Abstract
Statins are mainstream therapy in the treatment and prevention of cardiovascular disease through inhibitory effects on cholesterol synthesis. However, statins’ beneficial effects in cardiovascular disease may also be attributable to their role as anti-inflammatory mediators. Here, we investigated the effects of simvastatin treatment on expression levels of interleukin (IL) 1β in both patient with hyperlipidemia and healthy human peripheral blood mononuclear cells (PBMCs) using cholesterol crystals (CC), a cardiovascular pathogenic stimulus for activation of the NOD-like receptor pyrin domain–containing protein 3 (NLRP3) inflammasome. Cholesterol crystal-induced NLRP3 inflammasome activation was used to trigger maturation and release of IL-1β in PBMCs. Specifically, isolated PBMCs from patients with hyperlipidemia at baseline and following 8 weeks of in vivo treatment with simvastatin (10-20 mg) daily were stimulated with lipopolysaccharide (LPS; 100 ng/mL) for 3 hours to induce proIL-Iβ expression followed by CC (2 mg/mL) stimulation for further 18 hours to activate the NLRP3 inflammasome complex to induce maturation/activation of IL-1β. Peripheral blood mononuclear cells were also isolated from healthy donors and stimulated in vitro with simvastatin (50, 25, 5, and 2 µmol/L) prior to stimulation with LPS and CC as described above. The effects of simvastatin treatment on levels of IL-1β expression were determined by enzyme-linked immunosorbent assay and western blot. Both in vitro and in vivo treatments with simvastatin led to a significant reduction in the levels of expression of IL-1β in response to stimulation with CC. Simvastatin inhibits the expression and activation of IL-1β induced by CC in PBMCs, which may contribute to its protective role in patients with cardiovascular disease.
Introduction
Atherosclerosis is a progressive inflammatory disease of lipid accumulation in the arterial wall, which can result in vessel occlusion from plaque rupture and the formation of thrombosis. These thrombotic events can give rise to the clinical presentation of myocardial infarction (MI) or stroke. For many decades, it was thought that atherosclerosis was, at its core, a disease of progressive cholesterol deposition and storage, characterized by the presence of macrophage foam cells. 1 As such, many lipid-lowering drugs were developed in order to treat the disease, with statins leading the way as the therapeutic of choice today. 2 However, it has since emerged that atherosclerosis is also a disease of inflammation and not merely one of simple lipid storage. 3,4 Interleukin 1β (IL-1β), a proinflammatory cytokine derived mainly from monocytes, has emerged as having a key role to play in the initiation and progression of atherosclerotic inflammatory processes. 5 –9 Production of mature active IL-1β is controlled through the formation and activation of the NOD-like receptor pyrin domain–containing protein 3 (NLRP3) inflammasome, which involves active caspase 1. The inflammasome has been linked to various inflammatory diseases, including atherosclerosis. 10,11 Crystalline substances such as cholesterol crystals (CC) are capable of activating the inflammasome and subsequently IL-1β mediated inflammation in human cells. 12,13 These studies and others led to the design of the Canakinumab Anti-inflammatory Thrombosis Outcome Study (CANTOS) trial to test the efficacy of canakinumab, a fully human monoclonal antibody targeting IL-1β, in reducing secondary MI events in patients. The results for this trial have shown for the first time that modulating inflammation in atherosclerotic disease has a positive impact by reducing secondary cardiovascular events in patients. 14
Statins, which are 3-hydroxy-3-methylglutaryl-coenzyme A reductase inhibitors, exert beneficial antiatherosclerotic effects due to their ability to reduce cholesterol biosynthesis. 15 Large clinical trials served to provide comprehensive evidence that statins had a clear beneficial effect in reducing mortality in patients with atherosclerosis and also in reducing rates of nonfatal vascular events. 16,17 In several cases, these trials had unanticipated results; they indicated that there was a greater level of cardiovascular benefit obtained from statin therapy than expected. It was noted that, in fact, this degree of benefit did not entirely correlate with the degree of cholesterol lowering observed. 18 Coupling this observation with the fact that atherosclerosis is recognized as an inflammatory disease, 19 studies began to emerge detailing the effects of statins on reducing inflammation independent of effects on serum cholesterol level.
