Abstract
Conventional prothrombin time (PT) assays have limited sensitivity and dynamic range in monitoring the anticoagulant activity of direct factor Xa inhibitors. Hence, new assays are needed. We modified a PT assay by adding calcium chloride (CaCl2) to the thromboplastin reagent to increase assay dynamic range and improve sensitivity. Effects of calcium and sodium ion concentrations, and sample handling, were evaluated to optimize assay performance. Increasing concentrations of calcium ions produced progressive increases in PT across the factor Xa inhibitor concentrations of 0 to 2500 nmol/L for razaxaban and apixaban. The greatest effect was seen when the thromboplastin reagent was diluted 1:2.25 with 100 mmol/L CaCl2 (thus selected for routine use). The optimized assay showed an interassay precision of 1.5 to 9.3 percentage coefficient of variation (%CV) for razaxaban and 3.1 to 4.6 %CV for apixaban. We conclude that the modified PT assay is likely to be suitable as a pharmacodynamic marker for activity at therapeutic concentrations of factor Xa inhibitors.
Keywords
Introduction
Factor Xa plays a central role in the intrinsic and extrinsic pathways of blood coagulation; hence, inhibition of this factor is attracting increasing attention as a strategy for the prevention or treatment of thromboembolic events. 1 Orally active, direct factor Xa inhibitors offer a potential means of overcoming the limitations associated with existing anticoagulants, including the need for parenteral administration with unfractionated or low-molecular-weight heparin, and the unpredictable pharmacokinetics and pharmacodynamics and monitoring requirements with oral vitamin K antagonists. 2,3 A number of orally active, direct factor Xa inhibitors, including apixaban 4 and rivaroxaban, 5,6 are undergoing clinical trials or are currently marketed.
Owing to consistent and predictable pharmacokinetic and pharmacodynamic profiles and wide therapeutic index, the oral factor Xa inhibitors do not, or are not expected to, require routine anticoagulant monitoring during clinical use. 6,7 However, there may be clinical circumstances for which the availability of an assay to monitor anticoagulant activity is desired. Furthermore, it is important to be able to measure their pharmacodynamic activity during clinical trials, particularly as conventional anticoagulant assays have been shown to be subject to limitations in the presence of these agents. 8
Although traditional prothrombin time (PT) assays are widely used for monitoring the effects of conventional oral anticoagulants (such as warfarin), they are not suitable for accurate measurement of pharmacokinetic and pharmacodynamic profiles because they produce relatively small differences in clotting times between anticoagulant-treated and -untreated individuals. 9 Furthermore, a recent study has shown that commercial PT assays produce highly variable results in patients receiving factor Xa inhibitors and that conversion to international normalized ratio (INR) values actually increases this variability rather than decreases it. 10
During the early clinical development of a series of novel factor Xa inhibitors, including razaxaban 11 (formerly DPC 906; first-in-human study ~2000) and apixaban (December 2002), a modified PT (mPT) assay was developed in which calcium chloride (CaCl2) was added to the thromboplastin reagent in order to prolong clotting times and, hence, increase the sensitivity of the dose–response curve for the direct factor Xa inhibitor. 12 This article presents the results of studies evaluating modifications to this PT assay in order to optimize its performance across the anticipated range of therapeutic concentrations of factor Xa inhibitors in plasma in the first single ascending dose study in humans and to validate the optimized assay analytically and clinically in patients receiving a factor Xa inhibitor.
Material and Methods
Experimental
Ethics
All clinical studies, including the healthy volunteer sample collection for in vitro testing, were conducted in accordance with Good Clinical Practice (as defined by the International Conference on Harmonisation) and with the ethical principles of the Declaration of Helsinki, EU Directive 2001/20/EC, the US Code of Federal Regulations, Title 21, Part 50 (21CFR50), and all applicable local regulatory requirements. The protocol and participant informed consent forms received appropriate approval by the Institutional Review Board/Independent Ethics Committee prior to the initiation of the study at the site. Written informed consent was obtained from all the individuals prior to the participation in the study.
