Abstract
Approximately 40% of heart attack survivors remain at increased risk of recurrent cardiovascular events, despite the current treatment options showing that atherothrombosis is not exclusively a disorder of lipoprotein aggregation in the arterial wall. Clinical and experimental data suggest that inflammation plays an important role in atherothrombosis independent of the cholesterol level. Acute-phase reactants, such as C-reactive protein, increase in patients with coronary artery disease and are known to predict adverse outcomes in such patients. The recent CANTOS trial published in The New England Journal of Medicine provides evidence that interleukin-1β along with other cytokines play central roles in the inflammatory reaction that drives the interleukin-6 signaling pathway and have profound effects on cardiovascular outcomes. Several other ongoing studies are focused on multiple immune mediators involved in this process to support the inflammatory hypothesis of cardiovascular diseases. These new classes of drugs could represent the biggest breakthrough in cardiovascular medicine, which could have the greatest impact on cardiovascular mortality since the advent of statins. The drug canakinumab has shown promise in lowering atherosclerosis, and other drugs, such as colchicine and methotrexate, are gaining interest and are being investigated in multiple ongoing trials. A major concern is the affordability of these drugs, as most cardiovascular diseases are noted among people of lower socioeconomic statuses. The LoDoCo trial showed some benefits of colchicine, and whether this old drug can be marketed with a new label for cardiovascular disease remains in question. Therefore, a clear understanding of the different inflammatory pathways involved in atherosclerosis is needed to help develop more effective treatment modalities that will benefit humankind.
Introduction
Atherosclerosis is still the leading cause of morbidity and mortality in a large amount of the patient population. The pharmacological interventions that are currently used focus almost entirely on reducing plasma cholesterol levels and other risk factor modifications intended to slow the progression of atherosclerosis. 1 Further insights into the pathogenesis of atherosclerosis can be subsumed under the so-called inflammatory hypothesis. 2 Enhanced knowledge about the role of inflammation in atherosclerosis can lead to the development of more effective therapeutic agents and ultimately improve patient outcomes. Identification of many of the cytokines, chemokines, and adhesion molecules that arise from the vessel wall as well as from multiple cell lines, including macrophages and monocytes, led investigators to use agents that target them specifically and monitor patient outcomes. The aim of each of these studies was to mitigate and target cellular inflammatory pathways independently affecting atherosclerosis.
Effective cardiovascular drugs with anti-inflammatory effects, such as aspirin and statins, predominantly exert their therapeutic benefits by mechanisms other than inflammation. 3 Although aspirin is an anti-inflammatory medication, its mortality benefit in coronary artery disease (CAD) stems from its ability to prevent platelet adhesion and thrombus formation. Similarly, statins are considered to lower inflammation to some extent via their pleiotropic effects. However, its therapeutic benefit was correlated directly with reducing cholesterol levels, essentially the LDL levels. Therefore, the extent to which inflammation adds to heart disease risk has never been clarified. 3,4
Inflammation: A Curse or a Blessing?
Inflammation involves a series of events that occur in the human body in response to pathogen invasion or tissue damage. Inflammatory pathways involve multiple leukocytes exiting the blood vessels into the interstitial space along with fluid and plasma proteins to eliminate the pathogen and clear the necrotic debris. However, how this inflammation leads to atherosclerosis has always been questionable. The hypothesis regarding the involvement of inflammation in atherosclerosis dates back to the 19th century. The involvement of monocytes and macrophages in the early stages of atherosclerosis suggested this claim. However, there was no substantial evidence to back up the inflammatory hypothesis of atherosclerosis. Later, several inflammatory markers were noted to be increased in patients with CAD. In fact, traditional statin therapy has been shown to decrease inflammatory markers via these pleiotropic effects. Additionally, it was noted that patients with chronic inflammatory conditions were prone to recurrent cardiovascular events. However, a direct causal role of inflammation in these events was never established.
