Abstract
Background:
Patients hospitalized for first acute coronary syndrome (ACS) are frequently discharged on multiple new medications. The short-term tolerability of these medications is unknown.
Methods:
This single-center cohort study assessed 30-day health-care utilization and how it may be impacted by medication prescribing trends. We included Olmsted County patients presenting with ACS and previously undiagnosed coronary artery disease in 2008 to 2009. All health-care contacts were reviewed 30 days after index hospital discharge for potential adverse medication effects including documented hypotension or bradycardia, or symptoms likely attributed to the medications.
Results:
The study included 86 patients; their mean age was 63 (standard deviation: 15.5 years). Antianginal or antihypertensive cardiovascular (CV) medications were prescribed to 98% of patients at discharge; 76% were prescribed 2 or more. There were 233 health-care contacts in 30 days; 90 (39%) of these contacts were unscheduled. More CV medications tended to be prescribed to patients with unscheduled contacts, both pre-ACS (P = .045) and upon hospital discharge (P = .051). Hypotension and/or bradycardia at follow-up occurred in 52 patients (60%). Surprisingly, there was no association between hypotension and/or bradycardia at follow-up and increased health-care utilization (P = .12). Potential adverse drug effects were reported in 34 (40%) patients. These patients had significantly more total health-care contacts (P < .001) and unscheduled health-care contacts (median 0 vs 1.5; P < .001).
Conclusions:
Symptoms of adverse drug effects were associated with more frequent health-care utilization after ACS. Clinicians need to consider this while striving to increase patient compliance with post-ACS medications and optimize care transitions.
Introduction
Acute myocardial infarction (MI) treatment has advanced significantly in recent years. The American College of Cardiology/American Heart Association (ACC/AHA) 2014 Guideline for the management of patients with non–ST-elevation acute coronary syndromes (ACSs) include recommendations for initiation of at least 5 medication classes: 2 antiplatelet agents, HMG Co-A reductase inhibitors or other cholesterol-lowering agents, beta-blockers, and angiotensin-converting enzyme inhibitors or angiotensin receptor blockers. 1 The latter 2 lower blood pressure and can promote light-headedness. Beta-Blockers are associated with multiple other side effects. Patients with recent ACSs may be especially vulnerable to cardiovascular (CV) medication side effects and may be at risk for complications due to these side effects.
In post-ACS patients, clinicians often initiate and rapidly up titrate medications in the acute setting before patients can realize the full effects of these CV medications. The safety of this method has not been formally assessed. It is not surprising that such practices are raising concern among guideline authors, especially as the population on treatment ages. Recent hypertension guidelines issued the following warning: “caution is advised in initiating therapy with multiple agents, particularly in some older persons and in those at risk for orthostatic hypotension, such as diabetics with autonomic dysfunction. 2 (p1222) Greater than 50% of patients discontinue guideline-recommended therapy within 3 years of ACS. 3 The rationale for CV medication discontinuation is not entirely clear but adverse side effects may play a role.
Aggressive dosing of CV medications with antianginal or antihypertensive effects can lead to adverse effects such as symptomatic hypotension and bradycardia. Additionally, patients may experience frank syncope, fatigue, depressed mood, or light-headedness. These symptoms may result in increased health-care utilization. The frequency of potential CV medication induced side effects associated with guideline-recommended therapies and the sequelae of those symptoms have not been carefully studied to date. We believe that it is important to explore the incidence and prevalence of CV medication side effects in patients with recent ACS and to determine whether these side effects are associated with subsequent increased health-care utilization.
Therefore, we sought to determine adverse sequelae associated with the prescribing of antianginal or antihypertensive medications after hospitalization for ACS. We focused on 3 specific objectives: (1) establish the frequency of adverse drug effects in patients dismissed from the hospital after their first episode of ACS, (2) determine the clinical factors associated with adverse drug effects, (3) and evaluate the association of adverse drug effects with the utilization of health-care resources.
