Abstract
Markers of platelet activity (P-selectin), fibrinolysis (
Introduction
Although platelet function measured in the peripheral blood in patients with mitral stenosis (MS) has been studied in only few reports, it was suggested that MS has been associated with platelet hyperaggregatory state 1 that improves after percutaneous mitral valvotomy (PMV). 2 –4
The mechanism of reduced platelet activity by PMV remains unclear with nonconsistent results regarding the relation to the hemodynamic and echocardiographic factors such as increased mitral valve area (MVA), left atrial area, left atrial pressure, and pulmonary artery systolic pressure (PASP) after PMV. 3,5,6
Several reports have demonstrated elevated inflammatory markers in patients with MS having evidence of decreased levels of some these markers after PMV. 7 –9 The cause of change in these markers by PMV is not clear. A relationship between high-sensitivity C-reactive protein as a marker of persistent inflammation and the PMV procedural success rate has been reported. 10 The relationship between these inflammatory and hemostatic changes was not studied before.
This study aimed to study the changes in platelet, thrombin, and fibrinolytic activities as well as interleukin 1β(IL-1β) as an inflammatory marker after PMV and to find the relationship between the change in each of these hemostatic parameters and the changes in echocardiographic parameters (MVA, left atrial diameter [LAD], transmitral pressure gradient, PASP, left ventricular dimensions, and ejection fraction) as well as systemic inflammation measured by IL-1β.
Patients and Methods
Sixty-five patients with symptomatic MS and sinus rhythm undergoing PMV were included. Patients were excluded if they had atrial fibrillation, MVA >1.5 cm2, mitral regurgitation > grade II, left atrial thrombus, interatrial septum thickness > 4 mm, a previous thromboembolic event, associated other valvular lesions needing surgery, associated significant coronary artery disease, acute rheumatic activity in the last year, infective endocarditis, and patients not giving a written consent to the procedure. All patients were not on any anticoagulant or antiplatelet therapy 10 days before the procedure and during the follow-up period. This was a prospective study approved by our faculty ethical committee and was adherent to the regulations of the Declaration of Helsinki. Clinical data were collected in a personal interview 24 hours before the PMV procedure, just before doing the prepocedural echocardiography.
Echocardiographic Evaluation
Transthoracic echocardiography was done in the routine way, 24 hours before and 2 weeks after PMV using a Hewlett Packard HP Sonos 4500 cardiac – vascular ultrasonographic machine with a phased array multifrequency transducer 2 to 4 MHz. The standard echocardiographic measurements were done and averaged in 4 cardiac cycles. These measurements were taken while the patient was on supine and left lateral decubitus positions. Two-dimensional examination was done to assess Wilkins score, 11 MVA (planimetry) measured in parasternal short-axis view. 12 Continuous wave Doppler measurements were taken at a speed of 100 mm/sec. The values from 5 consecutive beats were averaged to measure transmitral pressure gradient using Bernoulli equation 13 and PASP from tricuspid regurgitation. 14 Color Doppler was done to assess the degree of mitral regurgitation, which was graded, qualitatively, on a scale of 1+ to 4+ with the use of color flow mapping according to the jet extension in the left atrium. 15 Transoesophageal echocardiography was done 24 hours before PMV to exclude left atrial appendage thrombus and measure interatrial septal thickness. Patients were examined in left lateral position after being sedated with intravenous diazepam using a T6210 Omniplane II TEE Transducer 4-7 MHz.
Valvuloplasty Procedure
Percutaneous mitral valvotomy was done using either Inoue balloon technique 16 or the Multitrack system. 17
Laboratory Investigations
Venous samples were obtained from all patients immediately before and 2 weeks after PMV. For each sample, 4 mL of venous blood was collected into a sterile syringe and immediately transferred to an evacuated 5 mL polyethylene terephthalate tube containing 3.8% sodium citrate (Vacuette, Greiner, Germany). Blood samples were centrifuged within 2 hours of collection at 2500g and 4°C for 10 minutes, and the collected plasma was then stored at −70°C. All plasma samples were assigned a numerical code and assayed without knowledge of their time of collection (before or after PMV). All samples were measured within the same assay run. Peripheral blood samples were collected and managed from age- and sex-matched 23 healthy controls using the same method as used for the patients’ group. The control group was recruited from healthy volunteer members of the hospital staff and healthy persons presenting to our hospital for routine laboratory checkup.
Standard enzyme-linked immunosorbant assay (ELISA) technique was used to measure plasma concentrations of plasma soluble P-selectin (R&D Systems, Inc, Minneapolis, Minneapolis), Prothrombin fragments 1, 2 (PF1,2; Enzygnost F1-2; Behringwerke AG, Marburg, Germany), thrombin–antithrombin III complex “TAT” (Diagnostica Stago, Asnieres sur Seine, France),
Statistical Analysis
Clinical and echocardiographic variables were prospectively collected to be analyzed using SPSS 21 statistical software. Categorical data were represented as number (%). Continuous variables are expressed as mean (standard deviation) or as median (25th to 75th percentile range), if otherwise indicated. Variables, before and after the procedure, were compared using paired t-test or Wilcoxon test for nonparametric data when appropriate. Unpaired t-test or Mann-Whitney U test was used for comparison with the control. Linear regression analysis was used to study the predictors of the change in the different hemostatic and inflammatory markers. A P value of <.05 was considered significant.