Materials
Razaxaban (formula weight: 564.93 g/mol) was synthesized by DuPont Pharmaceuticals (Wilmington, Delaware). Apixaban (formula weight: 459.51 g/mol) was synthesized by Bristol-Myers Squibb (Princeton, New Jersey). Sodium chloride (NaCl) and CaCl2 were from Sigma Chemicals (St Louis, Missouri). The thromboplastin reagent (Thromboplastin C Plus) and quality control samples (Ci-Trol Coagulation Control, levels 1 and 3, used to compare PT results with the manufacturer specified range to confirm reagent and assay performance) were obtained from Dade Behring (Newark, Delaware) and were stored and prepared according to the manufacturer’s instructions. The thromboplastin reagent was reconstituted by adding 10 mL deionized water to a vial of lyophilized Thromboplastin C Plus and mixing by gentle inversion. The coagulation control samples (Ci-Trol) were dissolved in 1 mL deionized water and mixed by gentle inversion. Level 1 samples were derived from the individuals who had not received oral anticoagulants, whereas level 3 samples were derived from patients treated with warfarin.
The pooled human plasma was obtained from Bioreclaimation (Hickory, New York). Individual plasma samples were obtained from healthy volunteers at Covance Laboratory (Vienna, Virginia) after appropriate consent. Healthy individuals were determined as having no clinically significant deviation from normal in medical history, physical examination, clinical laboratory determination, and electrocardiographs; a body mass index of 18 to 32 kg/m2; age 20 to 45 years, inclusive, and with no use of over-the-counter or prescription drugs within 1 week of study.
A total of 21 human plasma validation samples were prepared by spiking varying amounts of razaxaban or apixaban directly into pooled human plasma to achieve the final concentrations ranging from 0 to 2500 nmol/L. Conversion factors for razaxaban are 1 nmol/L = 0.565 ng/mL and 1 ng/mL = 1.77 nmol/L; and for apixaban 1 nmol/L = 0.46 ng/mL and 1 ng/mL = 2.176 nmol/L.
For apixaban clinical studies, blood samples were collected from an indwelling catheter or by direct venipuncture. Only saline was used to keep the catheter patent. If an indwelling catheter was being used, then a minimum of 3 mL of blood was initially withdrawn and discarded. Venous blood samples were drawn into 3.2% sodium citrate tubes, with each tube filled to its maximum capacity. Immediately after collection, the contents of each tube were gently mixed by inverting 6 to 8 times without shaking. All samples were processed within 30 minutes of collection. The sodium citrate tubes were centrifuged for 10 minutes at 2500g at 4°C twice, with the supernatant subsequently stored at or below −20°C in an upright position and shipped on dry ice to their respective analytical laboratories. On the day of assay, the samples and controls were thawed at room temperature and then stored in the instrument at 2 to 8°C for approximately 2 hours prior to testing.
Assay Procedures
Both the PT and mPT assays were performed using an MLA Electra 1600C/1800C Automatic Coagulation Analyzer (Medical Laboratory Automation Inc, Mount Vernon, New York). All samples were assayed in duplicate, and the coagulation control samples were assayed at the beginning and end of each assay day. For mPT, the thromboplastin reagents were prepared freshly each day in bulk, according to the manufacturer’s instructions.
Assay Optimization
Experiments were performed to modify the PT assay by adding calcium ions to the PT thromboplastin reagent so that the range of clotting time could be extended in the presence of factor Xa inhibitors. The CaCl2 solutions of 0, 50, 75, and 100 mmol/L were added to achieve a constant ratio of 1 part thromboplastin + 1.25 parts of CaCl2 (1:2.25). NaCl was added, as appropriate, to maintain a constant ionic strength of 100 mmol/L in the solution.
Analytical Validation
The performance characteristics of the mPT assay under the established “optimal” conditions were evaluated across the concentration range from 0 to 2500 nmol/L, which was the anticipated drug concentration range from multiple ascending dose studies of razaxaban or apixaban. Interassay precision was conducted by the repeat measurements of a set of validation samples on 3 different days with at least 2 measurements each day, which is used to calculate a percentage coefficient of variation (%CV). 13 The intra-assay precision was evaluated by repeat measurements of the above validation samples for at least 5 times and calculation of %CV. 13 The freeze/thaw (F/T) stability of the samples was assessed, as recommended by the American Association of Pharmaceutical Science Biomarker Committee, 13 by measuring mPT of human plasma samples containing 0, 300, 1300, and 2500 nmol/L of razaxaban and apixaban that had undergone 1, 2, and 3 cycles of freezing and thawing treatment. Sample-stability testing was conducted using plasma samples stored at −20°C or at −65°C for up to 1 year. The effect of sample processing time on mPT was evaluated using samples collected from 6 healthy volunteers, processed <3 hours, 3 to 4 hours, and 6 hours after collection, and then centrifuged at 1000, 2000, 3000, and 4000g for 10 minutes at 2 to 8°C. The reference range of the mPT assay was evaluated using individual samples from healthy volunteers from a phase I clinical study.
