Abstract
The vascular endothelium is a continuous layer of flat polygonal cells that are in direct contact with the blood and participate in responses to inflammation. Chrysin is a flavonoid compound extracted from plants of the genus Asteraceae with a wide range of pharmacological activities and physiological activities. Here, we studied the effects of chrysin on the regulation of the proadhesion and pro-inflammatory phenotypes of the endothelium both in vitro and in vivo. Our results revealed that chrysin strongly inhibited Tohoku Hospital Pediatrics-1 (THP-1) cell adhesion to primary human umbilical vein endothelial cells and concentration-dependently attenuated interleukin 1β-induced increases in intercellular adhesion molecule 1 (ICAM-1), vascular cell adhesion molecule 1 (VCAM-1), and E-selectin messenger RNA levels and ICAM-1 and VCAM-1 protein levels. Previous studies reported that nuclear factor κB (NF-κB) is important in the inflammatory response in endothelial cells, particularly in regulating adhesion molecules, and our data shed light on the mechanisms whereby chrysin suppressed endothelial inflammation via the NF-κB signaling pathway. In addition, our in vivo findings demonstrated the effects of chrysin in the permeability and inflammatory responses of the endothelium to inflammatory injury. Taken together, we conclude that chrysin inhibits endothelial inflammation both in vitro and in vivo, which could be mainly due to its inhibition of NF-κB signaling activation. In conclusion, chrysin may serve as a promising therapeutic candidate for inflammatory vascular diseases.
Keywords
Introduction
The vascular endothelium consists of a continuous layer of endothelial cells 1 that regulates permeability, vascular tension, and the balance between bleeding and coagulation through responses to the cell environment. 2 -4 It has the ability to secrete various factors and to regulate the exudation of leukocytes, plasma, and macromolecules. Under physiological conditions, the endothelium plays a role in anticoagulation, antiproliferation, and inhibition of leukocyte recruitment. Endothelial cells can be activated under circumstances of systematic or local injury, including inflammation, hypercoagulation, and blood stagnation. The early state of vascular injury known as endothelial dysfunction 5 can be followed by high expression of cell adhesion molecules, such as intercellular adhesion molecule 1 (ICAM-1), vascular cell adhesion molecule 1 (VCAM-1), and E-selectin, 6,7 that promote the activation and adhesion of leukocytes and platelets and can consequently contribute to inflammatory disorders such as atherosclerosis 8 and thromboangiitis obliterans. 9,10 In some animal models, the severity of inflammatory disorders, such as multiple sclerosis and inflammatory bowel diseases, can be reduced by blocking cell adhesion molecules. 11,12 As a result, medication inhibiting the expression of adhesion molecules can be considered a candidate to treat or prevent vascular inflammatory diseases.
Chrysin, also called 5,7-dihydroxyflavone (Figure 1), is a flavonoid compound extracted from plants of the genus Asteraceae with a wide range of pharmacological activities and physiological activities including antifibrotic, antioxidant, anti-inflammatory, antitumor, antiallergic, antiestrogenic, and neuroprotective activities. 13 -17 Anti-inflammatory activity is a promising pharmacological effect displayed by chrysin. 18,19 In vitro studies have shown that chrysin may affect several cell signaling pathways. 15 However, the potential impact of chrysin on vascular inflammation has not yet been elucidated. In the current study, we investigated whether chrysin could suppress vascular endothelial inflammation both in vitro and in vivo and the latent-associated regulatory pathways.

Chemical structure of chrysin.
Method
Reagents
Chrysin (Yuanye, Shanghai, China) was diluted to 4 different concentrations (1 mM, 3 mM, 10 mM, and 30 mM) in dimethyl sulfoxide (DMSO). Each solution was added to the cell culture medium at a proportion of 0.1%. The final concentration of DMSO in the medium did not exceed 0.1%, and DMSO alone was used as a vehicle control. Interleukin 1β (IL-1β) was dissolved in ultrafiltered water to a final concentration of 10 μg/mL. Lipopolysaccharide (LPS) and acetoxymethyl ester (BCECF-AM) were obtained from the Beyotime Institute of Biotechnology (Jiangsu, China).