Results
Patients included in this study were relatively young 26 ± 8 years; 32% were males, with a mean Wilkin score 6.9 ± 1.1. The patients’ baseline characteristics are shown in Table 1. The control group was matching with the patients’ group regarding age (mean 25 ± 7; range 13-43 years) and sex (7 of 23; 30% males). As shown in Table 2, PMV produced significant increase in MVA, LAD, and left ventricular end-diastolic diameter. Also, there was a significant decrease in right ventricular diameter, left ventricular end-systolic diameter, mean transmitral pressure gradient, and PASP.
Baseline Patients’ Characteristics.a
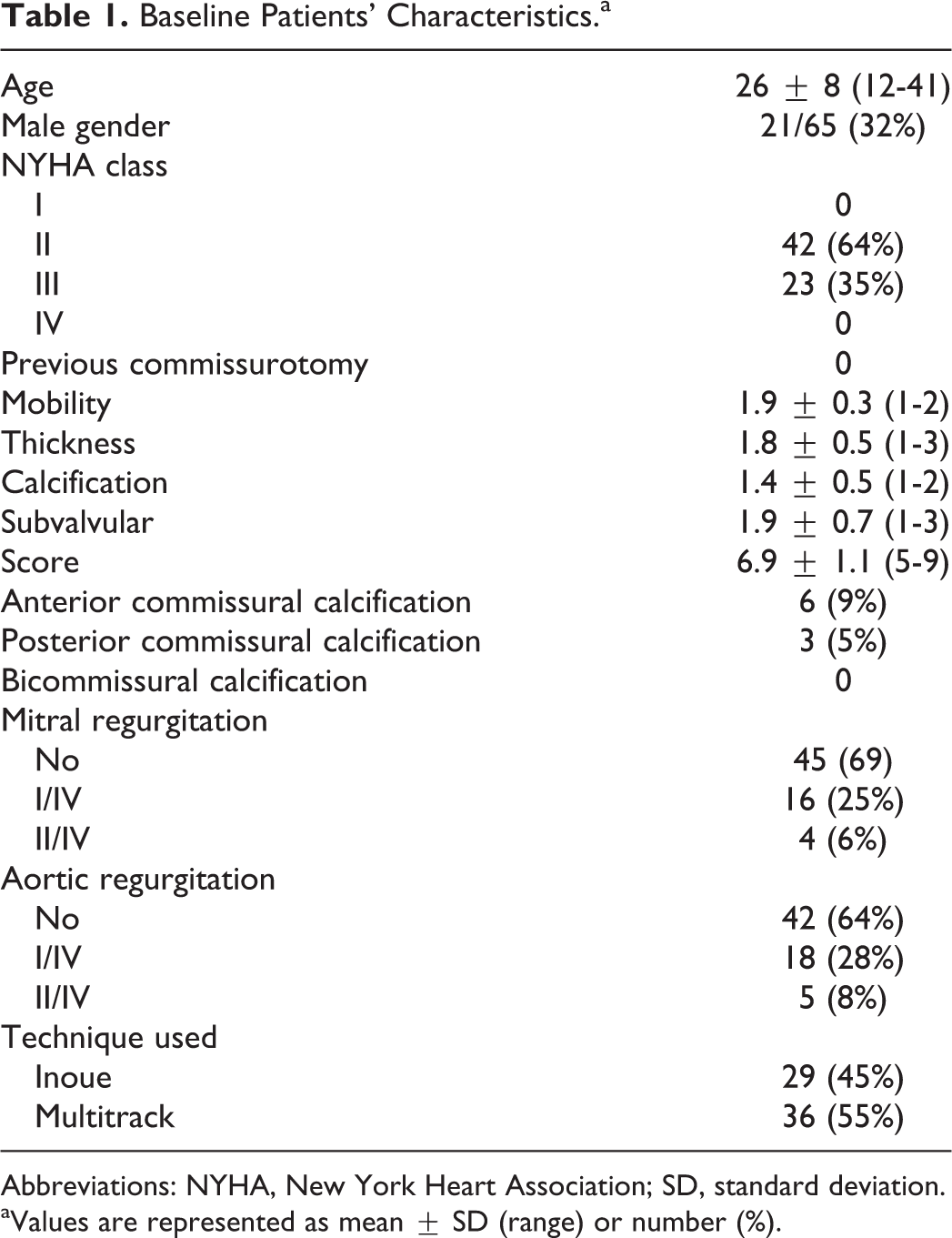
Abbreviations: NYHA, New York Heart Association; SD, standard deviation.
aValues are represented as mean ± SD (range) or number (%).