Inflammatory Pathways in Vascular Disease
Atherosclerosis was always considered a chronic disease with the deposition of excess cholesterol in the arterial intima. However, inflammation participates in atherosclerosis from its genesis to the grand finale. Normally, the endothelial surface is resistant to leukocyte adhesion. However, certain triggers, such as smoking, high-fat diet, hypertension, hyperglycemia, obesity, and insulin resistance, can cause tissue damage and initiate the process of inflammation, leading to the expression of adhesion molecules on the arterial Endothelial Cells (EC). One such culprit is vascular cell adhesion molecule 1 (VCAM-1). 5
The VCAM-1 is expressed on the endothelial cell surface by the activation of multiple immune response genes via the upregulation of NF-kB, stimulated by the oxidized lipid particles along with the production of multiple proinflammatory cytokines, such as IL-1, TNF-α, interleukin 6 (IL-6), and IL-8. This leads to the recruitment of monocytes via diapedesis through the endothelial cell layer toward the intima. The monocytes become macrophages expressing scavenger receptors that engulf these oxidized lipid particles, thus forming lipid-laden macrophages or foam cells. 6 This becomes the hallmark of the earliest stage of atherosclerosis. Another major chemokine that is involved in recruiting monocytes is MCP-1, or monocyte chemoattractant protein, which is shown to be overexpressed in experimental studies of early atheroma. 5 The macrophages then multiply and release several growth factors and cytokines, thus amplifying and sustaining proinflammatory signals. This earliest stage of atherosclerosis has even been shown to occur during the adolescent period, especially in those who follow a sedentary lifestyle. T lymphocytes, which are part of the adaptive immune system, have been shown to play a major role, as they enter the inflamed arterial wall via interaction with their CXCR3 receptors, which are also highly expressed in the plaques. This leads to the production of IFN-¥, which further activates the macrophages and sustains the immune response. Another cytokine CD-40 ligand (CD-154) has been shown to be overexpressed in atheromatous plaques in mouse models, and treatment with anti-CD154 in these mice over a period drastically reduced the progression of the lesion. This shows the role of inflammation in the progression of atherosclerosis from a fatty streak to a complex plaque. 7 –9
Critical stenosis does not cause most myocardial infarctions (MIs). Often, the rupture of a vulnerable plaque and subsequent thrombus formation leads to acute coronary syndrome. Patients with multiple plaques that are prone to rupture are at an increased risk of recurrent MIs. The vulnerability of a plaque to rupture depends on the strength of the fibrous cap. The fibrous cap is made up of thick collagen fibers that protect the blood from contact with the lipid core. Studies have shown that inflammation impedes the integrity of this collagen matrix by promoting the destruction of existing fibers and blocking the production of new fibers. This results in a thin fibrous cap that is subsequently prone to rupture. Thus, we see that inflammation plays a vital role in all 3 steps of atherosclerosis from initiation to progression and, finally, to weakening and rupture of the fibrous cap. 5,10,11 Thus, targeted therapy in specific areas of the inflammatory cascade can prevent cardiovascular events (see Figure 1).
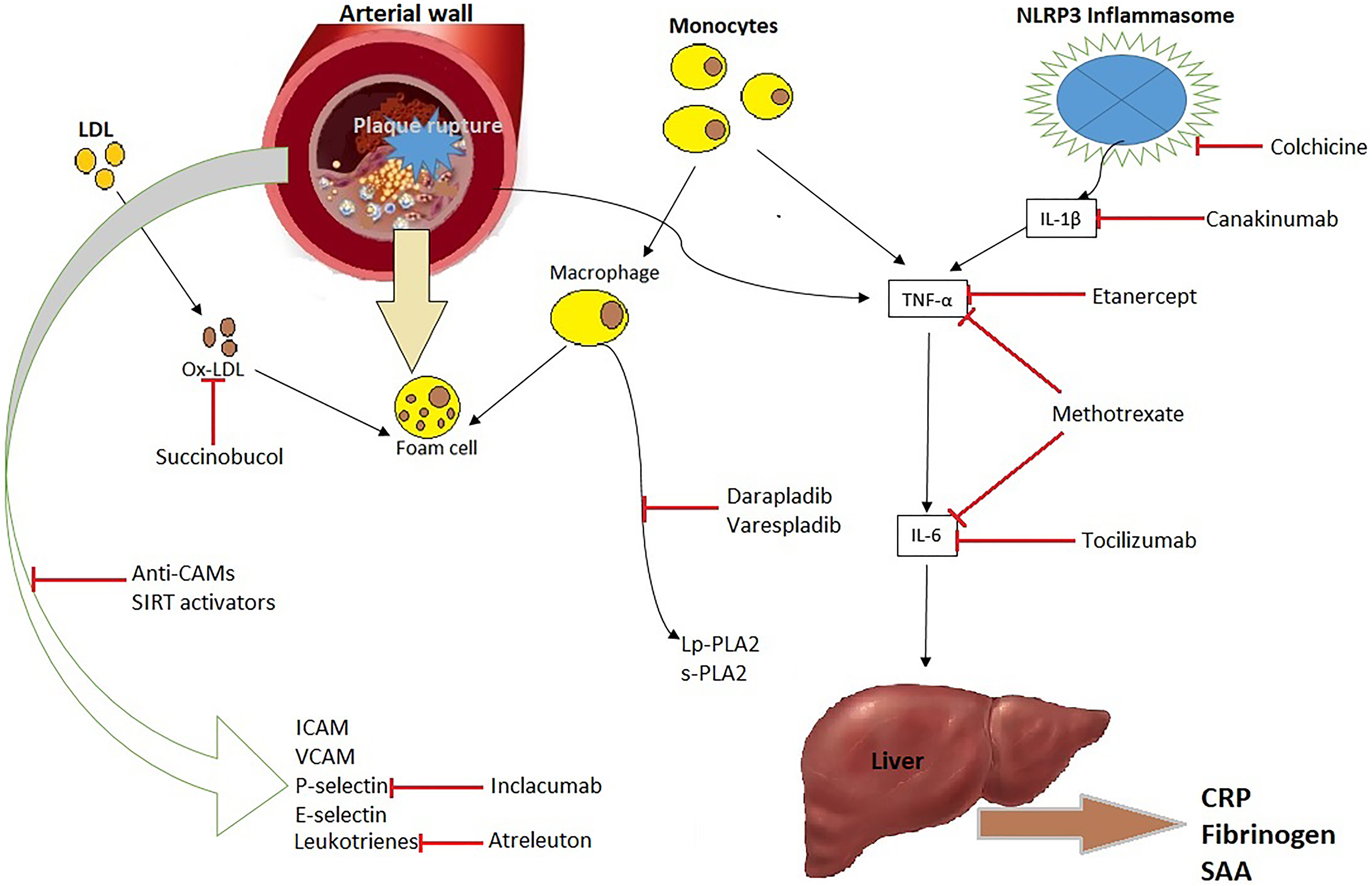
Potential targets of anti-inflammatory agents.
