Abstract
Using parenteral anticoagulants in compliance with their summaries of product characteristics (SPCs) and consensus guidelines is associated with decreased rates of adverse drug reactions. The objectives were to assess compliance of parenteral anticoagulants use with SPCs and 4 consensus guidelines in outpatients of general practice. A descriptive cross-sectional study was conducted among 112 teaching general practitioners’ offices, from May to October 2011. Among the 332 adult outpatients consecutively included, 108 (32.5%) had a parenteral anticoagulant therapy complying with SPCs for both indication and regimen. The rate of compliance increased when considering consensus guidelines (159 patients, 47.9%). Rate of adverse events was 5.4%, including 0.6% of serious adverse events. This study showed that the use of parenteral anticoagulants complies with indications and regimens of SPCs in only 1 of the 3 outpatients. Further studies should be conducted in primary care to explore reasons for such low compliance rates.
Introduction
Parenteral anticoagulants are used for prophylaxis and treatment of thromboembolism. In 2011, 1.7 million French patients received at least once low-molecular-weight heparins (LMWHs), 54 000 unfractionated heparins (UHs), and 53 000 factor Xa inhibitors (fondaparinux). 1 Anticoagulants (including oral and parenteral anticoagulants) are one of the main drugs involved in preventable serious adverse drug reactions (ADRs). 2 Incidence of major bleedings is 0% to 7% for curative doses of UHs, with a mortality rate of 0% to 2%. For curative doses of LMWHs, incidence of major bleedings is 0% to 3% with a mortality rate of 0% to 0.8%. Overall, incremental risk of major bleeding with UFH or LMWH ranges from 0% to 2% and increases with concomitant medication and intensity of treatment. For fondaparinux, incidence of major bleedings is 0.2% to 2.4%, and mortality rates are poorly assessed. 3
Summaries of product characteristics (SPCs) provide information on drugs to physicians. The SPCs contain essential data on approved indications, regimen, ADRs, drug interactions, and basic pharmacodynamics and pharmacokinetics. The SPCs are validated through marketing authorizations process. Use of parenteral anticoagulants in compliance with their SPCs is associated with a decrease in ADR occurrences. 4 However, daily parenteral anticoagulants use may shift over time to newer indications, 5,6 defining “off-label” use. 7 Consequently, consensus guidelines elaborated by scientific societies may propose indications or doses for parenteral anticoagulant use that differ from SPCs. 8 –12
Several observational studies have assessed the compliance of parenteral anticoagulants use in hospital settings. 13 –16 These studies suggested that quality and safety of parenteral anticoagulants use are warranted in most of inpatients. However, to our knowledge, no study has assessed this issue in primary care settings. We thus conducted a descriptive study aiming to assess compliance of parenteral anticoagulants use with SPCs in outpatients in general practice. Secondary objective was to assess compliance with French consensus guidelines.
Methods
A descriptive cross-sectional study was conducted among the offices of 112 teaching general practitioners (GPs) from Midi-Pyrénées area (South Western France), from May to October 2011. Fifty-six general practice residents in rotation in these offices were responsible for inclusion and data collection. Each outpatient encountered during the study period (at the office or during home visit), aged older than 18, and treated with parenteral anticoagulant (LMWHs, UHs, or fondaparinux) was consecutively included (maximum 10 patients per resident, to account for potential cluster effect). No exclusion criterion was used. Three reminders were sent to GPs by post during the study period.
Patients’ characteristics (age, gender, weight, and serum creatinine for estimation of glomerular filtration rate using the Cockcroft-Gault formula) were retrieved in GPs’ medical records and collected in standardized forms. Parenteral anticoagulant indications were classified according to the following classification: deep vein thrombosis prophylaxis (including postsurgical indications), vein thrombosis (deep and superficial) and pulmonary embolism treatment, arterial thrombosis prophylaxis, and other (including catheter thrombosis and blood coagulation disorders). The indications were then grouped as preventive or curative.
