samples (Figure 2 ).
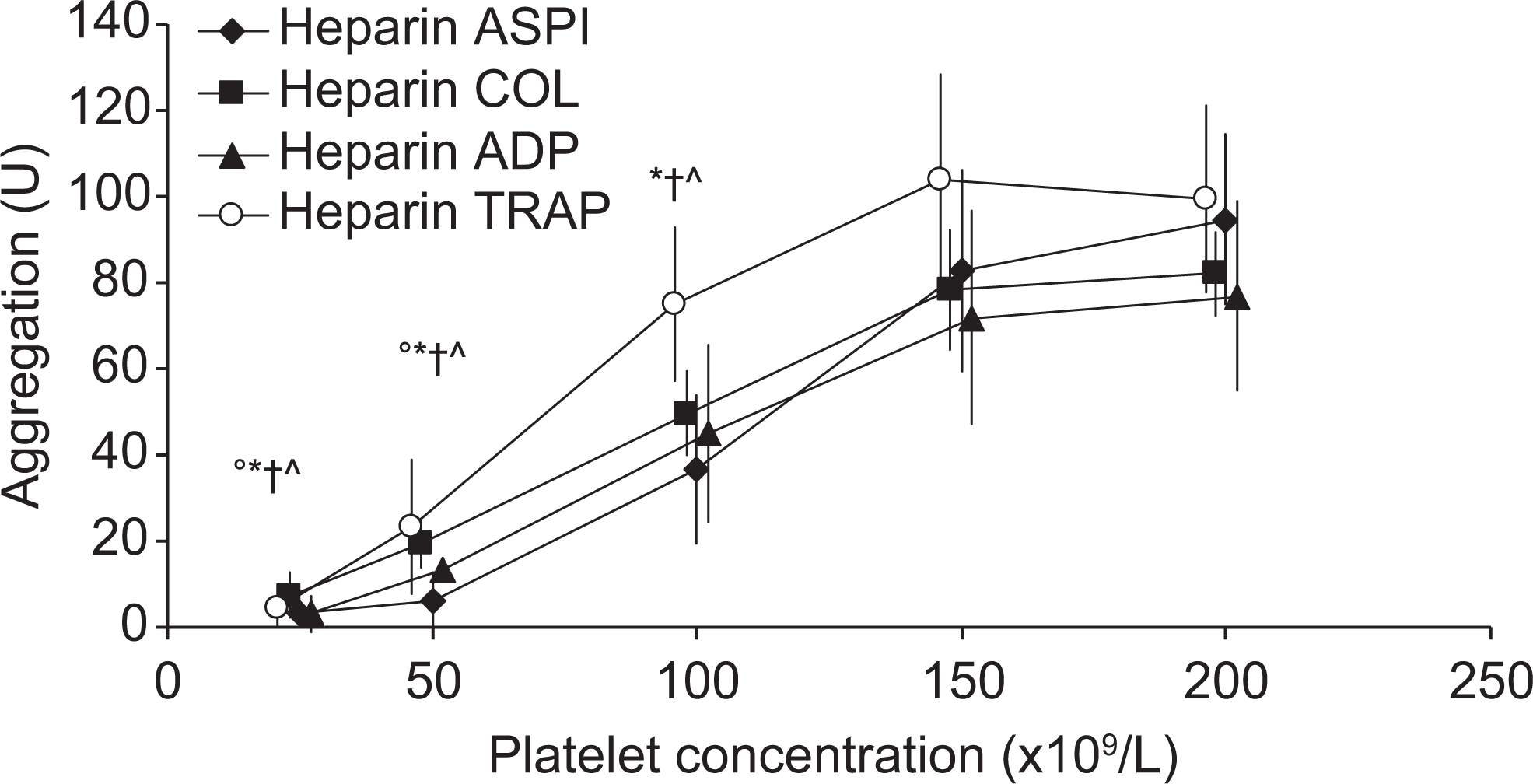
Platelet aggregation in heparinized whole blood (WB). Agonist-induced (arachidonic acid [ASPI], collagen [COL], adenosine diphosphate [ADP], and thrombin receptor-activating peptide [TRAP]) aggregation in heparinized WB (n = 10) with platelet concentrations of 200, 150, 100, 50, and 25 × 109/L. Values are given in mean ± standard deviation. °* ˆ †Significant difference from the highest concentration by Bonferroni adjusted (×4) by post hoc Wilcoxon signed rank test (P < .05).°TRAP, *ASPI, ˆCOL, and †ADP. U indicates area under curve.
