Abstract
We aimed to evaluate the relation among epicardial adipose tissue (EAT) thickness, angiographic presence of thrombus, and the no-reflow in the patients with non-ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. The study population consisted of 229 patients. The EAT thickness and neutrophil to lymphocyte ratio (NLR) were significantly higher in the patients with coronary thrombus than in those without coronary thrombus (6.1 ± 1.1 vs 5.1 ± 1.3 mm, P < .001 and 3.4 ± 0.9 vs 2.5 ± 0.7, P < .001, respectively) and in the patients with no-reflow compared to patients with reflow. The EAT thickness was found to be correlated positively with the degree of the thrombus burden, NLR, and waist circumference and negatively with high-density lipoprotein cholesterol. Multivariate logistic regression analysis demonstrated that EAT thickness and NLR independently predicted coronary thrombus formation and no-reflow. We have suggested that EAT can play an important role in the pathophysiology of coronary thrombus formation and the no-reflow.
Keywords
Introduction
The no-reflow phenomenon is observed frequently after primary percutaneous coronary intervention (PPCI) for acute coronary syndrome (ACS), and is strongly associated with adverse left ventricular remodeling and worse prognosis. 1,2 Various mechanisms are responsible for the development of the no-reflow phenomenon such as platelet aggregation, distal embolization of thrombus and/or plaque components from the lesion site, capillary vasospasm, and neutrophilic plugging. 3,4 The previous studies have shown that a high thrombus burden could predict the development of no-reflow in the patients who had an acute coronary event. 5 The no-reflow phenomenon and the coronary thrombus formation are well-known risk factors for short- and long-term adverse cardiovascular events in the patients with ACS. 6 Thus, the prediction and the early recognition of no-reflow phenomenon and the coronary thrombus formation are crucial in terms of both predicting adverse cardiovascular events and choosing the most appropriate treatment modalities to prevent procedure failure.
Epicardial adipose tissue (EAT) is the true visceral fat depot of the heart, located along the large coronary arteries and on the surface of the ventricles and apex. It has become evident that EAT is a metabolically active organ that produces proinflammatory and proatherogenic cytokines. 7 The previous studies demonstrated that the increased EAT thickness was associated with the presence and the severity of coronary artery disease (CAD), subclinic atherosclerosis, coronary slow flow phenomenon, hypertension (HT), and metabolic syndrome. 8,9 Because of the proximity of EAT to coronary arteries and the absence of fascial boundaries, its secretions may have paracrine effects on these vessels. 7
Inflammation has been demonstrated to be an important risk factor for the development of cardiovascular events. In the previous studies, it was reported that the blood serum markers of inflammation such as peripheral white blood cells (WBC) and high-sensitivity C-reactive protein (hs-CRP) were independently related to both no-reflow phenomenon and the increased thrombus burden in the patients with ACS. 10,11 Based on this, we hypothesized that a mechanistic relation could be found between EAT and no-reflow phenomenon and coronary thrombus formation because of the proinflammatory features of EAT. Therefore, the goal of the present study was to investigate whether the EAT thickness was associated with the no-reflow phenomenon and the presence of intracoronary thrombus in the patients with non-ST-segment elevation myocardial infarction (NSTEMI) undergoing PPCI.
Methods
Study Population
This study included 229 consecutive patients with NSTEMI who underwent PPCI within 24 hours after admission. The diagnostic criteria for NSTEMI were based on current guidelines. The exclusion criteria were the baseline anemia (<13 g/dL for males, 12 g/dL for females), the history of blood transfusion in the last 3 months, active and ongoing infection, chronic inflammatory disease, malignancy, renal insufficiency, liver cirrhosis, febrile disorders, and the history of recent surgery or trauma within the preceding 2 months. The study protocol was approved by our local ethics committee, and all patients gave their written informed consent to participate in the study.
After the assessment of detailed medical history and a complete physical examination, age, sex, the family history of CAD, current smoking status, the history of HT, dyslipidemia, diabetes mellitus (DM), the previous history of CAD, body mass index (BMI; weight (kg) /height squared (m2)), and waist circumference were recorded for all patients. Lipid profile, hematological indices, creatine kinase (CK), CK-myocardial band isoform (CK-MB), and troponin I levels were measured for all patients during the hospital admission. Hemoglobin, WBC, neutrophil to lymphocyte ratio (NLR), red cell distribution width, and the platelet counts were measured as part of the automated complete blood count using a Coulter LH 780 Hematology Analyzer (Beckman Coulter Inc, Miami, Florida). The NLR was obtained by dividing total count of neutrophils by lymphocytes count.