The main outcome was global compliance of parenteral anticoagulant use with 2011 SPCs and associated compliance with both indication and daily regimen (the 2 main criteria of SPCs). 7 Daily regimen was considered compliant if the dosage was in a ±0.05 mL range of the recommended daily dosage (based on the experience of a panel of clinician experts) and associated with the adequate frequency of administration (1, 2, or 3 injections a day).
We also assessed the compliance with indication according to 4 French consensus guidelines (French Society of Anesthesia and Intensive Care, 8 French Society of Cardiology, 9,10 French College of Obstetricians and Gynaecologists, 11 and Agence Nationale de Sécurité du Médicament et des Produits de Santé [ANSM, the French Drug Agency]). 12
Adverse events were recorded and classified “serious” (if lethal, life-threatening, requiring hospitalization or hospitalization prolongation, or causing persistent or significant disability/incapacity) or not. All data were made anonymous during form filling. No ethical approval was sought nor was patient informed consent obtained due to the observational, noninterventional nature of this study.
Descriptive statistics were computed for exploration and description of the results. All patients were considered, including those with some unavailable data. For the statistical analysis, Pearson chi-square test was performed at a significance level of 5%. For all analyses, Stata version 11.0 (StataCorp LP, College Station, Texas) was used.
Results
Population Description
A total of 53 general practice residents collected data on 343 patients (participation rate 94.6%, Figure 1). Among them, 11 were not included (7 patients aged younger than 18 and 4 patients without birth date). Over the remaining 332 patients, 170 (51.2%) were women. Mean age was 65.9 ± 18.9 years. Weight was filled for 211 (63.6%) patients, and serum creatinine for 134 (40.4%) patients. Overall, creatinine clearance could be calculated for 97 (29.2%) patients. In all, 41 (12.3%) patients had a creatinine clearance <60 mL/min including 4 (1.2%) with a creatinine clearance <30 mL/min.
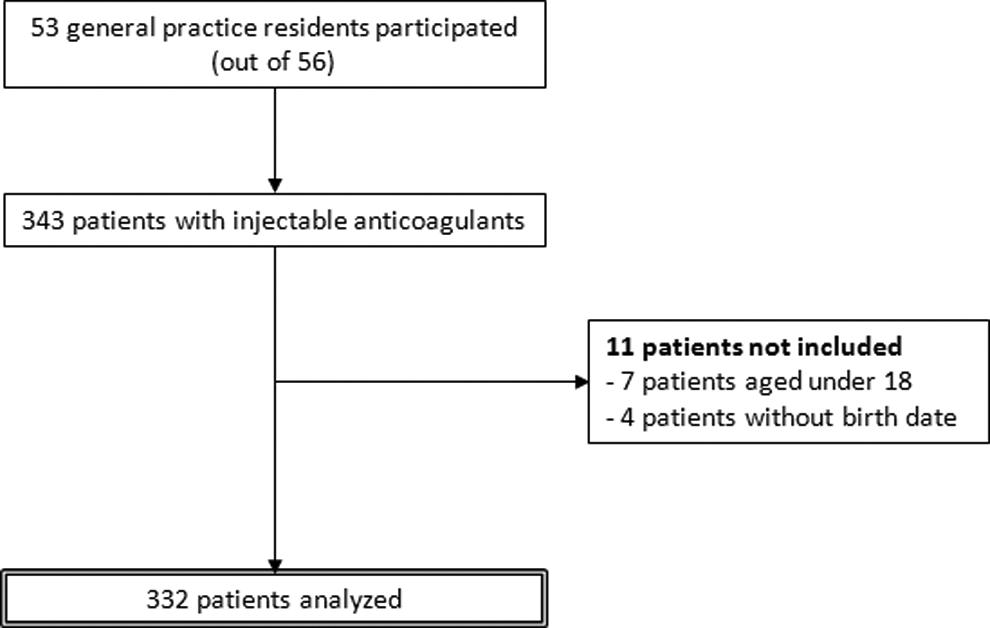
Patient selection flowchart.