Cell Cultures
Primary human umbilical vein endothelial cells (HUVECs) were cultured at 37°C in 5% CO2 in endothelial medium (No. 1001, ScienCell, San Diego, CA, USA), which contains 5% fetal bovine serum, 1% endothelial cell growth supplement, and 1% penicillin–streptomycin solution. Cells were used for experiments between passages 2 and 5. Human embryonic kidney-293 T (293 T) and human monocyte THP-1 cell lines were purchased from American Type Culture Collection (Mansassas, VA, USA).
Cell Adhesion Assay
Human umbilical vein endothelial cells were seeded into 12-well plates and cultured overnight until confluency was almost reached. Then, chrysin was added into the medium 6 hours before stimulation with IL-1β. Tohoku Hospital Pediatrics-1 (THP-1) cells were incubated with BCECF-AM for 30 minutes in advance. Fluorescently labeled THP-1 cells were added to the HUVEC monolayer. After incubating for 1 hour at 37°C, nonadherent cells were washed away with phosphate-buffered saline (PBS). Then the number of adherent THP-1 cells in each well was counted. The experiment was repeated 3 times.
Western Blotting
Western blotting was performed according to the procedures described previously. 20 The following primary antibodies were used: mouse anti-ICAM-1, mouse anti-VCAM-1, mouse anti-E-selectin (1:1000 dilutions; Proteintech, Wuhan, China), rabbit antiphospho-p65 (1:1000 dilutions; CST, Danvers, MA, USA), rabbit anti-p65 (1:500 dilutions; Selleck, Shanghai, China), mouse anti-phospho-IκBα (1:250 dilutions; Santa Cruz Biotechnology, Heidelberg, Germany), mouse anti-IκBα (1:1000 dilutions; CST), rabbit anti-β-tubulin, and mouse anti-β-actin (1:1000 dilutions; ABclonal, Cambridge, MA, USA). Horseradish peroxidase-coupled secondary antibodies (1:5000 dilutions; Abcam, Cambridge, UK) were used to detect primary antibody binding. Proteins were detected with enhanced chemiluminescence substrate (Millipore, Billerica, MA, USA). All Western blot experiments were carried out at least 3 times.
Extraction of RNA and Real-Time Polymerase Chain Reaction
Human umbilical vein endothelial cells were pretreated with chrysin for 6 hours followed by an additional 4-hour stimulation with IL-1β; then the cells were harvested and total RNA was extracted with TRIzol reagent (TaKaRa, Beijing, China). Reverse transcription kits (TaKaRa) were used to obtain complementary DNA (cDNA). The cDNA was then used for real-time polymerase chain reaction (PCR) under the following conditions: 95°C (20 seconds), 40 cycles of 95°C (3 seconds), and 60°C (30 seconds). β-Actin was used as the reference gene. Gene expression levels were analyzed by the 2−ΔΔCT method. 21 The primer sequences used for PCR are presented in Table S1.
Immunofluorescent Staining for Nuclear Factor κB
Immunofluorescent staining was carried out using a procedure described previously. 22 Human umbilical vein endothelial cells were pretreated with chrysin (10 or 30 μM) for 6 hours followed by stimulation with IL-1β (10 ng/mL) for another 2 hours. Immunofluorescent detection of rabbit antiphospho-p65 antibody (1:100 dilutions; CST) binding was carried out with an Alexa Fluor 594-conjugated secondary antibody (red; 1:800 dilutions; CST), and images were obtained using Olympus cellSens version 1.5 imaging software.
Isolation of Proteins From the Cytoplasm and the Nucleus
The medium was removed, and the cells were washed 3 times with ice-cold PBS. Then the cells were harvested by scraping in PBS, and 500 µL ice-cold hypotonic buffer was added to the suspended cells. The cells were left on ice for 20 minutes before the cell suspension was passed through a 25-G needle 25 times on ice using a 1-mL syringe. The samples were then left on ice for an additional 5 minutes. After centrifuging at 500g for 10 minutes, the supernatant was transferred to a new tube, and the pellet was dispersed with 500 µL ice-cold hypotonic buffer and then passed through a 25-G needle 50 times on ice. The pellet was centrifuged at 500g for 10 minutes and washed with 1-mL ice-cold hypotonic buffer. After centrifuging, the pellet was considered the nuclear fraction and was lysed by 30 µL lysis buffer. The lysed nuclear fraction was sonicated on ice briefly and left on ice for 5 minutes to release the proteins. On the other hand, the supernatant was centrifuged at 3000g for 10 minutes, centrifuged at 100 000g for 30 minutes twice, and then the final supernatant was kept as the cytosolic fraction. The samples (nuclear and cytosolic fractions) were ready to run by SDS-PAGE (sodium dodecyl sulfate-polyacrylamide gel electrophoresis).