Echocardiographic Analysis
Echocardiography was performed using a GE Vivid S5 system (GE Vingmed Ultrasound AS, Norten, Norway) with a 2.5-MHz phased-array transducer. All patients were imaged in the left lateral decubitus position and underwent a complete transthoracic echocardiographic examination. All examinations were made by an experienced cardiologist blinded to the clinical data of the patients. The left ventricular ejection fraction (LVEF) was calculated by the modified Simpson method. 12 The EAT thickness was measured according to the previously published methods. 7 The EAT was identified as an echo-free space in the pericardial layers on the 2-dimensional echocardiography, and its thickness was measured perpendicularly on the free wall of the right ventricle from both parasternal long-axis and short-axis views at end systole in 3 cardiac cycles. The average of the maximum values measured at each site was considered as EAT thickness. The intraobserver correlation coefficient was .96.
Angiographic Analysis
Before angiography, all patients received chewable aspirin (300 mg), clopidogrel (600 mg), and an intravenous bolus of heparin (100 U/kg). The PPCI procedures were performed using the standard femoral approach with a 7F guiding catheter, and angiographic thrombus burden was scored in 5 grades: 0 (no thrombus), 1 (possible thrombus), 2 (definite thrombus <0.5 reference vessel diameter), 3 (definite thrombus 0.5-2 reference vessel diameters), 4 (definite thrombus >2 reference vessel diameters), and 5 (complete vessel occlusion) as described previously. 13 Postprocedural thrombolysis in myocardial infarction (TIMI) flow grade, TIMI myocardial perfusion grade (TMPG), and the corrected TIMI frame count were assessed as previously described. 14 –16 The TIMI 0, I, and II or a TIMI flow grade 3 with a TMPG 0 or 1, at least 10 minutes after the end of the PPCI procedure were described as angiographic no-reflow. 17
Statistical Analysis
Continuous variables as mean + standard deviation or median (minimum–maximum) values and categorical variables as percentage were shown. The One-Sample Kolmogorov-Smirnov test was used to evaluate whether the distribution of continuous variables was normal. Continuous variables between 2 groups were compared with Student t test or Mann-Whitney U test. Categorical variables were compared appropriately with χ2 or Fisher exact test. The correlations between variables were tested by the Pearson correlation test for normally distributed variables and with Spearman correlation tests for the nonnormally distributed variables. In order to determine the independent predictors of angiographic thrombus and no-reflow phenomenon, parameters which were found to have significance (P < .05) in the univariate analysis were evaluated by stepwise forward logistic regression analysis. The 95% confidence interval and odds ratios (OR) were presented together. In order to predict cutoff value of EAT thickness and NLR, receiver–operating characteristics (ROC) curve analysis was performed. The area under curve (AUC) comparison of these scoring systems was performed using method of Delong et al method. 18 P < .05 was considered significant in all statistical analyses. All of the statistical analyses were performed by using SPSS 17 (SPSS Inc, Chicago, Illinois).
Results
A total of 229 consecutive patients (mean age 54.3 ± 10.1 years; 77.7% male) with NSTEMI who underwent PPCI were enrolled in the study. In the coronary angiography of 142 patients (62.1%), coronary thrombus was detected at various grades. Angiographic no-reflow during PPCI occurred in 51 (22.3%) patients and did not occur in 178 (77.7%) patients of the whole population. The comparison of clinical, laboratory, and angiographic findings between the patients with and without angiographic thrombus is shown in Table 1. Compared to the patients without coronary thrombus, the patients with coronary thrombus had significantly increased EAT thickness (6.1 ± 1.1 mm vs 5.1 ± 1.3 mm, P < .001), more family history of CAD, higher baseline CK-MB, baseline troponin, and NLR (2.5 ± 0.7 vs 3.4 ± 0.9, P < .001) values. Similarly, metabolic parameters such as BMI and waist circumference were found to be significantly higher in the patients with coronary thrombus. When the angiographic findings were compared, angiographic no-reflow during PPCI was observed more frequently in the patients with coronary thrombus than in the patients without coronary thrombus. The comparison of clinical, laboratory, and angiographic findings of the groups based on no-reflow and reflow is shown in Table 2.