Echocardiographic Parameters Before and After PMV.a
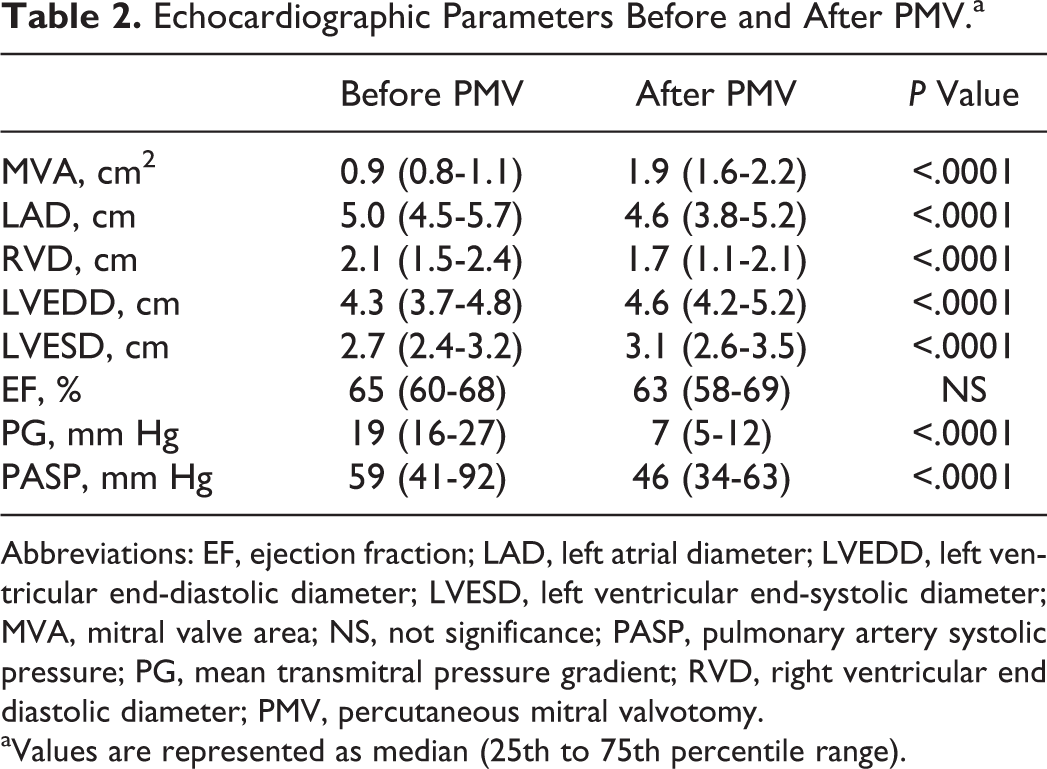
Abbreviations: EF, ejection fraction; LAD, left atrial diameter; LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; MVA, mitral valve area; NS, not significance; PASP, pulmonary artery systolic pressure; PG, mean transmitral pressure gradient; RVD, right ventricular end diastolic diameter; PMV, percutaneous mitral valvotomy.
aValues are represented as median (25th to 75th percentile range).