The main indication was deep vein thrombosis prophylaxis (161 patients, 48.5%). In all, 91 (27.4%) patients were treated for vein thromboembolism: 74 (22.2%) patients for deep vein thrombosis (including 22 [6.6%] waiting for diagnosis confirmation through ultrasonographic evaluation), 13 (3.9%) patients for superficial vein thrombosis (1 associated with deep vein thrombosis), and 8 (2.4%) patients for pulmonary embolism (3 associated with deep vein thrombosis). Fifty (15.1%) patients were treated for arterial thrombosis prophylaxis (atrial fibrillation or mechanical heart valve).
Compliance With SPCs and Consensus Guidelines
Table 1 shows compliance with SPCs according to classes of parenteral anticoagulants. One hundred and eight (32.5%) patients had a parenteral anticoagulant therapy compliant with SPCs (indication and daily regimen). In 196 (59.0%) cases, parenteral anticoagulant therapy was classified as noncompliant with SPCs because of a nonconform indication (163 patients, 49.1%) and/or a nonconform daily regimen (77 patients, 23.2%). Compliance with regimen could not be assessed for 60 patients (18.1%, see table footnotes for details). The rate of compliance increased when considering consensus guidelines (159 patients, 47.9%; Table 2), since compliance with indication increases to 77.4% (257 patients).
Compliance of Parenteral Anticoagulants Use With Summary Product Characteristics (Indication and/or Regimen), According to Injectable Anticoagulants.a
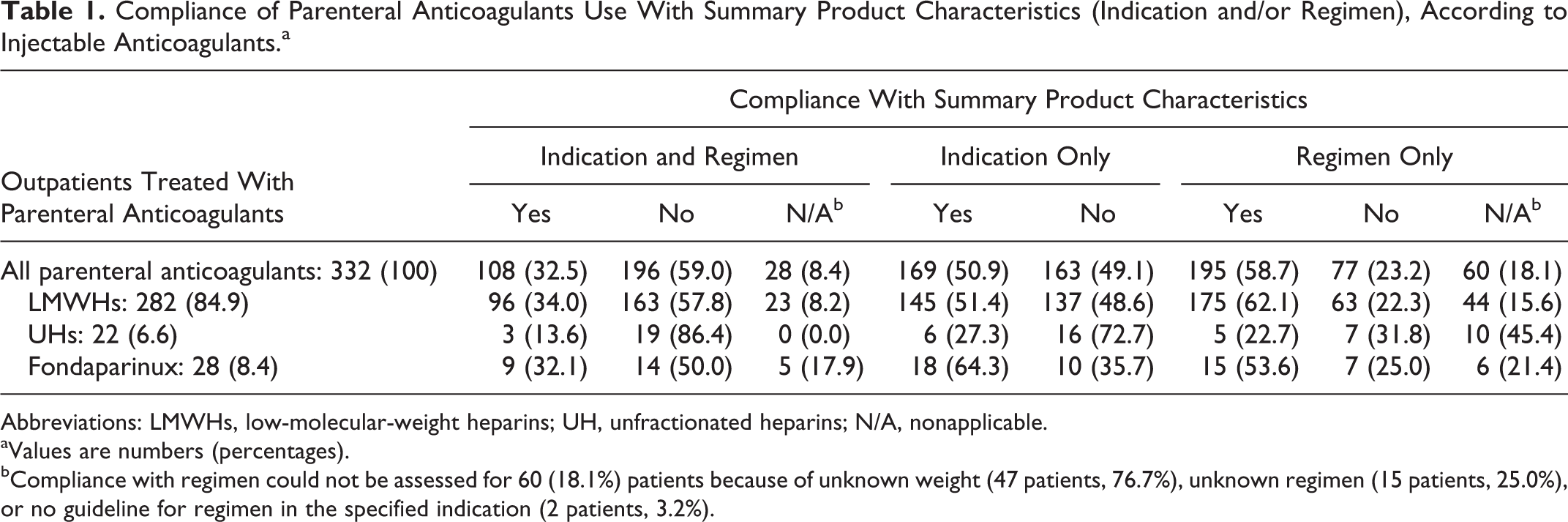
Abbreviations: LMWHs, low-molecular-weight heparins; UH, unfractionated heparins; N/A, nonapplicable.