Luciferase Reporter Assay
The promoters of ICAM-1, VCAM-1, and E-selectin were cloned into the pGL3 basic vector (Promega, Madison, WI, USA) upstream of the luciferase gene and then the recombinant DNA was introduced into 293 T cells. After growing for a period of time, the cells were treated with chrysin for 6 hours and then the activities of the promoters were measured by a luminometer using the Dual-Glo Luciferase Assay System (Promega).
Model of Acute Inflammation
Male C57BL/6J mice were randomly divided into 5 groups (n = 8 per group) and orally pretreated with vehicle (saline containing 0.1% DMSO), chrysin (40 mg/kg weight or 80 mg/kg weight), or dexamethasone (2 mg/kg weight) once a day for 7 days. 23 Lipopolysaccharide (20 mg/kg weight) was injected into the mice intraperitoneally to induce vascular inflammation. Eighteen hours later, the mice were killed to harvest the lungs, liver, and kidneys for H&E staining.
Statistical Analysis
All experiments were performed in at least triplicate. The Western blotting results were quantified by customized automated image processing software (ImageJ, version 1.47d; NIH, Bethesda, Maryland). The data were assessed by GraphPad Prism 7 (Graph Pad Software, Inc, San Diego, CA, USA). All values were expressed as the mean ± SEM (Standard Error of Mean). One-way analysis of variance and parametric t test were used for statistical comparisons within a group and between groups. A difference was considered to be significant if P < .05 ( #P < .05; ##P < .01; ###P < .001; *P < .05; **P < .01; and ***P < .001).
Results
Chrysin Inhibited Monocyte–Endothelial Cell Adhesion
To investigate the role of chrysin in endothelial inflammation, we first detected its effect on monocyte–endothelial cell interactions. Human umbilical vein endothelial cells, which were pretreated with chrysin for 6 hours and then stimulated with IL-1β, were cocultured with fluorescently labeled THP-1 cells. As shown in Figure 2, the numbers of THP-1 cells adhering to HUVECs were markedly increased when we pretreated HUVECs with IL-1β; however, the numbers were concentration-dependently reduced when chrysin was added to the HUVECs.
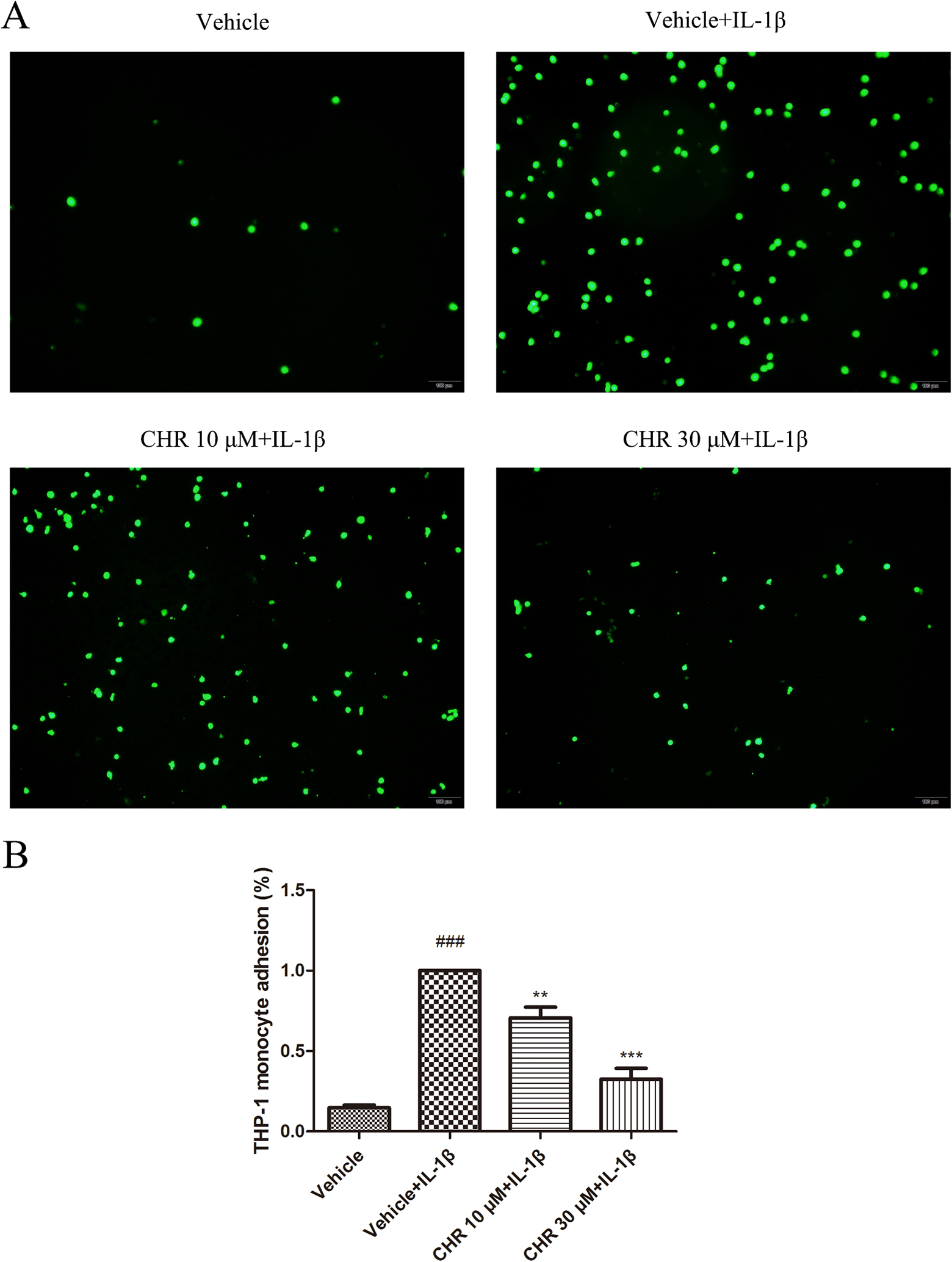