Changes in Hemostatic and Inflammatory Markers
As shown in Table 3, all markers were significantly higher than the control and significantly decreased after PMV. Using linear regression analysis, it was found that the change in any of the studied hemostatic variables was not predicted by the change in MVA (Figure 1), mean transmitral pressure gradient (Figure 2), left ventricular dimensions, and function. Interestingly, among the echocardiographic variables, LAD was the most strongly correlated with these hemostatic markers (Figure 3). The PASP also showed significant correlations with these hemostatic variables (Figure 4). The IL-1β as an inflammatory marker showed the strongest correlations with the studied hemostatic variables, even better than the echocardiographic variables (Figure 5).
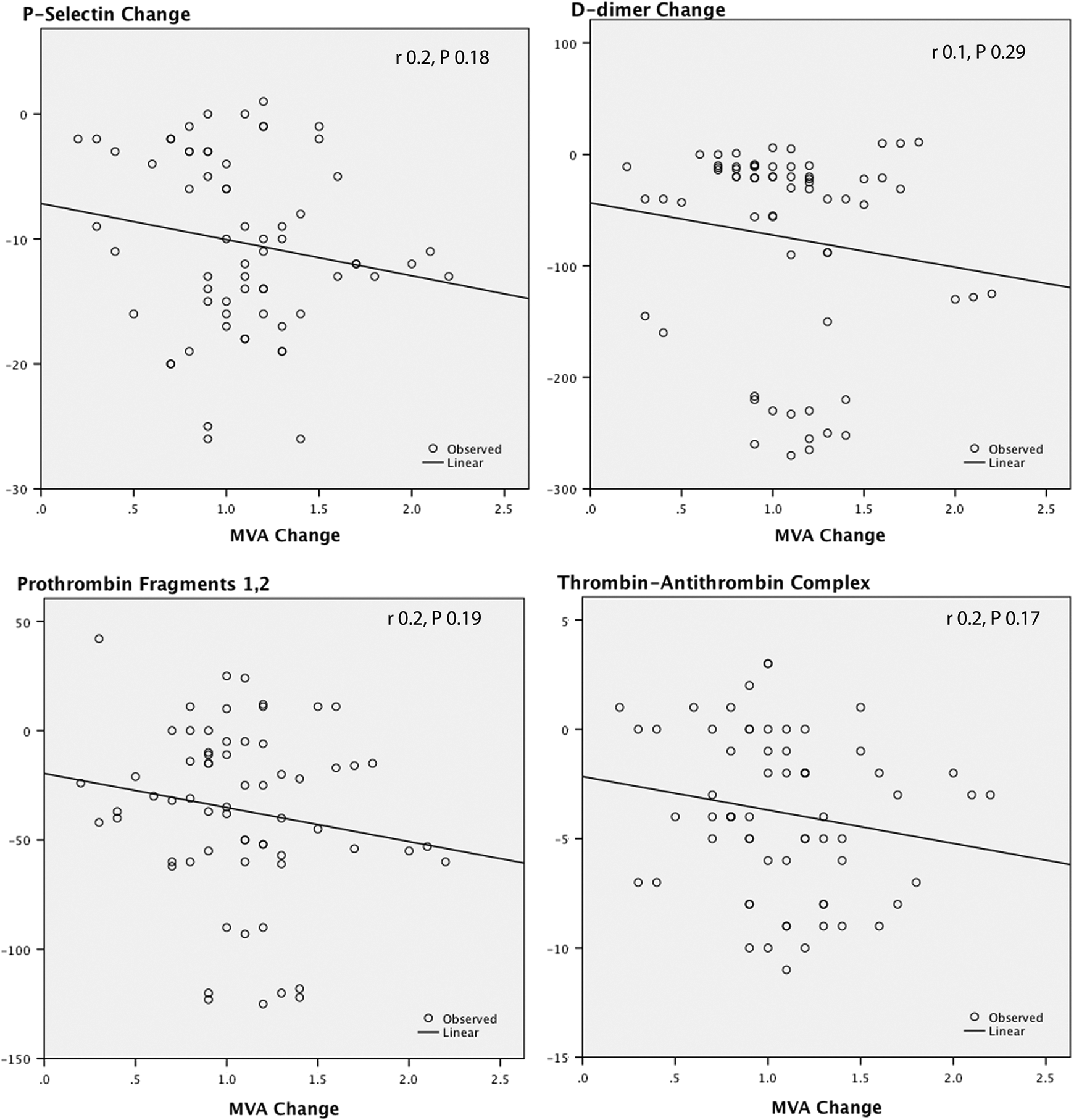
Correlations between the change in mitral valve area (MVA) and the changes in P-selectin,
