Abstract
After the first description of platelets more than a century ago, the knowledge about their origin and function grew continuously. The development of the impedance method as a completely automated assay allowed integrating mean platelet volume (MPV) measurement as a routine parameter of the complete blood count. This enabled us to focus more on the association of platelet function and size. Since then, many authors described MPV as a marker of platelet reactivity and risk factor for cardiovascular diseases. Hence, the preanalytical variability of this parameter is known from its introduction as standard laboratory value. Unfortunately no preanalytical standards have been implemented. This review shows the high variability in the literature with MPV as a risk factor for cardiovascular disease. After a brief description of the biology of platelets, we provide an in-depth survey of the measurement methods and their drawbacks. Finally, we propose a possible approach to standardization.
Background
More than 100 years ago platelets were called the dust of blood. They were thought to be disintegrated parts of leukocytes or fibrin. In 1882, Bizzozero was the first to acknowledge the importance of platelets and used the term piastrine (small plates). He showed that platelets play a key role in coagulation. Furthermore, he contributed to the exploration of the development of thrombopoiesis. 1,2 Since that time, platelets have been investigated intensively. In addition to their procoagulatory properties, platelets carry a myriad of receptors and contain miscellaneous mediators. In the 1970s, Karpatkin and Freedman associated platelet size with function. 3 Martin and colleagues showed in the 1980s that larger platelets demonstrate increased reactivity and a shorter bleeding time. 4 They express higher levels of P-selectin and glycoprotein IIb/IIIa (GPIIb/IIIa) receptors. 5,6 Consequently, platelet volume is thought to be a risk marker for cardiovascular disease. 7 Hence, in about 1000 publications throughout the past decade, mean platelet volume (MPV) was the subject of investigation as a potential marker of platelet reactivity and a surrogate parameter for a broad variety of diseases. Furthermore, several diseases are associated with MPV abnormalities. For example, a pathognomonic sign for the Bernard-Soulier syndrome is an increased platelet size (also called giant platelets). Moreover, an increased turnover is associated with changes in platelet size and function. 8 Large platelets are associated with cardiovascular disease and prothrombotic states, whereas small platelets are detected in chronic inflammatory diseases, for example, lupus erythematodes, inflammatory bowel disease, and rheumatoid arthritis. 9 This might be explained by the influence of immune mediators and growth hormones on platelet production. 10,11
Accurate measurements of platelet count and size are important for diagnostic, therapeutic, and research purposes. After hematological tests were revolutionized by the invention of the Coulter principle in the 1950s, the different parameters on platelets became more easily accessible. Ultimately, the MPV could be added to the standard full blood count in the late 1970s after it became fully automated. However, interpreting MPV value—thus, its use as a marker in a clinical setting—is not as straightforward as it might appear. Concern arose because of the drawbacks of the laboratory analysis and its interpretation. Currently there is no preanalytical standard when dealing with MPV measurements. 12
In the present article, we demonstrate the high technical diversity of MPV measuring. After a brief recapitulation of platelet production and physiology, the contributing factors are described and compared in more detail and in conclusion, a recommendation to standardize the measurement of MPV is proposed.
Literature Overview on MPV and Cardiovascular Disease
Initially, we conducted a PubMed literature search on MPV. This PubMed search was performed for studies using human participants, with abstracts in Dutch, German, or English using the following search terms: mean platelet volume (all subheadings) or MPV (all subheadings) or platelet sizing (all subheadings) and (cardiovascular diseases [MeSH] or atherosclerosis [MeSH] or coronary disease [MeSH] or hypertension [MeSH] or myocardial infarction [MeSH]). Due to the vast amount of publications on the topic, the search criteria chosen were limited to MPV publications related to cardiovascular disease between 2006 and 2011. Cardiovascular disease was selected because the relationship between it and MPV is the subject of many studies. 13 Many authors describe MPV as a risk factor for myocardial infarction, coronary artery disease, and stroke. 14,15
This search resulted in 126 articles, 63 of which were not about the MPV measurement method and as such were excluded. Full texts of the remaining 63 articles were obtained, analyzed, and the methodological design of the study was assessed on 3 topics:
Type of anticoagulant used for blood drawing tubes.