Correlation of mPT, PT, and Drug Concentrations in Clinical Trial Samples
Both mPT and PT were measured in a series of human plasma samples from healthy volunteers who were administered with a single dose of razaxaban (Study CV906-101). 11 To reach maximal drug exposure, the top 2 dose panels of 100 and 160 mg were selected for the evaluation. All samples were processed and split into 2 aliquots at the clinical site and then shipped in frozen form to an analytical laboratory for PT/INR and mPT or drug concentration analysis by liquid chromatography/tandem mass spectrometry. The razaxaban drug exposure was found to range from 0 to 2309 nmol/L.
Both mPT and PT were also evaluated from a phase I clinical study of apixaban (Study CV185-022), 12 where a total of 624 samples were obtained from 39 healthy volunteers, who were administered with a single dose of apixaban (20 mg). Blood samples were collected in citrate tubes. After centrifugation, plasma samples were split, and 1 aliquot was frozen at −70°C for mPT and 1 aliquot was frozen at −20°C for drug concentration analysis. The apixaban drug exposure was found to range from 0 to 600 ng/mL. A curvilinear regression analysis of mPT data was performed with a simplified E max model [Y = A + B × X D/(C + X D), D = 1]; PT data were analyzed with linear regression.
Statistical Methods
All analyses and figures were generated using GraphPad Prism (Version 4.00, Graph-Pad Software Inc, La Jolla, California). The standard regression method was used to explore the linear relationships of apixaban plasma concentration to PT or mPT and the associated coefficient of determination, R2 values of apixaban plasma concentration to PT or mPT. Statistical analyses were carried out using SAS/STAT Version 8.2 software (SAS Institute, Inc, Cary, North Carolina).
Results
Effect of Calcium Concentration