Methods
Study Sample
All consecutive adult patients (≥18 years) who were admitted to Mayo Clinic Saint Mary’s Hospital (Rochester, Minnesota) between January 4, 2008, and December 31, 2009, and discharged with a primary diagnosis of ACS as their first manifestation of coronary artery disease (CAD) were screened for enrollment. For the purposes of this study, a primary discharge diagnosis of ACS included all International Classification of Diseases, Ninth Revision codes 410-411. Only Olmsted County, Minnesota residents with scheduled follow-up at our institution were included to increase the likelihood that parameters being studied would be documented in the available medical record. Patients with a history of prior CAD, an index hospitalization greater than 10 days or including coronary artery bypass grafting (CABG), and whose MI occurred in the absence of atherosclerotic CAD were excluded (eg, profound gastrointestinal bleed resulting in type 2 MI). These exclusions were made to assess medication safety and side effects early after hospital discharge and limit confounding from cardiac surgery or other medical illnesses.
All clinical notes from the index hospitalization to 30 days after the index hospitalization discharge date were reviewed by the primary author. Applicable patient data were gathered retrospectively. All primary care and cardiology communications were reviewed including physician and nurse telephone calls, physician and nurse visits, emergency medical contacts, and hospitalizations. Initial visits to cardiac rehabilitation were noted but not included in analysis as additional health-care contacts. Contacts were defined as unscheduled if initiated by the patient rather than the health-care team. The primary end point of the analysis was the occurrence of unscheduled health-care contacts.
Patient Characteristics and Laboratory Measurements
We recorded the following baseline characteristics at the time of hospital admission: age, gender, race, and presence or absence of comorbid diseases including hypertension, hyperlipidemia, diabetes mellitus, and nicotine dependence. Blood pressure and heart rate were recorded at the time of hospital discharge and whenever present in the medical record over the 30-day period after hospital discharge. In the case of cardiac rehabilitation, emergency department visits, or hospital admission, only the first recorded vital signs were included. Hypotension was defined as systolic blood pressure <90 mm Hg or diastolic blood pressure <60 mm Hg. Bradycardia was defined as heart rate <60 bpm.
Cardiovascular Drug Therapy
Medication lists were reviewed upon index hospital admission, hospital discharge, and whenever included in follow-up documentation during the 30-day postdischarge study period. The use of CV medications in the following classes were documented: α-1 blockers, angiotensin-converting enzyme inhibitors (ACEIs), angiotensin receptor blockers (ARBs), beta-blockers, calcium channel blockers, diuretics, nitrates, clonidine, hydralazine, and minoxidil.
Symptom Assessment
Symptoms potentially related to CV medications were reviewed upon each occurrence in the medical record. The primary author reviewed all notes and determined potential adverse drug events based on clinical impression. Symptom review included the following key terms and their variations: weak, tired, fatigued, dizzy, light-headed, syncope, depressed, or lacking energy. Symptoms clearly attributed to a secondary cause were not included in this evaluation. Chart review included assessment for the following adverse events: oliguria, progressive azotemia, acute renal failure, ischemic colitis, ischemic hepatitis, falls resulting in medical care, syncope, and ischemic strokes clearly associated with hypotension or bradycardia.
Statistical Analysis
Statistical analyses were performed using SAS software (version 9.4; SAS Institute Inc, Cary, North Carolina). Descriptive statistics on patient and clinical characteristics were reported as mean (standard deviation [SD]) or median (interquartile range [IQR]) for continuous variables, as appropriate, and as mean and outer quartiles for discrete numeric variables representing counts. Differences between groups were determined by the t test and Cochran-Armitage trend test for continuous and ordinal variables, respectively, and χ2 test for categorical variables. For all analyses, a P < .05 was considered statistically significant.