Differences in time delay between blood drawing and MPV measurement (measurement in ethylenediaminetetraacetic acid [EDTA] after 120 minutes and in citrate after 60 minutes). 16,17
Cell count device or method used.
Totally, 2 articles were excluded because of the nature of the article: 1 was a meta-analysis and 1 a review. The remaining 61 articles mentioned at least 1 of the methodological items. More than half of the researchers (62%, n = 39/61) became aware of technical drawbacks and in their methods described all 3 topics (anticoagulant type, time interval of measurement, and counting method; see Figure 1). While researchers may have been aware of possible problems, most of them did not solve the challenge of standardizing in a proper way for the research that they presented. More specifically, 82.5% (n = 52/61) of the studies reported the anticoagulant which was used in its procedure, irrespective of concentration (heparin: 1; EDTA: 41; and citrate: 10). In 65% (n = 41/61) of the studies, the time delay between blood sampling and measurement was described, but only 3.2% (n = 2) followed the recommendation in the literature to measure after 120 minutes in EDTA and 60 minutes in citrate. 16,17 More than 10 different descriptions for time interval of the measurement were counted, ranging from immediately after blood sampling to within 24 hours after, and almost anything in between. Finally, 93.7% (n = 59/61) mentioned the device used for measurement. All kinds of techniques were used for this as well (see below for technical description). This high variability causes confusion within all these reports, making comparison impossible.
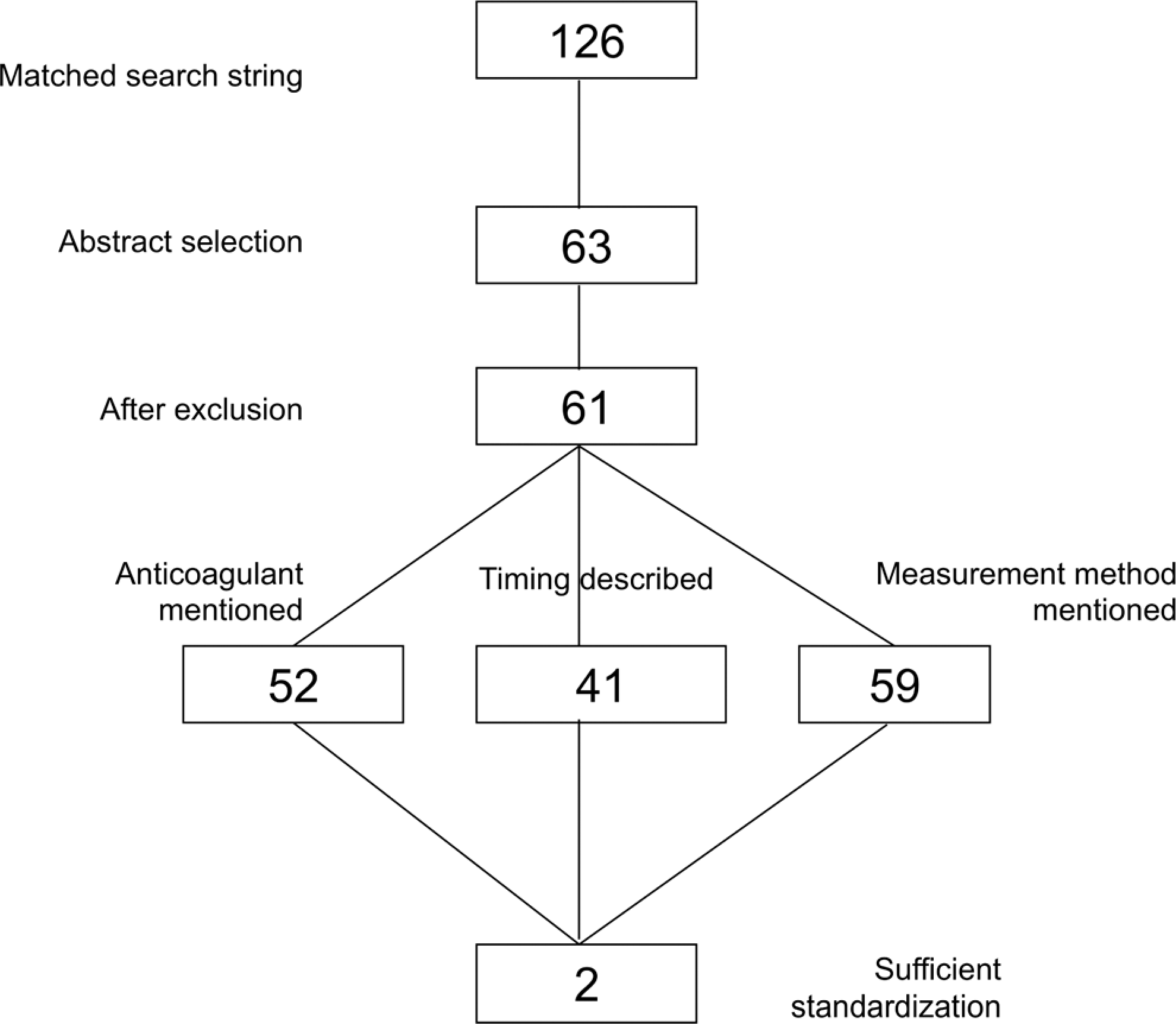
Search strategy, 126 articles met the initial search strategy criteria, 63 dealt with mean platelet volume (MPV) measurement methods, 2 exclusions (review and meta-analysis), 52 described the anticoagulant use, 41 included the time interval, and 59 reported the measurement method. Two articles met the standardization criteria (anticoagulant, correct time intervals and measurement method).
Platelet Production
The intraindividual platelet count is normally maintained within a narrow range throughout one’s lifetime, whereas the average platelet count differs between individuals about 3-fold (150-450 × 10 9 /L). 18 Nevertheless, the total platelet mass, which is the product of platelet count and MPV, is kept at a narrow level. 19 Hence, the platelet count is inversely related to the MPV, that is, in patients with low platelet count the MPV is larger and vice versa. 16,20,21 A normal range of platelet count is derived from the mean ± 2 standard deviation from a group of healthy individuals. 22 An adult generates approximately 10 11 new platelets per day that have a life span of about 8 to 10 days. The production may increase up to 10-fold in the event of higher demands (eg, blood loss). 11 Under these conditions, the MPV of the freshly released platelets is increased within 8 hours after onset of thrombocytopenia. 23 However, the relation between platelet size and platelet age is still being debated. Some authors argue that platelets decrease in size due to aging, whereas others do not support this theory. It is known that platelet size does decrease as a result of chemotherapy and radiation. The mechanism of this, however, is unclear. It may be due to the therapy (alteration of thrombopoiesis) or an effect of selective consumption of larger platelets. The latter has been shown in surgical and trauma patients. 