Abstract
Introduction
Deep vein thrombosis (DVT), a part of clinical diagnosis called venous thromboembolism, is an important cause of morbidity and mortality especially in hospitalized patients. Various predisposing clinical conditions for higher risk of DVT are prolonged immobility and postoperative bed rest, malignancy, advanced age, cardiorespiratory and other organ failures, neurological disorders, and a wide variety of inherited and acquired hematological diseases. 1
Circulating platelets are heterogeneous in size, density, and reactivity.2,3 Mean platelet volume (MPV) shows platelet size and may reflect platelet function and activity with platelet production and stimulation indirectly. Elevated values of MPV can result in cardiovascular diseases,4,5 since larger platelets are more reactive than the normal size ones. 6 However, less clear data are present whether MPV and other morphometric parameters including mean platelet mass (MPM) and mean platelet component (MPC) may be used as indicators of platelet function and activation in patients with DVT. Therefore, in the present study, we aimed to measure MPV, MPM, and MPC in patients with DVT and compare the results with controls.
Methods
Study Population
A total of 6311 consecutive ultrasound examinations performed on the lower extremities of 2629 patients (the mean study per patient was 2.4) during the 2-year study period, from January 2009 to October 2010, were possible candidates. A total of 4964 (78.7%) examinations had normal Doppler findings. Of these, randomly chosen 210 (4.2%) examinations were recorded as the control group. A total of 934 (14.8%) examinations had venous regurgitation and were excluded. Remaining 203 (3.2%) examinations (study group) had DVT in lower extremities. Therefore, finally, 413 examinations (200 male [48.4%] and 213 female [51.6%], mean age, 58.8 ± 16.4; age range, 19-94) admitted to the department of radiology for compression ultrasonography of the lower extremity were included in the study. Only patients having positive results for lower extremity DVT were included in the study group (n = 203). Patients with negative results formed the control group (n = 210). Distal (calf vein) and proximal (popliteal, femoral, and iliac) location of the DVT were recorded. A written consent was obtained from all patients, and our local ethical committee approved the study.
Duplex Ultrasonography
Patients were examined using a GE LOGIQ 9 duplex ultrasound machine equipped with a 12-MHz linear transducer. Duplex ultrasonography is a highly sensitive and specific diagnostic tool for the detection of DVT, either acute or chronic, although it is operator dependent. Invasive venography is the gold standard technique for diagnosis. However, noninvasive, safe, and less expensive characteristics of duplex ultrasonography have made this tool the preferred method. Lack of compressibility (the major criterion), increased cross-sectional diameter, heterogeneous intraluminal material, absence of Doppler signal, and collateralization were the diagnostic ultrasound criteria for DVT. Venous thrombosis was classified into 2 categories, depending on location: proximal (involving the iliac, femoral, and popliteal veins) and distal (involving the below-knee deep veins).7,8
Laboratory Data
Fasting peripheral blood sample was drawn from the antecubital vein using a 21-guage needle with a vacutainer and minimal stasis. The blood sample was collected into the tubes containing 2.7 mL EDTA for platelet count, MPV, MPM, and MPC analyses. Using an automated system (the ADVIA 120 Hematology System, Bayer HealthCare, Diagnostics Division, Tarrytown, New York), MPC (g/dL), MPV (in fL), and MPM (in pg) were measured. These indexes were obtained from the histograms of 2-dimensional light scatter signals that were converted into platelet count, volume, and granularity values. The MPM (in pg) is computed from the MPV and MPC. The intra-assay and inter-assay coefficients of variation were <3% and <7%, respectively. The measurements were performed within 60 minutes after vein puncture to rule out the possible swelling of platelets. 9 All the laboratory studies were carried out in blinded fashion with respect to the identity of the blood samples.
The reference ranges in our hematology laboratory are MPV is 7.5 to 11.5 fL; MPM is 1.7 to 2.5 pg; and MPC is 24.5 to 26.9 g/dL.
Statistical Analysis
Data were analyzed with the SPSS Software Version 15.0 for Windows (SPSS Inc., Chicago, Illinois). Continuous variables were presented as mean ± standard error and categorical variables as frequency and percentage. The Kolmogorov-Smirnov test was used to assess the distribution of continuous variables. Student’s t test was used to compare normally distributed continuous variables and the Mann-Whitney U test for variables without normal distribution. The Pearson χ2 test or the Fisher exact test was used to compare categorical variables. Any correlation between MPV and platelet count was analyzed with the Pearson analysis. Separate logistic regression analyses were performed to identify univariate predictors of DVT, and a subsequent stepwise (forward conditional) regression analysis was performed (parameters with a P value of <.1 in univariate analysis were included in the model). Receiver–operating characteristic (ROC) analyses were used to determine the cutoff values and the sensitivity/specificity of platelet indices. The odds ratios (ORs) and the 95% confidence intervals (CIs) were calculated. A 2-tailed P value of <.05 was considered statistically significant.