In the citrated WB samples, a significantly reduced platelet aggregation was observed at platelet concentration ≤100 × 109/L for the ADP-, COL-, and TRAP-test when compared to the control (all P<.03). In the ASPI-test, the platelet aggregation was significantly decreased already at platelet concentration of 150 × 109/L compared to the control (P=.03; Figure 3 ). Thus, except for the ASPI-test, no differences were observed between the 200 and 150 × 109/L concentrations in the citrated WB samples.
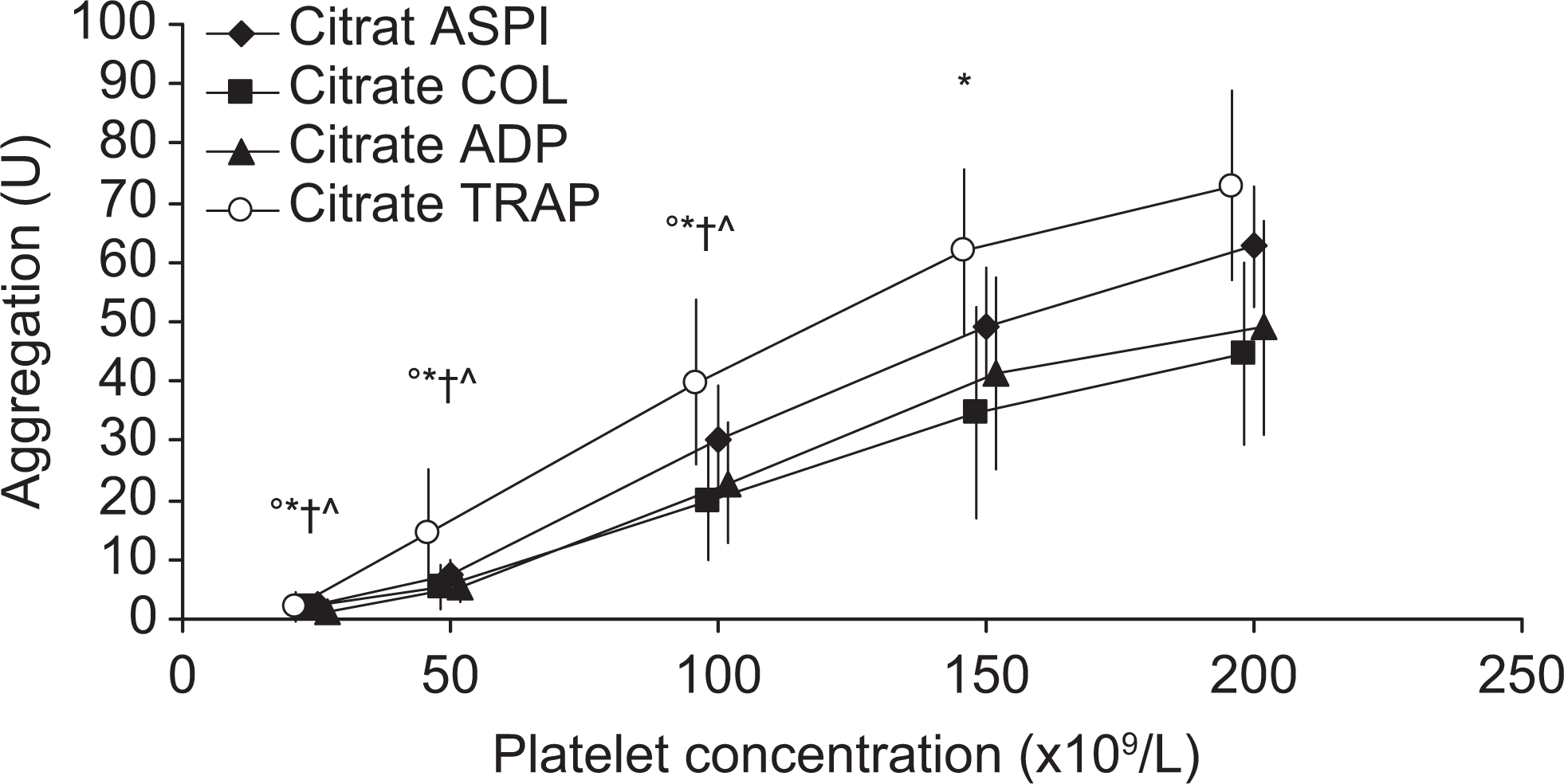
Platelet aggregation in citrated WB. Agonist-induced (arachidonic acid [ASPI], collagen [COL], adenosine diphosphate [ADP], and thrombin receptor-activating peptide [TRAP]) aggregation in citrated whole blood (n = 10) with platelet concentrations of 200, 150, 100, 50, and 25 × 109/L. Values are given in mean ± standard deviation. °*ˆ †Significant difference from the highest concentration by Bonferroni adjusted (×4) by post hoc Wilcoxon signed rank test (P < .05). °TRAP, *ASPI, ˆCOL, and †ADP. U indicates area under curve.
