Abstract
The antiphospholipid antibodies present in antiphospholipid syndrome (APS) are directed at a number of phospholipid-binding proteins: β2 glycoprotein I (β2GPI), prothrombin, and so on. Antibodies directed at β2GPI are accepted as a classification criterion for APS, while the presence of antiprothrombin antibodies is not. In the present article, we investigated the possible role of antiphosphatidylserine/prothrombin antibodies (aPS/PT) as marker of APS on a cohort of 295 individuals with APS (95 primary APS and 45 secondary APS) and APS-related diseases. We found aPS/PT to be highly associated with venous thrombosis (immunoglobulin G [IgG] aPS/PT odds ratio [OR], 7.44; 95% confidence interval [CI], 3.97-13.92 and IgM aPS/PT OR, 2.54; 95% CI, 1.35-4.77) and obstetric abnormalities (IgG aPS/PT OR, 2.37; 95% CI, 1.04-5.43), but not with arterial thrombosis. A very high degree of concordance between the concentration of aPS/PT and lupus anticoagulant activity was demonstrated. Therefore, we support the inclusion of aPS/PT determination as second-level assay to confirm APS classification.
Keywords
Introduction
Antiphospholipid syndrome is probably the most important cause of acquired thrombophilia and one of the most common autoimmune diseases. It is characterized by recurrent venous and arterial thromboses (VT and AT) or obstetric abnormalities (spontaneous abortion, fetal losses, and intrauterine fetal growth impairment), all in the presence of antiphospholipid (aPL) antibodies. It is known that aPL antibodies encountered in APS are not true antibodies to phospholipids but they are directed at phospholipid-binding proteins, such as β2 glycoprotein I (β2GPI), prothrombin (PT), annexine V, activated protein C, and protein S, and other proteins of the coagulation pathway. 1
Among the above-mentioned proteins, the most prominent targets for the aPL antibodies are β2GPI and PT. Antibodies to β2GPI and to PT have been described as independently responsible for the activity of lupus anticoagulant (LA) in phospholipid-dependent coagulation tests.2,3
The presence of antibodies directed to β2GPI (aβ2GPI), isolated, or cardiolipin bound β2GPI, together with the presence of activity of LA, is accepted within the revised criteria for APS classification. 4 Despite numerous problems concerning assay standardization, the presence of these antibodies is clearly associated with the major clinical manifestations of APS. However, the same cannot be stated for antiprothrombin antibodies. Studies have shown contradictory results regarding the association of antiprothrombin antibodies with thrombosis, especially VT.5–7 Reviewing the literature, we notice that 2 enzyme-linked immunosorbant assay (ELISA)-based methods are commonly employed for the detection of antiprothrombin antibodies: antiprothrombin (aPT-A) assays, in which gamma-irradiated plates are coated with purified PT, or anti-phosphatidylserine/prothrombin (aPS/PT) assays, in which PT is exposed to immobilized phosphatidylserine (PS).3,8,9 Recently, it was demonstrated that antibodies detected by the aPT-A and aPS/PT assays are in fact 2 distinct populations with partial overlapping identity10,11 as the mechanism of action of aPS/PT is mediated by increased thrombin generation. 11
Probably because the aPT-A method was the first one described, the bulk of studies on antibodies to PT were performed by means of direct antigen-binding ELISA (aPT-A). The results obtained with this method are poorly associated with APS manifestations. 5 A reduced number of investigators have performed assays with the aPS/PT ELISA, finding the presence of antibodies detected by this method to be more associated with major APS manifestations. 12
The small number of studies reporting results by the aPS/PT assay stimulated the present investigation, with the purpose of evaluating the clinical significance of aPS/PT antibodies in our patient population, as well as their reliability and diagnostic utility.