16,24 The spleen plays a central role in platelet homeostasis. It removes old platelets from the circulation. Moreover, one-third of the total platelet number is stored in the splenic pool and its release depends on adrenergic stimuli. 25 The amount of storage may increase up to 85% to 90% of the total platelets in cases of splenomegaly. 26,27 On the other hand, platelet size decreases in inflammatory diseases, such as systemic lupus erythematodes, inflammatory bowel disease, and rheumatoid arthritis. 9 This might be a result of the delicate interaction of thrombopoeitin and immune mediators on platelet production. It is known that tumor necrosis factor-α, interleukin (IL) 1 and IL-6, and granulocyte-macrophage affect megakaryocyte proliferation and platelet count. 9 –11
Platelet Physiology
Beside their well-known function in hemostasis, platelets also play a role in inflammatory and immunemodulatory processes. Hence, their surface holds a variety of receptors, among those are GP receptors. The presence of GPIIb/IIIa is unique to platelets and its absence is responsible for Glanzmann thrombasthenia. Important for the hemostasis is the GPIb-V-IX, whose absence is a pathognomonic sign for Bernard-Soulier syndrome. In addition, the platelet membrane contains the fibrinogen receptor (GPIIb/IIIa) and receptors for thrombin, adenosine diphosphate (ADP), collagen, and thromboxane. Platelets also contain secretory granules, which act in concert with the coagulation system and the vascular endothelium during hemostasis. The most numerous are alpha granules, in which fibrinogen, von Willebrand factor (vWF), thrombospondin, and fibronectin are stored. Their amount depends on the size of the platelets but is about 10 times higher than the amount of dense granules. 28 Also, at the level of the granules some disease-specific changes can be found; for example, giant alpha granules are associated with disorders such as Paris-Trousseau/Jacobsen syndrome and the hypogranular, White platelet syndrome. An early sign of storage injury seems to be the degranulation of alpha granules with exposure of P-selectin. 29 Normally, about 3 to 8 smaller dense granules can be found per platelet. They contain ADP, adenosine triphosphate, guanosine triphosphate, serotonin, and 60% to 70% of whole platelet calcium. 30 Further, platelets contain lysosomes, mitochondria, endoplasmatic reticulum, and a microtubular system. Again there seems to be a link between platelet size and platelet content. Larger platelets were supposed to contain more granules and hereby become more reactive. Though these results are contradictory and still there is no consensus about the relationship between platelet size and platelet age. However, large platelets are known to have more hemostatic capacity. 19,31,32