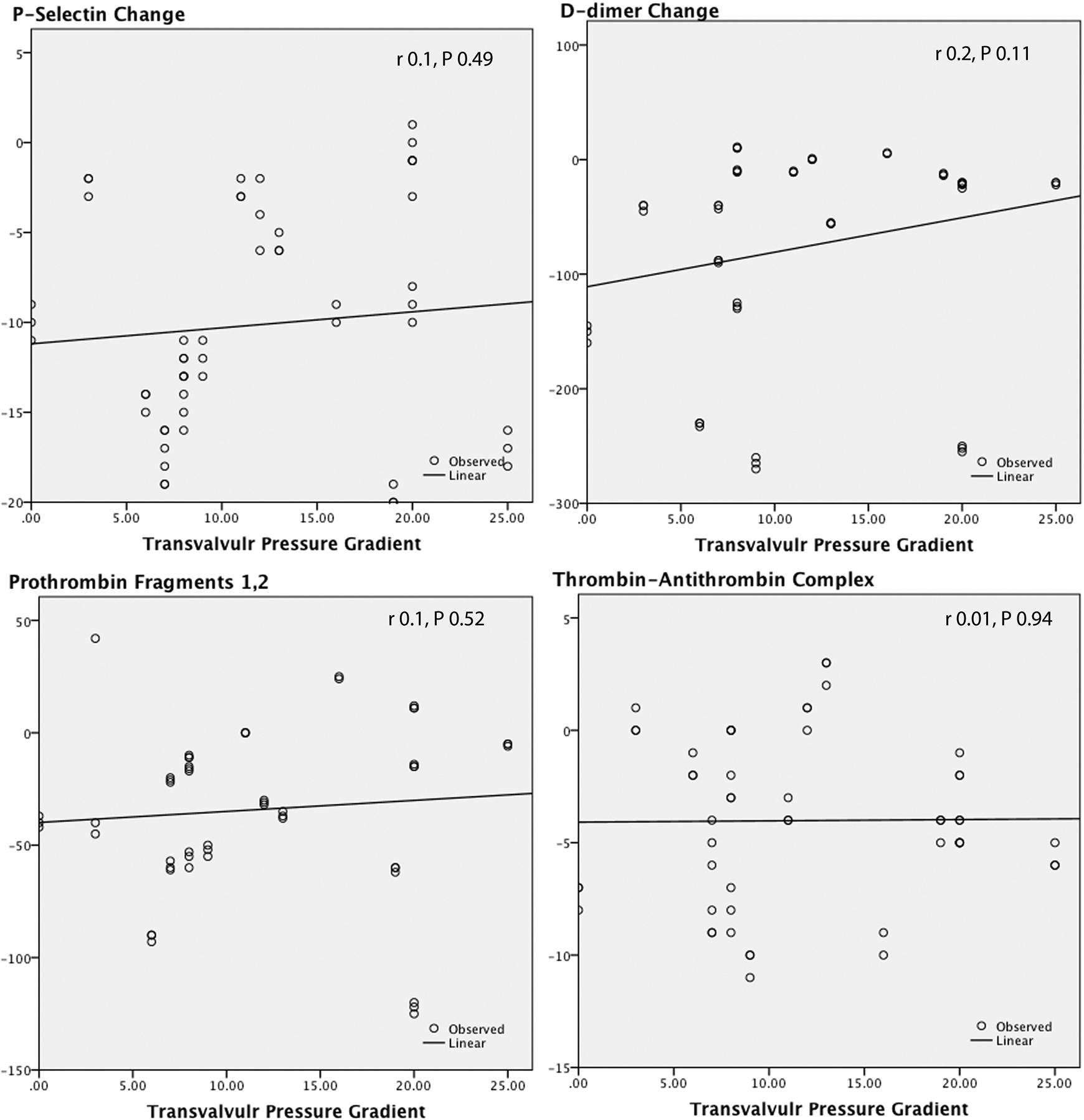
Correlations between the change in mean transmitral pressure gradient and the changes in P-selectin,
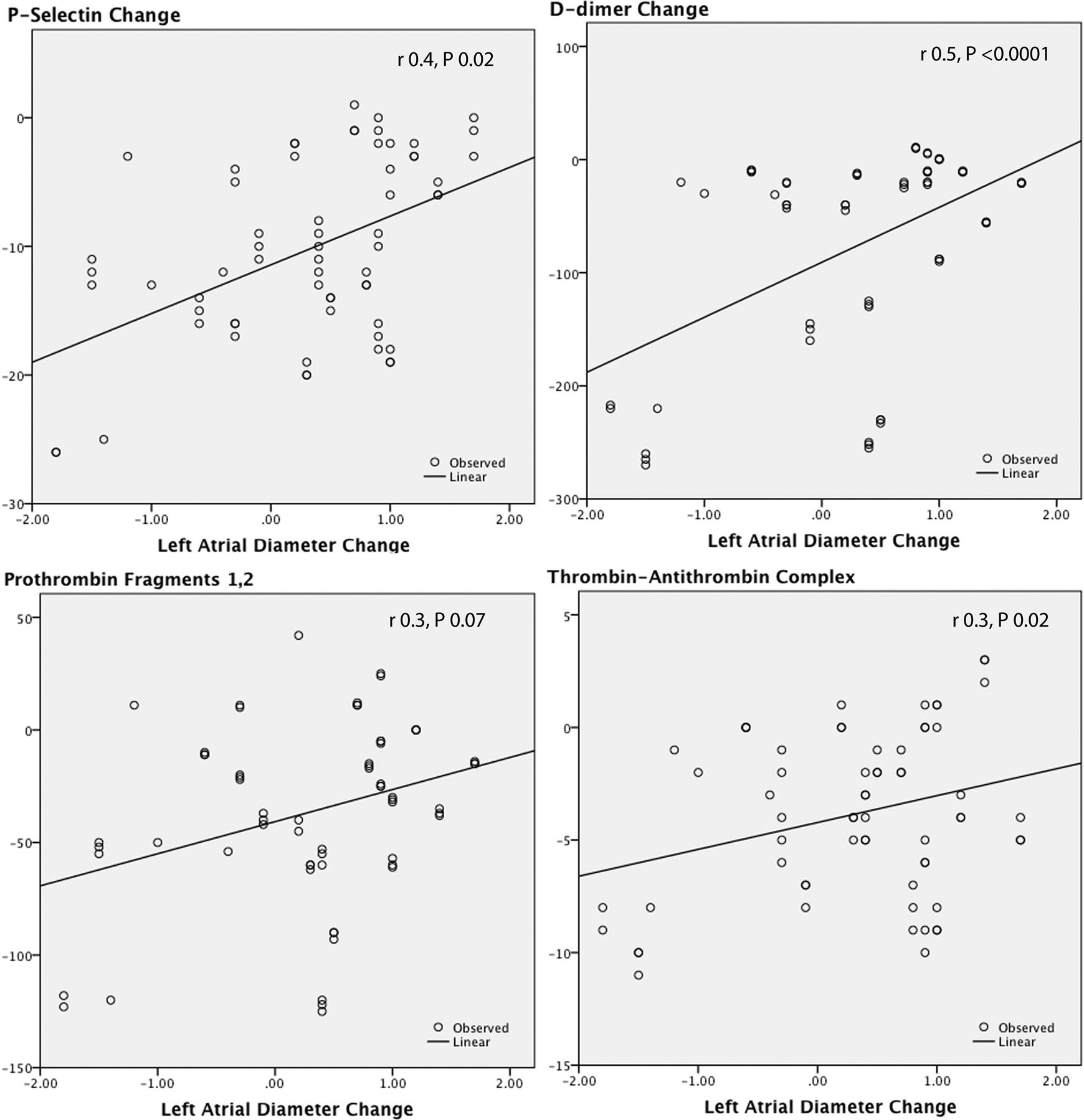
Correlations between the change in left atrial diameter and the changes in P-selectin,
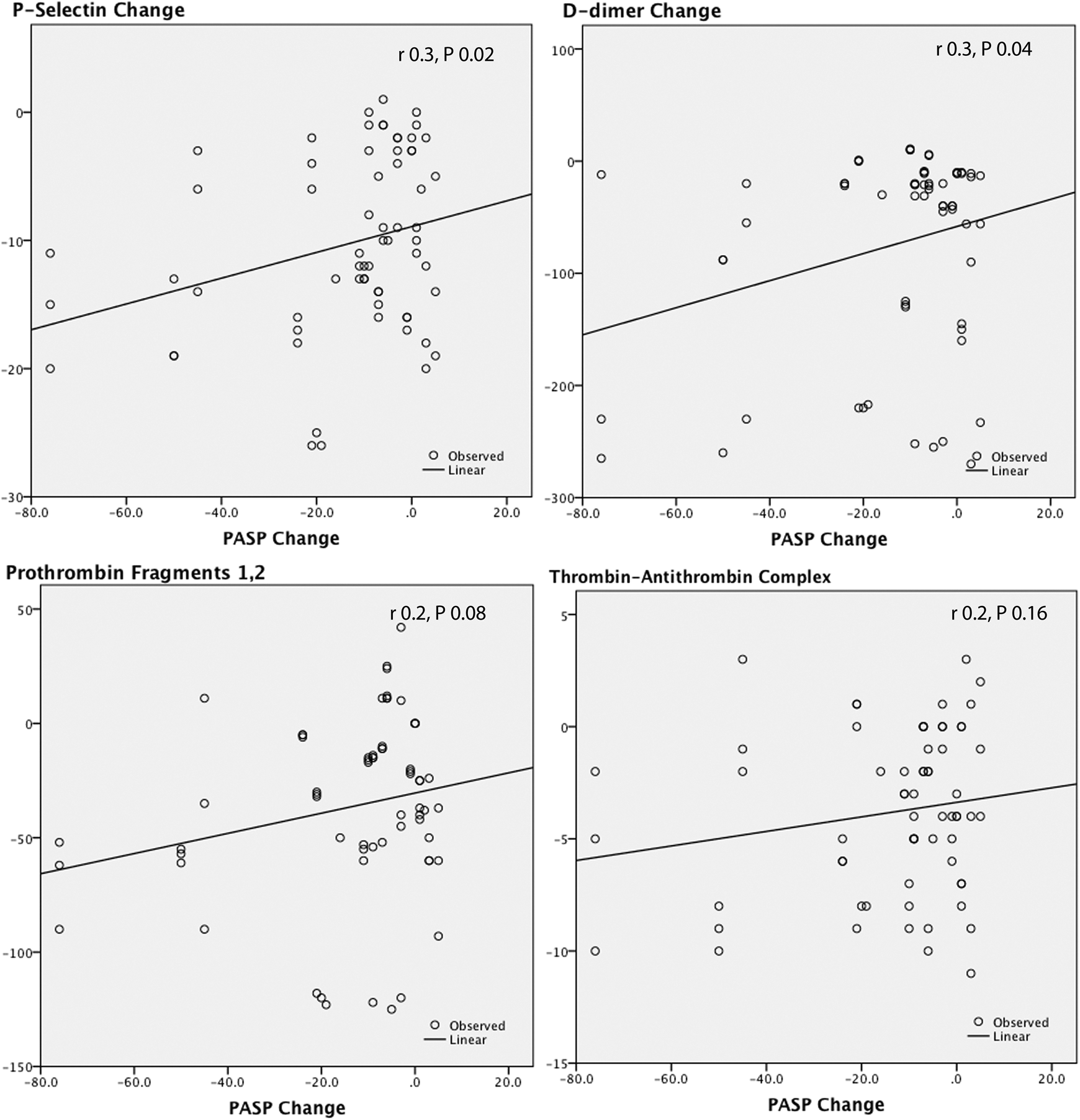
Correlations between the change in pulmonary artery systolic pressure (PASP) and the changes in P-selectin,