Comparison of Clinical, Laboratory, and Angiographic Characteristics Between Patients With and Without Angiographic Thrombus.
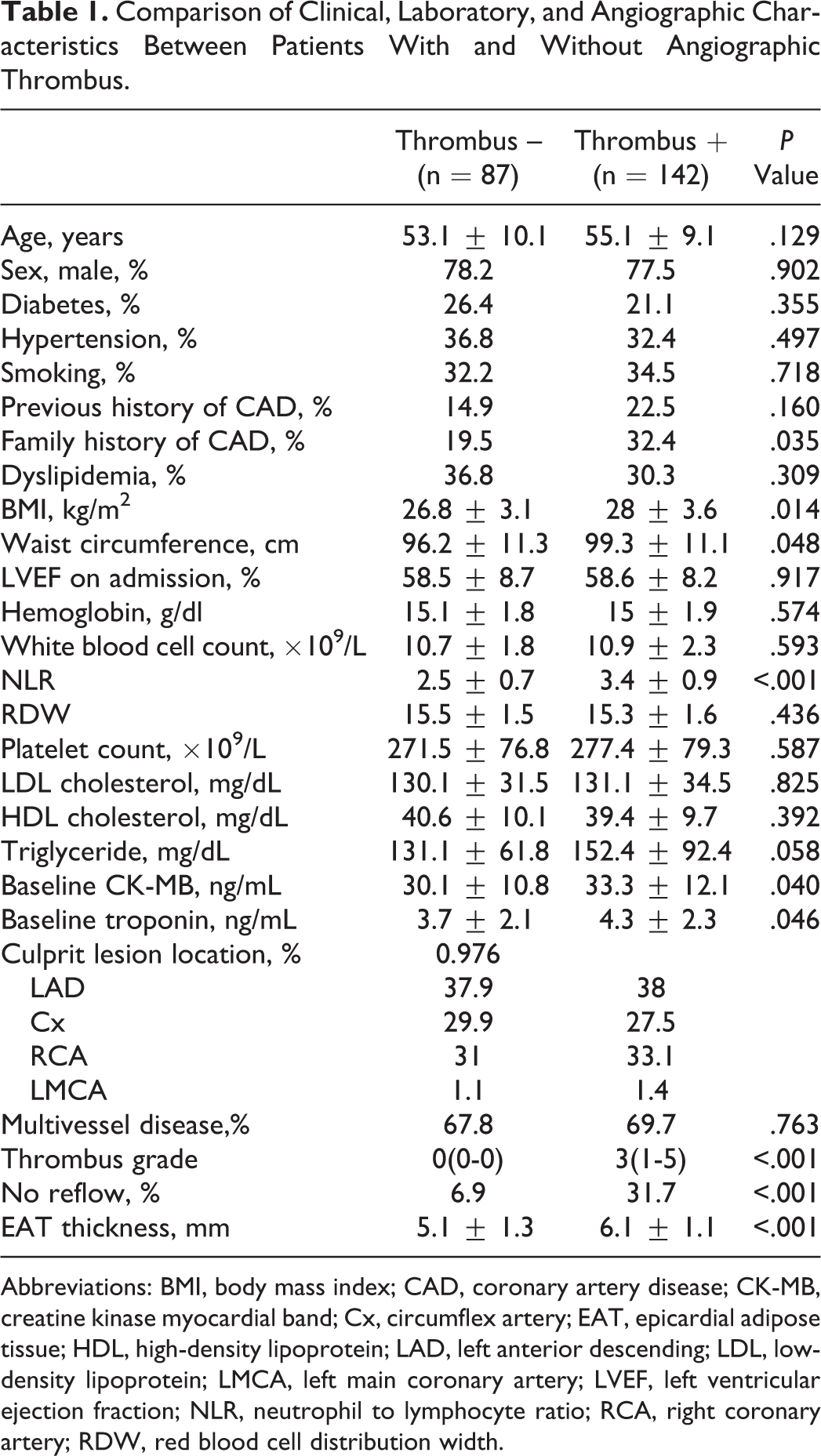
Abbreviations: BMI, body mass index; CAD, coronary artery disease; CK-MB, creatine kinase myocardial band; Cx, circumflex artery; EAT, epicardial adipose tissue; HDL, high-density lipoprotein; LAD, left anterior descending; LDL, low-density lipoprotein; LMCA, left main coronary artery; LVEF, left ventricular ejection fraction; NLR, neutrophil to lymphocyte ratio; RCA, right coronary artery; RDW, red blood cell distribution width.