Briefly, platelet function with respect to coagulation can be described as a sequence of several phases. In the resting phase, platelets are about 2 to 4 µm in diameter and have a discoid shape. The first step in activation is the adhesion of platelets to specific receptors. The cross-linking by the GPIb-V-IX receptor induced by vWF leads to shape change in the platelets. The binding to GPIb-V-IX receptors stimulates other receptors such as GPIIb/IIIa to adhere to fibrinogen and to enhance the aggregation. Moreover, this receptor facilitates the bridging of platelets and the stabilization of the clot. 33,34 Also the collagen binding GPVI receptor becomes activated in the early phase of thrombus formation. 34 Together GPIb-V-IX and GPVI promote different pathways, which induce secretion of the dense granules and alpha granules. The secreted thrombin, epinephrine, and ADP boost the secretion of the granules and directly interact with the coagulation system. 35
Platelet Counting and MPV Measurement
There are 4 different principles for platelet counting and MPV measurement:
Manual counting by phase contrast microscopy.
Impedance counting.
Optical light scatter counting.
Immunological flow cytometry techniques.
Manual counting by phase contrast of platelets was considered the gold standard until very recently. 36 Phase contrast microscopy used chambers in which the platelets were counted manually and their size was estimated. For a long time, this work-intensive technique did not allow for the addition of the MPV into the routine parameters, and automated methods still were too imprecise. Although the implementation of automated counters in the 1970s increased precision dramatically, problems in differentiating between platelets, and other small particles, such as fragmented red cells and immune complexes, especially in patients with low platelet count remained a source of uncertainty. As a result, manual microscopy remained the international reference method, and manual counting was used for calibration of the automated systems. Nevertheless, there were concerns about the imprecision, the interobserver variability is about 10% to 25%. 37
In the 1950s, W. H. Coulter developed and presented the Coulter principle using electronic impedance, which revolutionized platelet counting. 38 –40 The impedance method is based on conduction of the properties of cells. A suspension of blood diluted in isotonic saline is drawn through an aperture. A current flows through this aperture and as each cell passes through, the resistance increases. The poor conductive properties of a cell increase the impedance that is measured by an increase in voltage or pulse. In theory, the number of pulses corresponds with the amount of particles that pass through, and the amplitude of the pulse corresponds with the volume of the particles. This is comparable with the erythrocyte count and the MCV measurement. Two problems that immediately come to mind are simultaneous measurement of 2 particles and erroneous measurement of (fragmented or small) red blood cells. With impedance counting, particles ranging from 2 to 20 fL in volume are deemed platelets. This range is chosen because there are few other small cells, such as red blood cells, giant platelets, and cell fragments. From all the platelets counted and measured, a platelet size histogram is created and extrapolated to 70 fL, after which, log-normal curves are fitted to this histogram. The actual data are calculated from this superimposed curve, in which the MPV corresponds with the geometric mean. Fitting a curve is needed to exclude microcytic red cells and allows for the inclusion of giant platelets.
