Abstract
Objectives:
The aim of this study is to investigate the relationship between left ventricular thrombus (LVT) developments and the SYNTAX score (SS) in patients undergoing primary percutaneous coronary intervention (PPCI) for first anterior wall ST-segment elevation myocardial infarction (STEMI).
Methods:
We enrolled 160 patients. All participants were evaluated by serial transthoracic echocardiography. Baseline clinical, echocardiographic, and procedural features of PPCI were analyzed to find predictors of LVT development.
Results:
The LVT was detected in 32 (20%) patients. Left ventricular ejection fraction (LVEF) and SS-I were found to be independent predictors of LVT development. Receiver–operating characteristic curve analysis revealed a cutoff value >19.5 for SS-I (area under the curve: 0.697, 95% confidence interval 0.620-0.767, P < .001) with a specificity of 45.3% and a sensitivity of 84.3%.
Conclusion:
High SS which was obtained through diagnostic angiogram of PPCI may be associated with LVT development in patients with first anterior wall STEMI.
Keywords
Introduction
Left ventricular thrombus (LVT) is a well-known complication of ST-segment elevation myocardial infarction (STEMI). 1 Clinical importance of thrombus formation after STEMI is because of its relation with embolic complications, including embolic stroke. 2,3 Most of the LVT develop within early period after acute myocardial infarction (AMI). 4 In reports, its incidence varied due to different reperfusion strategy. It was reported up to 57% at 48 to 72 hours and 10.4% in the prereperfusion and the early reperfusion era, respectively. 5,6 The rate of occurrence of LVT decreased depending on new reperfusion strategy such as primary percutaneous coronary intervention (PPCI) and early anticoagulation therapy. 7
In previous studies, some predictors of LVT development after STEMI were revealed such as large anterior STEMI, low left ventricular ejection fraction (LVEF), anteroapical aneurysm, and increased wall motion score index (WMSI). 8,9 In thrombolytic and PPCI era, lowering the incidence of LVT development mainly depends on the early and successful reperfusion of infarct-related artery (IRA) that reduced infarct size and salvage of the myocardium. 10 Myocardial damage after STEMI has been determined by reperfusion time and strategy and patency of IRA. 7,11 Previous studies have shown that left ventricular function after STEMI is affected by some angiographic features such as patency and localization of culprit lesion at IRA and grading of collateral circulation during myocardial infarction. 11 –13 SYNTAX score (SS) is a widely accepted reproducible coronary artery disease (CAD) severity and complexity scoring system. 14 –16 It incorporates lesion morphology such as presence and first visualized segment of total occlusion, bifurcation, lesion length, and presence of thrombus. Thereafter, it is multiplied by a factor based on lesion localization that supplied myocardial area. The SS was previously evaluated in patients with STEMI, and there is growing data between SS and area at risk supplied by IRA and long-term mortality and major adverse cardiac events after STEMI. 15,17
The aim of our study was to investigate the relation of LVT development and complexity of CAD assessed by SS in patients undergoing PPCI for first anterior STEMI.
Methods
This was a single-center prospective study, between May 2011 and October 2012. A total of 213 consecutive patients with first acute anterior wall STEMI were enrolled in the study. All the patients underwent PPCI with/without stent implantation within 12 hours of the onset of symptoms. They were treated according to American College of Cardiology Foundation/American Heart Association (ACC/AHA) Guidelines for the Management of Patients With STEMI which was defined by the following criteria: typical chest pain, ST-segment elevation of 1 mm at least in 2 contiguous anterior electrocardiographic leads or new onset left bundle branch block, and transient elevation of serum cardiac biomarkers. 18,19 First anterior wall myocardial infarction was defined by past medical history, and patients with previous myocardial infarction were excluded from the trial. In all, 8 patients died in hospitality during postmyocardial infarction periods, 4 patients were referred to surgery because of the left main coronary artery disease or persistent chest pain due to extensive left anterior descending (LAD) CAD during hospitalization, 2 patients underwent emergency cardiac surgery due to unsuccessful PPCI and complication, and 2 patients died during the follow-up periods. Patients with stent thrombosis, rescue and facilitated PCI, vasospastic angina, having poor echocardiographic window, previous treatment with warfarin for other medical reasons, moderate to severe valvular heart disease, rhythm and conduction abnormalities, cardiogenic shock and history of previous coronary artery bypass grafting, unsuccessful recanalization of IRA, and previously having documented LVT were excluded from the study. Therefore, the remaining 160 patients constituted the study population.