Results
Baseline demographic, clinical, and laboratory characteristics of the study population were outlined in Table 1 There was no significant difference between the 2 groups regarding age, sex, clinical risk factors including hypertension, diabetes, and smoking, and laboratory parameters including cholesterols and fasting plasma glucose, blood pressure values, white blood cell count, hemoglobin, and platelet count.
Baseline Demographic, Clinical and Laboratory Characteristics of the Study Population
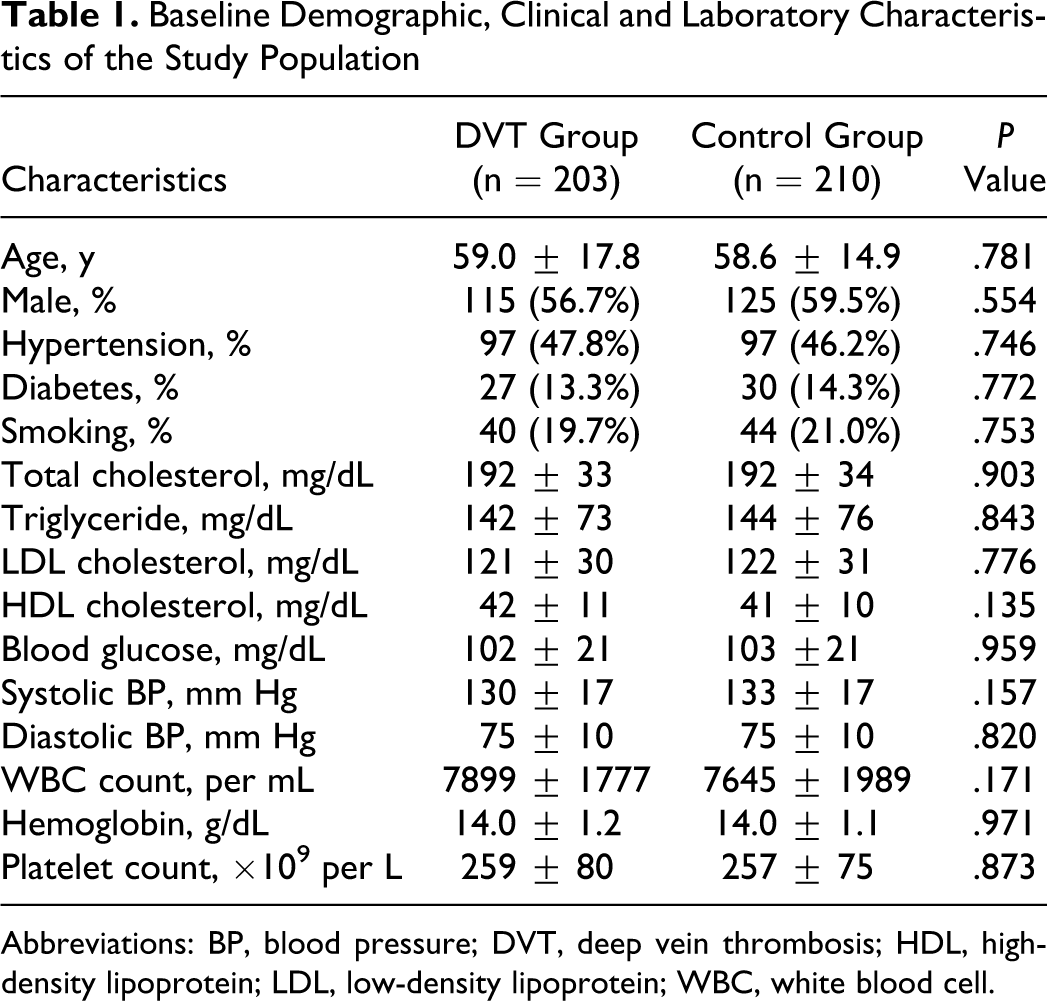
Abbreviations: BP, blood pressure; DVT, deep vein thrombosis; HDL, high-density lipoprotein; LDL, low-density lipoprotein; WBC, white blood cell.
According to the time scale of DVT, 84 (41.4%) patients had acute, 67 (33.0%) had subacute, and 52 (25.6%) had chronic DVT. In all, 81 (39.9%) of the DVTs were in the right-hand side, 105 (51.7%) in the left-hand side, and the remaining 17 (8.4%) had bilateral DVT.