aValues are numbers (percentages).
bCompliance with regimen could not be assessed for 60 (18.1%) patients because of unknown weight (47 patients, 76.7%), unknown regimen (15 patients, 25.0%), or no guideline for regimen in the specified indication (2 patients, 3.2%).
Compliance of Parenteral Anticoagulants Use With Consensus Guidelines (Indication and/or Regimen), According to Injectable Anticoagulants.a
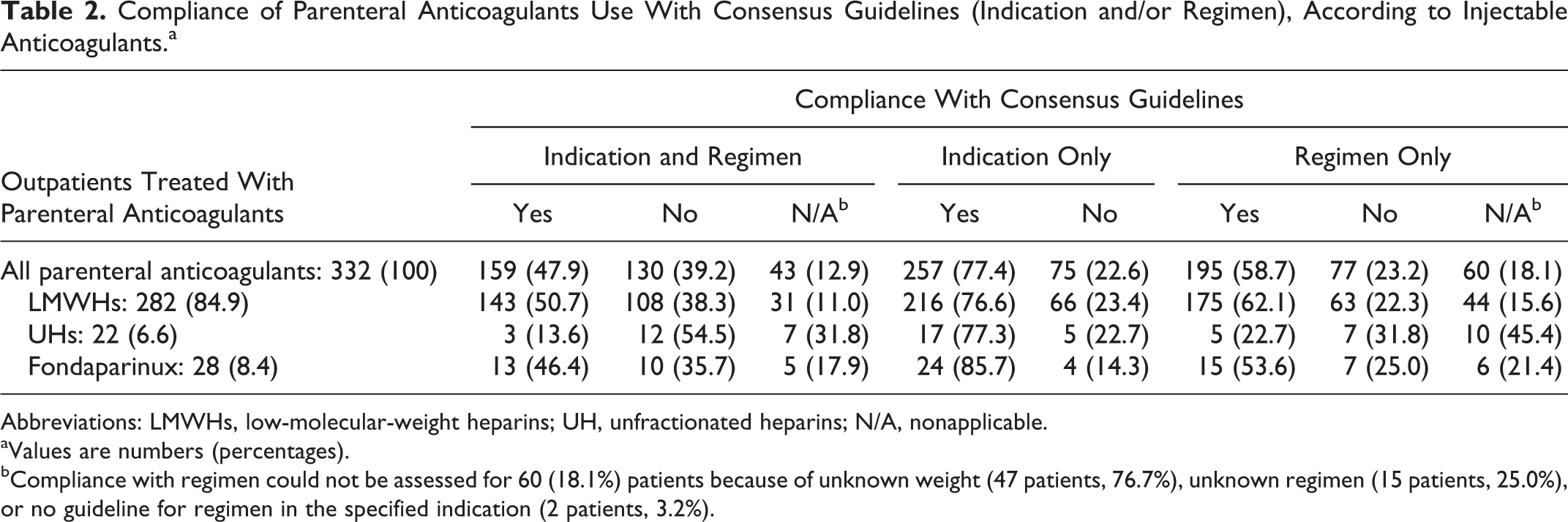
Abbreviations: LMWHs, low-molecular-weight heparins; UH, unfractionated heparins; N/A, nonapplicable.
aValues are numbers (percentages).
bCompliance with regimen could not be assessed for 60 (18.1%) patients because of unknown weight (47 patients, 76.7%), unknown regimen (15 patients, 25.0%), or no guideline for regimen in the specified indication (2 patients, 3.2%).