All patients gave informed consent before enrollment, and the study protocol was approved by the ethics committee. Baseline clinical and demographic characteristics were obtained from all the patients. A detailed physical examination was performed including past medical history. Blood sampling during presentation, including complete blood count, lipid profile, serum creatinine level, and serum cardiac biomarkers were obtained from all the patients. Hypertension was identified on the basis of prior receiving antihypertensive treatment or if the blood pressure exceeded 140/90 mm Hg in at least 3 measurements. Diabetes was defined as prior receiving oral antidiabetic medications or insulin or having fasting glucose levels above 126 mg/dL. The glomerular filtration rate was calculated using the Modification of Diet in Renal Disease formula. Killip classes, symptom onset-to-hospital admission time at presentation, and inhospital medications, including antiagregan and anticoagulation medications, were recorded. All the patients underwent PPCI within half an hour after the admission.
Baseline detailed 2-dimensional transthoracic echocardiography was performed in all patients within 48 hours after PPCI. For LVT diagnosis, serial 2-dimensional transthoracic echocardiography was performed in all patients at discharge and at 2 weeks after admission, and all images were stored for offline analysis. Patients’ angiographic characteristics during PPCI were analyzed.
Echocardiography
Baseline comprehensive 2-dimensional transthoracic echocardiography including M mode and Doppler echocardiography was performed by one experienced cardiologist blinded to angiographic data, using Vivid-S5, GE (United States) instrument, with 3.6 MHz transducer. Second harmonic imaging was used to optimize endocardial visualization. Apical views, 4-chamber, 2-chamber, long-axis, and short-axis views (SAX), basal SAX, mid SAX, and apical SAX were stored for 3 consecutive cardiac cycles.
Measurements were performed according to guidelines of American Society of Echocardiography. 20 The LVEF was calculated by the Simpson biplane formula. The LV WMSI was calculated according to 16-segment model. Each segment was scored with regard to endocardial motion and systolic thickening, and it was graded as follows: normal motion at rest (score = 1); hypokinetic (score = 2); akinetic (score = 3); and dyskinetic (score = 4). The WMSI was calculated by summation of individual segment scores divided by the number of interpreted segments.
All images that were obtained at discharge and at 2 weeks after admission were analyzed offline (EchoPAC 108.1.12, General Electric Medical Systems, Horten, Norway) to detect LVT by 2 experienced cardiologists blinded to angiographic data. The LVT was diagnosed when an echodense mass which was separated clearly from LV wall border was visualized at least in 2-different orthogonal echocardiographic plane, adjacent to an area of affected anterior wall of myocardium 21 (Figure 1). Each cardiologist separately interpreted echocardiographic views for LVT, and LVT was diagnosed if there were agreements between decisions of 2 cardiologists. Otherwise, patients with discrepancy diagnoses were interpreted by 2 cardiologists together and final decisions were made.
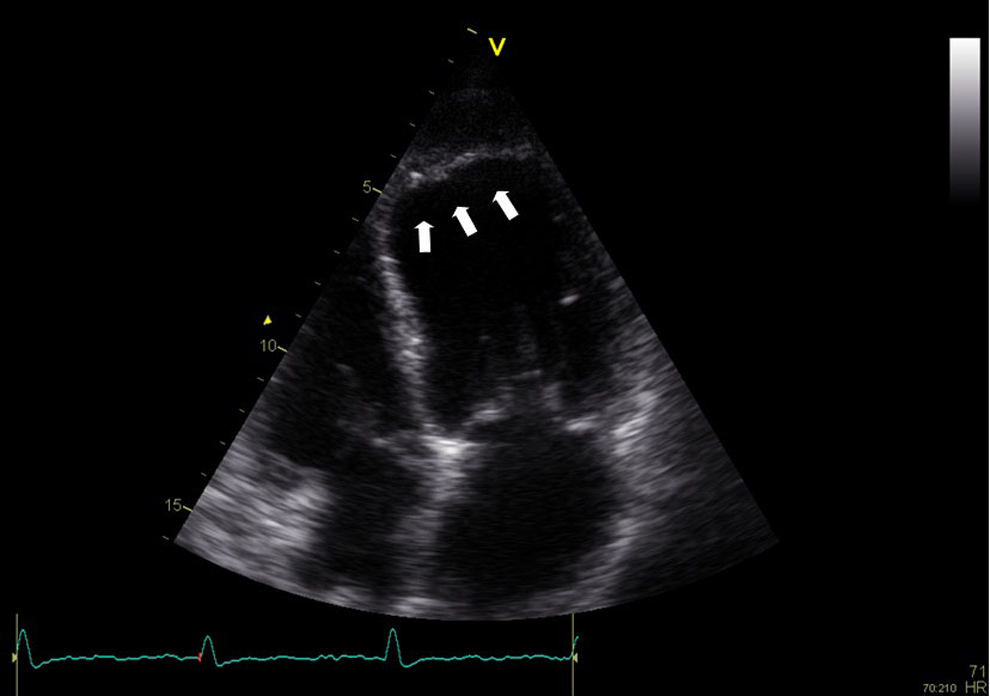
The LVT was diagnosed in left ventricle apex after first anterior wall STEMI, apical 4-chamber view, and white arrows indicate an echodense mass adjacent to akinetic LV apex with a distinct border from myocardial wall. LVT indicates left ventricular thrombus; STEMI, ST-segment elevation myocardial infarction.