One of the primary outcomes of MPV was significantly higher in DVT group compared with the control (8.6 ± 1.3 fL vs 7.9 ± 0.5 fL [95% CI −0.82 to −0.44], P < .001, respectively; Figure 1). The second primary outcome of MPM was also significantly higher in DVT group compared with the control (2.2 ± 0.3 pg vs 2.0 ± 0.1 pg [95% CI −0.20 to −0.11], P < .001, respectively; Figure 2). The last primary outcome of MPC was, however, significantly lower in DVT group compared with the control (24.9 ± 3.2 g/dL vs 26.3 ± 1.6 g/dL [95% CI 0.91 to 1.89], P < .001, respectively; Figure 3). In addition, there was a significant inverse relationship between MPV and platelet count (r = −.188, P < .001; Figure 4).
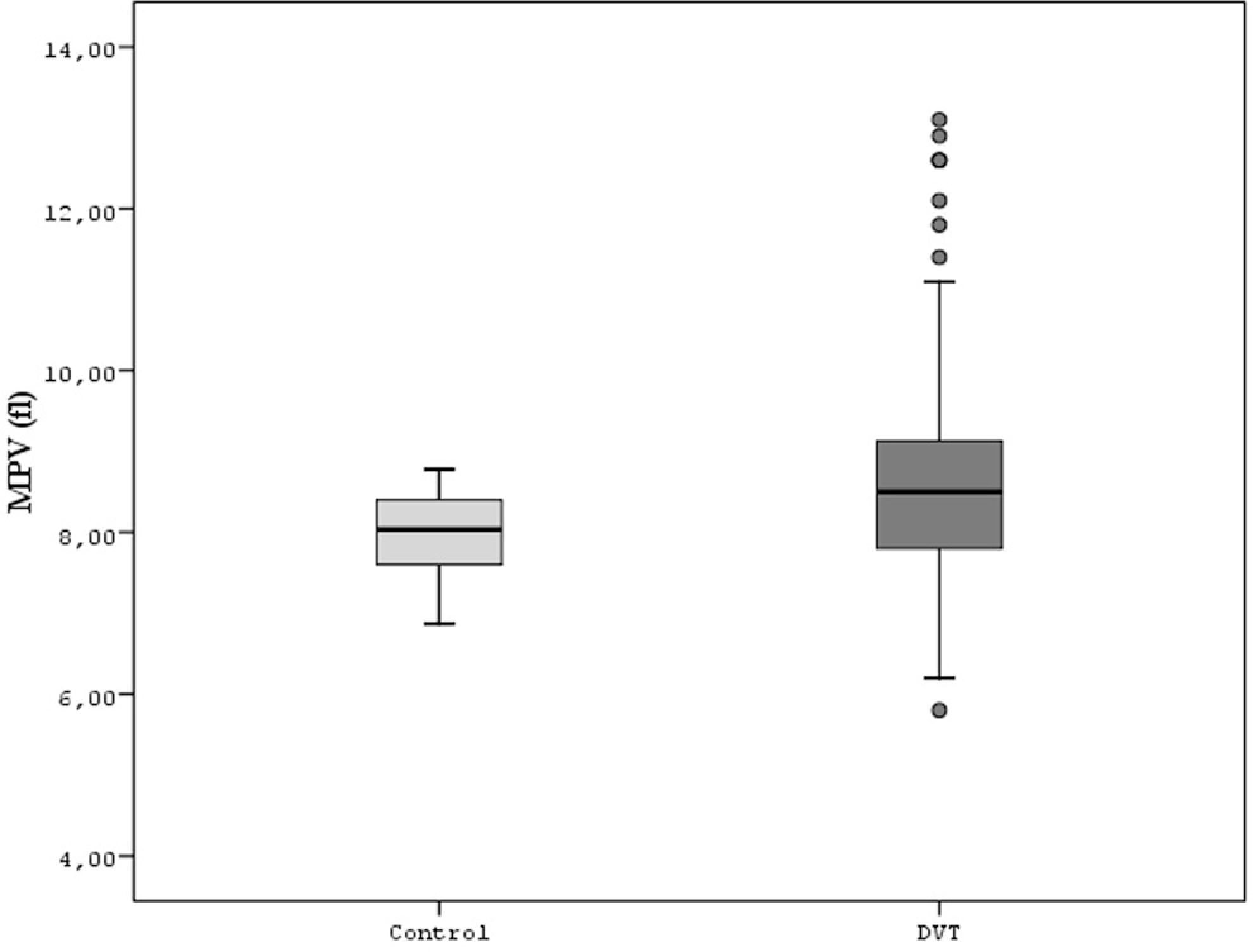
Comparison of both the groups for mean platelet volume.