Regarding the monitoring of inflammation that occurs in atherosclerosis, several proinflammatory markers have been associated with cardiovascular disease (CVD) risk and can be used to monitor a patient’s vulnerability for an acute event. The most important of these is the downstream acute-phase reactant C-reactive protein (CRP). 10 Other proinflammatory markers are IL-6 and TNF-α. The benefits of using CRP as a marker include its stability (only trivial circadian variations) and the fact that it can be measured easily and reliably. Statin therapy has previously been shown to significantly decrease CRP levels by 15% to 50%.
The JUPITER primary prevention trial apparently demonstrated that statins decrease the risk of cardiovascular events and all-cause mortality in patients with evidence of high inflammation levels even if they have low cholesterol levels. 3 This trial also identified that statins can reduce stasis thrombosis, which has no association with LDL levels. As a matter of fact, the JUPITER (Justification for the Use of Statin in Prevention: an Intervention Trial Evaluating Rosuvastatin) trial demonstrated that statin therapy is sufficiently adequate to prevent first-ever cardiovascular events among men and women who are not candidates for statin therapy but have high levels of the inflammatory biomarker high-sensitivity CRP (hsCRP). 12 How statins exert these anti-inflammatory effects is still unknown, but the mechanism is thought to be via the expression of transcription factors, such as Krüppel-like factor 2, which has antithrombotic, anti-inflammatory, and antiproliferative functions. 4,13
Generally, when considering the multiple pathways of inflammation involved in atherosclerosis, we can categorize anti-inflammatory agents under evaluation into those targeting the IL-6 signaling pathway as the classical pathway and those that take an alternative route as the alternative pathway.
Immunology and Overview of the Major Cytokines Involved in Atherothrombosis
Inflammation occurs in the body in response to infection or tissue necrosis. When appropriate, inflammation is the most effective process for the restoration of normal tissue architecture and eradication of the foreign entity. Acute inflammation operates via the mechanism of leukocyte infiltration and subsequent clearance. This is part of innate immunity that has limited specificity and an immediate response. A crucial step in resolving any inflammatory response is the transition of the immune system from innate to adaptive immunity. Adaptive immunity is more specific and involves lymphocytes and plasma cells. However, in chronic inflammation, this process becomes distorted and leads to the retention of activated mononuclear cells within the involved tissue. This results in chronic damage to the tissue. Therefore, improper regulation of the “immunological switch” from innate to acquired immunity can clearly impact disease outcomes. Although innate and acquired immunity were traditionally considered self-governing processes, recent advances in cytokine signaling, complement activation, and molecular biology have described several mediators participating in this immunological switch. 14 A better understanding of this concept can be achieved by identifying the major cytokines involved in this process (see Figure 2).

The classical pathways of inflammation in atherosclerosis.
The Role of IL-6 in Disease
Interleukin-6 is an inflammatory cytokine that exerts its effects via its membrane-bound or circulating soluble receptors. Interleukin-6 is produced by T cells, macrophages, and adipocytes, and its receptors are present on monocytes, hepatocytes, and endothelial cells. Several prospective observational studies have shown that high plasma IL-6 levels increase the risk of CAD. The effects of IL-6 on hepatocytes make it an activator of APR. Interleukin 6 is also considered a leukocyte-stimulating factor. Interleukin 6 along with its control over leukocyte recruitment, activation and apoptosis have surfaced as regulators of this immunological switch from innate to adaptive immunity. Predominantly, these activities have been identified based on an increased understanding of the regulatory properties of its soluble receptor and the subsequent IL-6 signaling 14 (see Figure 2).
The IL-6R complex consists of at least 1 subunit of signal-transducing glycoprotein gp130. Interleukin 6 can exert its effect by either directly binding to the transmembrane receptor or via its soluble receptor. 15 The term IL-6 trans-signaling is used when the mode of activation is via its soluble receptor. The IL-6–IL-6R complex binds directly to gp130 to exert its effect. This IL-6R signaling propagates a downstream inflammation cascade. Additionally, IL-6 signaling in liver parenchymal cells suppresses hepatic inflammation and enhances systemic insulin action. Thus, IL-6 exhibits both proinflammatory and anti-inflammatory properties. Upon reflection, IL-6 can be defined as a resolution factor that can balance the pro- and anti-inflammatory functions to facilitate the immunological response.