Patients and Methods
The study group included 295 consecutive patients, referred between 1997 and 2009 to the Systemic Autoimmune Diseases Unit of our Hospital, with a probable diagnosis of antiphospholipid syndrome (APS) or related autoimmune diseases. A complete medical history, physical examination, and complementary studies were systematically performed for all the patients at inclusion. After the initial visit, the patients were reviewed every 6 months, or sooner, if required by the patient’s clinical condition. The data were collected in a Microsoft Access database using 2 types of standardized forms: an inclusion form, summarizing the patient’s medical record, and a follow-up form, containing patient’s evolution and laboratory data between 2 consecutive visits. All the clinical events were collected by the physicians involved in the study.
The authors collected data on age, gender, AT (cerebrovascular event, myocardial infarction (MI), renal artery thrombosis, retinal artery thrombosis, cardiac and silent brain ischemia [SBI], ie, asymptomatic brain infarcts diagnosed by magnetic resonance imaging), VT (deep vein thrombosis [DVT], pulmonary thromboembolism [PE], renal vein thrombosis, etc), and obstetric abnormalities (abortion, fetal loss, premature delivery, intrauterine growth retardation, and preeclampsia, HELLP syndrome [hemolysis, elevated liver enzymes, and low platelets count], etc).
Thrombophilic risk factors for AT (smoking habit, diabetes, hyperlipemia, arterial hypertension, and corticotherapy) and VT (trauma, cancer, obesity, pregnancy, immobility, oral contraceptives, nephrotic syndrome, antithrombin deficiency, protein C and S deficiency, factor V Leiden, coagulation factor II G20210A mutation, and high levels of homocysteine) were thoroughly recorded for each patient.
In order to diagnose APS, β2GPI-dependent anticardiolipin antibodies (from now on referred to as aCL), aβ2GPI, and LA tests were performed. The same serum sample used to detect aCL and aβ2GPI antibodies was employed to assay aPS/PT.
The international consensus statement on an update of the classification criteria for definite APS was used as guideline for APS diagnosis. 4
Patients were diagnosed for the following 2 criteria: The presence of aPL and APS manifestations: primary antiphospholipid syndrome (PAPS—APS not accompanied by another autoimmune disorder); secondary antiphospholipid syndrome (SAPS—APS accompanied by another autoimmune disorder), silent presence of aPL/LA, and no aPL/LA. The presence of autoimmune disease: systemic lupus erithematosus (SLE), Sjögren syndrome (SS), mixed connective tissue disease (MCTD), and other autoimmune diseases (OAD).
Therefore, in our study group, a patient could have more than 1 diagnosis, for example, SAPS and SLE.
Patients were followed for a period of time between 6 months and 12 years. In order to be included in the study, 2 serum samples from each patient, obtained at least 12 weeks apart, were analyzed. After the patients entered the study, sera were obtained every 6 months. Both IgG and IgM isotypes of aCL, aβ2GPI, and aPS/PT antibodies were investigated.
Both aCL and aβ2GPI antibodies were assayed using commercial kits (INOVA, San Diego, USA). Positive cutoff values were established in 40 GPL/MPL for aCL 4 and in 20 U/mL for aβ2GPI.
For the detection of aPS/PT antibodies, a homemade ELISA method was performed. Briefly, 96-well polystyrene enzyme immunosorbant assay (EIA) plates (Costar 3591) were coated with soy phosphatidylserine (Sigma, St Louis, Missouri), previously reconstituted at 10 mg/mL with chloroform:methanol (4:1) and diluted to 50 μg/mL in ethanol. Plates were dried in the dark and then blocked with 20 mmol/L Tris, 1% bovine serum albumin, and 5 mmol/L CaCl2 (blocking solution). After washing with 20 mmol/L Tris, 5 mmol/L CaCl2, and 0.005% Tween-20 (washing solution for all steps), plates were incubated with human PT (Diagnostica Stago, Marseille, France) at 10 μg/mL in phosphate-buffered solution for 1 hour at 37°C. After washing, samples, calibrators, and controls diluted in blocking solution were incubated for 1 hour at room temperature. Then, a 1:4000 dilution of alkaline phosphatase-conjugated goat anti-human IgG and anti-human IgM antisera (Jackson ImmunoResearch, PA) in blocking buffer was prepared. In the last step, p-nitrophenilphosphate diluted in 1 mol/L dietanolamine, pH 9.8, was employed as substrate. Optical density (OD) at 405 nm was read after 15 minutes.
