Abstract
Introduction
The portal venous system consists of the mesenteric, splenic, and portal veins, and it is peculiar in that its communication with the circulation system is not direct but mediated by the hepatic sinusoids. When occlusion occurs in a portal vessel, it causes congestion of the organs it drains, with increased venous pressures and the development of collateral circulation. 1 The results of portal vein thrombosis (PVT) depend on the extension of the thrombus. Upstream from the thrombus, there is little effect on the intestine on condition that the mesenteric venous arches remain patent. Ischemia occurs from the extension of the thrombus into the mesenteric veins and the mesenteric venous arches. 2 It is likely that thrombosis of the arches prevents their function as a collateral circulation to drain intestinal blood toward the adjacent patent sites. In addition, reflex arteriolar vasoconstriction might occur when the arches are thrombosis. 2 If ischemia is prolonged for several days, intestinal infarction may occur, and in 20% to50% of the cases, intestinal infarction is responsible for death as a consequence of peritonitis and multiple organ failure, even in cases where the infarcted gut is resected. 3 One of the main causes of short bowel syndrome is extensive intestinal resection due to venous thrombosis. Short bowel stenosis can be a late sequel of mesenteric venous ischemia. 4 Downstream from the PVT, the consequences for the liver are hardly noticeable. Clinical signs of the liver disease are absent or transient unless thrombosis occurs in a patient with cirrhosis. 3 Many medical conditions can lead to PVT. Sarin et al 5 summarized these conditions into 3 categories: first, conditions that can lead to portal vein injury and subsequent obstruction such as intra-abdominal infection, including peritonitis; second, portal vein anomaly, including portal vein stenosis and atresia; and finally hypercoagulable states. These conditions are strongly associated with PVT (Table 1). 6 Mean platelet volume (MPV) is a blood parameter commonly used in determining thrombocyte sizes, which yields results in a short time, has a low cost, and can be detected in routine blood tests. 7 The MPV reflects the size of the thrombocytes, and it is accepted as a marker in determining thrombocyte function. Large platelets, when compared to smaller platelets, are enzymatically and metabolically more active and have more prothrombotic potential. 8 A rise in MPV levels increase platelets’ aggregation, thromboxane synthesis, b-thromboglobulin secretion, and expression of adhesion molecules. 9 Platelets secrete and express a large number of substances that are crucial mediators of coagulation, inflammation, thrombosis, and atherosclerosis. 10,11 Our aim in this study was to investigate whether MPV, which is associated with much vascular pathology, can be a new etiological factor in PVT.
Main Etiological Factors of Portal Vein Thrombosis.

Materials and Methods
The patients were separated into 2 groups: group I, control (n = 35) and group II, PVT (n = 34). The PVT group consisted of 34 patients who presented at the Dicle University Medical Faculty with stomachache of undetermined etiology and who were diagnosed with PVT with the support of the clinic, laboratory, and radiologic imaging techniques. The patients with PVT were further separated into 2 subgroups: acute and chronic thrombus. Patients’ files were scanned retrospectively. Age, gender, smoking, hypertension, diabetes mellitus (DM), hemoglobin (Hb) at the time of referral to hospital (g/dL), white blood cell (WBC; mm3 × 103), MPV (fL), platelet (PLT; mm3 × 103), and Doppler ultrasonography (USG) results were recorded. The acute–chronic identification of the thrombus on the portal vein was accomplished with Doppler USG. All patients involved in the study, except the control group, were given low-molecular-weight heparin treatment for anticoagulation.
Laboratory Data
In all patients, the blood samples were drawn from the antecubital vein after a fasting period of 12 hours. In order to avoid PLT swelling induced by EDTA, the measurements were performed immediately after blood sampling. The MPV, Hb, PLT count, and WBC count were measured. For analysis of the hematologic parameters, blood samples with K3 EDTA were processed by a Sysmex XT-2000i analyzer that uses the electric resistance detecting method with hydrodynamic focusing and fluorescence flow cytometry (Sysmex, Kobe, Japan).
Statistical Analysis
To analyze the study results statistically, the SPSS Windows 11.5 (SPSS Inc, Chicago, Illinois) program was used. The quantitative data were indicated as mean ± standard deviation. The Kolmogorov-Smirnov test was utilized for the compatibility of normal distributed data. In comparing the groups, the Mann-Whitney U test was used to analyze nonparametric data, and Student t test was used for parametric data, while the chi-square test was used in the analysis of categorical data. The P values <.05 were accepted as significant for all variables. Receiver–operating characteristic (ROC) analyses were used to determine the cutoff values and the sensitivity/specificity of the MPV.