Angiographic Characteristics
All patients were treated with PPCI within 12 hours of onset of symptoms. Medical treatment regimens that were administered in emergency room, referral center, and catheter laboratory were recorded including aspirin, loading dose of clopidogrel, and heparin doses. Glycoprotein IIb/IIIa inhibitors were administered at the discretion of operator. Significant lesion was defined as a ≥50% stenosis in the luminal diameter of major coronary artery. Basal angiographic characteristics of patients such as diseased vessel number and patency of IRA were recorded. Procedural features including final thrombolysis in myocardial infarction (TIMI) flow grade at IRA, presence of distal embolization, reperfusion methods, presence of postdilatation, performing of thrombus aspiration, total stent length and diameter, multiple stenting at IRA, final corrected TIMI frame count (cTFC), and coronary collateral circulation were recorded
SYNTAX Score
The SS is mainly related to the complexity of CAD. It was calculated using dedicated software that integrates 2 components (1) morphological features of each lesion such as dominance, chronic total occlusion, bifurcation, trifurcation, tortuosity, heavy calcification, lesion length, presence of thrombus, aorto-ostial, and diffuse lesion and (2) weighting factors of lesions based on myocardial area distal to lesion. Lesions with ≥50% luminal obstruction in vessels with a diameter ≥1.5 mm were added to provide SS. 14 The SS was calculated using dedicated software (version 2.11, www. syntaxscore. com), and it was first calculated before wiring IRA (SS-I) and then after the wiring or initial small balloon predilatation of IRA (SS-II). Finally, residual SS (r-SS) was calculated according to angiographic images after the culprit lesion revascularization. Both SS-I and SS-II were accepted as the same in patients with TIMI flow grade >1 at IRA. All morphological features of each lesion which are included in SS were recorded. All angiograms were scored by 2 experienced interventional cardiologists who were blinded to the echocardiographic data.
Statistical Analysis
SPSS 17.0 statistical software (SPSS Inc, Chicago, Illinois) and MedCalc software program, release 12.3.0.0 (MedCalc Software, Belgium) were used for statistical analysis. Continuous variables were expressed as mean ± standard deviation and categorical variables were expressed as percentage. The Kolmogorov-Smirnov test was used to test the normality of distribution of continuous variables. Group means for continuous variables were compared to the use of Student t test or the Mann-Whitney U test, as appropriate. Categorical variables were compared with the use of chi-square test. The Pearson correlation analysis was used for assessing correlation between LVEF and SS. Variables with a P value of ≤.05 at univariate analysis were selected for logistic regression analysis. Logistic regression model was performed to find independent associates of LVT development. Independent predictors of LVT development are presented as adjusted and unadjusted odds ratios (ORs) and 95% confidence intervals (CIs) due to the results of the model. Interobserver agreement of SS data was calculated using Bland-Altman analysis, and intraclass correlation coefficient was used to assess the intraobserver agreement. Interrater agreement of LVT diagnosis was calculated by weighted kappa test. Receiver–operating characteristic (ROC) curve analysis was performed to detect the cutoff value of SS in prediction of LVT development. A P value of ≤.05 was considered statistically significant.