Compliance by Indications
Tables 3 and 4 show compliance with SPCs and consensus guidelines according to the indications of parenteral anticoagulants (greater details for indication are given in Supplemental Tables S1 and S2). The main nonconform indication was arterial thrombosis prophylaxis (0% of global compliance according to SPCs and 8% according to consensus guidelines), followed by vein thromboembolism (33% of global compliance according to SPCs and 36% according to consensus guidelines). Indications compliance rates also differed between SPCs and consensus guidelines in indications of deep vein thrombosis prophylaxis for lower limb fracture/cast immobilization (Supplemental Tables S1 and S2).
Compliance of Parenteral Anticoagulants Use With Summary Product Characteristics (Indication and/or Regimen), According to Indication.a
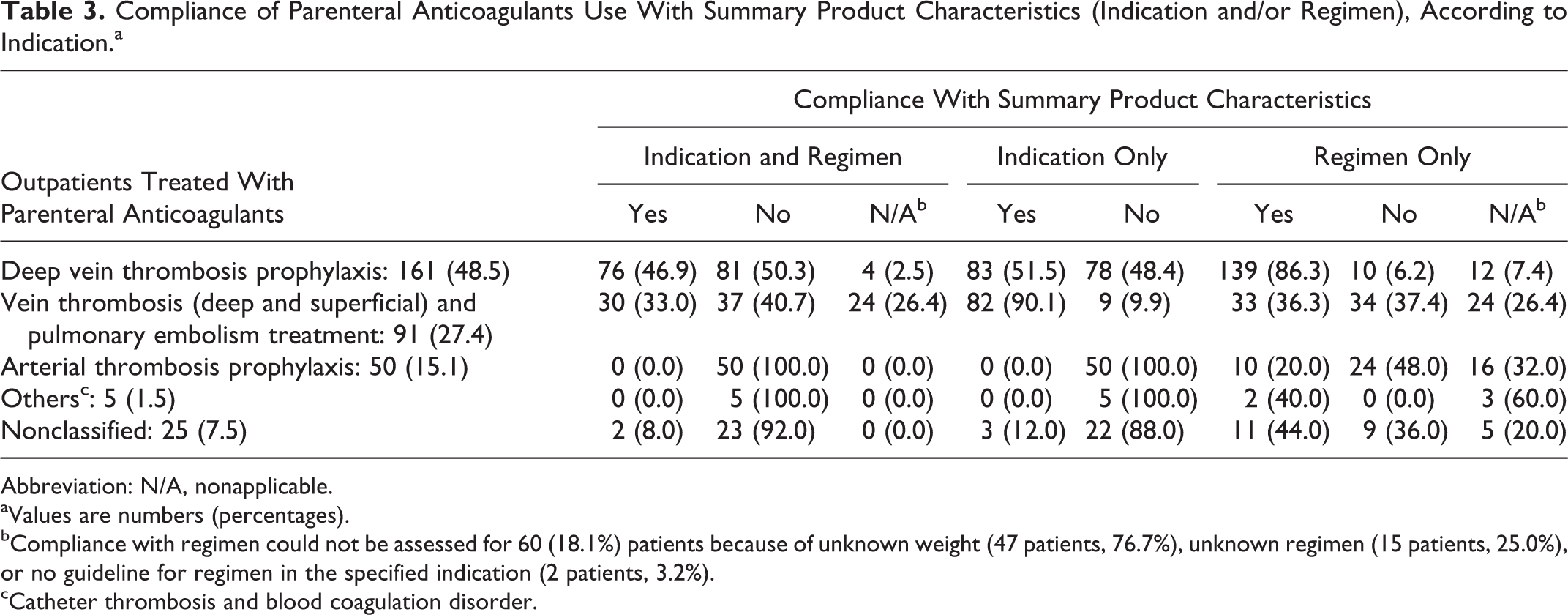
Abbreviation: N/A, nonapplicable.
aValues are numbers (percentages).
bCompliance with regimen could not be assessed for 60 (18.1%) patients because of unknown weight (47 patients, 76.7%), unknown regimen (15 patients, 25.0%), or no guideline for regimen in the specified indication (2 patients, 3.2%).
cCatheter thrombosis and blood coagulation disorder.