Increasing concentrations of calcium ions slowed the clotting reaction time and produced a progressive increase in the clotting time, resulting in increased assay sensitivity and broader dynamic range. In the standard PT assay, the clotting time increased from 12.3 seconds in the absence of razaxaban to 25.6 seconds at a razaxaban concentration of 2500 nmol/L, which is only a 2.1-fold change. In the presence of the highest calcium concentration studied (100 mmol/L), the corresponding increase was from 65.8 to 373 seconds (Figure 1 ), which is a 5.7-fold change. Similar clotting time prolongation was also observed for apixaban. The mPT was increased from 66.2 seconds in the absence of apixaban to 361 seconds at an apixaban plasma concentration of 2500 nmol/L, which is a 5.4-fold increase.
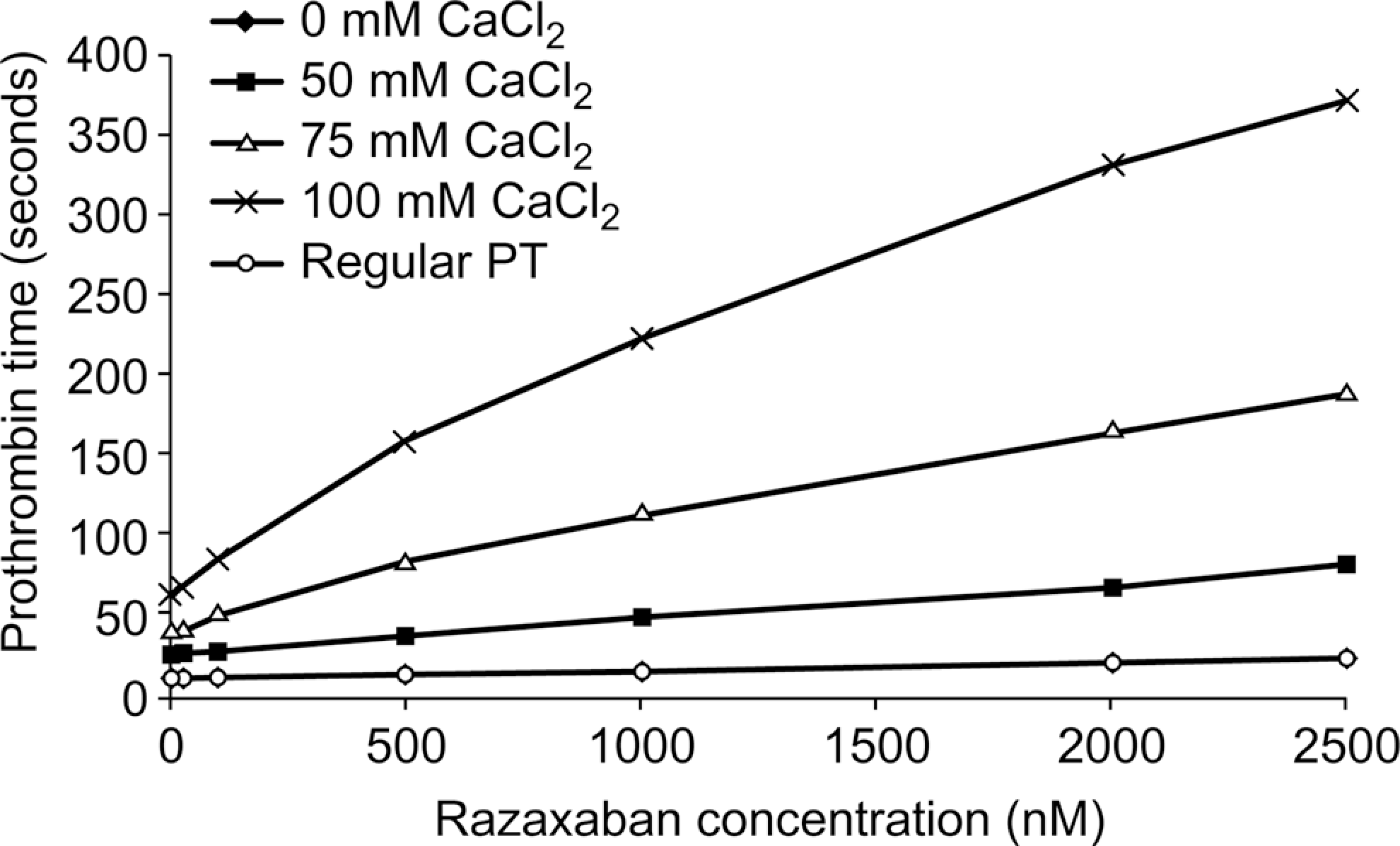
Effect of calcium ion concentration on PT. Razaxaban standard curves in the presence of increasing calcium ion concentrations were prepared by spiking razaxaban into pooled human plasma. The total ionic strength was maintained at 100 mmol/L by the addition of sodium chloride. Final thromboplastin reagent dilution was 1:2.25. Results are presented as the means of duplicate determinations. CaCl2 indicates calcium chloride; PT, prothrombin time.
The final conditions of thromboplastin reagent dilution of 1:2.25 with 100 mmol/L CaCl2 were selected so that the maximal instrument limit of 400 seconds would not be reached at the anticipated maximal concentration of the highest dose in apixaban and razaxaban dose-escalation studies.
Assay Analytical Validation
Interassay- and Intra-assay Precision
The razaxaban validation data showed interassay precision of the mPT assay ranging from 1.5 to 9.3 %CV, while the standard PT assay ranged from 1.5 to 4.2 %CV. The razaxaban intra-assay precision for mPT was 1.2 to 6.2 %CV, while the standard PT assay was 0.9 to 1.4 %CV. Similarly, apixaban validation showed an interassay precision of 3.1 to 4.6 %CV and intra-assay precision of 2.8 to 4.2 %CV for mPT assay (Table 1 ). Standard PT assay interassay precision ranged from 1.0 to 3.2 %CV.