For the most part, studies have highlighted a central role of IL-6 in inflammation and emphasized the potential effect on targeting its receptors. 16 The application of anti-IL-6 receptor antibodies, such as tocilizumab (TCZ) and atlizumab, in the treatment of Rheumatoid Arthritis (RA) 17 and Crohn’s disease 18 illustrates their efficacy. Mendelian randomization analysis conducted in 2012 demonstrated the involvement of IL-6R signaling in coronary heart disease and concluded that IL-6R would be a valuable target for the prevention and treatment of CAD. 19 Another collaborative meta-analysis of 82 studies showed that an IL-6R-related pathway was associated with coronary heart disease. 20 However, TCZ increases lipid levels, and studies have always been focused on its cardiovascular safety rather than efficacy.
Interleukin-1β and Vascular Inflammation
Along with the classical IL-6 signaling cascade, IL-1 exerts its effects on inflammation and is one of the major molecules functioning in atherothrombosis. IL-1 belongs to a family of cytokines consisting of 11 members, among which IL-1α and interleukin-1β (IL-1β) are the most important and widely studied. 21 Both IL-1α and IL-1β bind to the same receptor molecule, termed IL-1 type 1 receptor, and act as its activator. The receptor is also influenced by the naturally occurring inhibitor IL-1Ra (IL-1 receptor antagonist). 22
Interleukin 1β is produced by innate immune cells, such as monocytes, as an inactive precursor form (pro-IL-1β), which demands proteolytic cleavage. This is typically mediated by the NLRP3 inflammasome, which is an intracellular protein complex that consist of NLRP3, adaptor protein ASC, and caspase 1 (also called IL-1β converting enzyme). Numerous danger signals activate the NLRP3 inflammasome. These include pathogen-associated molecular patterns, such as LPS (bacterial endotoxins), and danger-associated molecular patterns, which are host molecules indicative of cellular damage, such as uric acid crystals. The NLRP3 inflammasome is also shown to be activated by cholesterol when precipitated from the soluble form to the crystalline form. This reveals that cholesterol crystals can trigger IL-1β production, which leads to vascular inflammation and supports the function of inflammation in atherosclerosis. 23,24
Cholesterol crystals are perceived as the tell-tale sign of atherosclerotic lesions, and their presence helps in the histopathological classification of advanced atherothrombotic lesions. Additionally, studies on mice fed a high-cholesterol diet showed that cholesterol crystals were deposited into subendothelial areas that were rich in immune cells and not only in the necrotic cores. 24 These studies demonstrate that cholesterol crystals emerge early in atherogenesis along with the appearance of immune cells in the subendothelial space. The NLRP3 inflammasome is acted upon by these crystals, resulting in a cascade of events with the production of IL-1β, which induces the upstream production of IL-6 and the downstream production of CRP.
Canakinumab is a human monoclonal antihuman IL-1β antibody that was initially developed by Novartis for the treatment of rheumatoid arthritis in 2009. It was later approved for the treatment of cryopyrin-associated periodic syndromes, systemic juvenile idiopathic arthritis, and other IL-1β overexpression disorders. Canakinumab was demonstrated to induce a >50% dose-dependent reduction in IL-6 and CRP as well as a 20% reduction in fibrinogen. When tested on patients with rheumatoid arthritis and diabetes, canakinumab obviously reduced inflammation and did not significantly change the lipid profile or platelet functions. Thus, canakinumab has been used as an innovative way to directly test the inflammatory hypothesis of atherothrombosis, which ultimately led to the Canakinumab Anti-inflammatory Thrombosis Outcome Study (CANTOS) trial. 3
Clinical Trials on Anti-Inflammatory Therapy for CAD Targeting the Classical Pathway
The CANTOS Trial
CANTOS (Canakinumab Anti-inflammatory Thrombosis Outcome Study) was a randomized, double-blinded, placebo-controlled trial that included stable patients who had previous episodes of MI. The trial selected candidates who experienced a persistent proinflammatory response with a high CRP level of 2 mg or more and evaluated whether canakinumab could effectively reduce recurrent vascular events. The trial was devised to directly test the inflammatory hypothesis of atherothrombosis. The investigator-driven, 5-year prospective trial was sponsored by Novartis. 25 The trial protocol along with the full text is available at NEJM.org. A total of 39 countries were involved, and the protocol was approved at the participating centers (see Table 1). 1
Important Clinical Trials of Anti-Inflammatory Therapy Targeting the Classical Pathway.

Abbreviations: CANTOS, Canakinumab Anti-inflammatory Thrombosis Outcome Study; CIRT, Cardiovascular Inflammation Reduction Trial; COLCOT, Colchicine Cardiovascular Outcomes Trial; ENTRACTE, Effects of IL-6 Receptor Blockade With Tocilizumab (TCZ) in Comparison With Etanercept (ETA) on the Rate of Cardiovascular Events in Patients With Moderate to Severe RA; IL-1β, interleukin-1β; IL-6; interleukin-6; LoDoCo, Low-Dose Colchicine.
A total of 10 000 patients with a history of MI and an elevated level of hsCRP (≥2 mg) were selected for the study. The trial used 3 doses of canakinumab (50, 150, and 300 mg), which were randomly assigned in addition to a placebo. Participants were followed prospectively over a period of 5 years for primary and secondary end points. The primary efficacy endpoint was nonfatal MI, nonfatal stroke, or cardiovascular death. The secondary end points were hospitalization for unstable angina that led to urgent revascularization, the incidence of new onset type 2 diabetes mellitus (T2DM), and all-cause mortality.