It is increasingly clear that statins exert their anti-inflammatory effects in a multifaceted manner. 20,21 Potentially, some of the reduction in inflammation arises from the reduction in low-density lipoprotein (LDL) cholesterol levels due to the inhibition of the mevalonate pathway. However, there is a large body of evidence supporting the hypothesis that statins can exert direct anti-inflammatory effects on the range of cells that are involved in atherosclerotic plaque development and rupture. 22 What is less clear is whether statins can affect the expression and activation of IL-1β in the context of cardiovascular disease. On the one hand, it has been reported that statins have the ability to downregulate the NLRP3 inflammasome, cathepsin B, and their downstream mediators like IL-1β, components that play a key role in the inflammation driving atherosclerosis. 23 In contrast, several reports have also indicated that statins can act to enhance IL-1β expression in response to lipopolysaccharide (LPS) in the absence of any NLRP3 stimulus. 24,25 However, such conditions may also result in deactivation of mature IL-1β through undetermined mechanisms of alternative cleavage. 26 Statins have also been shown to reduce the activity of transcription factors such as nuclear factor κB and activator protein 1, which are implicated in the regulation of a wide range of atherosclerotic inflammatory pathways 27 and are also subject to regulation by IL-1β. 7
Coupling this information with the knowledge that statins are known to have anti-inflammatory properties, targeting of inflammation, in particular IL-1β, in cardiovascular disease has produced promising results in both animal studies and, more recently, in the CANTOS trial. The aim of this study was to investigate whether simvastatin use in patients exerts an inhibitory effect on IL-1β expression using an appropriate cardiovascular disease modeling system by stimulation of the NLRP3 inflammasome in isolated peripheral blood mononuclear cell (PBMC) with CC.
Patients and Methods
Patient Recruitment
Ethical approval for the simvastatin study was approved by the Medical Research Ethics Committees at St James’s Hospital/Tallaght Hospital, Dublin, Ireland, and conforms to the ethical guidelines of the 1975 Declaration of Helsinki. Patients with hypercholesterolemia who visited the specialist Lipid Clinic at St James Hospital were recruited for the study. Before commencing the study, written informed consent was obtained from all patients entering the study. Patients selected for the study were over 18 years of age, had evidence of hypercholesterolemia (laboratory plasma lipid levels) for which the patient was indicated simvastatin therapy, and were able to donate a total of 40 mL of blood before and following 8 weeks of simvastatin treatment. Exclusion criteria included concomitant treatment with other lipid-lowering medication; previous statin therapy ceased <2 weeks; use of anti-inflammatory medication both prescription and over the counter agents; and a history of any inflammatory disorder like rheumatoid arthritis, psoriasis, asthma, diabetes, and pregnant women. All the subjects took simvastatin at a daily dose of 10 to 20 mg for 8 weeks. High-density lipoprotein, LDL, total cholesterol, and triglycerides levels before and after simvastatin treatment were measured for all patients enrolled in the study to monitor compliance.
Preparation of CC
Cholesterol crystals were synthesized as described previously. 13 Briefly, cholesterol (Sigma Aldrich, Poole, UK) was dissolved in a 95% ethanol water solution (12.5 g/L) by heating to 60°C, filtered, and incubated overnight for crystal formation. Crystals were again filtered, dried under vacuum conditions, and stored at 4°C. Crystal particles were visualized and photographed using a light microscope and size calculated using a laser diffraction instrument (Malvern Mastersizer 2000, Malvern, United Kingdom) to measure particle sizes between 0.02 and 2000 µm using laser diffraction. The mean size (d = 0.5) was used to denote average crystal particle size per preparation.
Activation of Simvastatin
Simvastatin 5 mg (Sigma, Poole, UK) was activated according to the manufacturer’s instructions by reconstituting in an ethanol/sodium hydroxide solution, incubated for 2 hours in a water bath preheated to 50°C. The drug was made to 1 mL with deionized water and pH adjusted to 7.
Isolation and Culture of PBMCs
Blood was drawn from each patient before and after 8 weeks of simvastatin treatment. Peripheral blood mononuclear cells were isolated from heparinized venous blood according to the manufacturer’s instructions using the density gradient Histopaque 10771 (Sigma Aldrich, Poole, UK). Cells were plated at 80 × 104 cells/mL in 48-well cell culture plates with RPMI-1640 media supplemented with 10% fetal bovine serum,
Cytokines Levels in Cell Culture Media
Levels of IL-1β (BioLegend-Human IL-1β, Medical Supply Co. Dublin, Ireland, Product Cat. No: 437004) and tumor necrosis factor (TNF) α (R&D Systems-Human TNF-α, Minneapolis, MN 55413, Product Cat. No: DY210-05) were measured by enzyme-linked immunosorbent assay using cell culture supernatants following standard manufacturer protocols. All measurements were performed in triplicate, and absorbances read on a plate reader (Bio Tek, Dublin, Ireland, EL808) using KC Junior software.