Intrarun and Interrun Precision of mPT for Apixaban in Human Plasma When Stored at ≤65°C.a
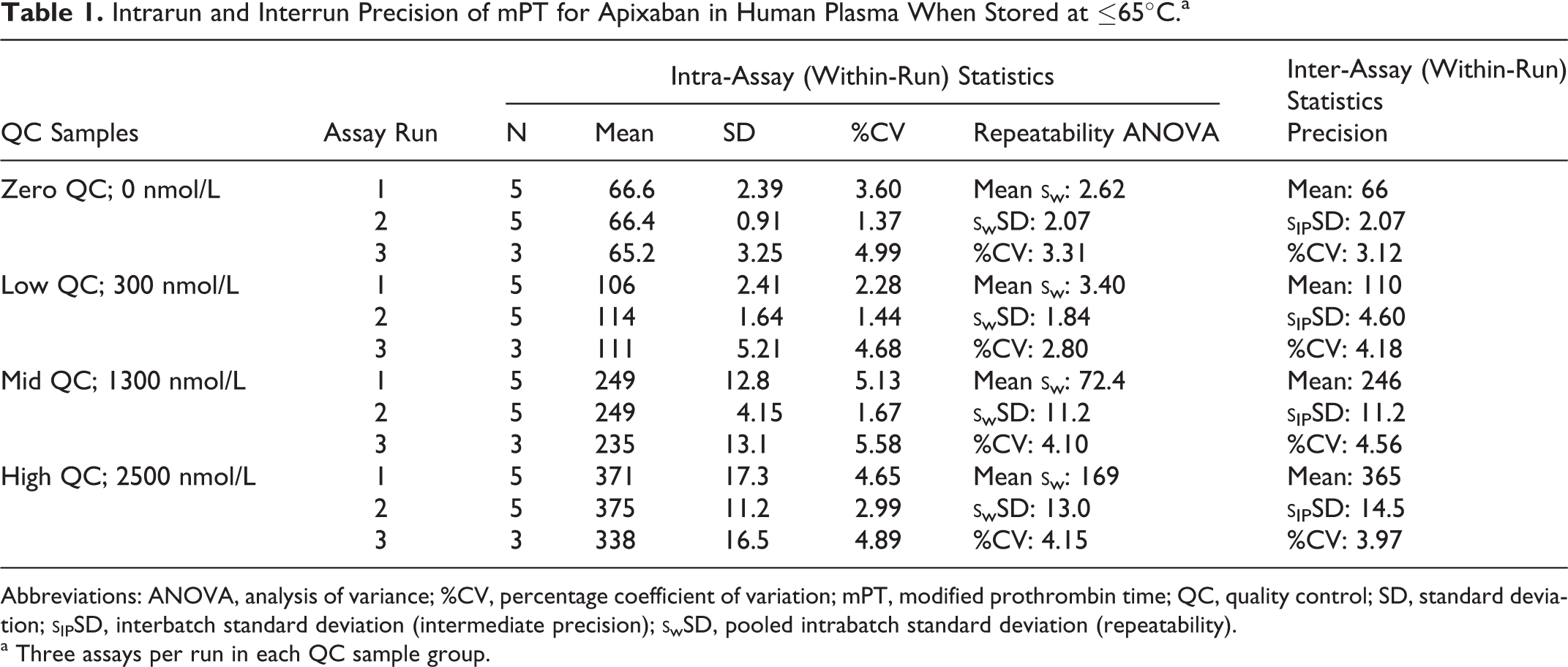
Abbreviations: ANOVA, analysis of variance; %CV, percentage coefficient of variation; mPT, modified prothrombin time; QC, quality control; SD, standard deviation;
a Three assays per run in each QC sample group.
Sample F/T and Long-Term Storage Stability
The relative changes in mPT values for razaxaban and apixaban after each cycle of F/T treatment were compared with the samples that had undergone F/T 1 time (baseline). The baseline samples were the day 1 samples used in the long-term storage stability study, in which mPT measurement was conducted 18 hours after sample preparation. The percentage of baseline values for samples stored frozen at −20°C ranged from 111% to 127% in all F/T cycles. The percentage of baseline values for samples stored frozen at −65°C ranged from 99% to 120% in all F/T cycles.
A statistical analysis using multiple linear regression showed that all the plasma samples, regardless of the concentration of apixaban, were stable for up to 364 days (~12 months) at −65°C (P = 0.32). The same statistical model was then applied to data from plasma samples under similar conditions, but with the samples stored at −20°C (P = 0.06). Although this P value was not statistically significant at the P = .05 level, it could be suggestive of a numerical trend that may become significant if the experiment was to be repeated with a larger number of individuals and samples. Upon visual inspection, there appeared to be a nonlinear trend in clotting times that became more apparent for the groups with higher concentrations. For this reason, it is recommended to minimize the storage time of samples at −20°C and to transfer samples to a storage location with a temperature of −65°C at the earliest convenience.