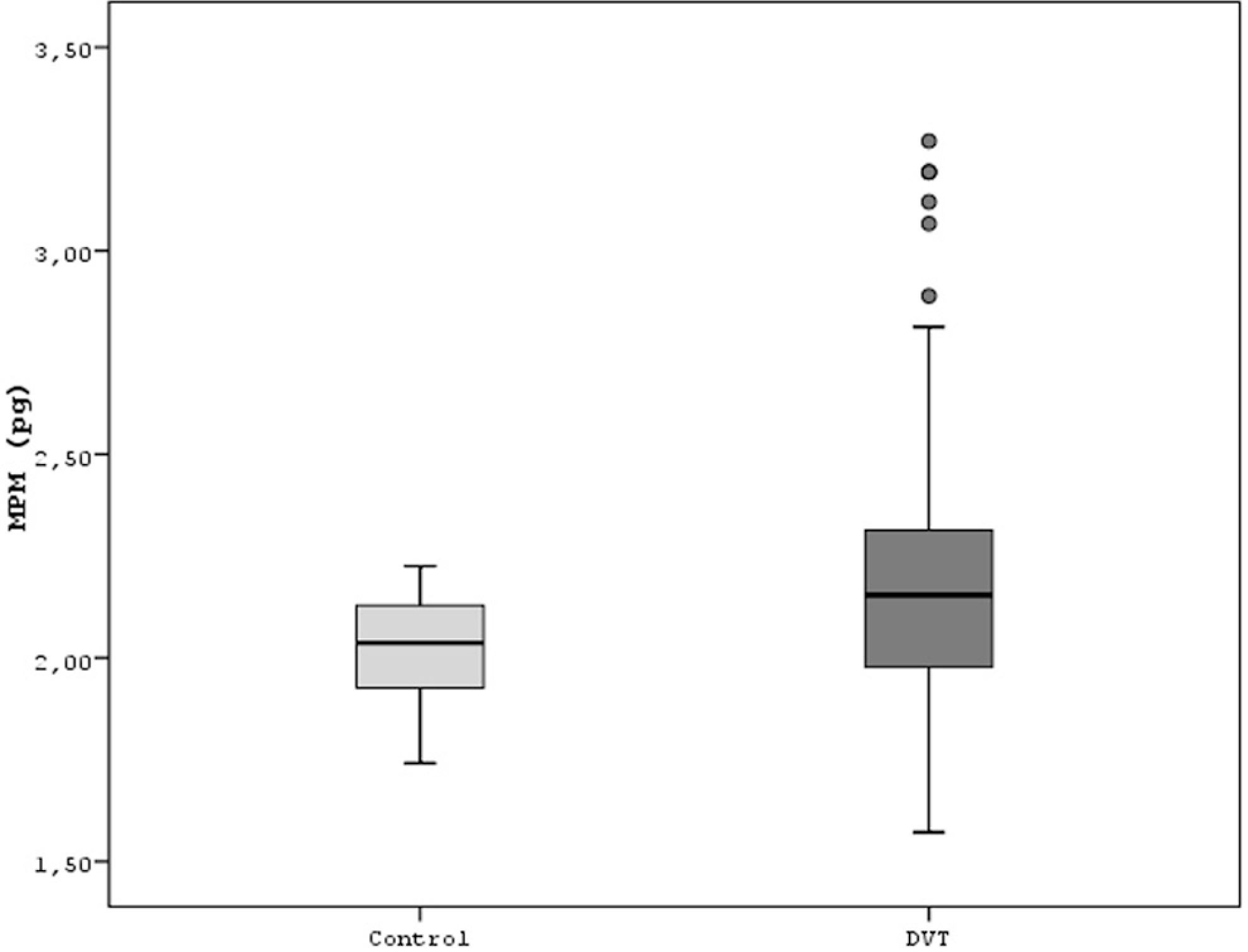
Comparison of both the groups for mean platelet mass.