Results
The PVT group consisted of 10 male and 24 female patients, while the control group consisted of 16 males and 19 females (P = .216). The average age of the PVT group was 43.7 ± 15.4 and that of the control group was 46.3 ± 9.6 (P = .255). There was no determined difference between the 2 groups in terms of DM, hypertension, or cigarette smoking (P > .05). Average Hb levels were 10.8 ± 2.1 in the PVT group, while they were 14.0 ± 1.7 in the control group (P < .001). There was no significant difference when leukocyte values and thrombocyte values were compared (P = .060; P = .680). The average MPV value of the PVT group was 8.2 ± 0.52, while the control group’s average was 7.8 ± 0.62 (P = .012; Table 2). According to Doppler USG results, only thrombocyte levels were different between the patients with acute and chronic thrombus; the other parameters were the same (P < .05; Table 3). In the ROC analysis performed, when the control group and patients with PVT were compared, the cutoff value was 7.9 (area under curve [AUC]: 0.674), sensitivity was 70.6%, and specificity was 65.7% for the patients with PVT (P = .013; Figure 1).
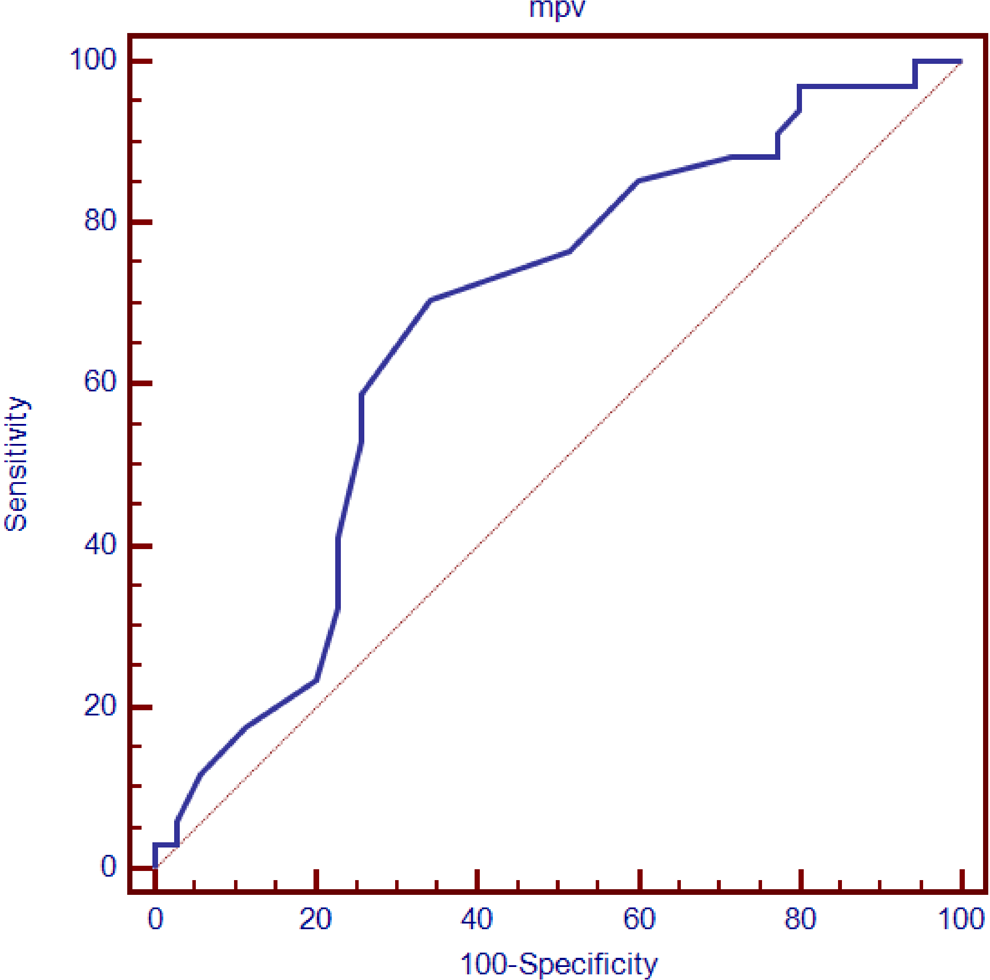
Receiver–operating characteristic curves for predictors of portal vein thrombosis. The mean platelet volume values area under the curve, 0.674; green line; reference line.