The study was approved by the Mayo Clinic institutional review board and conducted with full compliance of the Declaration of Helsinki. We also conducted the research in compliance with the Minnesota Research Authorization and Privacy Laws.
Results
Baseline Clinical Characteristics
One hundred and sixty-five patients were screened based on Olmsted County residency and ICD-9 diagnosis code. Seventy-nine patients were excluded from analysis; 45 patients with follow-up scheduled elsewhere, 14 patients with known history of CAD, 11 patients with type 2 MI, 8 patients who had CABG during their hospitalization, and 1 patient whose hospitalization was 25 days in the absence of the above were excluded. The final study group included 86 patients.
Baseline clinical data are summarized in Table 1. The mean age of the study cohort was 63 (SD = 15.5) years. The majority of patients were male (59 patients, 69%). Caucasian patients represented 92% (79 patients) of the total cohort. Index events of non-ST-elevation (51%) and ST-elevation MI (49%) were evenly represented.
Study Population Baseline Clinical Data.a
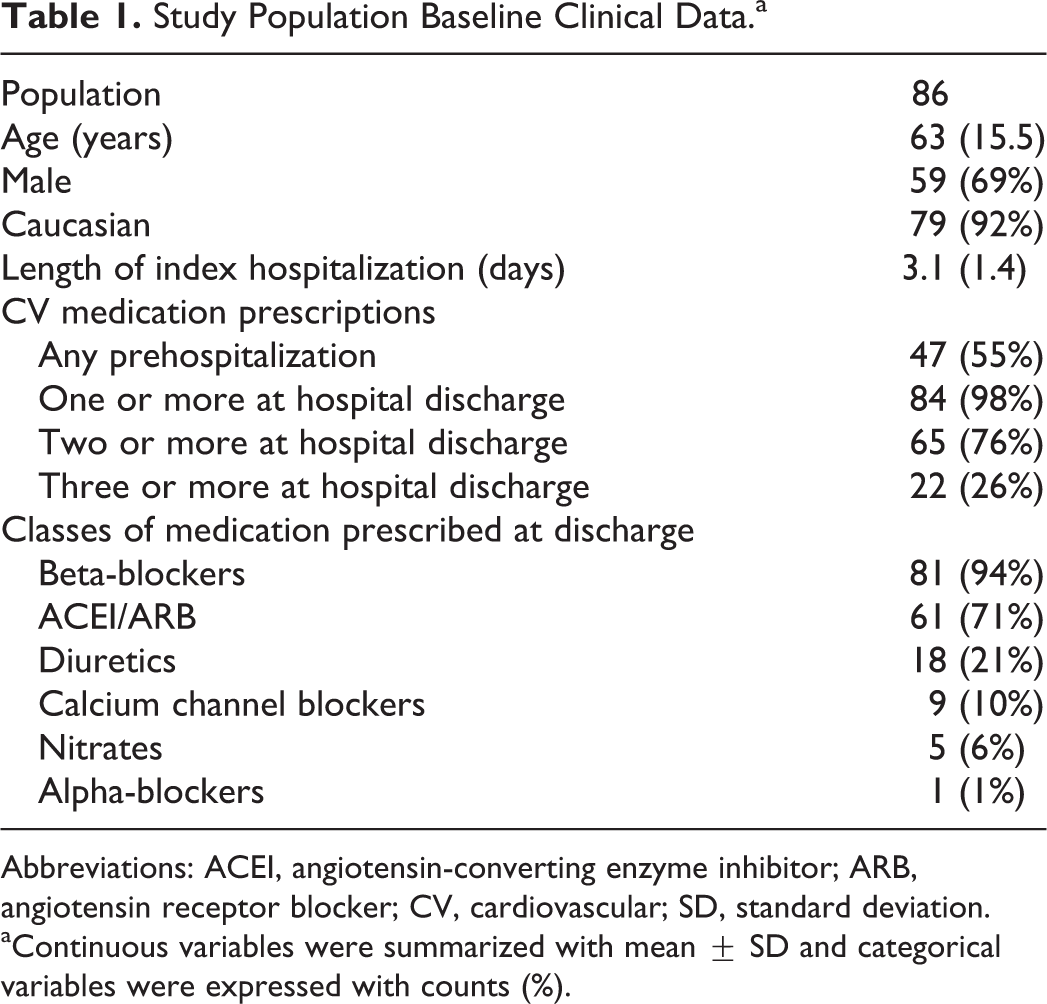
Abbreviations: ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; CV, cardiovascular; SD, standard deviation.