Platelet-Rich Plasma vs WB Aggregometry
When comparing the aggregation in heparinized and citrated WB and PRP (platelet concentration 100 to 200 × 109/L), the aggregation responses against all investigated agonists were higher in WB compared to PRP in the heparinized samples (all P < .04). In the citrated samples, the aggregation in the ASPI- and TRAP-test were increased significantly in WB vs PRP samples (all P<0.05), whereas no differences were found in the ADP- and COL-tests (P < .4; Figure 4A-D ).
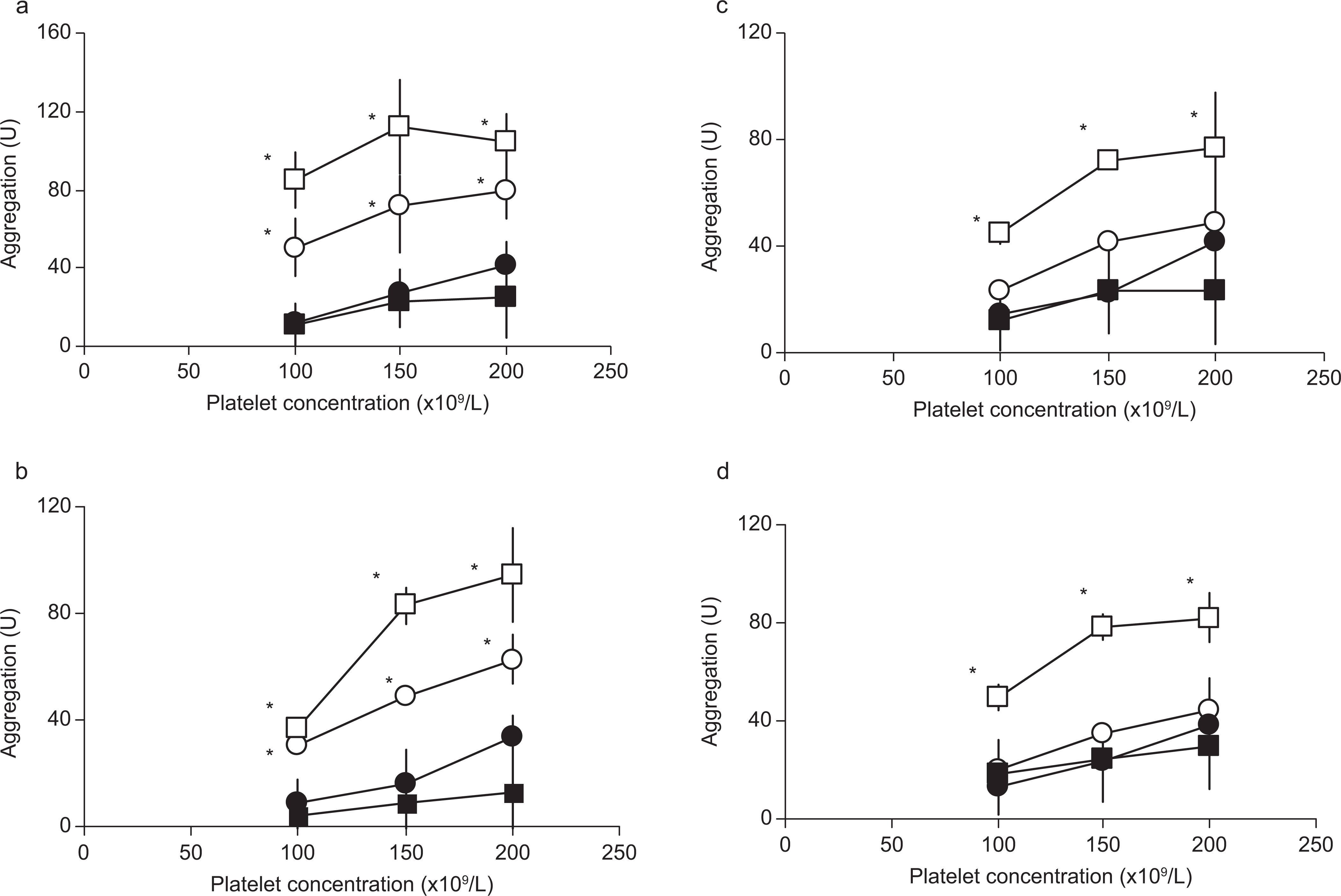
Platelet aggregation in whole blood (WB) and platelet-rich plasma (PRP). Thrombin receptor-activating peptide (TRAP; A), arachidonic acid (ASPI; B), adenosine diphosphate (ADP; C), and collage (COL; D)-induced mean aggregation in WB and PRP with plasma concentrations of 200, 150, and 100 × 109/L, (n = 7). Values are given in mean ± standard deviation. *Significant differences were analyzed by Wilcoxon signed rank test (P < .05) U indicates area under curve, □, WB-heparin; ˆ, WB-citrate; ▪, PRP-heparin; and •, PRP-citrate.