Effect of Sample Handling on PT and mPT
In the mPT assay, decreasing the centrifugation force from the 3000g recommended in current guidelines resulted in increased variability in PT. With razaxaban, 9 of 12 samples centrifuged at 1000g and 7 of 12 centrifuged at 2000g had >15% variability in mPTs compared with 0 of 12 centrifuged at 4000g. This increased variability was attributable to the contamination of the plasma by platelets at lower centrifugal forces: at forces ≤2000g, 5 or more samples had platelet counts >10 000 per mm3; whereas at 4000g, all samples had platelet counts below this figure. By contrast, altering the centrifugal force had no effect on PT variability in the standard PT assay.
Increasing the time between razaxaban sample collection and assay from <3 to >6 hours had no significant effect on PT in either the mPT or standard PT assays (<15% difference).
Reference Intervals
Reference intervals, which define a test result’s variability in healthy individuals, are frequently derived from data obtained from blood donors, laboratory workers, or healthy control populations. The central 95% interval is bounded by 2.5 and 97.5 percentiles, so that 2.5% of the values are cut from both the ends of the reference distribution. 14,15 Here, we evaluated reference intervals from predose samples obtained from an apixaban single-dose study (CV185-022), where 39 healthy volunteers, ranging from 21 to 74 years old, were assessed. The PT reference interval (2.5%-97.5%) was determined as 10.9 to 15.5 seconds, and the mPT reference interval was determined as 42.4 to 65.0 seconds.
Correlation of mPT versus PT as well as Apixaban and Razaxaban Plasma Concentrations
The correlation between apixaban plasma concentration and mPT or PT was calculated using data from an apixaban phase I study of 39 individuals and 620 plasma samples. The mPT showed a dose-related increase in clotting time with strong concordance with apixaban concentration (0-600 ng/mL). In this study, a curvilinear relationship between mPT and apixaban plasma concentration with R 2 = .75 (Figure 2a) was observed and a linear fit of PT data provided R 2 = .41 (Figure 2b). Similar to in vitro data, the mPT showed a broader dynamic range and higher sensitivity than the standard PT assay, which makes differentiation between drug exposure levels easier. For example, at the expected peak (~110 ng/mL) of apixaban 5 mg twice daily and trough (~30 ng/mL) of apixaban 2.5 mg twice daily, 16 mPT values were ~66 and ~93 seconds, respectively, a 27-second window of change versus PT values of ~13.8 and ~15.2 seconds, a change of 1.4 seconds.
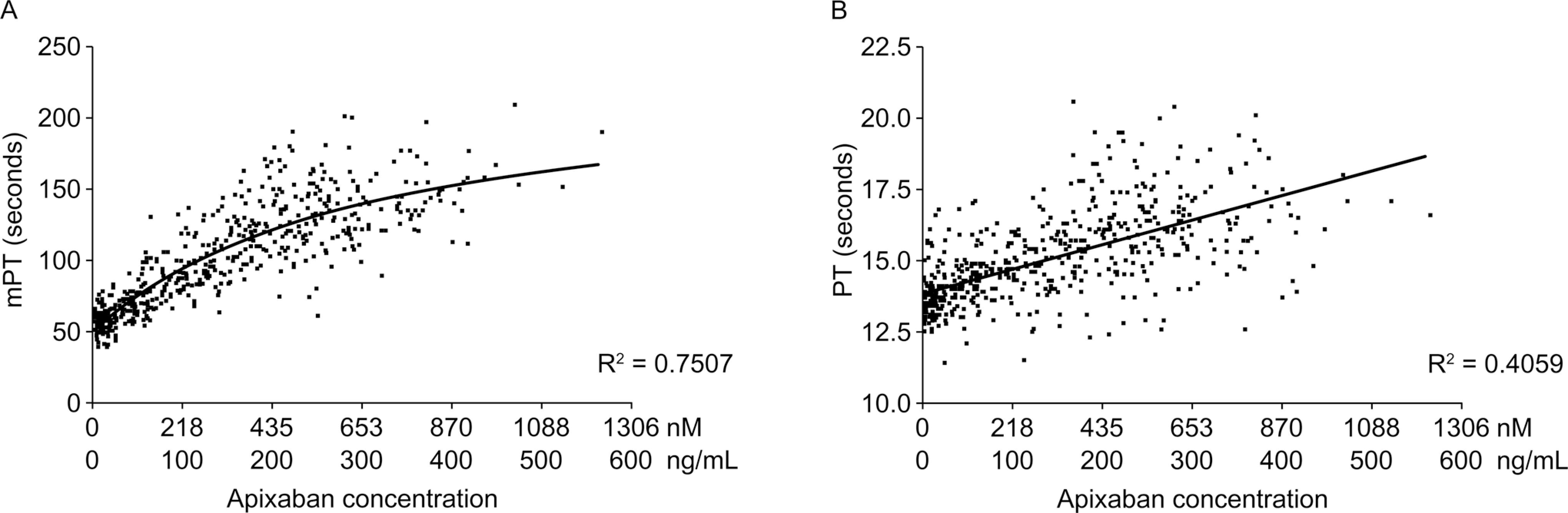
Scatter plot of (A) modified prothrombin time (mPT) and (B) prothrombin time (PT) versus plasma apixaban concentration. R 2 for mPT = .7507 (A); R 2 for PT = .4059 (B).