aContinuous variables were summarized with mean ± SD and categorical variables were expressed with counts (%).
Cardiovascular medications were prescribed to 47 patients (55%) at the time of hospitalization for their index event due to a comorbid diagnosis of hypertension. Three or more predefined CV medications were prescribed to 26% of patients, at hospital discharge. Table 1 details the prescribed CV medications. Among those patients prescribed beta-blockers, metoprolol tartrate was prescribed most commonly (62 patients). Atenolol was prescribed to 8 patients, metoprolol succinate to 8 patients, carvedilol to 2 patients, and labetalol to 1 patient. No patients were discharged from the hospital on clonidine, hydralazine, or minoxidil.
Symptomatic Potential Adverse CV Medication Events
Potentially adverse CV medication events occurred in 34 (40%) patients. These included fatigue/weakness in 17 patients, dizziness in 15 patients, depressed mood in 11 patients, and syncope in 1 patient. Patients with potential adverse drug events and those without were no different in regard to age (P = .8), gender (P = .88), or length of hospitalization (P = .29). Both groups received a similar number of prescriptions for CV medications at hospital discharge (P = .13). Patients who experienced potential adverse drug effects did not have significant differences in the number of CV medications prescribed (P = .13), nor did they have more frequently documented vital sign abnormalities at discharge (P = .59) or follow-up (P = .84) compared to patients not reporting symptoms.
Objective Evidence of Potential Adverse CV Medication Effects
Bradycardia and/or hypotension at follow-up were present in 52 patients (60%). Patients who were older (67.2, SD = 14.3 vs 56.5, SD 15.3; P = .001) or who had bradycardia and/or hypotension at hospital discharge (60% vs 29%; P = .006) were more likely to experience bradycardia and/or hypotension at follow-up. Patients prescribed an ACEI/ARB at dismissal were more likely to have documented bradycardia and/or hypotension at follow-up (67% vs 44%; P = .046). The occurrence of any symptomatic potential adverse drug effects was not significantly different in those with documented bradycardia and/or hypotension at follow-up (P = .84).
Thirty-Day Health-Care Utilization
Outpatient primary care or cardiac follow-up occurred within 30 days at our institution for 84 patients (98%). One patient did not attend the scheduled follow-up and a second patient’s first follow-up was scheduled more than 30 days after hospital dismissal. There were 233 total health-care contacts in the 30 days after hospital discharge from index events. Type and frequency of health-care contacts were widely distributed as outlined in Table 2.
Thirty-Day Health-Care Utilization.a

aContinuous variables were summarized with median (interquartile range) and categorical variables were expressed with counts (%).
Unscheduled health-care contacts accounted for 39% (90 contacts) of all contacts 30 days after index hospitalization. Forty patients (47%) had 1 or more unscheduled health-care contacts. There were a total of 16 emergency department visits among 14 patients and 14 hospital admissions among 12 patients. Chest pain evaluations accounted for 9 emergency department evaluations all of whom had associated hospital admissions. Rapid atrial fibrillation complicated by heart failure resulted in 2 emergency department evaluations and subsequent hospitalizations. Two hospital admissions occurred for planned staged revascularizations. One emergency department evaluation and subsequent admission was for prolonged loss of consciousness that was felt to be bradycardia mediated and resulted in intubation in the field. In this elderly gentleman, his beta-blocker was discontinued and he was discharged 3 days later without complication. Night terrors, vertigo, mechanical right shoulder pain, and dysuria were the final diagnoses in the remaining 4 emergency department evaluations.