Discussion
The results of the present study demonstrate that the platelet aggregation response in WB as evaluated by Multiplate may be influenced by the platelet concentration, with reduced aggregation response observed at platelet concentrations <150 × 109/L.
To our knowledge, this is the first study describing the association between low-platelet count and platelet aggregation responses assessed by Multiplate in heparinized and citrated WB. The relationship between low platelet count and platelet aggregation has previously been studied in the first model of an impedance-based aggregometer, the Chronolog, 14 but differences regarding the concentration of the agonists, cleaning techniques, etc., makes it difficult to compare the results with the Multiplate. Recently, Mengistu et al analyzed the impact of low platelet count on platelet aggregation in hirudin-anticoagulated WB by Multiplate. This study found significantly decreased platelet aggregation responses at platelet concentrations <150 × 109/L compared to the control (native blood sample, 150-400 × 109/L) using ADP, COL, and TRAP as agonists. 10 It is notable that the platelet aggregation responses in the present study were reduced at the same platelet concentration (<150 × 109/L), although methodological differences between the 2 studies makes it difficult to compare them. The previous studies have reported variations in platelet aggregation responses with different anticoagulants illustrating the importance of standardized methods when comparing results.12,15 Another important difference is the adjustment of the platelet concentrations. Mengistu et al used plasma-adapted crystalloid solution for the WB dilution, whereas the present study used autologous plasma as the diluting fluid in order to mimic the physiological environment in the patient with thrombocytopenic PCI. Due to the absence of plasma coagulation factors and residual cells in plasma-adapted crystalloid solutions, the use of this fluid instead of plasma will dilute the concentration of plasma coagulation factors considerably and this may influence the aggregation response. Furthermore, the concentration of RBCs and WBCs may probably be diluted, which may all influence platelet aggregation. 10
Considering the association between platelet aggregation and platelet concentration in healthy participants with a normal platelet count (150-400 × 109/L), the relationship has been studied previously with conflicting results. Seyfert et al 16 did not find any correlation between platelet count and aggregation response in citrated WB in 120 healthy participants in the ASPI-test, but found a weak correlation between platelet count and aggregation in the ADP- and COL-test. This correlation could not be confirmed in the present study nor in a study by Tóth et al with 22 healthy participants using ADP and COL in both citrated- and hirudin-anticoagulated WB. 12 The discrepant results may be explained by the different sizes of the study populations, since the investigation of very few participants in the present study and the study by Tóth et al may introduce a type II error. However, since the study by Seyfert et al only presented data as a ρ correlation, it cannot be assumed that the relationship between platelet count and aggregation was simply linear.
Comparing PRP with WB, lower platelet aggregation responses were clearly demonstrated for platelet concentrations of 100 to 200 × 109/L in all investigated PRP heparinized and citrated samples, against all agonists, with the only exception being the ADP- and COL-test in citrated samples. The lower platelet aggregation responses in PRP are probably multifactorial. The centrifugation step in PRP may selectively influence the composition of the remaining platelet population and activate the remaining platelets, thereby impairing the aggregation activity as compared to WB. 12 Furthermore, the hemostatic role of RBCs in WB may influence the platelet aggregation. In vivo, RBCs contribute significantly to platelet activation and furthermore support thrombin generation.17,18 In vitro, the relationship between hematocrit and platelet aggregation is less clearly described.14,16
Although the present study used a modified WB matrix compared to natural WB, the different components in natural WB were still present in the WB matrix. Since hemostasis in human is attributed to the function of platelets in WB and not in PRP, 19 the results of the present studies indicate that platelet function measurements performed in an artificial PRP matrix may not be a “true” reflection of the platelet function capacity and hence hemostasis in vivo.