Chrysin inhibits monocyte–endothelial cell adhesion. Human umbilical vein endothelial cells were incubated with DMSO or chrysin for 6 hours and then stimulated with IL-1β (10 ng/mL) for 4 hours. A, Representative images used for quantification of Tohoku Hospital Pediatrics-1 (THP-1) cells (green, BCECF-AM) attached to HUVECs. B, Quantification of adhesion. The results are expressed as the percentage of THP-1 cells adhered to HUVECs in the chrysin treatment group compared to the IL-1β-stimulated group. Data represent the mean ± SEM (Standard Error of Mean) (n = 12 per group) of 3 independent experiments. ### P < .001 versus HUVECs treated with neither IL-1β nor chrysin; **P < .01 and ***P < .001 versus treatment with IL-1β alone. BCECF-AM indicates acetoxymethyl ester; CHR, chrysin; DMSO, dimethyl sulfoxide; HUVECs, human umbilical vein endothelial cells; IL-1β, interleukin 1β.
Chrysin Decreased the Expression of Adhesion Molecules in HUVECs
We detected the expression of adhesion molecules in HUVECs to determine whether chrysin also has an inhibitory effect on these molecules during endothelial cell inflammation. As shown in Figure 3, the messenger RNA (mRNA) expression levels of ICAM-1, VCAM-1, and E-selectin were markedly increased when we pretreated the cells with IL-1β. However, treatment with chrysin suppressed the increase in mRNA expression of ICAM-1, VCAM-1, and E-selectin. Similarly, the protein levels of ICAM-1 and VCAM-1 were significantly reduced by chrysin in HUVECs; the protein levels of E-selectin tended to decrease with chrysin treatment but were not significantly different.

Chrysin decreased the expression of adhesion molecules in HUVECs. Human umbilical vein endothelial cells were incubated with DMSO or chrysin for 6 hours and then stimulated with IL-1β (10 ng/mL) for 4 hours. A-B, Western blot analysis and quantification of the fold changes in ICAM-1, VCAM-1, and E-selectin expression in HUVECs. C, The RT-PCR analysis of ICAM-1, VCAM-1, and E-selectin expression. # P < .05 and ### P < .001 versus HUVECs treated with neither IL-1β nor chrysin. *P < .05; **P < .01; and ***P < .001 versus treatment with IL-1β alone. CHR indicates chrysin; DMSO, dimethyl sulfoxide; HUVECs, human umbilical vein endothelial cells; ICAM-1, intercellular adhesion molecule 1; IL-1β, interleukin 1β; RT-PCR, real-time polymerase chain reaction; VCAM-1, vascular cell adhesion molecule 1.