Factors related to unscheduled health-care contacts
Baseline clinical characteristics of the patients stratified by the presence or absence of unscheduled health-care contacts are given in Table 3. There were no significant differences in baseline clinical variables including age, gender, or comorbid CV risk factors between these groups. The first scheduled outpatient follow-up was a median of 7 (5-11) days from the date of hospital discharge. There was no difference between patients with outpatient follow-up ≤7 days and >7 days from hospital discharge in terms of total health-care contacts (P = .35) or unscheduled contacts (P = .89) over the 30-day follow-up period.
Summary of Clinical Factors Associated With Unscheduled Health-Care Contacts.a
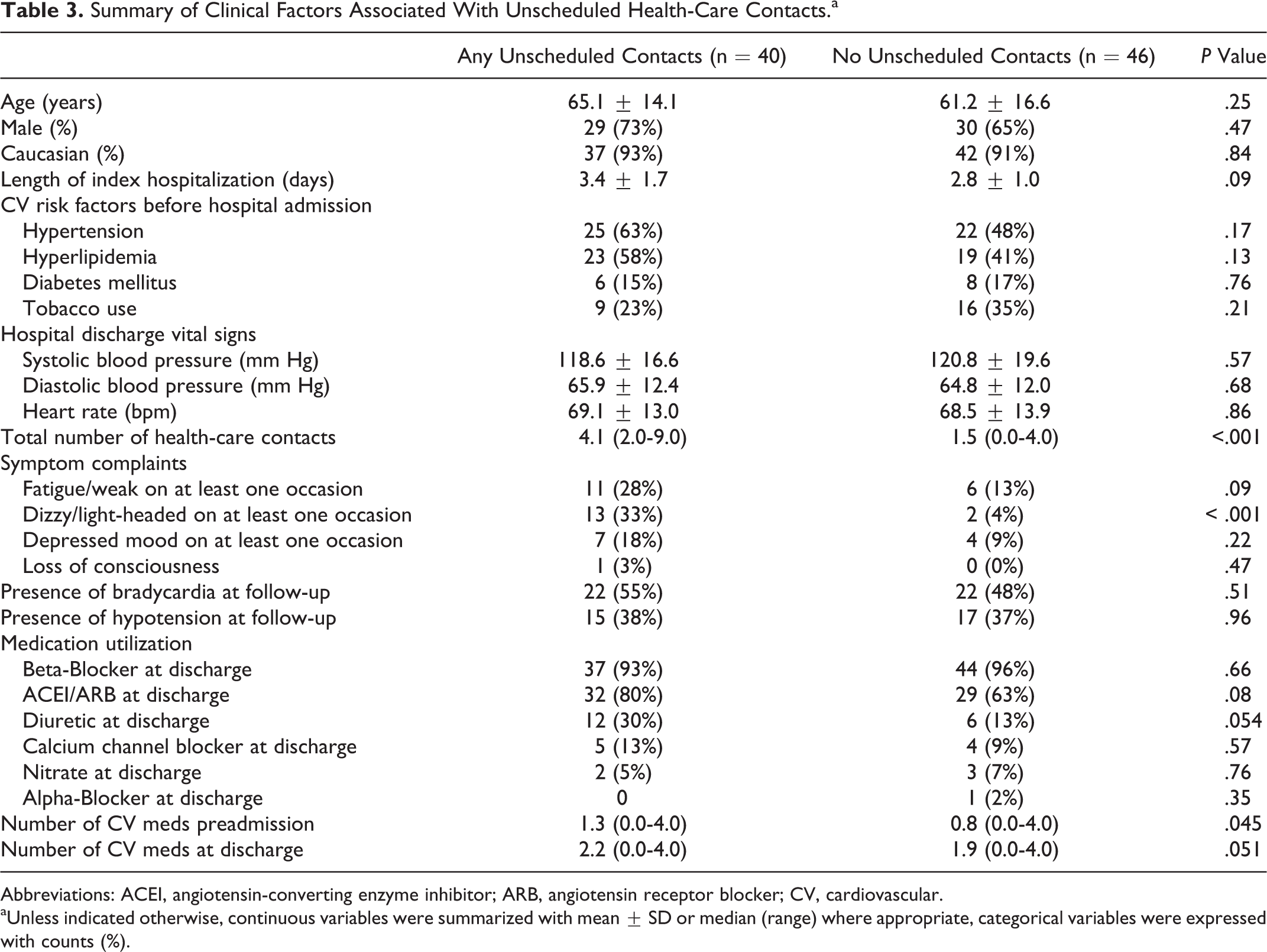
Abbreviations: ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; CV, cardiovascular.