Western Blot Analysis
Standard western blot techniques were performed. In brief, protein from cell culture supernatants was precipitated using methanol/chloroform and resuspended in RIPA buffer. Peripheral blood mononuclear cells cultured as above were lysed in RIPA buffer containing Sigma protease inhibitor cocktail. Protein concentration of cell lysates was determined by BCA (Sigma Aldrich, Ireland) assay; protein cell lysates (20 µg) or total protein precipitates from cell culture supernatants were resolved on a 15% SDS-PAGE gel followed by semi-dry transfer onto PVDF membrane (Amersham, Buchinghamshire, United Kingdom). Membranes were blocked for 2 hours with TBS-T containing 5% w/v Marvel and was probed with primary antibody directed against human IL-1β (R&D Systems, AF-201-NA, 1:1000 dilution) for 24 hours at 4°C followed by incubation with secondary goat immunoglobulin horseradish peroxidase-conjugated antibody (R&D Systems, HAF017, 1:1000 dilution); signal detection was performed via enhanced chemiluminescence and autoradiography using a Fusion Fx imaging system. Anti β-actin antibody (Sant Cruz Biotechnology, Heidelberg, Germany, 1:1000 dilutions) was used as a loading control for cell lysate samples.
Statistical Analysis
Data are expressed as either mean (standard error of the mean) of absolute values or as a percentage of the appropriate control. All statistical analyses were performed using Graph Pad Prism 5 (Graph Pad Software, Inc, San Diego, California). Differences were considered statistically significant at P < .05 using paired and unpaired Student t-tests where indicated. Correlation analysis was performed using Pearson correlation in Graph Pad Prism 5.
Results
Simvastatin Treatment Reduces Plasma Lipid Levels and CC-Induced IL-1β Expression Levels in PBMC From Patients With Hypercholesterolemia
In order to assess the in vivo effects of simvastatin therapy (10-20 mg daily) on the induction of IL-1β expression in response to CC stimulation, we analyzed the ex vivo responses of patient PBMCs isolated both pre and 8 weeks after the initiation of simvastatin therapy. Cholesterol crystals act as a secondary signal for NLRP3 inflammasome activation which results in the cleavage and secretion of the active form of IL-1β from monocytes, which have been primed with a primary stimulus (eg, LPS). 12 Patients assessed were not taking any other anti-inflammatory medications (eg, Aspirin) during the 8-week period of the study, nor did they have any illness or infection that are known to cause changes in IL-1β levels. Table 1 outlines the demographics of the patients (n = 8) monitored in the study. Patient plasma lipid levels were measured both before commencing and after 8 weeks of simvastatin treatment to act as an indicator of patient adherence and therapeutic efficacy to the simvastatin regimen. Table 2 illustrates the means and ranges of lipid levels observed, with reductions seen in both total cholesterol and LDL levels. In general, a reduction in total cholesterol of approximately 10% to 15% was seen (Figure 1A and B), while LDL levels were, on average, reduced by about 20% (Figure 1C and D ) with no detectable difference between simvastatin doses. These reductions are in line with what would be expected for these drug doses. 28 Although levels of IL-1β in the serum of these patients were below the range of detection (not shown), CC-induced expression of IL-1β in patient’s PBMCs was also assessed pre- and post-simvastatin treatment. Interestingly, treatment with simvastatin did not significantly alter the levels of IL-1β observed upon stimulation of patient’s PBMCs with LPS alone (Figure 1E). As expected, the levels of IL-1β expression detected were significantly increased in a dose-dependent manner when an initial period of LPS stimulation was followed by the addition of CC (Figure 1E). However, the levels of IL-1β released in response to LPS + CC stimulation were significantly decreased among all patients’ post-simvastatin therapy (Figure 1F and G) with no detectable difference between simvastatin doses. Furthermore, no correlation was detected between changes or total LDL levels and IL-1β levels in the patient cohort using a Pearson correlation analysis (Figure 1H). Altogether, we demonstrate that in vivo simvastatin treatment produced a significant reduction in CC-induced IL-1β expression by patient PBMCs, and it is likely independent of simvastatin lipid-lowering effect.