Chrysin Inhibited IL-1β-Induced Activation of Nuclear Factor κB
The transcription factor, nuclear factor κB (NF-κB) is essential to the inflammatory response in endothelial cells, particularly for regulating adhesion molecules. 24,25 To investigate the effect of chrysin on the NF-κB signaling pathway in HUVECs, the expression levels of p-IκBα, IκBα, p-p65, and p65 in HUVECs were evaluated by Western blot analysis. The results showed that total p65 levels remained unchanged under different treatments, while total IκBα levels were first reduced by IL-1β and then increased with 10 μM or 30 μM chrysin. P-p65 and p-IκBα levels were increased by IL-1β in HUVECs, but the induction was inhibited by treatment with 10 μM or 30 μM chrysin (Figure 4), indicating that chrysin suppressed the IL-1β-induced activation of NF-κB.

Chrysin inhibited the phosphorylation of p65 and IκBα. A, Western blot analysis of phospho-NF-κB p65, total p65, phospho-IκBα, and total IκBα extracted from HUVECs pretreated with DMSO or chrysin with or without IL-1β treatment for 4 hours. B, Quantification of phospho-NF-κB p65 and phospho-IκBα expression. # P < .05 versus HUVECs treated with neither IL-1β nor chrysin; *P < .05 and **P < .01 versus treatment with IL-1β alone. CHR indicates chrysin; DMSO, dimethyl sulfoxide; HUVECs, human umbilical vein endothelial cells; IL-1β, interleukin 1β; NF-κB, nuclear factor-κB.
Chrysin Prevented Nuclear Localization of p65 in IL-1β-Stimulated HUVECs
To determine the effects of chrysin on the phosphorylation of p65, we detected the influence of chrysin on NF-κB nuclear translocation by immunostaining. A decrease in the accumulation of nuclear p65 was observed in HUVECs treated with chrysin (10 or 30 μM) compared to the cells pretreated with IL-1β alone. To confirm this result, nuclear proteins were separated from plasma proteins (Figure 5). Western blotting results showed a concentration-dependent decrease in the nuclear p65 levels in HUVECs pretreated with chrysin; conversely, the plasma p65 levels in chrysin-treated cells exceeded that of cells pretreated with IL-1β alone.

Chrysin prevented nuclear localization of the p65 subunit in IL-1β-stimulated HUVECs. A, The HUVECs were treated with or without CHR (10 or 30 μM) 6 hours prior to IL-1β treatment (10 ng/mL) for 2 hours. Human umbilical vein endothelial cells were stained for p65 (red) and DAPI (4′,6-diamidino-2-phenylindol) (blue; 0.5 μg/mL, 15 minutes). B, Western blot analysis for p65, β-tubulin, and histone 3 in cytosolic (left) and nuclear (right) protein fraction lysates from HUVECs (scale bar: 50 μm). CHR indicates chrysin; HUVECs, human umbilical vein endothelial cells; IL-1β, interleukin 1β.
Chrysin Suppresses the Luciferase Activity of the Promoters of ICAM-1 and VCAM-1
To determine whether chrysin affects the transcriptional activity of the ICAM-1, VCAM-1, and E-selectin genes, reporter gene constructs cloned into the Renilla plasmid were introduced into separate 293 T cell cultures. The results demonstrated that the promoters of ICAM-1, VCAM-1, and E-selectin were activated in 293 T cells. As shown in Figure 6, the results of the ICAM-1 and VCAM-1 assessments indicated that luciferase activity decreased remarkably and concentration-dependently with chrysin treatment (10 or 30 μM). The luciferase activity of the ICAM-1 promoter in the groups treated with 10 μM and 30 μM chrysin was 36% and 70% lower, respectively, than that of the 293 T cells not treated with chrysin. Similarly, the luciferase activity of VCAM-1 was reduced by chrysin by 21% and 52%, respectively. Although the luciferase activity of the E-selectin promoter tended to decline, the difference was not statistically significant.