Because of the above-mentioned problems with impedance measurement, another automated technique was proposed as being superior. Optical cell counting was introduced in the 1970s. With this technique, cells are focused in a flow cell and what is measured is the light scatter produced by a laser that illuminates the cells. In order to count and size the cells, the light scatter is measured at 2 angles and converted to electrical pulses. Similar to the impedance technology, the number of pulses corresponds with the cell count and the amplitude is proportional to the cell volume. A platelet histogram is also produced from these measurements, but no log-normal curve is superimposed. In addition, the MPV is calculated from the geometrical mode instead of the geometrical mean, which means the maximum distribution of the measured volumes is represented. Optical fluorescence platelet counting is a variation of this method. Here, the reticulated cells and platelet membranes are first stained with a fluorescent. The intensity of fluorescence allows platelets, including giant platelets, to be determined. As with the impedance measurement, MPV is measured immediately by using the calculated mode, instead of a calculated mean. 41 The difference between optical and impedance measurement techniques may result in different MPV values using the same sample. Differences of up to 40% have been reported. 42 –44
In 2000, Harrison et al presented what they called immunoplatelet counting. The high precision of this method prompted the authors to propose this as new reference technique. 37 Briefly, immunological measurement uses monoclonal antibodies to platelet cell surface antigens (mainly anti-CD41 and anti-CD61). These are covalently attached to fluorescent substances, an example of which is fluorescein isothiocyanate. The antigens are registered by their fluorescence when running through a flow cytometer. Due to the high costs and the inaccessibility of the flow cytometer technique for some laboratories, this method is a calibrating method rather than a standard assay. Newer fully automated immunological methods have recently become available, but still, their use is not yet widespread because only one instrument is currently able to perform this method automatically. The main advantage of the immunoplatelet counting method is high accuracy, especially in samples with low platelet counts. However, there are some concerns regarding this advantage. Harrison already mentioned that their method might overestimate patients who had high levels of platelet antibodies or who were treated with antiplatelet GPs (eg, anti-GPIIb/IIIa antibodies). An underestimation of the true platelet count could be seen in absence of platelet receptors for the target antigen, for example, Bernard-Soulier syndrome. 37 Further, Sandhaus and colleagues observed a mild underestimation of the platelet count due to large platelets and platelet clumps. 45
Regardless, this method is still highly accurate, particularly when patients are thrombocytopenic; making it the new gold standard and its use is recommended for this population. 46,47
In addition to the MPV another parameter—the mean platelet component (MPC)—was introduced by Macey et al as a surrogate parameter for platelet activation. 48 This parameter derives from automated flow cytometric measurements, which are provided the ADVIA system. The authors reported an inverse relation between CD62 as a marker of platelet activation and the MPC. Still there was an interference with EDTA as anticoagulant. The results in EDTA samples are not stable. In contrast to MPV, the MPC increases slightly in the first hour and decreases from that point until more than 24 hours. 49 In a recent article by Cooke and coworkers, the authors could not detect a positive correlation between MPC and the severity of coronary artery disease. 50 However, Diaz-Ricart et al reported some additives to EDTA to make the results for MPC more stable. 49 In this light, MPC as a parameter for platelet activation seems interesting but currently not recommendable as standard parameter.
Factors Influencing the MPV Results
Beyond the methodological variables affecting the results, some preanalytical drawbacks are also of importance. Notably, the method of venipuncture and the degree of accuracy of filling and gently mixing the sampling tubes may cause platelet activation and produce clumping. The choice of anticoagulant, time interval of measurement, and temperature at which MPV is analyzed seem to be more relevant. In general, for all hematological laboratory tests and also for cell counting, the whole blood sample needs to be anticoagulated to inhibit coagulation before measurement. Commonly, this is achieved with either EDTA or sodium citrate as standard substances. They both bind calcium, thus preventing coagulation in an irreversible (EDTA) or reversible (sodium citrate) manner. There are also other substances, which are not used standardly, such as theophylline or wortmannin (see Table 1).
Summary of Trials With Different Anticoagulants, Methods, Temperature at Measurement, and Time Delay to Analysis

Abbreviations: ACD, acid citrate dextrose; CPDA, citrate phosphate dextrose adenine; CTAD, citrate, theophylline, adenosine, dipyridamole; E/C, EDTA and CTAD; EDTA, ethylenediaminetetraacetic acid; Imp, impedance; LS, light scatter; K, potassium; Na, sodium; NA, data not available; RT, room temperature.