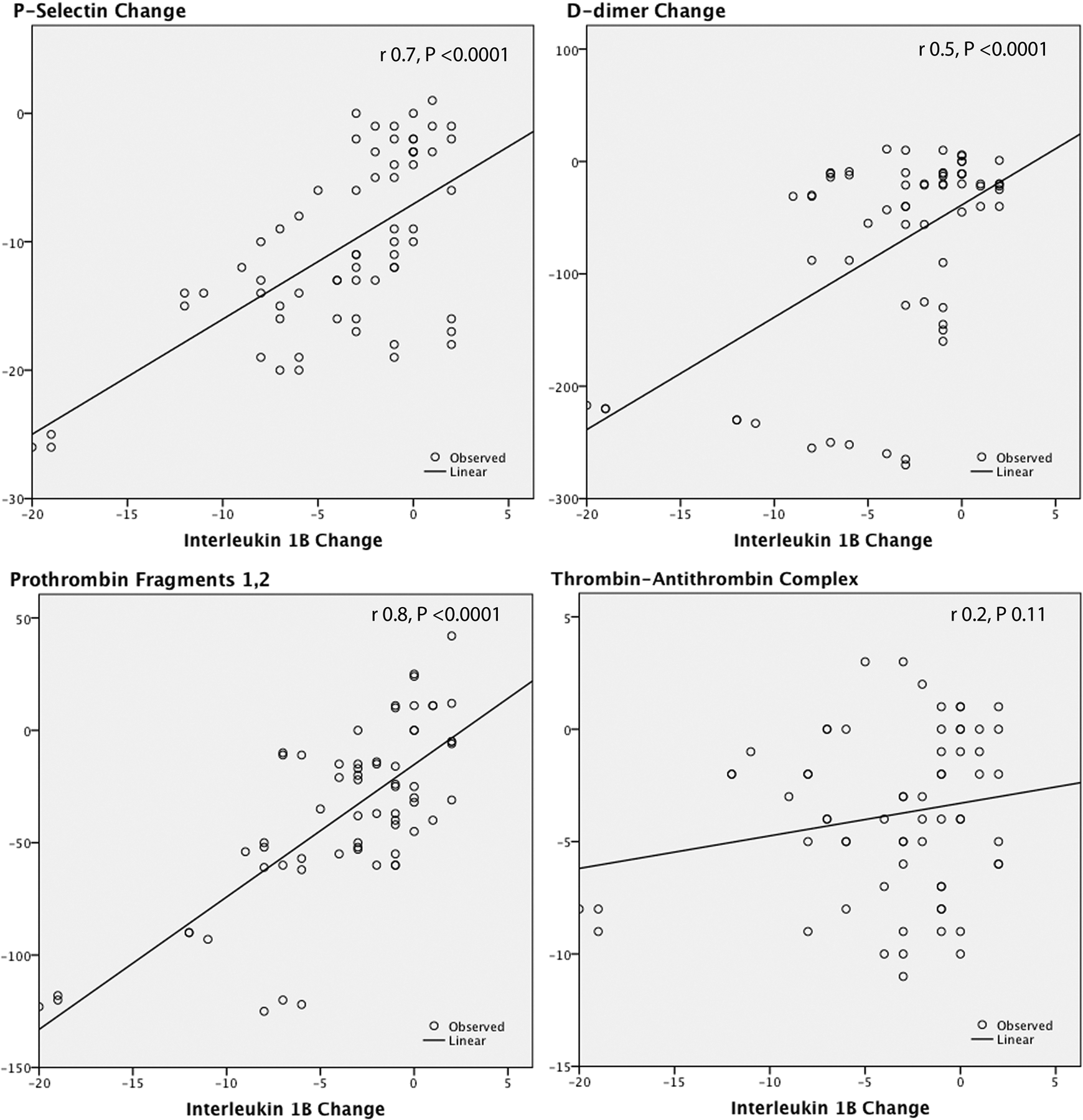
Correlations between the change in interleukin 1β and the changes in P-selectin,
Hemostatic and Inflammatory Variables Before and After PMV.a
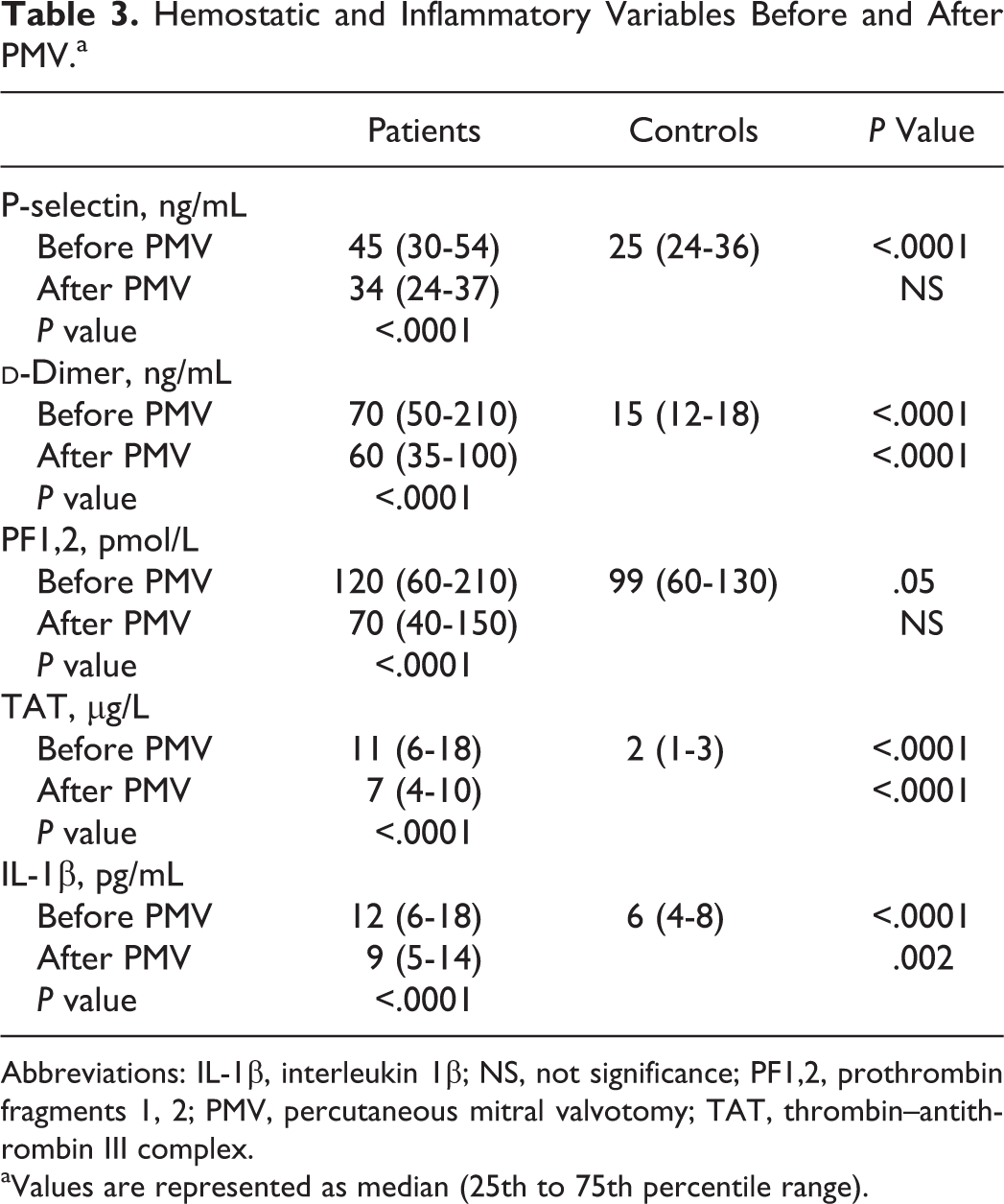
Abbreviations: IL-1β, interleukin 1β; NS, not significance; PF1,2, prothrombin fragments 1, 2; PMV, percutaneous mitral valvotomy; TAT, thrombin–antithrombin III complex.
aValues are represented as median (25th to 75th percentile range).