Results
Study population included 160 patients, 131 (81.9%) male and mean age was 60.2 ± 14.6. The LVT was detected in 32 (20%) patients and it was localized to LV apex in all the patients. Thrombi were initially detected at the discharge and control transthoracic echocardiographic evaluation in 25 of 32 and 7 of 32 patients, respectively. Stenting was performed in 146 (91.3%) patients. Baseline clinical, laboratory, and echocardiographic features were demonstrated in Table 1. Peak creatine kinase MB values (226.9 ± 93.1 vs 159.0 ± 107.2, P = .001), WMSI (1.5 ± 0.2 vs 1.3 ± 0.2, P < .001), presence of hypertension (71.9% vs 49.2%, P = .021), and Killip class II-III-IV (18.8% vs 5.5%, P = .014) were significantly higher among patients with thrombi, while LVEF was significantly lower (38.5 ± 4.2 vs 45.2 ± 6.9; P < .001). Procedural features were demonstrated in Table 2. The cTFC (36.5 ± 19.8 vs 25.7 ± 10.7, P < .001) and final TIMI flow grade 0 or 1 (9.4% vs 1.6%, P = .023) were significantly higher in patients with thrombi. In the calculated SS, SS-I and r-SS were significantly increased in patients with thrombi while SS-II was not significantly increased (Figure 2).
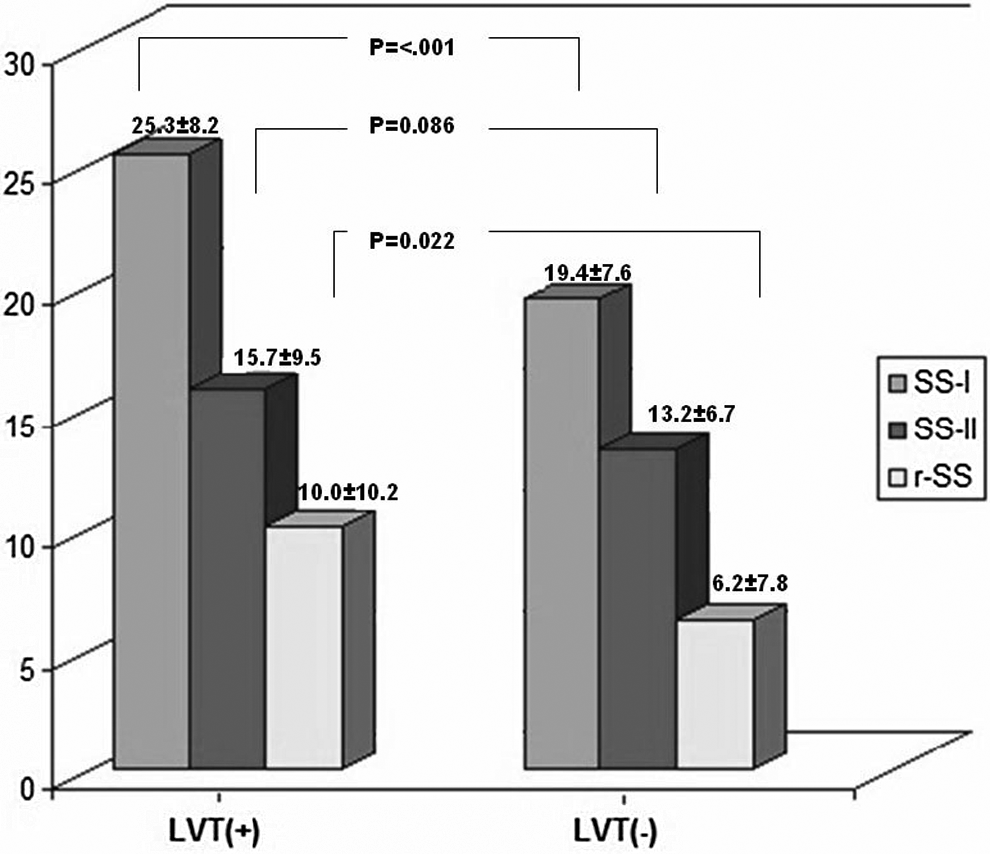
The mean values of SS-I, SS-II, and r-SS between groups with/without LVT. (SS-I and SS-II were SYNTAX score which was calculated before and after wiring of infarct related artery, respectively. r-SS was residual SYNTAX score that was calculated according to angiographic images after the culprit lesion revascularization.)
Baseline Clinical, Laboratory, and Echocardiographic Features of Patients.