Compliance of Parenteral Anticoagulants Use With Consensus Guidelines (Indication and/or Regimen), According to Injectable Anticoagulants.a
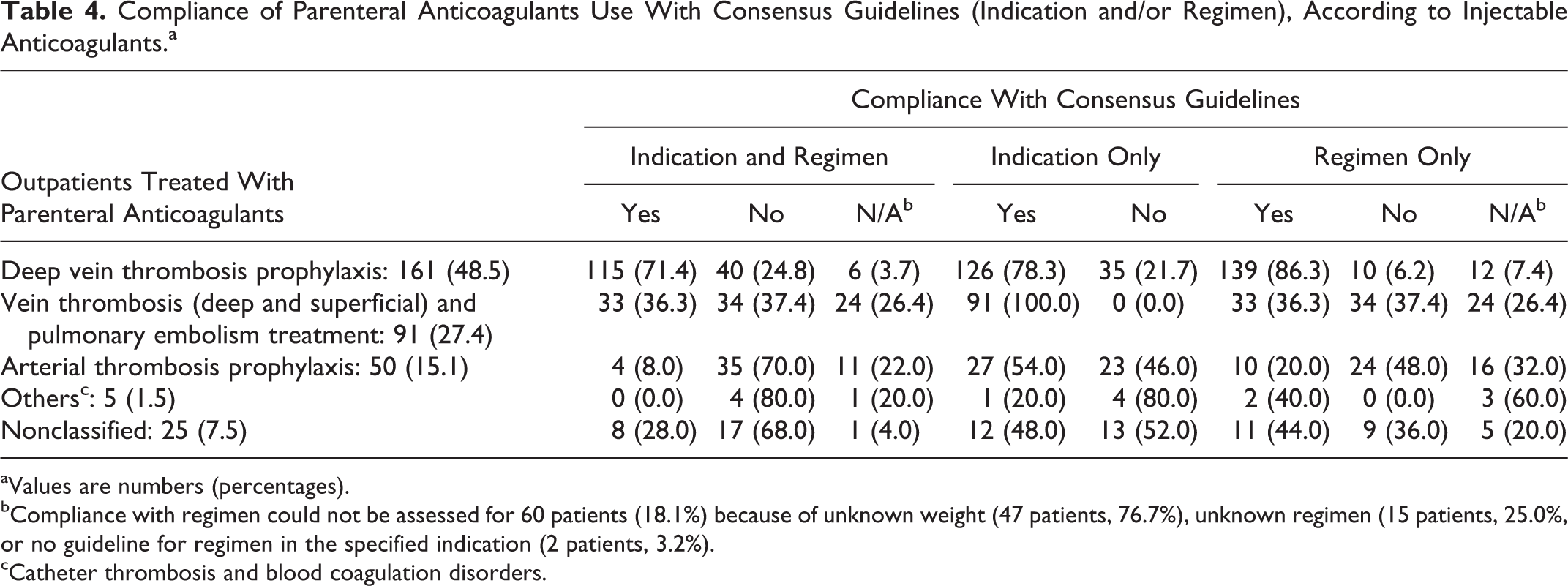
aValues are numbers (percentages).
bCompliance with regimen could not be assessed for 60 patients (18.1%) because of unknown weight (47 patients, 76.7%), unknown regimen (15 patients, 25.0%, or no guideline for regimen in the specified indication (2 patients, 3.2%).
cCatheter thrombosis and blood coagulation disorders.