Comparison of Clinical, Laboratory, and Angiographic Characteristics of Patients in No-Reflow and Reflow Groups.
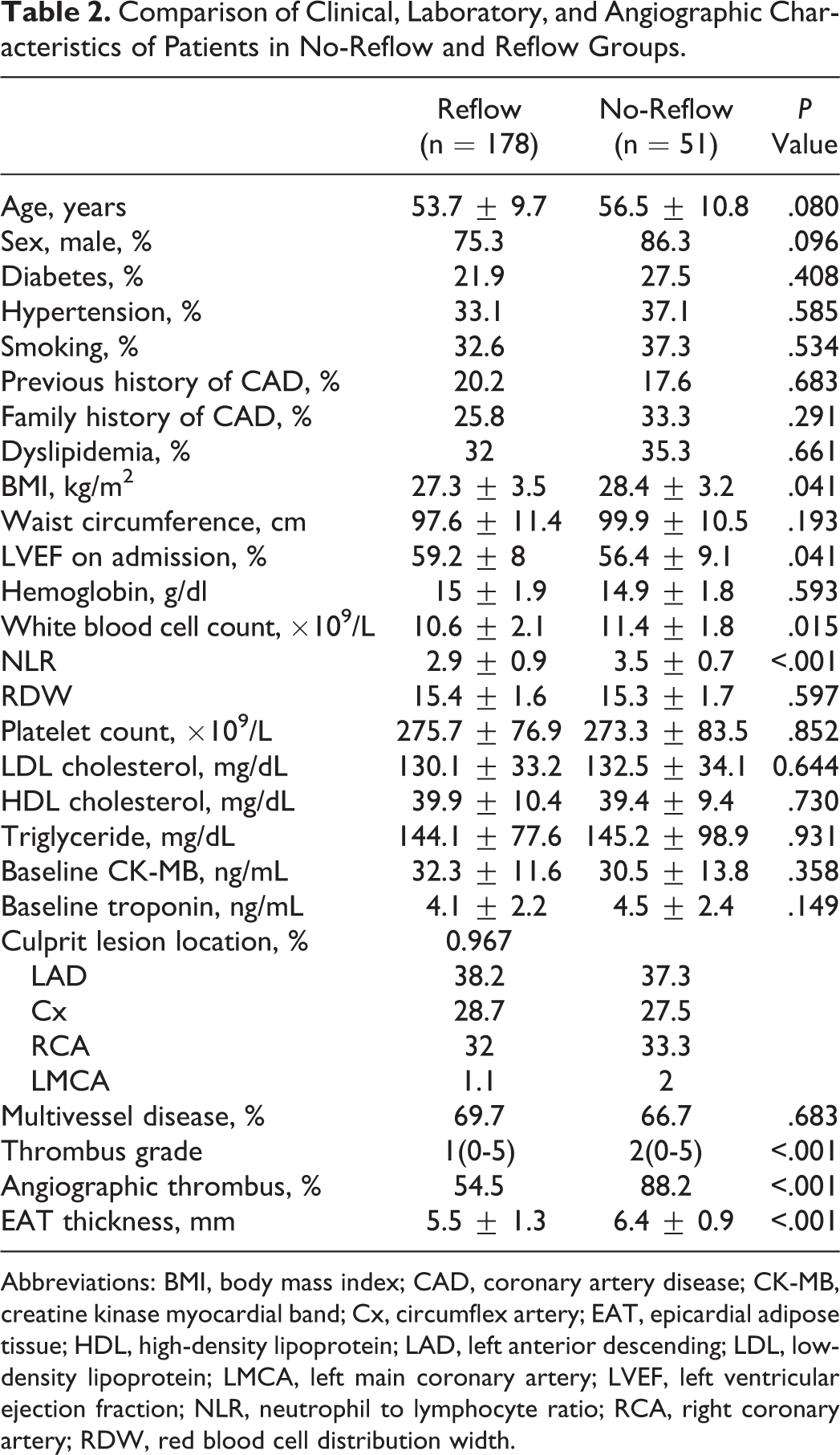
Abbreviations: BMI, body mass index; CAD, coronary artery disease; CK-MB, creatine kinase myocardial band; Cx, circumflex artery; EAT, epicardial adipose tissue; HDL, high-density lipoprotein; LAD, left anterior descending; LDL, low-density lipoprotein; LMCA, left main coronary artery; LVEF, left ventricular ejection fraction; NLR, neutrophil to lymphocyte ratio; RCA, right coronary artery; RDW, red blood cell distribution width.