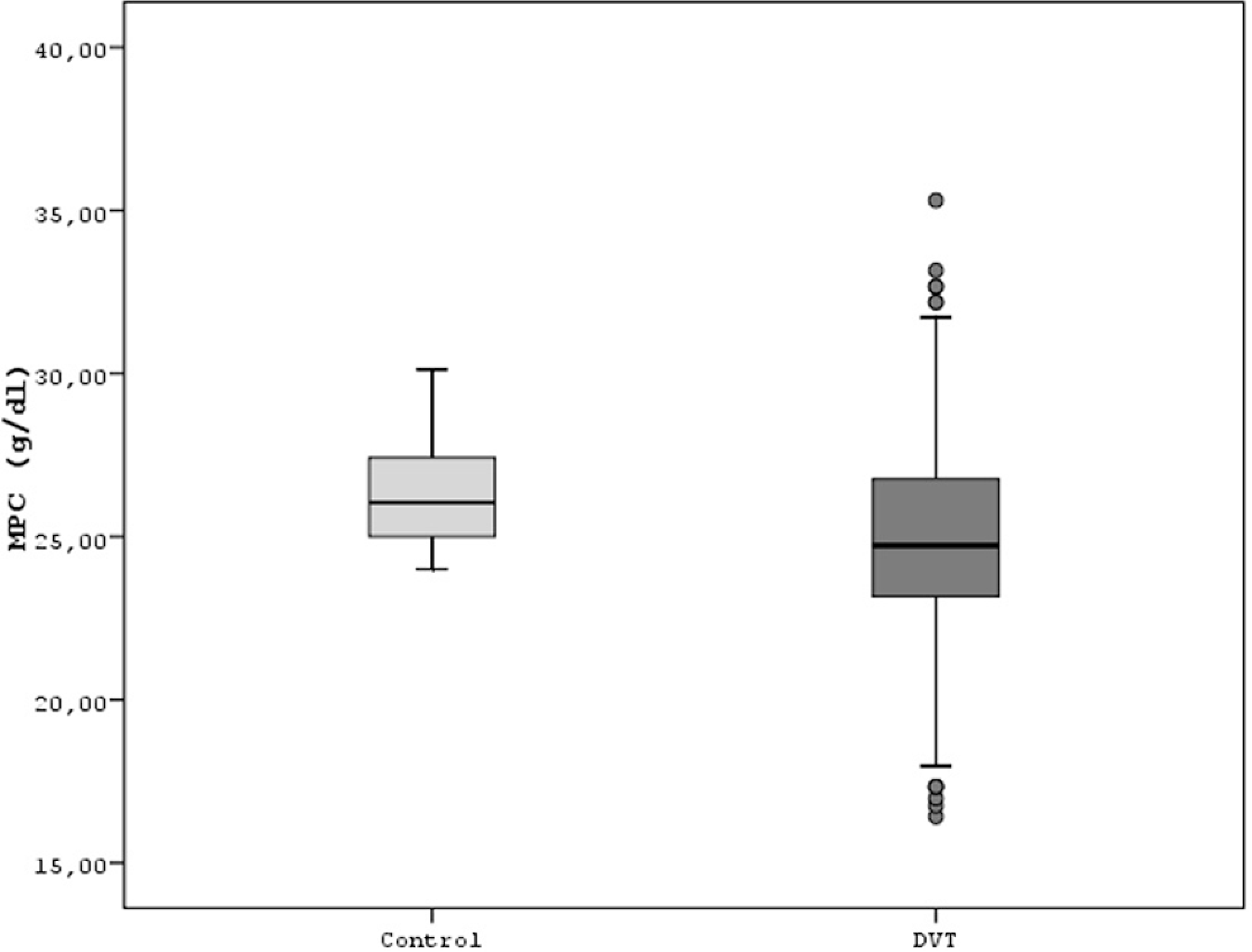
Comparison of both the groups for mean platelet component.