Nevertheless, platelets will swell and continue to do so due to most anticoagulants. The exact mechanism is not completely known, but it is probably caused by chelation of membrane-bound calcium in platelets. 53 In particular, EDTA causes ultrastructural morphological changes in platelets. The canalicular system opens and platelet volume will increase and the GPIIb/IIIa receptors dissociate irreversibly. 54 –56 Unfortunately, the reaction toward the anticoagulants is not uniform. For example, Na-EDTA as an anticoagulant results in less pronounced swelling than K-EDTA. 51 Further, even the concentration of anticoagulants may vary (eg, for sodium citrate), which results in more distinct swelling in “low-concentration” citrate (0.12 mmol/mL, ratio 9:1, blood to citrate) and almost no swelling in “high-concentration” citrate (0.12 mmol/mL, ratio 4:1, blood to citrate). 16 Thompson et al and Trowbridge et al were the first to quantitatively compare the effects of anticoagulants on platelet swelling. 42,51 They proposed standard mediums and also fixed time points after sampling for best measurement of MPV. Since that time many other authors reported different anticoagulants (see Table 1). In 1993, the International Council for Standardization in Hematology published their recommendation. Here, the use of K2-EDTA (1.5-2.2 mg/mL blood) as reference method is proposed. However, many mediums are still used in clinical and investigational settings. 57
Another important issue seems to be the time interval of the measurement. As already mentioned, Thompson and Bath described their experience with MPV analysis. 16,51 The authors put forward that the dependency of the time interval is negligible in high-concentration Na3-citrate (1:4, citrate to blood), whereas in low-concentration Na3-citrate (1:9, citrate to blood) and K-EDTA, the best results are seen between 60 and 90 minutes, respectively. Others recommend to measure within 1 hour regardless of anticoagulant. 21 Finally, some authors suggest using additives for accurate measurements. 49,52
In a recent study, our group recommended a reference interval for time-adjusted MPV measurement for 2 commonly used standard media (K2-EDTA and sodium citrate) defined for the Coulter LH 750. The aim was to create a simple standardization of the MPV measurement in commonly used anticoagulants. 17 In conclusion, an optimal stability was detected in K2-EDTA after 120 minutes and in sodium citrate after 60 minutes, which resulted in a reference range of 7.2 to 10.8 fL for K2-EDTA and 6.1 to 9.5 fL for sodium citrate.
Recently, we determined new MPV reference ranges for the Sysmex XE-5000 in K2-EDTA after 120 minutes (n = 120), and this reference range was completely different compared to the Coulter method (unpublished data): 9.2 to 12.7 fL which proves that MPV results are not only dependent on time but also on method.
Another preanalytical condition, the storage temperature of sample tubes, has only been the subject of 2 publications. Both studies showed that cooling citrated blood samples from 37°C down to 4°C increases MPV results by 18%. 58,59 Even though this item is mentioned frequently, it is not considered by most researchers nor in the laboratory. 8 In actual daily practice, MPV measurements are performed at room temperature.
Some limitations of this review are that there is not much literature on preanalytical differences in blood drawing techniques and their influence on MPV. While we know that the sampling technique is of interest for standard laboratory tests, their impact on platelet function analysis recently was reported to be minor. 60,61 On the other hand, there are no reports on specimen transport and its influence on MPV. Although this might be less important as there is only a small change due to pneumatic tube transport on standard laboratory analysis and thromboelastometry. 62,63 Finally, the literature search was limited to PubMed and cardiovascular disease in association with MPV. This might be a disadvantage. However, due to the large amount of literature found by this restriction it seems a good reflection of the current research.
Conclusion
As explained earlier, it is impossible to compare results from different measurement methods unless there are valuable correction coefficients. Because the use of MPV as a diagnostic marker is becoming increasingly popular, it is important that a standard is developed and implemented soon. Moreover, for studies, a uniformity of measurement should be used to make the results comparable with each other. A standard method should include recommendations for optimal temperature, commonly used anticoagulant, time delay from sampling to measurement, and technique implemented. For most laboratories, it should facilitate the measurement of MPV as a routine parameter by avoiding uncommon anticoagulants or concentrations, and they would not have to implement a wide variety of techniques. Until then, each laboratory should determine its own reference values considering the aforementioned recommendations.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