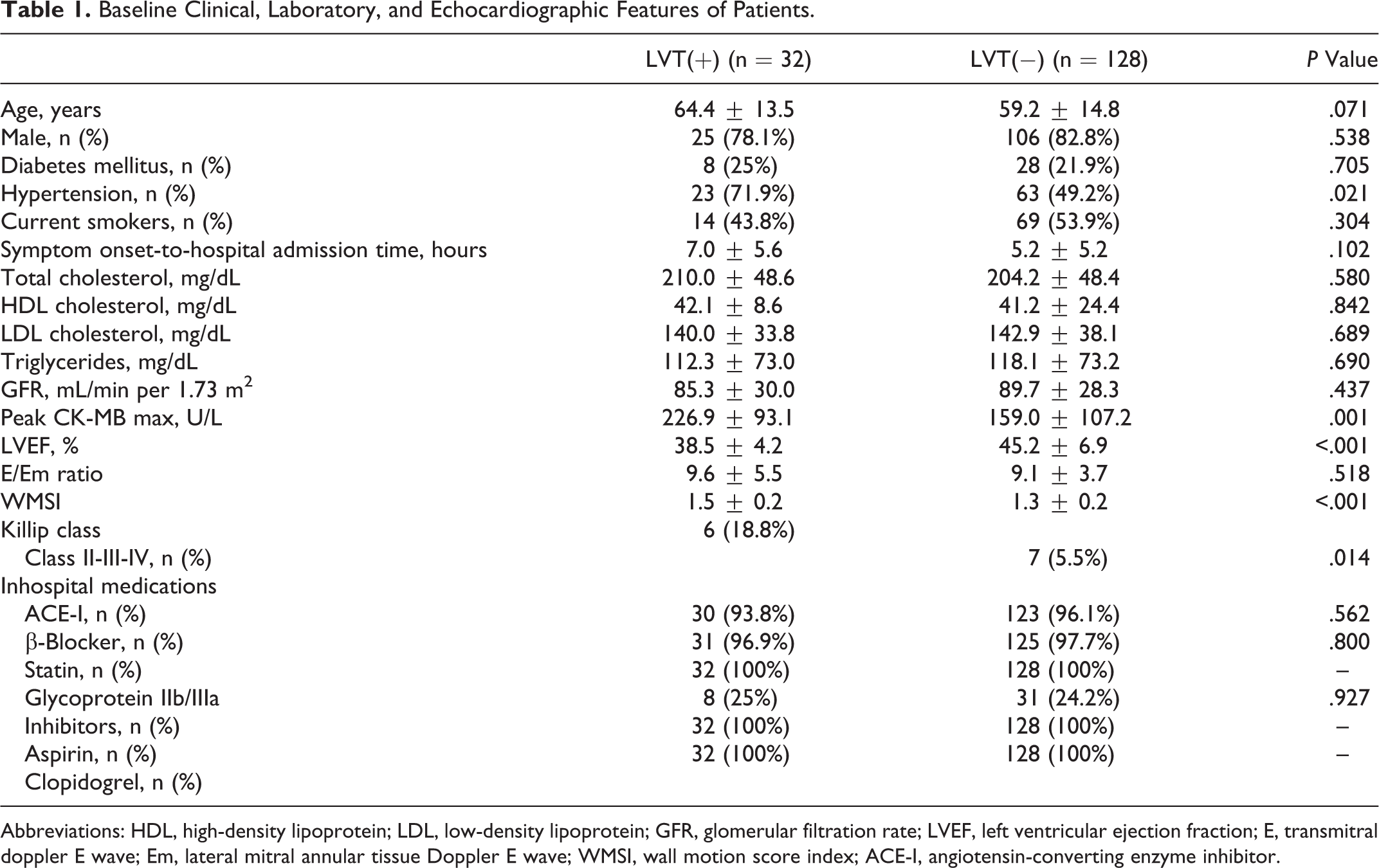
Abbreviations: HDL, high-density lipoprotein; LDL, low-density lipoprotein; GFR, glomerular filtration rate; LVEF, left ventricular ejection fraction; E, transmitral doppler E wave; Em, lateral mitral annular tissue Doppler E wave; WMSI, wall motion score index; ACE-I, angiotensin-converting enzyme inhibitor.
Procedural Features of Patients Obtained During PPCI.