Similarly, a curvilinear correlation between razaxaban plasma concentration and mPT was also observed at the razaxaban concentration range of 0 to 2500 nmol/L (Figure 3 ). No formal statistical analysis on correlation was conducted, as only a small number of individuals participated in the study, with 9 individuals contributing 42 PT samples and 63 mPT samples. In addition, the razaxaban samples were mostly concentrated toward the lower end of the concentration range (PT: 32 samples at razaxaban concentration of 0-1000 ng/mL and 9 samples in the 1000-2000 ng/mL range; mPT: 54 samples at razaxaban concentrations of 0-1000 ng/mL and 9 samples in the 1000-2000 ng/mL range). This imposes an imbalance to the data set, which renders the results of any formal statistical analysis potentially less meaningful.
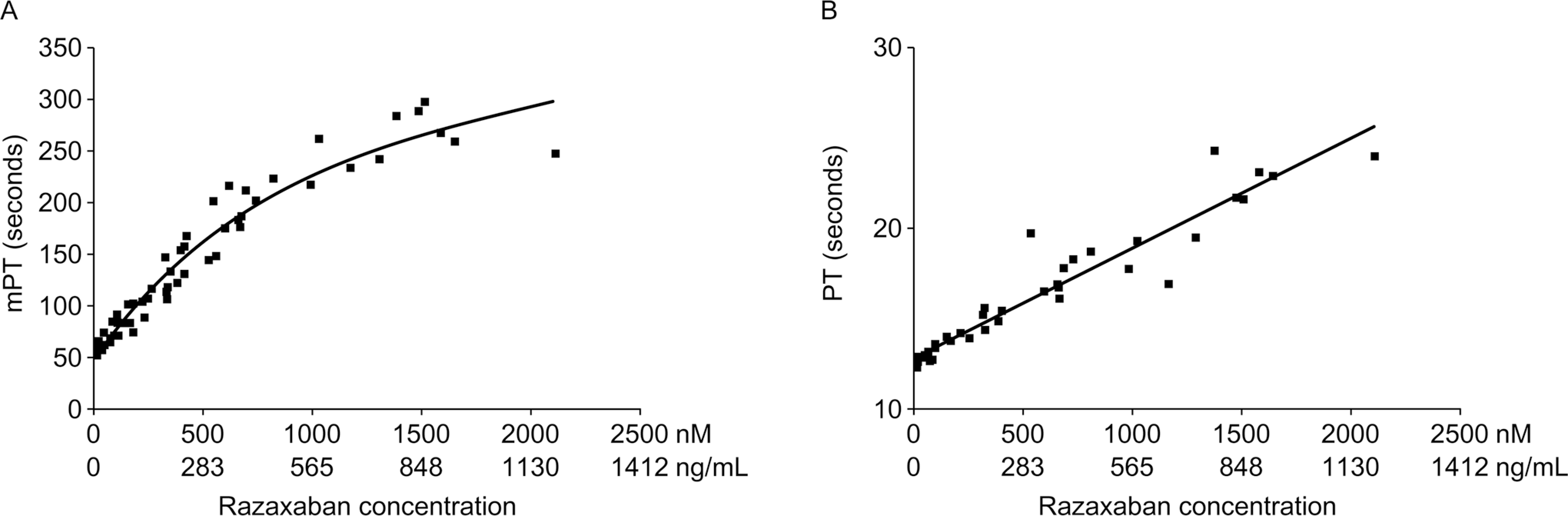
Scatter plot of (A) modified prothrombin time (mPT) and (B) prothrombin time (PT) versus plasma razaxaban concentration.