Regimens were more frequently conform in the 173 patients treated for preventive indications (142 patients, 82.1%) than in the 135 patients treated for curative indications (41 patients, 21.0%). In the latter, 42 patients treated with tinzaparin were more often treated with the adequate regimen (19 patients, 45.2%) than the 93 other injectable anticoagulants (22 patients, 23.7%, P = .039).
In all, 12 patients were treated for isolated superficial vein thrombosis: 9 with LMWH and 3 with fondaparinux, with conform regimens for 3 patients treated with LMWH (25.0%). Thus, in 3 (25.0%) patients with isolated superficial vein thrombosis, parenteral anticoagulant therapy was classified compliant with consensus guidelines, and 0 (0.0%) compliant with SPCs.
Adverse Events
Nineteen adverse events were reported in 18 (5.4%) patients. In all, 17 were classified nonserious, including 16 bleedings (15 hematomas and 1 hematuria) and 1 thrombocytosis. The 2 serious adverse events (0.6%) were bleedings (hematuria and intracranial hemorrhage necessitating hospitalization), with no case of thrombocytopenia. Among the 19 adverse events reported, 6 parenteral anticoagulants prescription (33.3%) were compliant with SPCs and 8 (44.4%) were not compliant with SPCs. The 2 serious adverse events were not compliant with SPCs or consensus guidelines.
Discussion
Summary of Main Findings
Our study assessed daily use of parenteral anticoagulant therapy in general practice. We showed that parenteral anticoagulants were used in compliance with SPCs (indication and regimen) in only one-third of the patients and in compliance with consensus guidelines in around half of the patients. Weight of the patient was known in only 64% of cases and serum creatinine in 40% of patients. Parenteral anticoagulant regimens were compliant with SPCs and consensus guidelines in 59% patients. The rate of adverse events was 5.4%, with 0.6% of serious adverse events.
Meaning of the Study
Our study showed that parenteral anticoagulants are appropriately used in only one-third of the outpatients, mainly because of nonconform indication. Our study stressed out a great gap between SPCs and consensus guidelines, regarding compliance with indication. This can be explained mainly by consensus guidelines recommending prophylaxis of deep vein thrombosis after lower limb fracture/cast immobilization (French Society of Anesthesia and Intensive Care). 8 Another explanation is the use of LMWH/UH in atrial fibrillation/mechanical heart valve (probably for bridging with oral anticoagulants, although this precision was not specified in our form), validated by the European Society of Cardiology 17,18 and thus by the French Society of Cardiology. 9,10 The recommendation of deep vein thrombosis prophylaxis after cesarean surgery (French College of Obstetricians and Gynaecologists) 11 or after abdominal surgery (French College of Obstetricians and Gynaecologists and French Society of Anesthesia and Intensive Care) 8,11 had little impact on compliance rates.
Such discrepancies in the use of injectable anticoagulants reflect the conflict between stringent marketing authorizations and daily use of parenteral anticoagulants, for which there is no “one-size-fits-all.” Moreover, SPCs do not take into account risk assessment scores for venous thromboembolism (VTE) developed in the recent years, which have been showed effective in improving VTE prophylaxis with parenteral anticoagulants in primary care. 19 This phenomenon could confuse prescribers and have an impact on the safe prescribing of parenteral anticoagulation by GPs. Further studies are required to fully assess motivations for “off-label” use of parenteral anticoagulants.