Chrysin suppresses the luciferase activity of the promoters of ICAM-1 and VCAM-1, but not E-selectin. Luciferase activity is represented as fold change versus empty vector. Error bars represent SEM (Standard Error of Mean) of triplicate experiments. **P < .01 and ***P < .001 versus treatment without chrysin. CHR indicates chrysin; ICAM-1, intercellular adhesion molecule 1; VCAM-1, vascular cell adhesion molecule 1.
The Effect of Chrysin on the Acute Inflammatory Response In Vivo
A model of acute inflammation was built to examine the inhibitory effect of chrysin on inflammatory injury in vivo, including assessing leukocyte infiltration and increased vascular permeability. We observed the potential role of chrysin in regulating acute inflammatory responses in murine lungs, liver, and kidneys that were challenged with LPS. As shown in Figure 7, LPS caused a dramatic increase in leukocyte infiltration in the lungs, liver, and kidneys 18 hours after injection. However, chrysin (40 mg/kg weight or 80 mg/kg weight) pretreatment alleviated injuries, such as leukocyte infiltration, erythrocyte aggregation, and interstitial edema, compared to the vehicle control (saline containing 0.1% DMSO). These results corresponded to our in vitro findings, indicating that chrysin suppressed leukocyte–endothelial cell adhesion and inflammation.
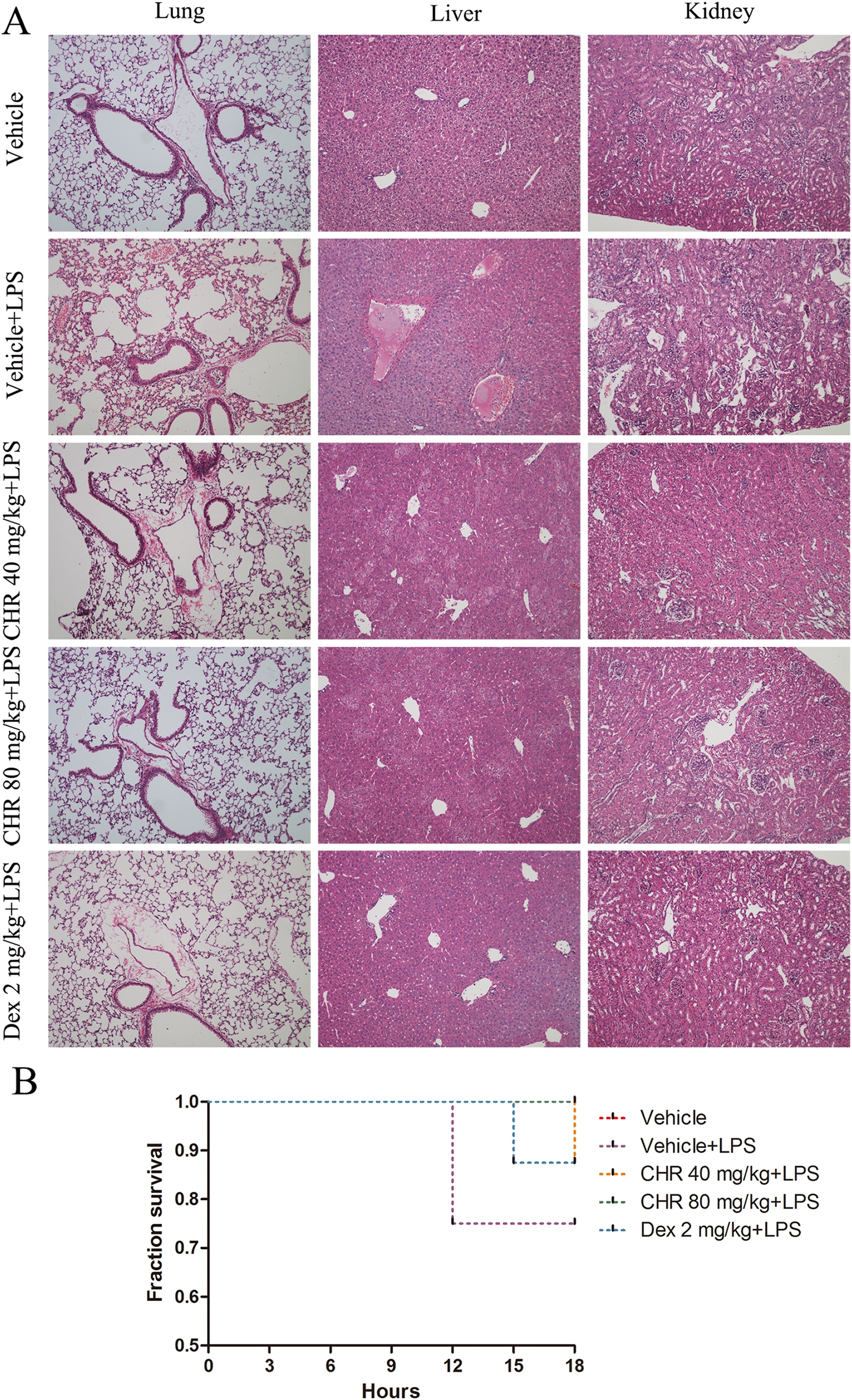
Effect of chrysin on the acute inflammatory response in vivo. A model of acute inflammation was used to investigate leukocyte infiltration in vivo. Male C57BL/6J mice were orally pretreated with vehicle (saline containing 0.1% DMSO) or chrysin (40 mg/kg weight or 80 mg/kg weight) or dexamethasone (2 mg/kg weight) once a day for 7 days. Lipopolysaccharide (20 mg/kg weight) was injected into mice intraperitoneally to induce vascular inflammation. Eighteen hours later, mice were killed to harvest the lungs, liver, and kidneys for H&E staining. Representative images show H&E staining for leukocyte accumulation in the lungs, liver, and kidneys. (n = 8 mice per group). B. Survival curves for mice injected intraperitoneally with a lethal dose of LPS (20 mg/kg; n = 8 mice per group). DMSO indicates dimethyl sulfoxide; LPS, lipopolysaccharide.