Discussion
It is essential for new drug development that a biomarker assay is in place for assessment of pharmacodynamic activity during early clinical studies. A conventional PT assay is readily available but has limitations in dynamic range and sensitivity when used for direct factor Xa inhibitors. Here, we report the modification of a conventional PT assay by diluting the thromboplastin reagent in 100 mmol/L CaCl2. This modification prolonged the PT measurements at all evaluated concentrations of apixaban and razaxaban and improved the sensitivity of the assay, thus making it possible for use as a variable biomarker to assess pharmacodynamic effects in early clinical studies.
The exact mechanism of this clotting time prolongation is not well understood, as interactions between calcium ions, prothrombinase, and downstream reactions in coagulation are complex. It has been speculated that increasing the concentration of calcium ions changes the ionic strength of the thromboplastin solution and thus interferes with the thrombin and fibrinogen interaction. 17 This, in turn, disrupts the cascade of the coagulation reactions and prolongs the thrombus formation. Further experiments by adding sodium or magnesium ions in place of the calcium ions to thromboplastin reagents also showed prolongation of the clotting time measurements. However, the same extent of clotting time prolongation required a higher concentration of sodium or magnesium ions than that of the calcium ions. 18
The improvement in sensitivity in the mPT assay made it possible to assess the pharmacodynamics of the novel factor Xa inhibitors razaxaban and apixaban. In animal studies with apixaban, the sensitivity of the modified assay was ~10- to 20-fold higher than that of conventional PT assays. 18 Similarly, in pharmacokinetic studies in healthy volunteers, the PT measured by the modified assay increased 3.2-fold after administration of apixaban, 50 mg for 7 days, compared with a 1.5-fold increase when PT was expressed in terms of the INR. 19 This assay was successfully implemented in early clinical studies of both razaxaban and apixaban, and the pharmacodynamic effects were used as evidence for factor Xa mechanism of action. 11,19 There was evidence also from other small molecule factor Xa inhibitors being developed at Bristol-Myers Squibb to show that this assay can be used in a variety of factor Xa inhibitors. 12
The sensitivity improvement of the assay modification is at the expense of variability. The PT assay has been known to be easily influenced by sample handling conditions and thromboplastin reagent changes 20 –22 ; the mPT might have amplified that effect. Decreasing the centrifugation force below 3000g as recommended by the Clinical and Laboratory Standards Institute’s (CLSI) 1998 guidelines 23 increased the variability, apparently as a result of contamination of the plasma with platelets. Increased variability was also seen when the time from collection to assay was prolonged beyond 6 hours. Thawing plasma samples at room temperature, as opposed to a 37°C water bath as recommended by the CLSI guidelines, could also impact on mPT values as cryoprecipitate may have formed during the slower thawing process, which could have consumed certain clotting factors such as fibrinogen. By contrast, these factors had less effect in the standard PT assay, presumably because of the lower sensitivity of the assay. Similarly, sample stability measured by mPT was limited to lower than −65°C. However, for research purposes, the variability of the mPT assay is reasonable (<10 %CV for interassay precision); thus, with careful controlling of assay reagents and sample conditions, this assay can be used successfully in a research setting. While the clinical utility of this assay outside of research is unclear, there may be a need for simple, fast clinical tests to assess drug levels of factor Xa inhibitors as they become more available for clinical use. With further development and standardization, this assay could provide a potential option, should the need for such a test arise.
In conclusion, the addition of a high concentration of calcium ions to an automated PT assay increased the measured PT and improved the sensitivity of the assay, making it suitable for the measurement of anticoagulant activity in individuals receiving direct factor Xa inhibitors. This assay should, therefore, facilitate pharmacokinetic and pharmacodynamic studies with such agents.
Footnotes
Acknowledgments
The authors thank Harry Law and Andy Shepherd, PhD of Caudex Medical Ltd, Oxford, UK, funded by Bristol-Myers Squibb and Pfizer, for assistance in preparing the initial draft of the manuscript, collating the comments of authors and other named contributors, assembling tables and figures, and coordinating submission requirements. The authors gratefully acknowledge the contribution of the staff at Covance laboratories.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: YCB, ZW and RK are employees of, and owns stocks in, Bristol-Myers Squibb.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research of the study was funded by Bristol-Myers Squibb. Professional writing support was funded by Bristol-Myers Squibb and Pfizer, Inc.