aUnless indicated otherwise, continuous variables were summarized with mean ± SD or median (range) where appropriate, categorical variables were expressed with counts (%).
Patients who had symptom complaints had almost 2 times more total (3.9 [1-9] vs 2.0 [0-7]; P < .001) and 4 times more unscheduled health-care contacts (1.9 [0-7] vs 0.5 [0-4]; P < .001; Figure 1). Dizziness and light-headedness were more common in patients with unscheduled health-care contacts (P < .001). Patients who reported fatigue or weakness were more likely to experience an unscheduled health-care contact, although this result was not statistically significant (P = .09). Documented bradycardia and/or hypotension at follow-up was not more common in patients with unscheduled contacts (P = .21).

Box plots of relationship between total (A) health-care contact (P < .001) and unscheduled (B) health-care contact (P < .001) stratified by presence of symptom complaints. Boxes display the IQR with the horizontal bar inside the box demarcating the median, diamonds represent the mean, whiskers for the range of nonoutliers and outliers represented by circles. IQR indicates interquartile range.
Patients with any unscheduled health-care contacts were prescribed a marginally higher number of CV medications both pre-ACS (P = .045) and upon hospital discharge (P = .051). The majority of patients prescribed a beta-blocker were prescribed metoprolol tartrate (79%) at discharge with the median (IQR) dose being 25 mg (21.9-50) in patients who experienced unplanned health-care contacts and 25 mg (15.6-50) in patients who did not experience unplanned health-care contacts (P = .48 from Wilcoxon rank sum test). There was a trend toward increased frequency of ACEI/ARB prescribing in those with unscheduled contacts (80% vs 63%; P = .08).
Discussion
The present study demonstrates a high incidence of health-care contacts within 30 days after first ACS events. The frequency of health-care contacts was greater in patients with symptom complaints potentially attributed to guideline-directed medical therapy. This was particularly true for patients with orthostatic complaints. Patients experiencing potential CV medication side effects had almost 4 times more unscheduled health-care contacts (from a mean of 0.5-1.9 contacts).
It is certainly possible that medication side effects may have triggered some of these unscheduled contacts. It is also conceivable that less aggressive medication titration in the acute setting may mitigate these side effects and subsequent unscheduled health-care contacts. Future studies should assess whether changing patterns of medication initiation and titration during the hospitalization might limit the increased health-care utilization associated with CV medication side effects.
Previous reports suggest a rate of adverse medication events in the outpatient setting of 27%. 4 Our study documented higher potential adverse events which may reflect the addition of multiple therapies simultaneously. With new trial data supporting ever increasing blood pressure lowering goals, more attention to medication side effects may be required. 5
Our study shows minimal early objective risk from adverse medication effects with only 1 serious patient event occurring (1.2%). We did, however, observe a high rate of documented bradycardia and/or hypotension during follow-up (60%). A significantly higher proportion of patients prescribed ACEI/ARBs at discharge had bradycardia and/or hypotension at follow-up. Notably, these objective vital sign abnormalities were not associated with increased health-care utilization. This suggests that a patient’s symptoms are more important than an absolute heart rate or blood pressure threshold when assessing their tolerance of post-ACS medications. Defining patients at risk for adverse medication effects will be imperative to the chronic management of ACS survivors.