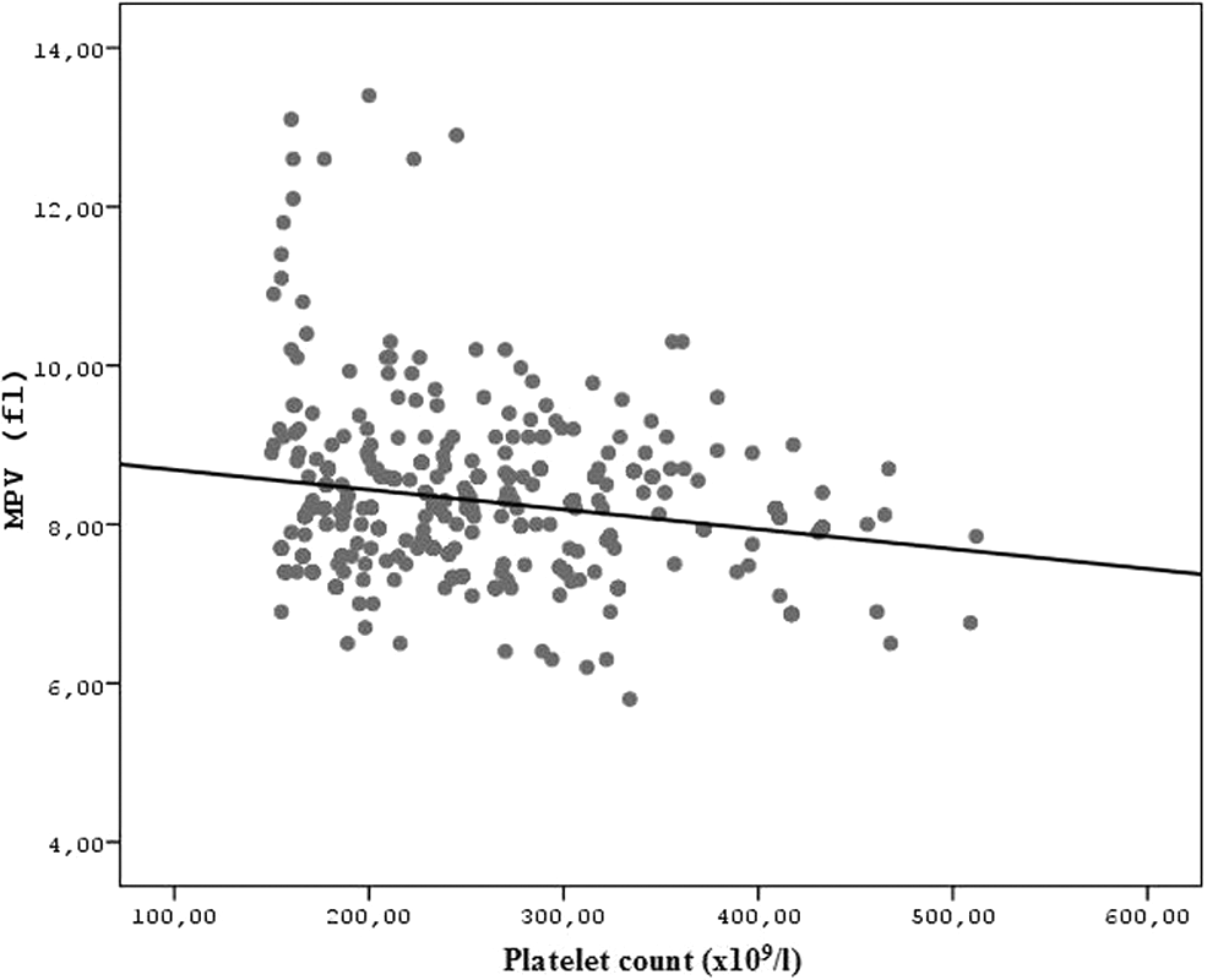
Correlation between platelet count and mean platelet volume.
In a multivariate logistic regression analysis, after the addition of variables of age, sex, cardiovascular risk factors including hypertension, diabetes, smoking, laboratory parameters including white blood cell count, hemoglobin, fasting plasma glucose, cholesterols, platelet count, MPV, MPM, and MPC, significant predictors of the presence of DVT were only MPV, MPM, and MPC (Table 2). For MPV, the multiple-adjusted OR of the presence of DVT was 2.179 (95% CI, 1.673-2.837); for MPM, the multiple-adjusted OR of the presence of DVT was 24.448 (95% CI, 8.435-70.855); and for MPC, the multiple-adjusted OR of the presence of DVT was 0.795 (95% CI, 0.729-0.867).
Multivariate Regression Analysis of the Presence of DVT
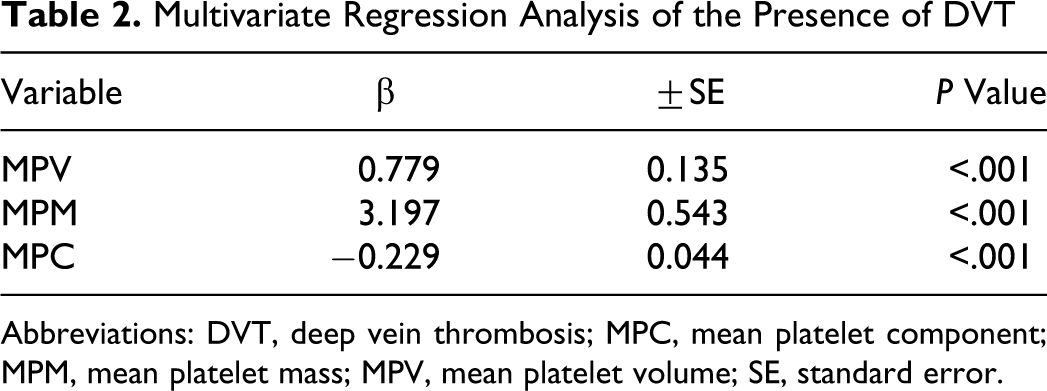
Abbreviations: DVT, deep vein thrombosis; MPC, mean platelet component; MPM, mean platelet mass; MPV, mean platelet volume; SE, standard error.