P-selectin change was predicted by the changes in LAD (r = .4, P .02), PASP (r = .3, P .02), and IL-1β (r = .7, P < .0001).
Discussion
This study suggests that MS induces a heightened inflammatory condition as well as a prothrombotic state irrespective of atrial fibrillation. We found that patients with MS had increased platelet activity (P-selectin), thrombin activity (TAT, PF1,2), fibrinolytic activity (
It has been reported that platelet activity is increased in peripheral blood 1,5,18 as well as centrally 6 in patients with rheumatic MS, with a significant decrease immediately, 6 4 weeks, 3 and 2 to 3 months after PMV. 5
These studies had major limitations. All of them had much smaller number of patients. The effect of heparin used on platelet activity in some of these studies 3,6 was not ruled out. Also, the sampling method used before the procedure was different from that used after. 3 In addition, these studies either measured the secretory substances of platelets in plasma (platelet factor 4 and β-thromboglobulin), 5,6 which are rather crude methods with many limitations, 19 or measured surface P-selectin (CD 62p antigen), 3 which is a biologically active adhesion molecule that is released to the platelet surface from α-granules on activation. 3 Surface P-selectin is expressed on unstimulated platelet surface and was measured using flow cytometry that doesn’t necessarily reflect changes in platelet physiologic function. 3
In this study, we assessed soluble P-selectin by ELISA, which better reflects platelet activity and physiologic function. 20 Also, samples were taken before and 2 weeks after PMV with complete absence of the possible effect of heparin used during the procedure. Sampling method was the same before and after the procedure.
In contrast to our data Chen et al reported in a small patient series (19 patients) that the reduction in platelet activity was associated with increased MVA instead of hemodynamic and echocardiographic factors such as left atrial area, left atrial pressure, and PASP after PMV.
3
This difference can be explained in part by the methodological differences detailed in the above-mentioned limitations. Also, the change in MVA does not necessarily reflect the actual hemodynamic improvement because subvalvular obstruction may be another variable with a remarkable effect on transmitral flow in some patients even in the presence of a large MVA.
21,22
Platelet activation with consequent high prothrombotic condition in patients with MS is a complex multifactorial process with different complementary mechanisms. Shear stress with turbulent flow as a result of MS itself is an important factor.
23
Another factor is stasis of blood in the left atrium and pulmonary circulation. High left atrial pressure
6
especially when accompanied with high levels of endothelin is another important factor.
24
Also, a significant hypercoagulable state in the form of increase in
It is an intriguing question whether such MS-associated prothrombotic state is derived from atrial dilation (stasis) or stenosed valve (high shear). The strong association of hemostatic markers with LAD but not MVA or PG in our data may suggest the former possibility.
Yetkin and colleagues demonstrated increased levels of circulating adhesion molecules (intracellular adhesion molecule, vascular cell adhesion molecule, E-selectin) in patients with MS stenosis before PMV. They assumed this to either increased shear stress and/or smoldering immunological activity in these patients. 8 Changes in the levels of circulating soluble adhesion molecules has been reported after PMV with a decreased E-selectin 24 hours after the procedure 8 and vascular cell adhesion molecule 1 one week after. 7 The relationship between the inflammatory markers and the hemostatic changes has not been studied before.
In our study, IL-1β as an inflammatory marker was found to be higher in patients with MS before PMV compared to the control with a significant reduction after the procedure. The change in IL-1β strongly correlated with the changes in platelet activity (P-selectin), thrombin activity (PF1,2) and fibrinolysis (
The relationship between inflammatory condition and hemostasis is a complex process. It is suggested from the current available data that inflammation can induce platelet activation with subsequent thrombosis and vice versa. P-selectin expressed by activated endothelial cells as well as by activated platelets has been shown to be implicated in the rolling of leukocytes at the sites of inflammation. Platelet–leukocyte aggregates were reported to play a critical role in inflammation present in vascular diseases. 26 Leukocytes binding to the activated platelets have shown increased levels of proinflammatory mediators such as IL-1β. 27 Moreover, activated platelets, via P-selectin, can induce monocytes to express tissue factor and increase the hypercoagulability 28,29 Also, platelet P-selectin with Regulated upon activation normal T cell expressed and presumably secreted (RANTES; a family of chemokines) can significantly increase cytokine levels in monocytes. 30 Also, it was found that increased levels of circulating microparticles resulting from apoptotic platelets, monocytes, and endothelium are associated with quite a large number of diseases implicating vascular, inflammatory, and/or coagulation problem. 31
In conclusion, MS is associated with heightened inflammatory, platelet, thrombin, and fibrinolytic activities that decrease after PMV. Altered hemodynamics play a possible role in these changes. Reduced inflammatory activity might have a role in reduced platelet, thrombin, and fibrinolytic activities after PMV.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