In addition, we calculated the survival rate of mice in the 18-hour period after being challenged with a lethal dose of LPS (20 mg/kg). All mice pretreated with chrysin at the high dose (80 mg/kg weight) recovered from this challenge, giving the group an 18-hour survival rate of 100% (Figure 7B), which was the same as the vehicle control group. However, the 18-hour survival rate in the group treated with LPS alone was 75%. When we pretreated mice with a low dose of chrysin (40 mg/kg weight), the survival rate reached 85%.
Discussion
The vascular endothelium is of great importance in inflammation due to its barrier function separating the blood and all the tissues. 26,27 During inflammation, cytokines confer a permeable, proadhesive and prothrombotic phenotype on the endothelium. 28 -30 However, if vascular inflammation persists, these sustained responses may result in endothelial dysfunction, which contributes to the pathogenesis of vascular diseases, 31 -33 such as atherosclerosis. 34
Adhesion molecules participate in the adhesion of monocytes to vascular endothelial cells, making the adhesion molecules pro-inflammatory during inflammation. 35 Adhesion molecules, including ICAM-1, VCAM-1, and E-selectin, play an important role in activating, promoting, and sustaining leukocyte recruitment to lesion areas in inflammatory vascular diseases. 36 -38 This study demonstrated for the first time that chrysin, an active compound isolated from the genus Asteraceae, dynamically inhibited the adhesion of THP-1 cells to HUVECs and concentration-dependently attenuated the IL-1β-induced increase in the mRNA levels of ICAM-1, VCAM-1, E-selectin and the protein levels of ICAM-1 and VCAM-1. Medication regulating the expression of adhesion molecules and pro-inflammatory cytokines may be a potential therapy for inflammatory vascular diseases.