In subgroup analysis of patients with DVT, there was no significant difference in MPV, MPM, and MPC regarding DVT duration. In addition, there was no significant difference in these values regarding DVT side.
Finally, to determine the best cutoff values of platelet indices for predicting DVT, ROC analyses were performed. The areas under the ROC curves for these indices, which were used to detect DVT, were calculated (Table 3) and the sensitivities and the specificities of the best cutoff values were determined (Table 4). The cutoff values of 8.1 fL, 2.0 pg, and 25.9 g/dL for MPV, MPM, and MPC, respectively, were found to be moderately sensitive and specific for predicting DVT.
The Receiver Operating Characteristic Analysis for Platelet Indices to Predict DVT
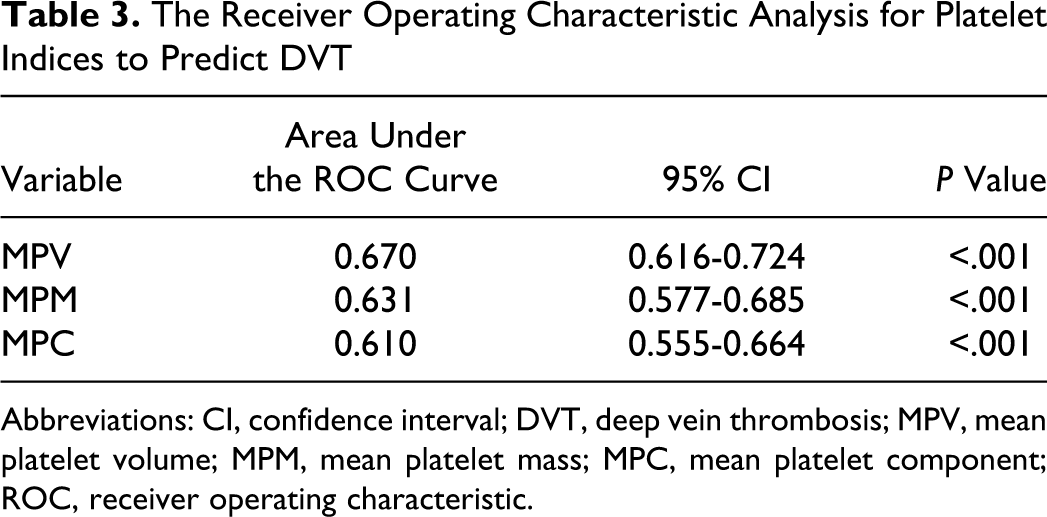
Abbreviations: CI, confidence interval; DVT, deep vein thrombosis; MPV, mean platelet volume; MPM, mean platelet mass; MPC, mean platelet component; ROC, receiver operating characteristic.
The Sensitivity and Specificity of the Cut-Off Values of Platelet Indices
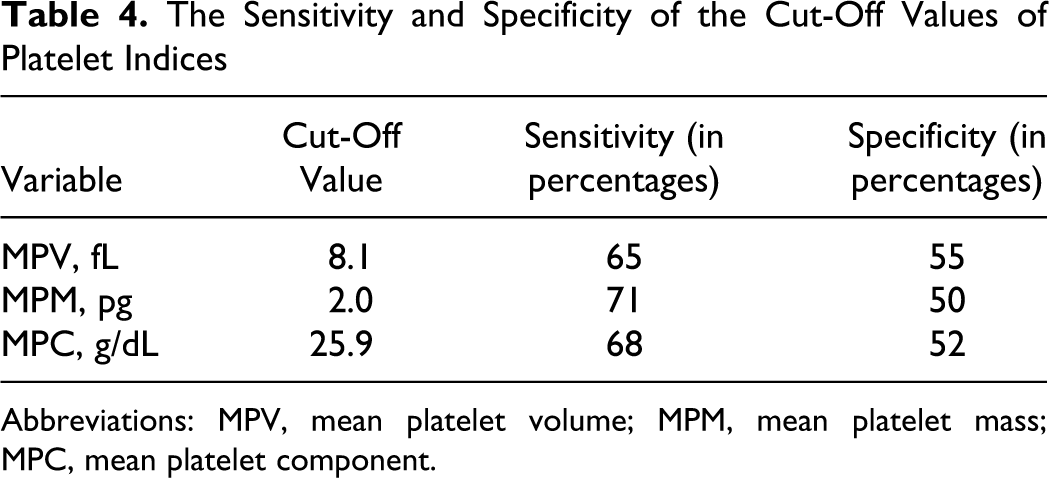
Abbreviations: MPV, mean platelet volume; MPM, mean platelet mass; MPC, mean platelet component.