Demographic Features and Laboratory Values of the Patients and Controls.
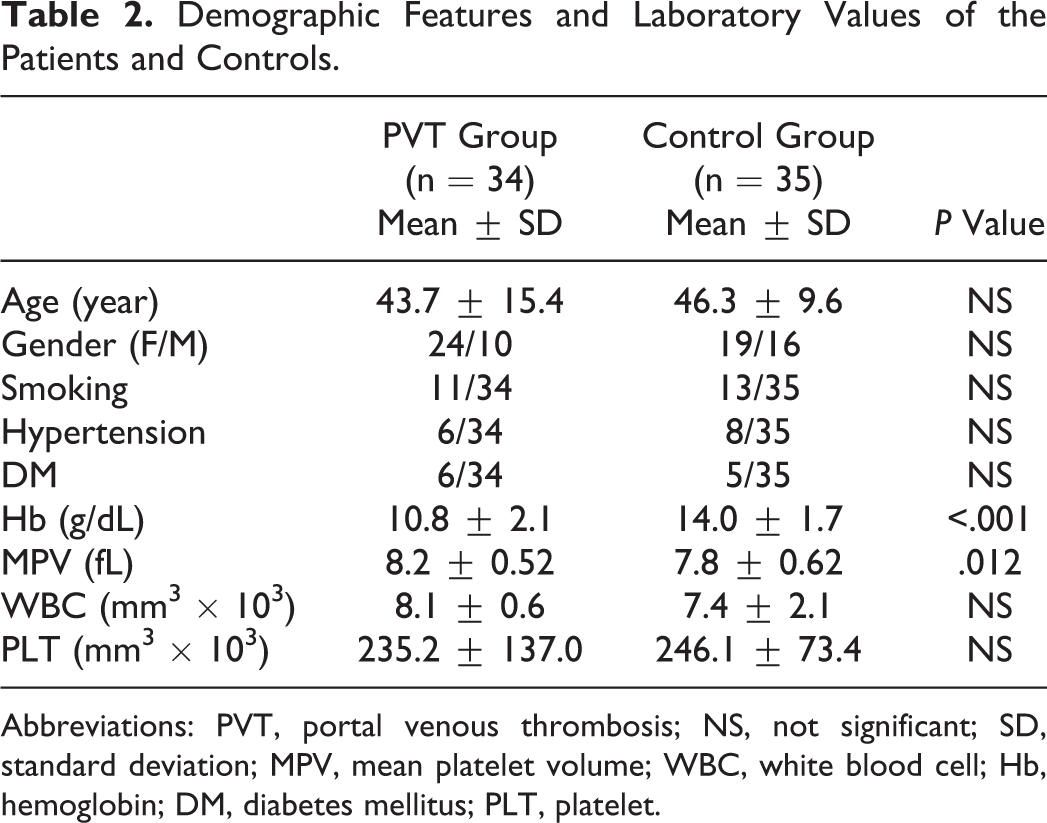
Abbreviations: PVT, portal venous thrombosis; NS, not significant; SD, standard deviation; MPV, mean platelet volume; WBC, white blood cell; Hb, hemoglobin; DM, diabetes mellitus; PLT, platelet.
Comparison of Laboratory Values of Patients With Acute and Chronic Portal Venous Thrombosis.
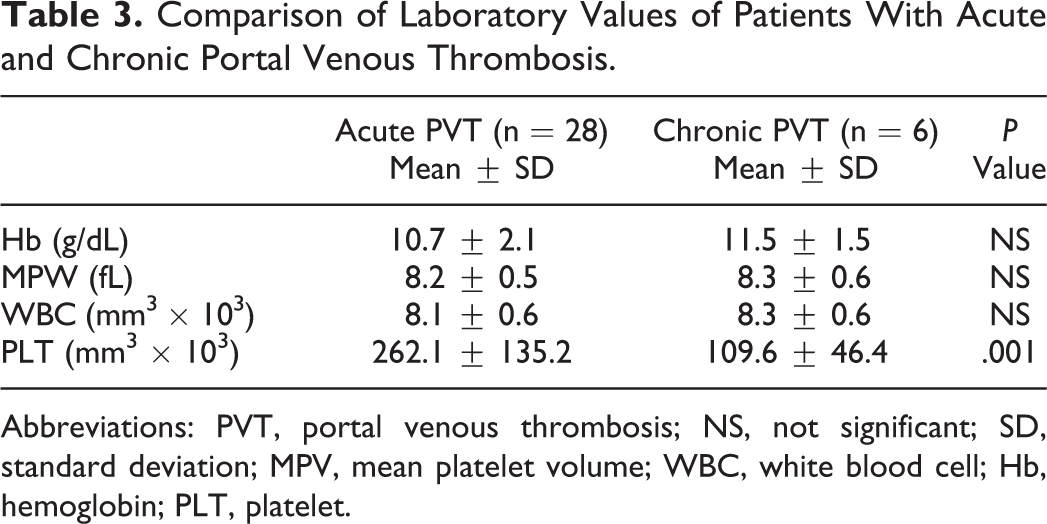
Abbreviations: PVT, portal venous thrombosis; NS, not significant; SD, standard deviation; MPV, mean platelet volume; WBC, white blood cell; Hb, hemoglobin; PLT, platelet.