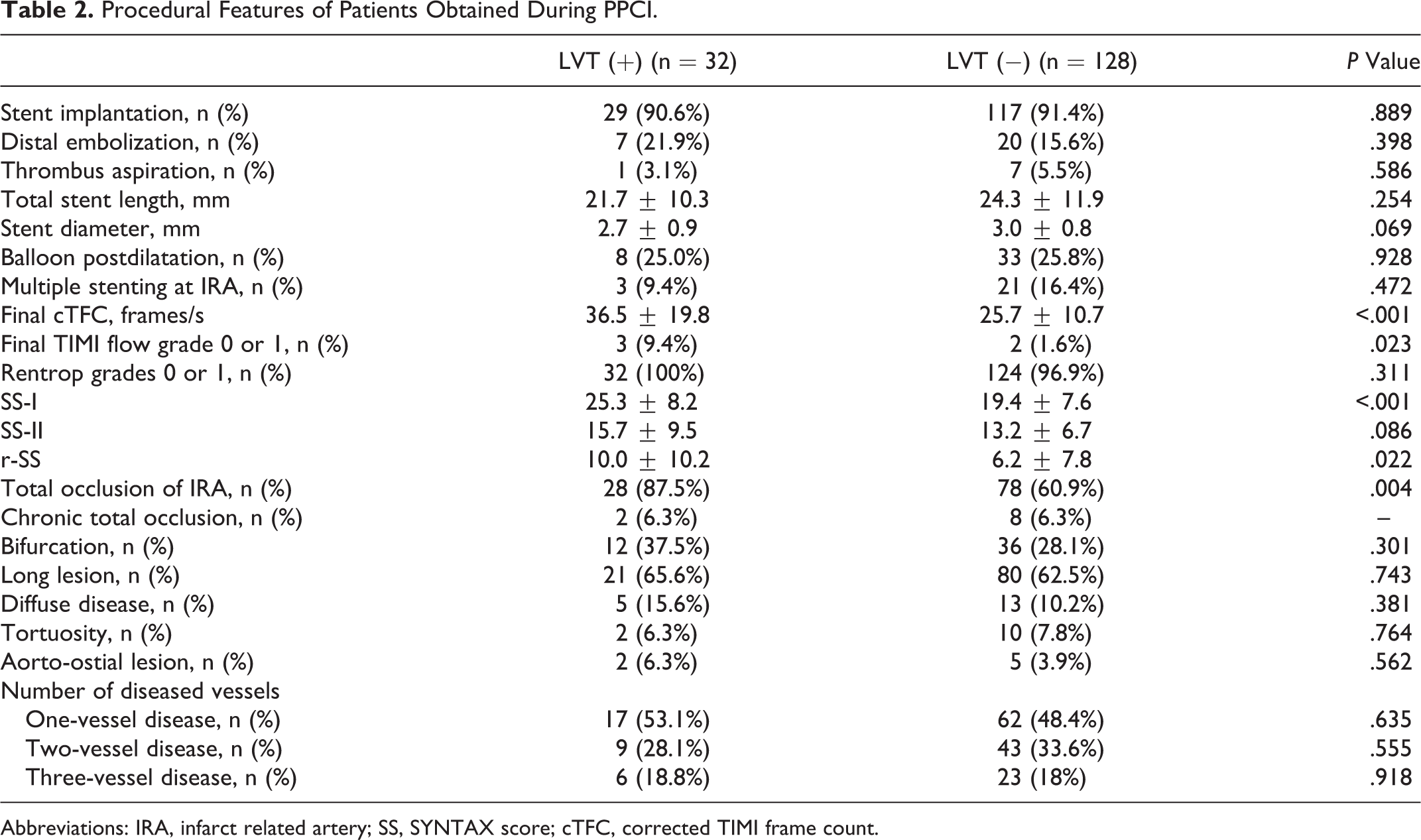
Abbreviations: IRA, infarct related artery; SS, SYNTAX score; cTFC, corrected TIMI frame count.
Independent Predictors of LVT Development
To assess the independent predictors of LVT, clinical, laboratory, echocardiographic, and procedural features that were significantly different between groups at univariate analysis were further evaluated with logistic regression model. In multivariate analysis, LVEF (OR: 0.83, 95% confidence interval [CI]: 0.71-0.98, P = .027) and SS-I (OR: 1.14, 95% CI: 1.01-1.29, P = .025) were found to be the independent predictors of LVT development (Table 3). At univariate analyses, LVEF was found to be negatively correlated with SS-I (r: −.325, P < .001).
Independent Predictors of LVT Development in Multivariate Analysis.

Abbreviations: LVEF, left ventricular ejection fraction; SS, SYNTAX score.
Assessment of Cutoff Point of SS-I for LVT Development
The ROC curve analysis was performed to detect the cutoff value of SS-I in the prediction of LVT development (Figure 3). The SS-I identified patients with LVT with a specificity of 45.3% (95% CI: 36.5%-54.3%) and a sensitivity of 84.3% (95% CI: 67.2%-94.7%) and with a cutoff value of >19.5 (area under the curve: 0.697, 95% CI: 0.620-0.767, P < .001).