Symptom complaints consistent with adverse drug effects were present in 40% of patients. It is unknown whether such side effects result in higher rates of medication nonadherence. Melloni et al found persistence to all prescribed evidence-based medications was only 72% at 3 months after ACS. 6 The majority (62%) of patients had self-discontinued therapy. As the amount of evidence-based medical therapies for ACS increases, it will be important to assess patient-level risks associated with therapy. Side effects of otherwise beneficial medications may increase the risk of nonadherence effectively nullifying the potential for benefit. Our data show no association between patients with potential adverse CV medication effects and the number of CV medications prescribed or vital sign abnormalities at dismissal. This suggests that identifying patients at higher risk of adverse CV medication symptoms may be challenging. Future studies to allow prospective identification of patients at risk for adverse drug effects of guideline-directed therapy are necessary for optimal long-term management of patients with CAD.
Limitations
This was a single-center study representing a narrow spectrum of patients and care providers with a culture that may affect the frequency of health-care contact. The study excluded patients who did not plan for ongoing care at our institution which may select patients with more perceived “need” than the general population. Patients with more health-care contacts may have pursued those contacts for evaluation of symptoms likely explaining some of the association between these variables. We also restricted the study population to Olmsted County, Minnesota. This has limitations with regard to racial diversity and included patients largely insured with private or public health insurance. The uninsurance rate within Minnesota is significantly below the national average. We elected not to do multivariable modeling because the sample size was insufficient.
Physician assessment of symptom complaints is often poorly documented in sufficient detail to ascertain whether symptoms are definitely medication induced. Iatrogenic hypotension and/or bradycardia are often transient phenomena such that patients will have normal vital signs at the time of medical evaluation but still may have symptoms secondary to their medications. This may explain why our patients did not have a correlation between symptom complaints and measured hypotension and/or bradycardia. Unfortunately, standardized measures of hypotension/bradycardia with ambulatory blood pressure and Holter monitoring were not utilized in the patients studied herein. Further prospective assessments utilizing objective tools to ascertain adverse drug effects would help inform in this regard. It is also important that the electronic health records of the future make it easier to capture medication and treatment-related adverse events so that associations and causality may be more easily evaluated.
Conclusions
Our study demonstrates a high incidence of health-care contacts within 30 days after ACS in a largely insured and Caucasian population. Cardiovascular medications were prescribed in almost all (98%) post-ACS patients upon hospital discharge. Only 1 (1.2%) significant adverse event occurred. Despite this, patients with symptoms associated with their CV medication utilized significantly more health-care resources during follow-up. The interaction of medication prescribing, potential adverse effects, and frequent health-care contacts after ACS deserves ongoing attention in future studies.
Footnotes
Authors’ Note
All authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
Authorship Statement
R. Le contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; M. Cullen contributed to design, contributed to interpretation, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy; B. Lahr contributed to analysis, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; R. S. Wright contributed to design, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy; S. Kopecky contributed to design, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Mayo Clinic Rosenow Award. Dr Wright has received support from Sanofi-Regeneron, Pfizer, The Medicines Company, Boehringer Ingelheim/Lilly unrelated to the current work. Dr Kopecky has received personal fees from Applied Clinical Intelligence, Prime Therapeutics, Pfizer and Fusion Conferences; he has received nonfinancial support from Genzyme, Regeneron, and Amgen, and has received other support from the American Society for Preventive Cardiology, American Society for Men’s Health, Mayo Clinic Support Services Texas Non Profit Health Organization, and the Familial Hypercholesterolemia Foundation outside the submitted work.