Discussion
Although patients with acute PVT are presented to the hospital with symptoms such as stomachache, distension, diarrhea, nausea, vomiting, and bleeding in, patients with chronic PVT are usually asymptomatic and present with portal hypertension. 12 Liver cirrhosis in 24% to 32% of the patients, malignancies in 21% to 24%, myeloproliferative diseases and coagulation disorders in 10% to 12%, and intra-abdominal infections in some were responsible for the etiology of PVT; in 8% to 15% of the patients no reason was determined. 13 According to a recent hypothesis, venous thromboses generally occur only when several factors are combined. 14 These factors include inherited or acquired prothrombotic disorders, other thrombophilic factors, and local factors. Prothrombotic disorders are characterized by features of coagulation activation, while thrombophilic factors refer to a more general tendency toward thrombosis 15 (Table 1). These etiological factors were detected in the patients included in this study. Diagnosis should be suspected in many different situations such as abdominal pain, abdominal sepsis, gastrointestinal bleeding due to portal hypertension, and fortuitous finding of the portal hypertension. In the past, thrombosis of the portal vessels was diagnosed with angiography or splenoportography. Today, the vessels of the splanchnic area can be accurately explored with noninvasive diagnostic methods such as ultrasound or color Doppler USG. 1 In our study, all patients were diagnosed using Doppler USG. In addition, we used Doppler USG to distinguish between acute and chronic PVT. The PLTs secrete and express a large number of substances that are crucial mediators of coagulation, inflammation, thrombosis, and atherosclerosis. 10,11 The MPV is a simple marker that indicates PLT function and activation that can be determined by complete blood count (CBC) counts, at no additional cost; it is influenced by inflammation. Increased MPV levels are determined in conditions such as metabolic syndrome, myocardial infarction, acute ischemic stroke, DM, 16 hypertension, 17 hypercholesterolemia, 18 smoking, 19 and obesity. 20 In our study, the control group was chosen to be homogenous with the PVT group in terms of risk factors. Therefore, there were no significant differences (P < .05) between the control group and the PVT group in age, gender, smoking, DM, or hypertension. When Hb levels were compared, it was determined that Hb levels were lower in the PVT group (P < .001). No differences were detected between the 2 groups in terms of WBC and PLT (P > .05). Berger et al 21 found that the MPV levels increase in peripheral artery disease; Gulcan et al 22 found that the MPV levels increase in deep venous thrombosis; and Varol et al 23 found that the MPV levels increase in patients with aortic stenosis. Braekkan et al 24 reported that MPV is a risk factor in venous thromboembolism. In our study, we determined that the average MPV values were higher in the PVT group when compared to the control group (P = .012). In the ROC analysis we performed in the study, when the control group and the PVT group were compared, for the MPV levels in the PVT group, the sensitivity was determined as 70.6%, specificity as 65.7%, and AUC as 0.674 (P = .013). No differences between parameters, other than PLT, were determined when the laboratory findings of patients with acute PVT and chronic PVT (P = .001) were compared (P > .05). The limitations of the study are the retrospective nature of the study and the lack of a very large number of patients. Studies conducted on a large prospective series would help us to understand the role of MPV in the etiology of PVT.
Conclusion
The MPV is a low-cost blood parameter that can be determined in routine blood tests, yields results in a short time, and is used commonly in determining platelet sizes. The risk of thrombosis is increased due to the platelets’ larger size and increased aggregation capabilities in higher MPV. In our study, we showed that MPV levels were significantly increased in patients with PVT of undetermined etiology compared to the control group. This result indicated that MPV could be a new etiologic risk factor for PVT, with the support of new clinic and laboratory studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