Baseline Patient Demographics.

Abbreviations: SEM, standard error of the mean, SBP, systolic blood pressure; DBP, diastolic blood pressure.
Lipid Parameters at Baseline and Week 8 of Simvastatin Therapy.a
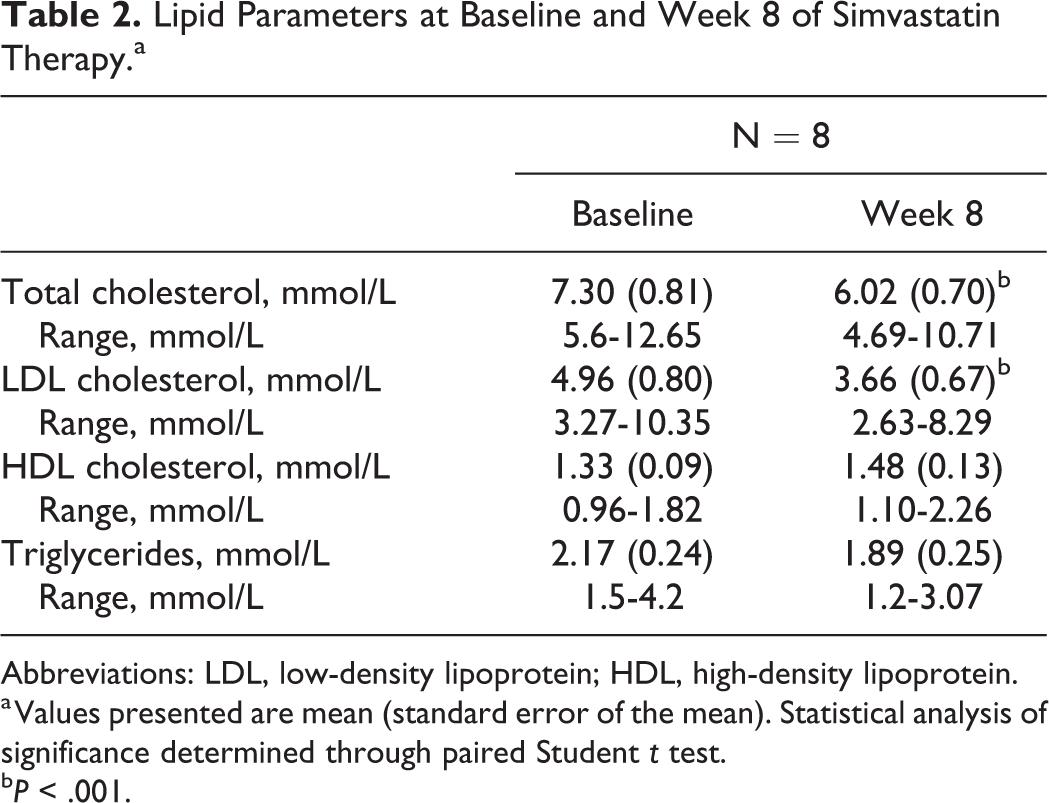
Abbreviations: LDL, low-density lipoprotein; HDL, high-density lipoprotein.
a Values presented are mean (standard error of the mean). Statistical analysis of significance determined through paired Student t test.
b P < .001.

Simvastatin treatment significantly reduces ex vivo cholesterol crystal-induced interleukin (IL) 1β secretion from patient peripheral blood mononuclear cells (PBMCs). A-D, 8 weeks of simvastatin treatment results in a significant reduction in levels of total cholesterol and low-density lipoprotein (LDL) levels among patients enrolled in the study. E, Cholesterol crystal (CC)-induced dose-dependent induction of IL-1β secretion from PBMCs in a single representative patient (patient # 5) prior to simvastatin treatment and significant inhibition of these responses 8 weeks post-treatment. F and G, Levels of inhibition of CC (2 mg/mL)-induced IL-1β secretion in 8 individual patients pre and after 8 weeks (post) simvastatin therapy. H, Pearson correlation visualized as a scatterplot of IL-1β levels versus LDL levels from patients both pre- and post-simvastatin treatment. Statistical analysis of significance determined through paired Student t test. ***P < .001, **P < .01, *P < .05. Pearson correlation r = 0.01, nonsignificant.