Epicardial adipose tissue thickness was found to be correlated positively with the degree of the thrombus burden (r = .240, P = .003), NLR (r = .269, P < .001), and waist circumference (r = .156, P = .019) and found to be correlated negatively with high-density lipoprotein cholesterol (r = –.145, P = .028).
Multivariate logistic regression analysis was performed in order to determine the independent predictors of both coronary thrombus and the no-reflow phenomenon. The multivariate logistic regression model including family history of CAD, baseline CK-MB and troponin I, BMI, waist circumference, NLR, and EAT thickness demonstrated that EAT thickness and NLR were independent predictors of coronary thrombus (Table 3). Additionally, the EAT thickness, NLR, and the presence of angiographic thrombus were found to be independent predictors of no-reflow phenomenon in the analysis performed for the variables found significant in univariate analysis (LVEF, angiographic thrombus, WBC, NLR, and EAT thickness; Table 4). The cutoff values of EAT thickness and NLR for coronary thrombus were 5.3 with a sensitivity of 83.1% and a specificity of 78.2% (AUC, 0.762; 95% CI, 0.701-0.815; P < .001) and 3.25 with a sensitivity of 76.1% and a specificity of 89.7% (AUC, 0.794; 95% CI, 0.735-0.844; P < .001), respectively, in the ROC curve analysis (Figure 1). The cutoff values of EAT thickness and NLR for the no-reflow phenomenon were 5.7 with a sensitivity of 86.3% and a specificity of 61.2% (AUC, 0.709; 95% CI, 0.645-0.767; P < .001) and 3.26 with a sensitivity of 82.4% and a specificity of 62.4% (AUC, 0.725; 95% CI, 0.663-0.782; P = .001), respectively, in the ROC curve analysis (Figure 2). However, AUC comparison of these parameters for coronary thrombus and the no-reflow phenomenon did not reach statistical significance (P > .05).
Independent Predictors of Thrombus Formation in Multivariate Logistic Regression Analysis.

Abbreviations: BMI, body mass index; CAD, coronary artery disease; CK-MB, creatine kinase myocardial band; EAT, epicardial adipose tissue; NLR, neutrophil to lymphocyte ratio; OR, odds ratio; CI, confidence interval.
Independent Predictors of No-Reflow Phenomenon in Multivariate Logistic Regression Analysis.
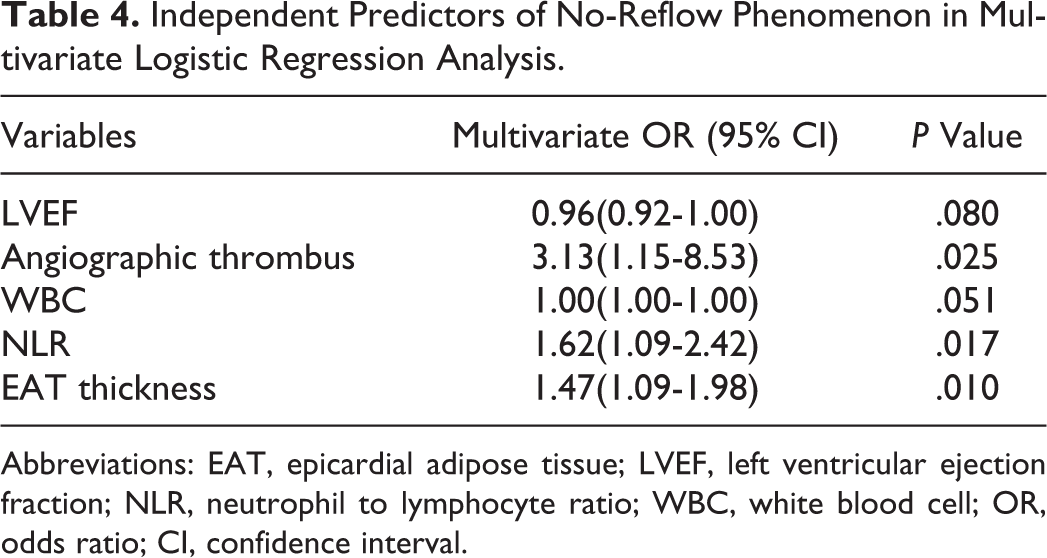
Abbreviations: EAT, epicardial adipose tissue; LVEF, left ventricular ejection fraction; NLR, neutrophil to lymphocyte ratio; WBC, white blood cell; OR, odds ratio; CI, confidence interval.