The study noted a significant reduction in the levels of hsCRP and IL-6 in those taking canakinumab compared to those administered the placebo, and no confounding changes were found in the baseline lipid levels. At 48 months, the median reduction in hsCRP from the baseline was 37% greater in the 150-mg and 50-mg groups and 41% greater in the 300-mg group compared to that in the placebo group (P < .001). By the end of the follow-up period, the incidence rates for the primary end point were 4.50, 4.11, and 3.86 events per 100 person-years in the 50-, 150-, and 300-mg groups, respectively. No significant effect was observed in the 50-mg group compared to the placebo group (hazard ratio [HR] = 0.93; P = .30), while a significant effect was observed for the primary end point in the 150-mg group (HR = 0.85; P = .020). In the 300-mg group, the HR was similar to that in the 150-mg group, but the P value did not meet the prespecified threshold for significance (HR = 0.86; P = .0314).
The major adverse reaction observed was neutropenia, which resulted in infections and thrombocytopenia. Six confirmed cases of tuberculosis occurred in the canakinumab group. Although significantly more deaths occurred due to infection and sepsis in the canakinumab group, the patients who died were older and more likely to have diabetes. No significant difference in the incidence of hemorrhage was observed in the canakinumab group compared to that in the placebo group. Canakinumab also resulted in fewer reports of arthritis and gout compared to those in the placebo group.
Thus, the risk of the primary end point was almost 15% lower in the 150-mg canakinumab group compared to that in the placebo group and almost 17% lower for the secondary end point with a statistically significant P value. 1 The fact that canakinumab significantly reduced the hsCRP level without reducing the LDL cholesterol level provided evidence for the inflammatory hypothesis in atherosclerosis. The only factor limiting its use in the general public would be its cost. A cheaper anti-inflammatory medication needs to be derived to increase the popularity of anti-inflammatory therapy in cardiovascular medicine.
The CIRT Trial
Low-dose methotrexate (LDM; 15-20 mg per week) is the recommended treatment for RA and Psoriatic Arthritis (PsA) and is taken safely over a long period of time by thousands of patients worldwide. The mechanism underlying the anti-inflammatory effect of LDM in these patients is by decreasing the levels of IL-6 and CRP, and LDM has no confounding effects on lipid levels, hemostasis, or platelet function. Thus, LDM is an appropriate agent to directly test the inflammatory hypothesis of atherothrombosis. Additionally, compared to canakinumab, methotrexate is a drug that physicians are familiar with, and the risks are well established. 4,26
Low-dose methotrexate directly blunts the central IL-6 signaling pathway. Mechanistic studies suggest that some of the atheroprotective effects of methotrexate may accrue from the enhanced release of adenosine and antagonism of the adenosine A2A receptor. 2,16,27 The receptor stimulation induces the expression of several proteins linked to reverse cholesterol transport, such as 27-hydroxylase and ATP-binding cassette transporter. Other work suggests that methotrexate has direct effects on apoptosis and suppressing the function of adhesion molecules, such as ICAM and VCAM, 28 both of which play relevant roles in atherothrombosis. The trial is event driven and concludes after the accumulation of at least 530 primary end points or extreme effects.
Funded by the National Heart Lung and Blood Institute in 2013, the Cardiovascular Inflammation Reduction Trial (CIRT) involves the randomization of 7000 men and women from the United States and Canada who have chronic atherosclerosis and either diabetes or metabolic syndrome. 29 The primary aim of the CIRT trial is to identify whether LDM will reduce the rates of recurrent myocardial infarction, stroke, and cardiovascular death among stable postmyocardial infarction patients with T2DM or metabolic syndrome, which are conditions associated with an enhanced proinflammatory response.