The present study has several limitations, such as a small sample size and furthermore, it has been demonstrated that adjusting the platelet count with PPP in a PRP matrix may affect platelet aggregation independently of the induced change in platelet count. A theory is that substances may be released by blood cells during high-speed centrifugation and inhibit platelet aggregation. One such substance could be ADP released from platelets or RBCs which could desensitize the ADP receptor on platelets and thus impair platelet responses to exogenous ADP. 20 Though this phenomenon could explain the decreased platelet aggregation independently of the platelet concentration in both the PRP and the WB study, Mengistu et al have found a decreasing aggregation with decreasing platelet count using a noncentrifugation crystalloid solution. 10 If the possible inhibitory effect of PPP only influences a PRP matrix, this phenomenon may be another explanation of the more pronounced inhibition of the platelet seen in the PRP study as compared to the WB study. Another limitation in the study is the concentrations of RBCs and WBCs which were not standardized along with platelet dilution. A criticism could be that the reduced platelet aggregation did not reflect the lower platelet concentrations but the diluted amount of RBCs or WBCs instead. Concerning the effect of RBCs on platelet aggregation, these cells may be able to stimulate aggregation through the release of ADP and ATP, the latter being converted to ADP by ecto-ATPase in blood cells. Furthermore, RBCs may remove adenosine, the end product of ATP breakdown, which may inhibit platelet aggregation. On the other hand, very high concentration of ATP may have an inhibitory effect on platelet aggregation, possibly because of antagonism by ATP of ADP interaction with the ADP receptor. 21 In the present study, there was a change in mean RBC concentration from 7.7 to 3.7 mmol/L and in the mean hematocrit from 39% to 19% after dilution of the platelet concentration from 200 to 100 × 109/L. The problem with analyzing the influence of RBCs on platelet aggregation is the manipulation of WB to an artificial matrix with standardized hematocrit and platelet concentrations, which could influence hemostasis as mentioned earlier. Our group has analyzed the influence of hematocrit on platelet aggregation in stored buffy coat platelets (BCP) with a standardized platelet concentration. Addition of RBCs (to a final hematocrit of 0.15 or 0.29) to the BCP did not increase aggregation against COL and TRAP. However, increasing the hematocrit to 0.29 increased the platelet aggregation against ADP and ASPI significantly. 22 In another study by Bochsen et al, 23 the results obtained by Ostrowski et al 22 were confirmed, by again demonstrating that the hematocrit did not have an impact on platelet aggregation using TRAP and COL as agonists. A recently published study by Hanke et al 24 analyzed the influence of platelet count (50-250×10 9 /L) on aggregation in Multiplate while maintaining hematocrit and hence using an artificial matrix instead of WB. Comparing the results with platelet count within normal range, the aggregometry results decreased significantly with platelet concentrations of 100× 109/L and 50 × 109/L, respectively. Interestingly, though it is difficult to compare the study by Hanke et al with the 2 WB studies, platelet concentration of 100 × 109/L seems to be critical, concerning the influence of platelet counts on platelet aggregation. Considering the study by Hanke et al, 24 Ostrowski et al, 22 and Bochsen et al, 23 the present aggregation results cannot exclusively be explained by the hematocrit. Regarding the WBCs, the dilution of the platelet concentration from 200 × 109/L to 100 × 109/L reduced the mean WBCs from 5.5 to 2.7 × 109/L, and thereby below the normal WBC level (3.5-8.8 × 109/L). Stafford et al have shown that WBCs may influence platelet aggregation through a conversion of ATP to ADP, released from other blood cells. 21 On the other hand, Oleksowisc et al demonstrated that cytokine release, IL-2, from lymphocytes may enhance platelet secretion but may also inhibit platelet aggregation. 25 Regarding the neuthrophils and platelet aggregations interaction in vitro, there may be both a platelet activation and a platelet inhibitory effect of the neuthrophils depending on the experimental conditions.26,27 Hence, in vitro WBCs may both exert stimulatory or inhibitory effect on platelet aggregation upon different physiological stimuli. Considering these opposite influences of WBCs on platelet aggregation, it is difficult to estimate the possible influence of the WBCs on the platelet aggregation in the present study.
When applying the Multiplate to estimate and regulate the effect of standardized antiplatelet therapy in patients, it is important to critically evaluate the results in relation to the limitations of the point-of-care test. In the present study, the platelet aggregation measured by Multiplate in heparinized and citrated WB may be affected by platelet concentrations of <150 × 109/L. This should be taken into consideration when interpreting the Multiplate responses in clinical settings.
Footnotes
Acknowledgment
The technical assistance from laboratory technicians José A. Salado-Jimena, Karen Dyeremose, Charlotte Månsson, and Rola Ghadban is greatly acknowledged.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