In Vitro Treatment With Simvastatin Suppresses CC-Induced IL-1β Expression in PBMCs From Healthy Patients
In an effort to confirm our data from patients with hyperlipidemia described above, we investigated whether similar effects could be observed upon in vitro treatment of PBMCs derived from healthy individuals with simvastatin. In agreement with our patient data, the addition of simvastatin upon initiation of priming with LPS led to a dose-dependent reduction in the levels of IL-1β secretion detected after subsequent stimulation with CC (Figure 2A). Interestingly, levels of TNFα secretion detected under these conditions were not significantly altered in the presence of simvastatin (Figure 2B). As the induction of TNFα expression in this system occurs predominantly as a result of the initial LPS priming signal, these data demonstrate that simvastatin treatment does not act to broadly suppress LPS-induced responses. In addition, these data indicate that cell viability is not adversely affected by the doses of simvastatin used in this assay and was further confirmed by assessing 24-hour simvastatin-treated PBMCs viability as a function of redox potential using MTT, data not shown.

In vitro simvastatin treatment inhibits cholesterol crystal-induced expression and activation of interleukin (IL) 1β from healthy peripheral blood mononuclear cells (PBMCs). Simvastatin treatment of healthy PBMCs in vitro inhibits the secretion of (A) total IL-1β in a dose-dependent fashion without affecting levels of (B) tumor necrosis factor α secretion-induced in response to lipopolysaccharide (LPS; 100 ng/mL) and cholesterol crystals (CC) (2 mg/mL). In vitro treatment of PBMCs with simvastatin (50 μmol/L) significantly reduces levels of (C) total IL-1β secretion, (D) intracellular proIL-1β expression, and (E) secretion of both pro and active IL-1β forms detected in response to stimulation with LPS in combination with CC (2 mg/mL). Data are shown as mean (standard error of the mean [SEM]). Statistical analysis of significance determined through unpaired Student t test. *P < .05, **P < .01.
As described above, LPS provides an initial signal to prime subsequent proteolytic activation of proIL-1β in an NLRP3/caspase 1-mediated fashion upon activation of this pathway with CC. 12,13 Therefore, we next sought to determine whether simvastatin altered the induction of pro-IL-1β expression and/or its subsequent cleavage into its active form. In agreement with previous reports, 24 –26 we found that in vitro treatment with simvastatin resulted in a small increase in the levels of IL-1β secretion in response to stimulation with LPS alone (Figure 2C). These effects were also clearly evident upon analysis of active and proforms of IL-1β in the supernatants by western blot which also demonstrated the appearance of an intermediate sized IL-1β band as reported previously (Figure 2E). 26 As expected, stimulation with CC resulted in significantly increased expression of intracellular pro-IL-1β as well as its secreted pro and active forms (Figure 2D and E). Importantly, treatment with simvastatin led to significantly decreased expression of intracellular pro-IL-1β as well as both active and inactive secreted forms (Figure 2D and E). Together, these data demonstrate that simvastatin can act as a potent inhibitor of CC-induced secretion of active IL-1β in vitro and may act to restrict the enhanced expression of intracellular pro-IL-1β induced by CC to mediate these effects.
Discussion
Based upon their lipid lowering properties, statins have become the gold standard approach for treating patients with cardiovascular risk and disease. 2 Since the approval of lovastatin in the late 1980’s, statin use has provided undisputable benefit to millions of patients worldwide prompting further investigation into their beneficial properties. 29 These efforts have revealed substantial pleiotropic effects beyond their capacity to lower cholesterol and potentially impact cardiovascular health. 30,31 Arguably, the most prominent of these effects are the reported anti-inflammatory properties associated with statin use. 32 Recently, statins have also been attributed to driving production of pro resolvins such as 15-epi-lipoxin A4 through adenosine-induced effects. 20 Inflammatory mechanisms have long been associated with the pathogenesis of atherosclerosis in preclinical studies, 33,34 and their central role in mediating disease have been confirmed through the recently published results of the CANTOS trial. 14 This trial evaluated the efficacy of a monoclonal antibody, canakinumab, aimed at neutralizing the inflammatory cytokine IL-1β on the recurrence of cardiovascular events among patients exhibiting high titers of the inflammatory marker, C-reactive protein. Importantly, the results of this trial have validated the inflammatory hypothesis of cardiovascular disease and established that inhibiting inflammation can have a positive effect on disease outcomes, independently of lipid-lowering therapy. However, it is noteworthy that >90% of patients enrolled in the CANTOS trial were taking statins at the commencement of the study. 14
The design of the CANTOS trial was informed by significant advances in our understanding of the role of inflammation and of the central role of IL-1β, in particular, in the pathogenesis of cardiovascular disease from preclinical studies. Interleukin Iβ has been shown to promote the development of atherosclerotic lesions by stimulating both inflammatory and stromal cell subsets; animal studies have confirmed the key role that this cytokine can play in disease pathogenesis. 6,35 Similarly, studies targeting the NLRP3 inflammasome pathway, which mediates proteolytic activation of the IL-1β cytokine, have demonstrated its importance as a mediator of disease progression. 12,13 In the context of atherosclerotic lesions, CC are thought to represent a key endogenous danger signal in driving activation of the NLRP3 inflammasome pathway and subsequent cleavage of proIL-1β into its active form.