After a 5- to 6-week open-label run-in, eligible participants are selected as those who have had a documented myocardial infarction or had multivessel disease diagnosed via coronary angiography in the past 5 years. These patients are randomly assigned to usual care plus LDM (15 mg orally per week) or usual care plus placebo over a period of 3 to 4 years. In addition, all participants are administered folic acid supplements to decrease some of the side effects associated with LDM. The initial 5- to 6-week open-label run-in for all participants is conducted to ensure safety and improve long-term compliance. It was assumed that further complications of LDM can be minimized by excluding those with known hepatic disease, pulmonary dysfunction, or HIV-related diseases and limiting enrollment of those with any evidence of malignancy, renal dysfunction, chronic infection, or other methotrexate risk factors. The monitoring of liver function, blood count, and creatinine is performed on a regular basis for all study participants. A centralized methodology was designed to ensure participant safety and undertake issues of compliance and follow-up on a cost-effective basis. The primary end point of CIRT is recurrent nonfatal cardiovascular events and mortality. Secondary end points are a decrease in the progression of Diabetes Mellitus (DM) and a decreased need for hypolipidemic agents in diabetics. 30
The COLCOT and LoDoCo Trials
Recent data regarding the utility of colchicine, an agent used for gout, supports the hypothesis of anti-inflammatory therapy for vasculoprotection. Colchicine is a powerful spindle poison that has several anti-inflammatory properties, such as inhibiting neutrophil function via its antitubulin effects. It has also been shown to have limited effects on macrophages via the inhibition of NLRP3 gene expression and the activation of caspase 1 and IL-1β. Additionally, the fact that it reduces CRP levels independent of aspirin and statins makes it a suitable agent to test the hypothesis. 31,32
Colchicine Cardiovascular Outcomes Trial (COLCOT) is an ongoing study that evaluates whether colchicine reduces cardiovascular events in patients after MI. The patients receive either colchicine or a matched placebo for a 2-year period or until the target 301 primary end points have been met. The primary end points are cardiovascular death, resuscitated cardiac arrest, acute MI, stroke, and hospitalization for angina requiring coronary revascularization. The study is expected to be completed in September 2019 with an estimated enrollment of 4500 participants. By disrupting the cytoskeleton, colchicine is believed to suppress the secretion of cytokines and chemokines as well as platelet aggregation, which ultimately induces cardiovascular benefits by inhibiting atherosclerosis. 33
The LoDoCo (Low Dose Colchicine) trial assigned 532 patients with stable CAD to receive either colchicine 0.5 mg/d or a matched placebo. The benefits were observed early and constantly throughout the median 3-year follow-up period, with a reduction in the primary end point of recurrent ACS/cardiac arrest/ischemic stroke (HR = 0.33, 95% confidence interval = 0.18-0.59; P < .001). Adverse effects of the drug were a major concern, as nearly 20% of those on colchicine were discontinued from the study due to anticipated adverse gastrointestinal effects. 2,16
Colchicine has been shown to be beneficial in a number of inflammatory conditions, such as pericarditis and atrial fibrillation. In the past, colchicine has also been tested as an antirestenotic agent in patients undergoing percutaneous coronary intervention (PCI) due to its anti-inflammatory and antimitotic effects. 34 This trial suggests that the mechanism benefits de novo lesions and not stented lesions; thus, large-scale fully blinded trials of colchicine in secondary prevention are warranted. 35
The ENTRACTE Study
Cardiovascular disease accounts for approximately 31% of deaths in patients with RA. 36 Large retrospective studies show that the risk factors for CVD are increased by nearly 2-fold in patients with RA compared to control participants, with a Relative Risk (RR) ranging from 1.5 to 4.0. Moreover, IL-6 and TNF-α are significantly elevated in RA and are typically associated with subclinical atherosclerosis in RA as shown by increased carotid intima–media thickness. 37 In light of these observations and evidence from the CANTOS trial, we can state that the key driving force of this increased risk is the high inflammatory burden associated with RA.
Both RA and atherosclerosis share the same inflammatory pathway, which involves IL-6 signaling. Thus, blocking IL-6 signaling has potential therapeutic benefits on the prevention of CAD. 38 Tocilizumab blocks IL-6R and blunts IL-6 signaling, which exerts a strong and sustained effect on inflammation, eventually normalizing the CRP levels. Although TCZ has been shown to increase lipid levels, these levels are rapidly normalized by lipid-lowering therapies, and whether it increases the risk of CVD in RA remains unclear. The ENTRACTE study was actually conducted to compare the risks associated with TCZ versus the TNF inhibitor etanercept. The use of a TNF inhibitor is usually recommended as an add-on therapy in patients with moderate-to-high disease activity along with a DMARD. Tumor necrosis factor, a pivotal cytokine in chronic inflammation, plays a role in the classical pathway, as it is located adjacent to IL-6 and affects other metabolic functions, such as lipid metabolism, insulin resistance, and endothelial function. 4,38
Tumor necrosis factor α mediates its effects via 2 of its receptors, TNFRI and TNFRII. Both TNF receptors can activate the transcription factor NF-kB, inducing the transcription of a wide range of genes involved in inflammation. The neutralization of TNF-α has anti-inflammatory and disease-modifying effects on a wide range of immune-mediated diseases, such as Crohn’s disease, 39 ankylosing spondylitis, psoriasis, and rheumatoid arthritis. Etanercept is a recombinant DNA fusion protein that mimics the naturally occurring soluble TNF2 receptor, which deactivates TNF and blunts the immune response. 40 Thus, etanercept has increased its popularity as an agent for the treatment of RA.
Tocilizumab’s profound effects on lipids have been confirmed in all of its pivotal trials, with average increases in total cholesterol, LDL, HDL, and triglycerides of 17% to 25%. The MEASURE study tried to determine the quantitative changes in these lipid subfractions. This study evaluated 132 patients treated with TCZ or placebo. 41 The study showed that even though TCZ increased the total LDL, no statistically significant increase in the concentration of small LDL or oxidized LDL was observed compared to that achieved with the placebo. Moreover, a remodeling of the HDL particle from the proinflammatory to anti-inflammatory phenotype was observed in TCZ recipients. This supports a potential detrimental effect of IL-6R signaling on CAD. 38
ENTRACTE is the first trial, however, to compare the cardiovascular safety profile of TCZ to that of another RA medication. The study consisted of 3080 patients with active seropositive RA from 353 centers across 31 countries. The mean age of the patients was 61 years. All patients had at least one cardiovascular RF, including current smoking (30%), hypertension (71%), and diabetes (18%). 42 The participants were randomized to open-label TCZ 8 mg/kg infused every 4 weeks or etanercept 50 mg injected weekly. The mean follow-up was 3.5 years with some up to 5 years. This was considered enough time to see early atherothrombotic complications if to develop.