Discussion
In the present study, we have found that the platelet activity indices were significantly and independently associated with the presence of DVT. Platelet activity can be influenced by a multitude of variables. Therefore, it is difficult to predict the extent of platelet reactivity in an individual. A practical, reliable, and available index of platelet activation has not been discovered although considerable amount of research has been performed.
Various laboratory methods including platelet count and size, aggregates, and released substances from activated platelets have been introduced for the detection of platelet activity. However, they have different advantages and disadvantages.
Platelets are one of the main actors in the process of thrombosis and hemostasis. When platelets are activated, they adhere to each other and this interaction is mediated by soluble adhesion molecules of activated platelets. If explained in detail, following endothelial damage, the subendothelial tissue was exposed directly to the circulating blood components. Circulating platelets are activated and adhere to the subendothelial tissue, aggregate and release vasoactive substances, and further aggregation occurs with fibrin production finally resulting in thrombotic occlusion, whereas venous thrombi found in low-flow or during stasis have greater proportions of erythrocytes and fibrin. 10 Similarly, antiplatelet agents inhibiting platelet adhesion and aggregation may play a role in the treatment of DVT. Accordingly, aspirin use has been found to be related to a significantly lower incidence of venous thrombosis, demonstrating that the platelets played an important role in the pathogenesis of DVT. 11
Within an individual, the circulating platelets are heterogeneous in size, metabolism, functional activity, and density. Larger platelets are metabolically and enzymatically more active than smaller platelets, containing more prothrombotic material, with increased thromboxane A2 and B2 per unit volume and glycoprotein IIb-IIIa receptor expression.12,13
Regulation of platelet size is multifactorial. Platelet volume is determined both during megakaryopoiesis and thrombopoiesis and may not correlate with the age of the platelet. 14 Environmental factors as well as genetic influences contribute to a substantial variation in platelet activity in healthy participants. Transcription and post-transcriptional modifications seem to be responsible from the genetic component. 15
The platelet count can predict the risk of major adverse cardiovascular events. 16 In healthy participants, there is a nonlinear inverse correlation between MPV and platelet concentration: MPV tends to decrease in participants with higher platelet counts as in our study. 17 Only a small number of large platelets is needed to obtain the same overall effect that a higher number of small platelets might obtain. 18
MPV is an inexpensive and easily available biomarker. It is routinely available in the inpatient and outpatient setting at a relatively low-cost. Increased MPV values are related to various cardiovascular risk factors; disorders and inflammatory processes resulted in arterial and venous thromboses. 19 No consensus is present whether the relationship between platelet size and cardiovascular risk is continuous or categorical.
In a study performed by Paramo and Rocha, platelet activity indices including MPV increased significantly following total hip replacement surgery. These changes, however, were not related to the development of DVT. 20
It has been shown that reductions in the MPC (reflecting a decrease in platelet granularity) are associated with an increase in CD62 expression and are indicative of platelet activation.21–24 In addition, the MPC decrement is correlated with anticoagulant-induced and thrombin-stimulated platelet activation.21,22 The MPC has been evaluated to demonstrate the role of platelet activation in coronary heart disease and for monitoring the efficacy of antiplatelet therapy.24,25 Availability and use of the MPV, MPM, and MPC on an automated system in a routine hematology laboratory without additional equipments are clear advantages over other complex methods to measure in vitro platelet activation.
Limitations
The study group was composed of a highly heterogeneous population, although participants using antiplatelet drugs were excluded. An important limitation concerning the predictive value of MPV, MPM, and MPC is the absence of well-defined limits to differentiate between activated and nonactivated platelets. Other platelet activity indices such as radio-labeling methods, aggregometry procedures (although the cornerstone for the assessment of platelet function is aggregometry), 26 determination of platelet contents released in serum, platelet-specific eicosanoids, other release reactions such as serotonin and histamine, adhesion molecules, flow cytometric investigations, platelet function analyzers, thromboelastography, and plasma factors influencing platelet activation such as fibrinogen were not used in our study. In addition, platelet rich plasma was not used. Although the platelet rich plasma method has higher sensitivity, the disadvantage is centrifugation resulting in platelet activation in vitro.
Conclusion
DVT patients demonstrate some abnormalities of platelet activation compared with healthy participants, and these platelet abnormalities may partly contribute to the pathophysiology of DVT. Platelet activity indices including MPV, MPM, and MPC may be used in predicting DVT. However, these results have to be confirmed in larger, prospective, and more homogenous study groups.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