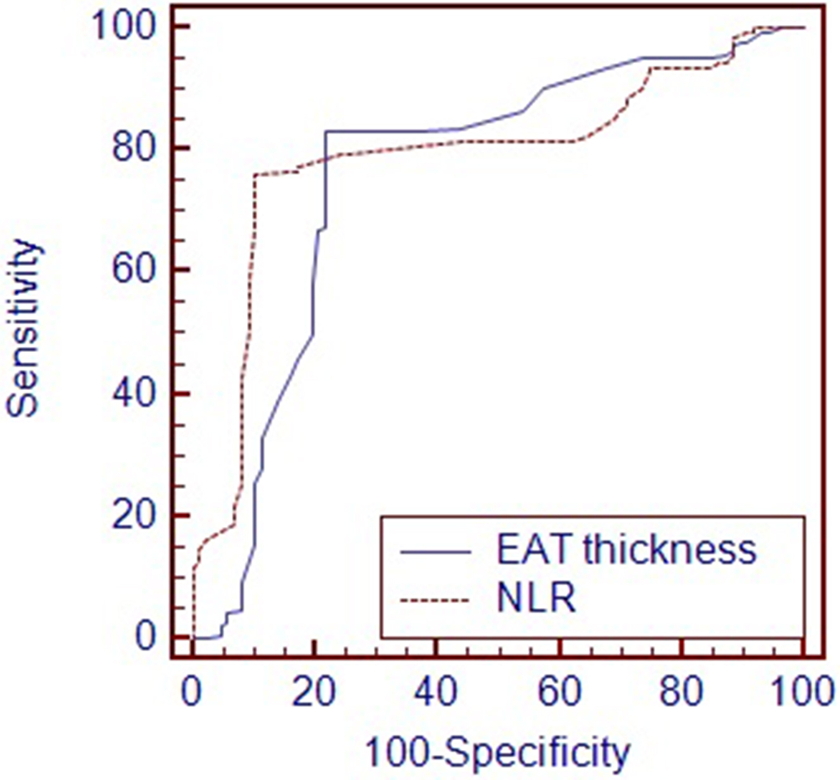
Comparison receiver–operating characteristics (ROC) curves for epicardial adipose tissue (EAT) thickness and neutrophil to lymphocyte ratio (NLR) in prediction of coronary thrombus formation.