The primary end point was a combination of major cardiovascular outcomes including cardiovascular death, nonfatal MI, or nonfatal stroke. Secondary end points comprised of individual components of the primary outcome, as well as all-cause mortality, heart failure hospitalization, fatal and nonfatal myocardial infarction and stroke, and lipid levels.
Since this was a safety study, the investigators were interested in the uncertainty in their risk estimates. The study was designed to exclude at most an 80% increase in cardiovascular events in patients treated with TCZ compared to those treated with etanercept. From the study, it was estimated that if a larger sample was studied, the risk of such events in the TCZ group would not exceed 43%. The clear drawback of the study was that there was no placebo group, as all the study participants needed anti-inflammatory therapy. Another multidatabase cohort study comparing TCZ to a TNFi showed no evidence of increased cardiovascular risk among RA patients who switched from a biological agent to TCZ versus a TNFi. 43
Potential Alternate Pathways and Trials in the Anti-Inflammatory Hypothesis
Secretory Phospholipase Inhibitors
Phospholipase A2 (PLA2) is an enzyme responsible for phospholipase hydrolysis, leading to the formation of potentially atherogenic lipid factors and increased exposure of the vessel wall to oxidative stress. This enzyme is derived mainly from macrophages and circulates in plasma by merging with LDL and HDL cholesterol. Data obtained from more than 80 000 patients suggest that plasma PLA2 activity and levels are associated with an increased risk of cardiovascular mortality. The PLA2 superfamily includes lipoprotein-associated PLA2 (Lp-PLA2) and secretory PLA2 (sPLA2). Both sPLA2 and Lp-PLA2 can activate multiple inflammatory pathways and are associated with oxidized LDL, which makes them pertinent in atherosclerosis. Based on these observations, varespladib and darapladib, which inhibit sPLA2 and Lp-PLA2, respectively, were developed (see Table 2). 2,16
Important Clinical Trials of Anti-Inflammatory Therapy Targeting Alternate Pathways.

Abbreviations: ARISE, Aggressive Reduction of Inflammation Stops Events; STABILITY, Stabilization of Atherosclerotic Plaque by Initiation of Darapladib Therapy.
To test the impact of these drugs, a few phase III outcome trials were conducted with both varespladib and darapladib. Among them, the 5000-participant VISTA-16 study was conducted to estimate the effects of an sPLA2 blockade with varespladib on cardiovascular outcomes. The double-blinded, randomized, multicenter trial was terminated early by the data and safety monitoring board due to futility and possible harm. It was shown that the primary outcome of cardiovascular events occurred in 136 candidates on varespladib but in only 109 participants receiving the placebo. Thus, varespladib has an association with an increased risk of MI. 44,45 No association was observed in patients receiving varespladib in the Fewer Recurrent Acute Coronary Events with Near-term Cardiovascular Inflammatory Suppression study or the Phospholipase Level And Serological Markers of Atherosclerosis (PLASMA) II trials.
The 15 000-participant STABILITY trial (Stabilization of Atherosclerotic Plaque by Initiation of Darapladib Therapy) observed a small reduction in vascular events, which was not clinically significant. Moreover, the SOLID-TIMI 52 trial, which randomized 13 000 participants to evaluate major coronary events, did not show any beneficial effect of darapladib. Because both trials failed, moving forward with the concept of PLA2 in coronary heart disease will be challenging. 4,15,46,47
Adhesion Molecule Inhibitors
As mentioned earlier, adhesion molecules play a vital role in the initiation of plaque formation. Molecules such as ICAM and VCAM are involved in the adhesion and transmigration of leukocytes across the vascular cell wall. The tethering and rolling of leukocytes via the vascular endothelium are also mediated by another class of cell surface glycoproteins called selectins. 2,16 Selectins have 2 subtypes, P-selectins and E-selectins. P-selectins are released by Weibel-Palade bodies and mediated by histamine, and E-selectins are induced by TNF and IL-1. The most important role in the development of CVD is played by P-selectin, which mediates multiple cell–cell interactions for the initiation and formation of atherosclerotic plaques.
Promising results from preclinical studies led to a prospective randomized phase III trial called SELECT-ACS. Inclacumab, a recombinant monoclonal antibody against P-selectin, was randomly allocated and compared to the placebo on 544 NSTEMI patients. Inclacumab at a 20 mg/kg body weight administered in continuous infusion over 1 hour in these patients was associated with less myocardial damage as expressed by serum troponin and CK-MB levels compared to that associated with the placebo. 48
Therefore, to further analyze the effects of P-selectin antagonists, the SELECT-CABG trial was conducted in patients who tolerated coronary artery bypass graft surgery. The trial assessed the effect of inclacumab on patients with saphenous vein graft (SVG) and monitored SVG disease progression over 1 year after CABG surgery. However, the study did not show significantly favorable effects on SVG disease progression. 49,50 Further evaluation might be necessary to assess the activity of inclacumab.