How or whether statins alter the expression and activity of NLRP3 inflammasome-mediated IL-1β activity remains controversial. It has long been established that statins including simvastatin can reduce cytokine production in monocytes from patients with hypercholesterolemia under toll-like receptor (TLR) stimulation conditions. 36 –38 More recently, statin therapy has been shown to reduce levels of serum IL-1β and to reduce the levels of NLRP3 gene expression in PBMCs from patients with cardiovascular disease. 23 However, these studies do not include an analysis of known prominent mediators of sterile atherogenic inflammation such as CC used in this study, which we now know to be involved in NLRP3 inflammasome activation and subsequent IL-1β processing and activation. In contrast, several reports have indicated that statins may act opposingly to enhance caspase 1 activity and promote IL-1β cleavage and activation. 24,25,39 This effect to date has primarily been reported with simvastatin and whether it extends to others in the statin class remains to be seen. Indeed, these effects have been linked with a reported increased risk of diabetes among a small number of patients receiving statin therapy. 40 This reported ability of statins to enhance IL-1β activity occurred after initial priming with LPS leading to the hypothesis that statins may stimulate the NLRP3 pathway. However, whether statin treatment impacts NLRP3 activation through more established NLRP3 activating stimuli, for example, CC, was surprisingly not addressed. This is of particular significance in the context of atherosclerotic disease but also as TLR4 stimulation through LPS in isolation has also been recently shown to induce a limited degree of IL-1β expression and activation in human PBMCs independently of the NLRP3 inflammasome. 41 In this study, we confirm earlier observations that statin treatment can enhance IL-1β expression and activation in vitro in response to LPS stimulation (Figure 2C and E). However, we also demonstrate that simvastatin acts to dramatically inhibit the higher levels of IL-1β expression and activation induced upon stimulation of the NLRP3 pathways with the atheropathogenic danger signal, CC. Importantly, these observations are confirmed in a small cohort of patients undergoing treatment with simvastatin over an 8-week period (Figure 1). Interestingly, no correlation was detected between plasma LDL levels and ex vivo PBMC-derived IL-1β levels in the patient cohort, which may suggest an independent drug effect in PBMC cells outside of simvastatin lipid-lowering effects in the liver. Together, these data offer a novel insight into the anti-inflammatory role of statins in the context of cardiovascular disease and for the first time demonstrate inhibitory effects upon stimulation with CC. To elucidate simvastatin effects in CC-induced activation of the NLRP3 pathway, further studies are needed to reveal these molecular mechanisms. The results presented here are, however, significant given the recently reported positive outcome of the CANTOS study and suggest that statin treatment can also suppress IL-1β activation among patients with cardiovascular disease. 14 As the overwhelming majority of patients evaluated in this trial were undergoing statin therapy, it is tempting to speculate that neutralization of IL-1β activity using canakinumab may exhibit even greater efficacy in the absence of simultaneous statin therapy.
Footnotes
Author Contributions
Boland contributed to acquisition, analysis or interpretation, drafted the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Gangadharan contributed to acquisition, analysis or interpretation, drafted the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Kavanagh contributed to acquisition, analysis or interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Hemeryck contributed to acquisition, analysis or interpretation, drafted the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Kieran contributed to acquisition, analysis or interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Barry contributed to acquisition, analysis or interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Walsh contributed to conception or design, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Lucitt contributed to conception or design, contributed to acquisition, analysis or interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded in part by a Special Merit Award from the Haematology Association of Ireland to M.L.