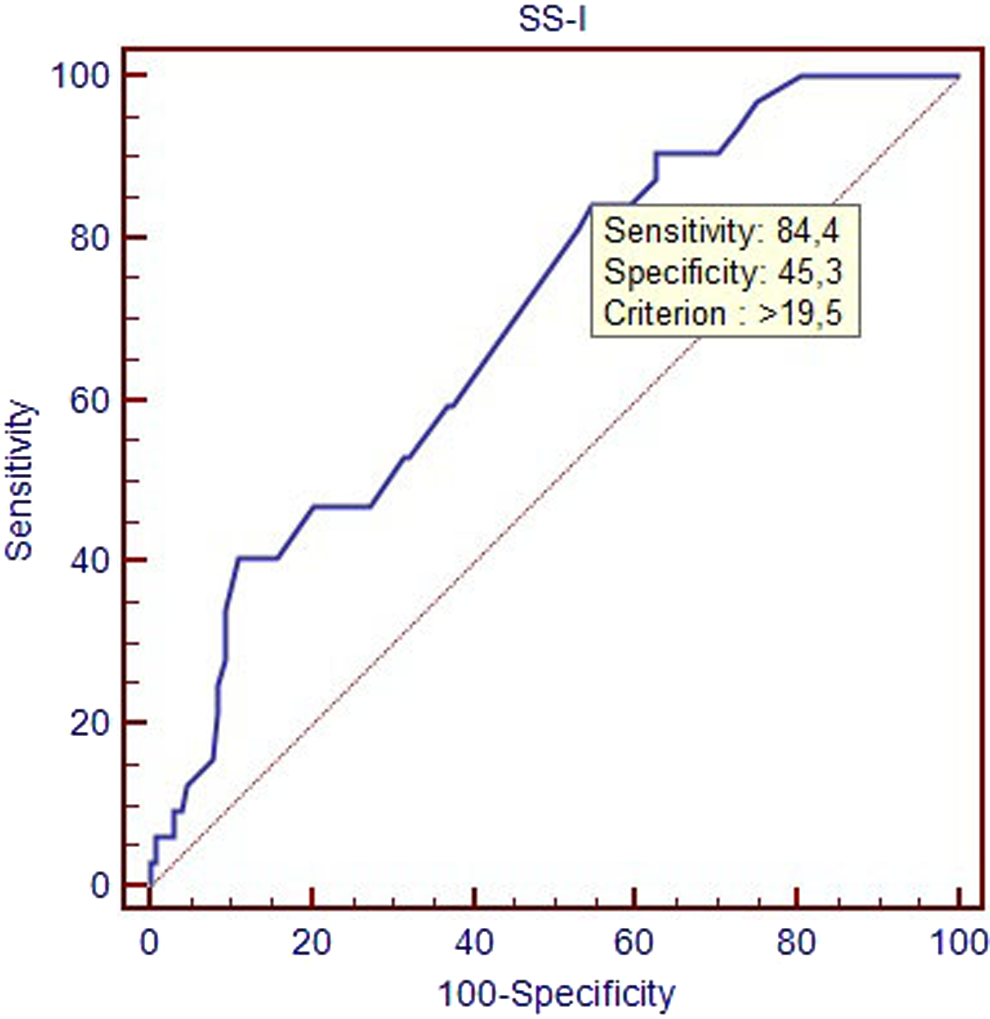
Receiver–operating characteristic (ROC) curves of SS-I for identification of patients with thrombi. SS indicates SYNTAX score.
Agreement analysis of interobserver and intraobserver for SS measurements revealed high agreement; a mean difference of 0.3 (95% limit of agreement −5.1, 5.8) and an intraclass correlation coefficient of 0.98 (95% CI 0.97-0.98), respectively. Interrater agreement of LVT diagnosis was substantial and weighted kappa coefficient of 0.733 (95% CI: 0.601-0.865).
Discussion
Our study indicates that LVEF and SS which were obtained during diagnostic angiogram of PPCI were independently associated with LVT development in patients undergoing PPCI for first anterior STEMI. A cutoff value >19.5 with a specificity of 45.3% and a sensitivity of 84.3% were found for identification of patients with thrombi.