Vascular-Targeted Antioxidants
Another major factor presumed to be involved in atherosclerosis in addition to inflammation is oxidative stress. Oxidized LDL is a critical component of atherosclerosis that triggers further inflammation in the vascular wall. Succinobucol is a drug that inhibits key oxidant signals within the endothelial cell wall and prevents the formation of oxidized LDL particles. The Aggressive Reduction of Inflammation Stops Events (ARISE) trial was designed to assess whether adding succinobucol to standard medical therapy would reduce the incidence of ACS. A total of 6144 patients with recent ischemia were randomized to either a succinobucol or a placebo group and followed for cardiovascular outcomes. 51
Even when the study showed a significant reduction in secondary end points, such as cardiovascular death, cardiac arrest, MI, and stroke, no change in primary end points were observed. Notably, a significant lowering of new-onset diabetes was noted in the succinobucol group with a modest reduction in Hba1c. However, no significant difference in overall serious adverse events was noted between the 2 groups. Although the result of ARISE failed to achieve their primary end point, the beneficial effect of the reduction in new-onset diabetes is certainly a promising outcome that warrants further study.
Leukotriene Inhibitors
Leukotrienes are arachidonic acid metabolites capable of activating multiple inflammatory pathways. They play a prominent role in acute and chronic inflammation. Leukotriene-targeted therapy has been used in conditions such as asthma, RA, IBD, psoriasis, and allergic rhinitis. The major enzyme involved in the pathway is 5-lipoxygenase (5-LOX). 2,16
Patients with unstable CAD show an increase in 5-LOX activity, and 5-LOX-activating protein gene polymorphisms have been reported to affect the risk of MI. Thus, excessive leukotriene synthesis by 5-LOX may significantly contribute to the progression of atherosclerosis. Zileuton is the only 5-LOX inhibitor currently available on the market and is used for the treatment of asthma. An N-hydroxyurea derivative, atreleuton, is being developed by VIA Pharmaceuticals and could be a potential alternative in this field. A phase II study on the use of atreleuton for the treatment of CVDs and vascular inflammation was completed. 52
The acute coronary ischemia study randomly assigned 191 patients to either the atreleuton group (VIA-2291) or the placebo group. The results showed a significant reduction in LTE4 in the treatment group, with a reduction in the development of new coronary plaques being observed in those who had coronary CT examinations before and after treatment. Another subset of patients undergoing carotid endarterectomy was evaluated over 12 weeks in the carotid study with VIA-2291, but no difference in the primary end point of a reduction in inflammatory macrophages was observed between the treatment and the placebo groups.
Antiplatelet Agents and Salicylates
Another area of clinical research interest is the effects of using aspirin, clopidogrel, and other P2Y12-receptor antagonists as anti-inflammatory agents. The Aspirin Versus/Or Clopidogrel in Aspirin-resistant Diabetics inflammation Outcome (AVOCADO) study showed that both aspirin and clopidogrel reduced inflammatory markers, such as hsCRP, IL-6, and CD40 ligand, in patients with T2DM. 53 Recently, another study found that clopidogrel had favorable effects on the suppression of hsCRP levels in patients undergoing PCI. 54 Furthermore, prasugrel demonstrated a superior effect on the reduction in hsCRP levels compared to that of clopidogrel. 55 Similar effects of ticagrelor 56 are currently under evaluation.
In addition to its antithrombotic effects, aspirin is one of the earliest agents to demonstrate that anti-inflammatory therapies might be effective for the prevention of CVD. Aspirin was shown to decrease the CRP levels in earlier studies (1997). Later, even more studies investigated this effect of aspirin on CRP levels. Although some studies showed a significant effect, 57 others did not. 58 The analysis of data from a recent population-based cohort called the CoLaus study indicated that low-dose aspirin does not significantly impact plasma hsCRP or other proinflammatory cytokine levels. 59 Thus, the anti-inflammatory effect may not play a major role in the cardioprotective effect of aspirin.
Meanwhile, salicylate, which is nonacetylated, is neither a COX inhibitor nor has any effect on bleeding time or platelet aggregation. Salicylate has been shown to have anti-inflammatory effects and to potentially reduce leukocyte counts, possibly due to its effects on NF-kB. The TINSAL-T2D trials conducted on diabetics showed that salicylate reduces HbA1c and CRP. 2 As an inexpensive and well-tolerated agent, its effects on vascular inflammation and atherothrombosis are promising and have yet to be explored.
Conclusion
There is substantial emerging evidence that links inflammation with atherothrombosis. The CANTOS trial demonstrated the clinical efficacy of anti-inflammatory therapy in reducing the thrombotic complications of CVD with no confounding effects on the lipid profile. This helps us to better define the role of antithrombotic and antiplatelet therapy in inflammatory states. Emerging published studies suggest that anti-inflammatory therapy could potentially become a crucial target for preventing cardiovascular events in high-risk patients. Further research studies and analysis are necessary to define how to bring this concept into clinical practice to further reduce mortality and rehospitalizations and transform these agents to be more patient friendly. An important question will be whether efficacy is sufficient to counterbalance potential side effects, such as increased risk for infection.
Footnotes
Authors' Contributions
SJK contributed to the acquisition, analysis and interpretation of the data and drafted the manuscript. RRA made substantial contribution to the concept and design of the study, by critical reading of the article with critical revision and approved the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