Off note, almost one-third of the patients in the deep vein thrombosis indication were still waiting for a confirmatory examination. This empiric treatment has already been described in Germany. 20 In a descriptive study assessing diagnostic work-up for suspected deep vein thrombosis, heparins had been given to 15.5% of patients before referral to the specialist. Moreover, in a similar context of uncertainty of DVT diagnosis, a therapeutic dose of LWMH was assessed effective and safe to avoid urgent ultrasonographic. 21
General practitioners tended to prescribe more frequently LWMH for the treatment of superficial venous thrombosis. However, fondaparinux marketing authorization for superficial vein thrombosis of the legs was approved during the study period, consequently to the results of the Comparison of Arixtra in Lower Limb Superficial Vein Thrombosis with Placebo (CALISTO) study, that showed effectiveness of fondaparinux in the treatment of patients with acute, symptomatic superficial vein thrombosis of the legs. 22 It is uncertain how GP prescriptions may have since shifted to fondaparinux.
Comparison With Literature
The rate of compliance of regimens in our study (58%) is lower than in hospital settings (64.9%-82.7%). 14 Similar to in-hospital studies, rates of regimen compliance are higher in thrombosis prophylaxis than in curative indications. 14,15 However, the interpretation of regimen compliance should be cautious in patients with therapeutic indication, since dosages could have been adapted after results of biological monitoring (activated partial thromboplastin time for UHs or anti-Xa activity for LWMHs). Also, we chose a stringent range for compliance with dose (±0.05 mL, based on clinicians’ experience). We performed a post hoc sensitivity analysis showing that choosing a wider range (±0.1 mL) was associated with an increase in regimen compliance (eg, for patients with deep vein thrombosis: 36.5% to 50% of regimen compliance, Supplemental Table S3). The comparison between rates of compliance with regimen should be interpreted with caution, since we chose to include in the analysis patients with unavailable data, thus reflecting the reality of the daily practice of GPs. Restricting the study population to the 272 patients with sufficient data to assess compliance with regimen led to slight increases in global compliance rates (SPCs: 32.5% to 39.7% and guidelines: 47.9% to 51.0%, Supplemental Tables S4-S7). Weight was unknown in 3 of the 4 patients whom compliance with regimen could not be assessed. Yet, weight evaluation is essential to calculate the dosage, especially when curative dose is needed. This low rate could be explained whether by an unfilled variable in the form or more probably by unfilled weight in GP’s medical records. Indeed, weight filling in GPs’ medical records is known to be insufficient (eg, 62% for the UK primary care database The Health Improvement Network). 23 Similar concerns could be raised upon insufficient monitoring of serum creatinine.
The prevalence of adverse events related to parenteral anticoagulants therapy (5.4%) is also in range with previous studies (3.7%-5%). 14,15 Bleedings (0.6% of patients) remain the most frequent serious adverse events related to parenteral anticoagulants therapy. However, our sample size was insufficient to assess the association between compliance with SPCs/guidelines and adverse events related to parenteral anticoagulants.
Strengths
To our knowledge, this study is the first one assessing the use of parenteral anticoagulants in real-life primary care setting. Moreover, participation rate of residents was very high (94.6%), thus minimizing nonresponse bias.
Limitations
Using teaching doctors’ offices to include patients could have led to an overestimation of compliance rates. It has been shown that errors in drug prescriptions occurred more frequently in nonteaching GPs (odds ratio 1.39, 95% confidence interval: 0.97-2.01). 24 Moreover, the criterion of global compliance with SPCs was limited to indication and regimen. Duration of use is another criterion of SPCs but could not be assessed with the cross-sectional design of our study.
Conclusion
The use of parenteral anticoagulants complies with SPCs in only 1 of the 3 outpatients and with consensus guidelines in around half of the outpatients. Further studies are required to understand the reasons for such low rates of compliance with SPCs and guidelines and to correlate these rates with occurrences of serious ADRs in general practice.
Footnotes
Acknowledgments
The authors thank all the teaching general practitioners and residents who participated in the study.
Authors’ Note
This article has been presented at the 2013 Congrès Physiologie, Pharmacologie, et Thérapeutique (Angers, France, April 22-24, poster) and at the 2013 Congrès de la Médecine Générale (Nice, France, June 27, oral presentation).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