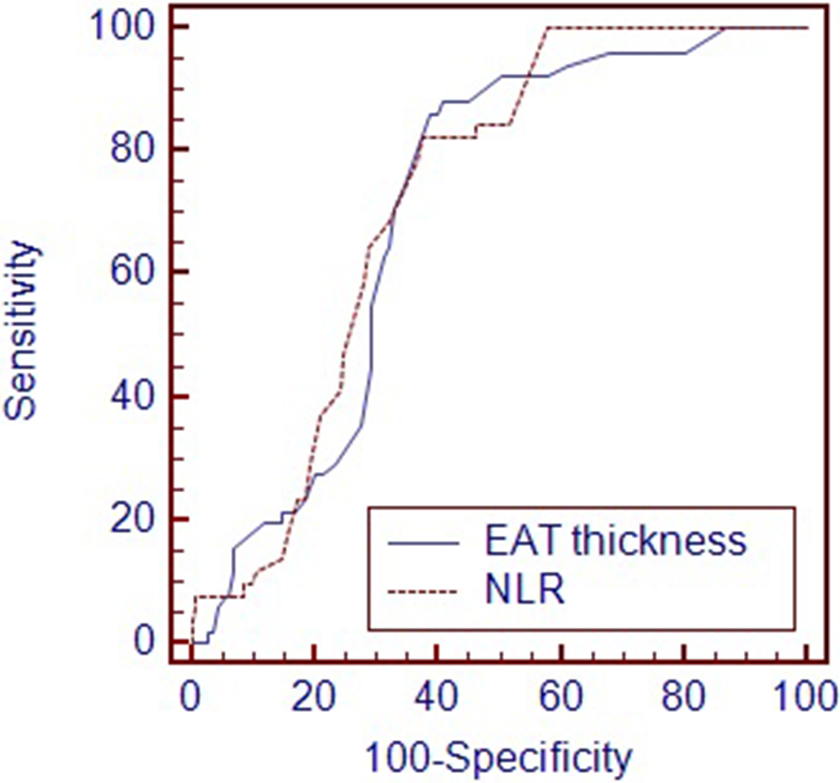
Comparison receiver–operating characteristics (ROC) curves for epicardial adipose tissue (EAT) thickness and neutrophil to lymphocyte ratio (NLR) count in prediction of the no-reflow phenomenon.
Discussion
Our study results indicate that increased EAT thickness and NLR were independent predictors of coronary thrombus formation and the no-reflow phenomenon in the patients with NSTEMI who underwent PPCI. Besides, we found that coronary thrombus formation was independently associated with the no-reflow phenomenon. To the best of our knowledge, this is the first study to evaluate the relationship between EAT thickness and coronary thrombus formation and no-reflow phenomenon in the patients with NSTEMI treated with PPCI.
Epicardial, mesenteric, and omental fat all share the same origin from the splanchnopleuric mesoderm. The EAT exists mainly in the atrioventricular and interventricular groove along the major coronary arteries and branches and can be quantified readily by transthoracic echocardiography. It has been shown that body fat distribution, particularly abdominal fat tissue, is strongly correlated with epicardial fat. 7 The EAT could locally modulate the heart and vasculature through paracrine secretion of pro- and anti-inflammatory cytokines. It is known that EAT is related to the presence and the severity of CAD, coronary plaque morphology, myocardial ischemia, and major adverse cardiac events (MACE). 19,20 It was shown in the previous studies that EAT was in relation to impaired coronary and myocardial flow reserve and coronary slow flow phenomenon. 21 In the literature, there is only 1 study focusing on the relationship between EAT and no-reflow phenomenon. 22 In this study carried out with 114 patients with STEMI, ST-segment resolution was used as no-reflow marker; and it was detected that there was an independent relation between the increased EAT thickness and the electrocardiographic no-reflow. In our study, we used TIMI flow grade and TMPG as no-reflow marker and demonstrated that there was an independent relation between increased EAT thickness, the angiographic no-reflow, and the coronary thrombus in the patients with NSTEMI.
Intracoronary thrombosis developed after plaque disruption (plaque rupture or superficial plaque erosion) is the basic pathophysiologic event in the patients with ACS. 23 Inflammatory cells play a critical role both in plaque disruption through the elaboration of matrix degrading metalloproteinases and in triggering thrombosis following plaque disruption through the tissue factor pathway. 24 Several studies have reported that a systemic inflammatory response occurs in the patients with ACS and that inflammation may directly contribute to coronary thrombosis and impaired perfusion. 25,26 The NLR reflects the balance between neutrophil and lymphocyte levels in the body and is an indicator of systemic inflammation. 27 It has been shown that NLR was an independent predictor of adverse cardiac events in the patients with ACS. 28 Yılmaz et al 29 have showed that NLR, neutrophil count, and lymphocyte count are the independent predictors of coronary thrombus formation in the patients with ACS. Similarly, Li et al 30 have showed that NLR was independently predictive of thrombus formation in the patients with STEMI. Barron et al 10 have demonstrated that the elevation in the WBC during STEMI is associated with a greater thrombus burden, reduced epicardial blood flow, and myocardial reperfusion. In an another study, Youssef et al 31 have reported that the circulating level of soluble CD40 ligand, an important proinflammatory mediator, is the most independent predictor of angiographic morphologic features that indicate high-grade burden thrombus formation. In line with the results of these studies, we detected in our study that the NLR as an indicator of systemic inflammation was independent predictor of coronary thrombus formation in the patients with NSTEMI. Furthermore, we revealed that the patients with coronary thrombus had increased EAT thickness compared to those without coronary thrombus and that increased EAT thickness was an independent predictive factor for coronary thrombus formation. The EAT has paracrine and endocrine functions and produces several proinflammatory and proatherogenic cytokines as well as tumor necrosis factor α (TNF-α), monocyte chemoattractant