Inflammatory responses have been found in all the stages of inflammatory vascular disorders, such as atherogenesis, 39 and the transcription factor NF-κB is regarded as an effective regulator of inflammation due to its ability to regulate the transcription of pro-inflammatory genes. 40 Previous studies reported that NF-κB activation is a crucial part of the development of atherosclerosis and is proatherogenic in endothelial cells. 41,42 Conversely, inhibition of NF-κB signaling may have potential effects on the prevention of atherosclerosis. Our data shed light on the mechanisms used by chrysin to suppress endothelial inflammation via inhibiting the NF-κB signaling pathway. Nuclear factor κB in the cytoplasm is regulated by inhibitors of κB (IκB), and the phosphorylation and ubiquitin-mediated degradation of IκBα are involved in receptor-mediated NK-κB activation. 43,44 As shown in Figure 4, chrysin significantly suppressed the IL-1β-induced phosphorylation and subsequent degradation of IκBα and the phosphorylation of p65 in HUVECs. To further confirm whether chrysin also has an inhibitory effect on the nuclear translocation of p65, we determined the location of p65 under different circumstances, and the results showed that a large quantity of p65 transferred from the cytoplasm to the nucleus after treatment with IL-1β, but p65 in cells pretreated with chrysin was mainly found in the cytosol. Similarly, the results of Western blotting revealed that nuclear p65 levels were decreased by chrysin treatment, while cytoplasmic p65 levels in chrysin-treated HUVECs outpaced the levels in purely IL-1β-stimulated cells. These results suggested that chrysin suppressed IL-1β-induced phosphorylation and degradation of IκBα, phosphorylation of p65, and p65 nuclear translocation in HUVECs. Therefore, chrysin has a protective effect on the endothelium against inflammation by inhibiting NF-κB signaling (Figure 8).

Schematic illustration of possible underlying mechanisms responsible for the anti-inflammatory potential of chrysin. Chrysin inhibited IL-1β-induced phosphorylation and degradation of IκBα, phosphorylation of p65, and subsequent NF-κB p65 nuclear translocation, thereby attenuating inflammation in endothelial cells. In addition, chrysin suppressed the activity of the promoters of adhesion molecules, such as ICAM-1 and VCAM-1, by transcriptional regulation. ICAM-1 indicates intercellular adhesion molecule 1; IL-1β, interleukin 1β; NF-κB, nuclear factor κB; VCAM-1, vascular cell adhesion molecule 1.
On the other hand, to examine the possible mechanisms of transcriptional regulation by which chrysin decreases ICAM-1, VCAM-1, and E-selectin levels, we asked whether chrysin could reduce the activity of the promoters of these adhesion molecules. To determine this, reporter gene constructs cloned into a Renilla plasmid were transfected into separate 293 T cell cultures. The results of the ICAM-1 and VCAM-1 assays showed that the luciferase activity decreased progressively and concentration-dependently with chrysin treatment, while the luciferase activity of the E-selectin promoter merely tended to decline with no statistical significance. Further studies will be required to determine whether there are binding sites for chrysin and NF-κB.
In addition, vascular endothelial cells are crucial in LPS-induced inflammatory responses. 40 Dysfunction of the endothelium results in increases in pro-inflammatory cytokine production and permeability/leakage. 23 Using a mouse model of acute inflammation, we have shown that 18 hours after injection with LPS, mice that were not treated with chrysin responded to LPS injection by recruiting leukocytes into the tissues. However, mice pretreated with chrysin dramatically decreased vascular permeability in several vascular beds. This blunted permeability response could contribute to the faster resolution of vascular inflammation observed in the mice treated with a high dose of chrysin. Our in vivo findings demonstrated that chrysin has a reducing effect on the permeability and inflammatory responses of the endothelium to injury, and further investigations into the potential uses of chrysin, such as in diabetic angiopathy or atherosclerosis, are required.
Altogether, our current studies indicate that chrysin reduces endothelial inflammation both in vitro and in vivo, and the results of the in vitro studies imply that the effects of chrysin could be mainly due to suppression of NF-κB signaling activation. These findings suggest that chrysin could attenuate the proadhesion phenotype and permeability of the endothelium during inflammation and identify chrysin as a promising therapeutic candidate for inflammatory vascular diseases.
Supplemental Material
Supplemental_Additional_Document - Chrysin Suppresses Vascular Endothelial Inflammation via Inhibiting the NF-κB Signaling Pathway
Supplemental_Additional_Document for Chrysin Suppresses Vascular Endothelial Inflammation via Inhibiting the NF-κB Signaling Pathway by Shengnan Zhao, Minglu Liang, Yilong Wang, Ji Hu, Yi Zhong, Jia Li, Kai Huang and Yiqing Li in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
Shengnan Zhao and Minglu Liang contributed equally as first authors. Y. Li and K. Huang conceived and designed the experiments. S. Zhao and M. Liang performed the experiments and prepared the manuscript. Y. Wang, J. Hu, J. Li, and Y. Zhong participated in discussions of data analysis. Y. Li and K. Huang revised critically the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been supported by the National Natural Science Foundation of China (81570405) and Major Key Technology Research Project of Science and Technology Department in Hubei Province of China (2016ACA151).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