The LVT is one of the more common complications of myocardial infarction, and it is most often seen in patients with anterior wall STEMI. 2 Predictors of LVT development after STEMI must be well known in terms of its relation with embolic stroke and increased bleeding complications with respect to oral anticoagulant therapy that was added to dual antiplatelets regimen after PPCI, and incidence of embolic stroke in patients with documented LV thrombi on echocardiography was increased 5.5 times after anterior STEMI. 24,25 In previous reports, some clinical and echocardiographic parameters were well established for LVT development. 8,9 However, relation of angiographic characteristics with LVT development is not well studied. The SS indicates complexity and severity of CAD and validated for patients with STEMI. 14,15 It is based on 3 parameters including dominance artery of coronary circulation, morphological features of lesions, and jeopardized myocardium distal to lesions. These parameters are the main determinants of myocardial area at risk during STEMI. When our study results were handled according to these parameters, left and right dominant coronary artery system has different impact on the flow to left ventricle. Thus, myocardial damage during anterior wall STEMI may not be the same in patients with LAD proximal culprit lesion with TIMI flow grade (0-1) depending on left or right dominant coronary artery system and amount of supplied flow to left ventricle by dominant coronary artery system. Right coronary artery (RCA) has supplied only 1% of the flow to left ventricle in left dominant coronary artery system. 15 The SS assigns different multiplying factor in terms of lesions in LAD (3.5), RCA (1), and circumflex coronary artery (1.5) due to functional impact; also, lesions at diverse segments of the same coronary artery were scored differently based on jeopardized myocardial area distal to lesions. 15,26 Therefore, the relation of SS-I with LVT development can be explained by the presence of having more proximal culprit lesions at IRA and other coronary arteries in patients with thrombi, all of them were included in SS-I. The morphologic features of culprit lesions during STEMI enclosed at IRA, according to patency or total occlusion of the IRA, SS was scored additionally 5 points in patients with TIMI flow grade (0-1). Nesković et al and Pizzetti et al have shown that total occlusion of IRA was related to the LVT development that was consistent with our study results, because of higher SS in patients with total occlusion of IRA. 8,11 The SS does not only contain occlusion of IRA; additionally, it provides the information about first visualized segment and the presence of side branch ≥1.5 mm when total occlusion was present at IRA. Higher SS was calculated, when first visualized segments were the most distal segment of total occluded IRA or presence of side branch ≥1.5 mm. Absence or poor collateralization during STEMI was associated with higher SS. Lønborg et al and Ortiz-Pérez et al have reported that angiographically visible collateral flow was associated with myocardial salvage index and reduction in infarct transmurality during AMI. 27,28 Our study results were compatible with these findings owing to relationship between myocardial damage and LVT development. Presence of side branch ≥1.5 mm close to the occluded IRA adds 1 point to SS; it may be associated with thrombus embolization to side branch and increased infarct size during PPCI. Zielinska et al reported that angiographic severity of CAD was not related to LVT development. 29 In that study, severity of CAD was only assessed by diseased vessel number, a simple method to evaluate severity of CAD, SS takes into account the lesion morphology and weighting factor in terms of myocardial area supplied by lesions in addition to diseased vessel number. Therefore, elevated SS was associated with LVT development.
In our study, SS-II was not associated with LVT development. The Main reason why SS-II did not associate may be restoration of flow distal to IRA and reduction of myocardial damage due to reperfusion of myocardium. Généreux et al reported that residual stenosis after PCI was associated with short- and long-term outcome in patients with acute coronary syndrome. 30 In our study, r-SS was not associated with LVT development in patients with first acute anterior wall STEMI. It may be attributable to relatively long-term effect of r-SS over left ventricle remodeling rather than acute effect over the left ventricle function within early days after myocardial infarction. Our study findings were compatible with the previous reports with respect to relationship between left ventricular function and LVT development. 31 The SS-I negatively correlated with LVEF.
Incidence of LVT was relatively high in our reports. It may be due to serial echocardiographic follow-up and study population consisted with anterior wall myocardial infarction. The study population was relatively older, and GUSTO-1 trial revealed that elderly patients have increased LV dysfunction despite successful reperfusion. 32 It may be other reason for high thrombus incidence. Our hospital is a referral center for PPCI, although there was no difference between the groups according to symptom onset to hospital admission time; incidence of LVT may be affected by late hospital admission.
Conclusion
The LVT is still one of the more common complications of myocardial infarction. Predictors of LVT development after myocardial infarction, especially anterior wall STEMI, must be known well, because of its relation to embolic complications and treatment challenge. High SS that was obtained by diagnostic angiogram of PPCI associated with LVT development in patients undergoing PPCI for first anterior STEMI.
Limitations
A main limitation of our study was the determination of LVT. Recent studies suggest that cardiovascular magnetic resonance may be more sensitive for LVT diagnosis. Contrast echocardiographic assessment was recommended by some reports in patients with suboptimal echocardiographic windows after STEMI. In our study, patients with suboptimal windows were excluded, and in order to increase the diagnostic accuracy several images were obtained and stored for the same patients in each echocardiographic evaluation. All images were interpreted by 2 cardiologist blinded to angiographic data. Another limitation of the study was that patients with high risk of LVT development such as cardiogenic shock or ongoing chest pain due to extensive CAD were not included in the study.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