protein 1, interleukin 1β (IL-1β), interleukin 6 (IL-6), nerve growth factor, leptin, resistin, plasminogen activator inhibitor 1, and angiotensinogen. However, epicardial fat also secretes anti-inflammatory, antiatherogenic adipokines, such as adiponectin and adrenomedullin. It is well known that tissue concentrations of proinflammatory adipocytokines (IL-6, IL-1β, TNF-α) in EAT are significantly higher in the patients with CAD than in those without CAD. 7 Interleukin 6 and TNF-α are known to stimulate the expression of tissue factor secretion from monocytes to increase platelet reactivity and to induce plasminogen activator inhibitor 1 secretion from the liver. Moreover, it was reported that IL-6 and TNF-α increase the risk of thrombosis development by stimulating ultralarge von Willebrand multimer secretion. 32 Interleukin 1β, another proinflammatory cytokine secreted from EAT, decreases the endothelial cell thrombomodulin expression and promotes thrombus formation. 33 The effects of these adipocytokines secreted from EAT on vascular hemostasis may explain the relationship between EAT and the coronary thrombus formation.
The role of inflammation and the coronary thrombus formation in the pathogenesis of the no-reflow phenomenon is well established. 25,34 –36 In the previous studies, it was showed that in the patients with ACS, there had been an increase in procoagulant activity because of the increase in Mac 1 (CD11b-CD18) expression in circulating leukocytes, which catalyze the conversion of factor X to Xa. It was also demonstrated that the platelet–leukocyte aggregation increased by Mac 1 may have a role in thrombus formation and decreased perfusion. 10 The leukocytes may also lead to reperfusion injury by free oxygen radicals and to capillary plugging by itself. In line with this pathobiological process, in our study, we demonstrated that the increased EAT thickness, NLR, and the coronary thrombus formation were independent predictors of the no-reflow phenomenon in patients with NSTEMI who underwent PPCI. The EAT can play a role not only in the development of intracoronary thrombus contributing to the inflammatory process in the patients with NSTEMI but also in the development of no-reflow phenomenon.
The intracoronary thrombus burden is related to the increased incidence of in-hospital MACE in the patients with NSTEMI. Besides, the no-reflow phenomenon carries a poor prognosis and an increased mortality rate. 37 Thus, the prediction of the presence of coronary thrombus and the determination of the patients with high risk of the no-reflow phenomenon in the patients with NSTEMI undergoing PPCI before the intervention may contribute to coronary thrombus and no-reflow management. This is carried out by enhancing utilization of preventive measures, which may include pharmacological agents such as glycoprotein IIb/IIIa antagonists and intracoronary vasodilators or interventional strategies such as mechanical thrombectomy.
There are several limitations to the present study. First, this was a cross-sectional study; therefore, we did not perform an analysis related to long-term events. Second, we did not evaluate microvascular no-reflow using myocardial contrast echocardiography or nuclear scintigraphy. Third, the measurement of EAT thickness using cardiac magnetic resonance imaging or computed tomography rather than echocardiography may give a more accurate assessment of epicardial fat amount. 7 However, the widespread use of these methods is not practical. Fourth, adipocytokines were not analyzed, and the exact mechanism of the relation between EAT and thrombus burden and the no-reflow phenomenon could not be clearly demonstrated.
Conclusion
In the current study, we demonstrated that the increased EAT thickness and NLR were independent predictors of the coronary thrombus formation and no-reflow phenomenon in the patients with NSTEMI undergoing PPCI. Our study also indicated that the coronary thrombus formation was independently related to the no-reflow phenomenon. On the basis of this study finding, we have suggested that EAT can play an important role in the pathophysiology of the coronary thrombus formation and the no-reflow phenomenon due to its close proximity to the heart and coronary vasculature and its capacity to secrete high amount of proinflammatory adipocytokines.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