Standard curves were constructed with the sera of 2 APS patients containing elevated levels of IgG and IgM aPS/PT, titrated in arbitrary units. Positive and negative controls were run in every plate.
In all the assays, the samples were run in parallel on the PS/PT-coated plate and in a “blank” plastic plate, coated only with PS. For each sample, OD of the PS-coated wells was subtracted from the OD of the PS/PT-coated wells. Cutoff was established at 10 AU/mL for IgG isotype and 15 AU/mL for IgM isotype, corresponding to mean OD + 10 standard deviation (SD) of 150 blood donors.
Activity of LA was determined in platelet-free plasma (platelet count is less than 10 × 109 per liter) obtained by double centrifugation at 2500g for 15 minutes, from blood samples collected into polypropylene tubes containing 0.129 mol/L sodium citrate (9:1 ratio). A sample of 20 pooled normal plasmas was used in the mixture assays as described above. Plasma from each patient was obtained on at least 2 occasions 12 weeks apart.
The LA was evaluated by screening and confirmatory procedures following the recommendations by the Scientific and Standardization Committee (SSC) Subcommittee of the International Society on Thrombosis and Haemostasis (ISTH). 13 Screening tests included the dilute Russell viper venom time (dRVVTS; Siemens LA1 screen) and kaolin clotting time, the variant described by Exner, which included the mixture assays. As the confirmatory test, the dilute Russell viper venom time (dRVVTC) and phospholipid-dependent test (Siemens LA2 confirmation) was used. The LA was considered positive if the ratio of dRVVTS/dRVVTC >1.3. Activity of LA was quantified as follows: low positive (dRVVTS/dRVVTC = 1.3-1.35), medium positive (dRVVTS/dRVVTC = 1.35-1.8), and high positive (dRVVTS/ dRVVTC >1.8).
Statistics
Data were collected using Microsoft Access database and statistical analysis was performed with SPSS v9. Descriptive study of mean and SD was calculated for continuous quantitative variables. Qualitative variables were described as absolute frequencies and as relative frequencies, expressed in percentages. For univariate analysis, parametric tests were employed when continuous quantitative variables were analyzed, utilizing the Student t test for 2-group comparisons. Spearman correlation coefficient was applied for correlations between variables of the same type. Analysis of qualitative variables was performed with the chi-square (χ 2 ) test or using Fisher exact test if necessary (N < 20 or if any expected values was <5). Yates correction was applied for the χ2 test. Multivariate analysis was performed in order to identify the independent factors that are positively associated with the dependent variable and to control for possible confounding parameters, various multivariate logistic regression models were designed. The magnitude of each factor was expressed as an odds ratio (OR). A stepwise model was followed, computing only the variables associated with the dependent variable in a previous univariate analysis or variables that could act as confounding factors. The model’s goodness of fit was expressed as an area under receiver operating characteristics (ROCs) curve and 95% confidence interval (95% CI). Results were grouped in tables showing the final step of the variables together with the corresponding β coefficient, standard error (SE), statistical significance, and eβ term, which represents the magnitude of effect adjusted by the rest of the variables and expressed as OR.
Results
Clinical Manifestations
Of the 295 patients, 237 (80.3%) were women and 58 (19.7%) were men, with a median age of 45.63 years (range: 18-87 years). Of the 237 women, 159 were considered “fertile”: appropriate age (menopause excluded) and not using contraception. The demographic data concerning prothrombotic risk factors are summarized in Table 1.
Prevalence of Prothrombotic Risk Factors in the Study Group.
The PAPS was present in 98 patients, SAPS was detected in 45 patients, 57 individuals presented aPL without clinical manifestation, and 95 patients did not present aPL at all.
The SLE was diagnosed in 130 patients, SS was encountered in 15 patients, MCTD was diagnosed in 4 patients, and 29 patients were diagnosed with other autoimmune diseases (OAD).
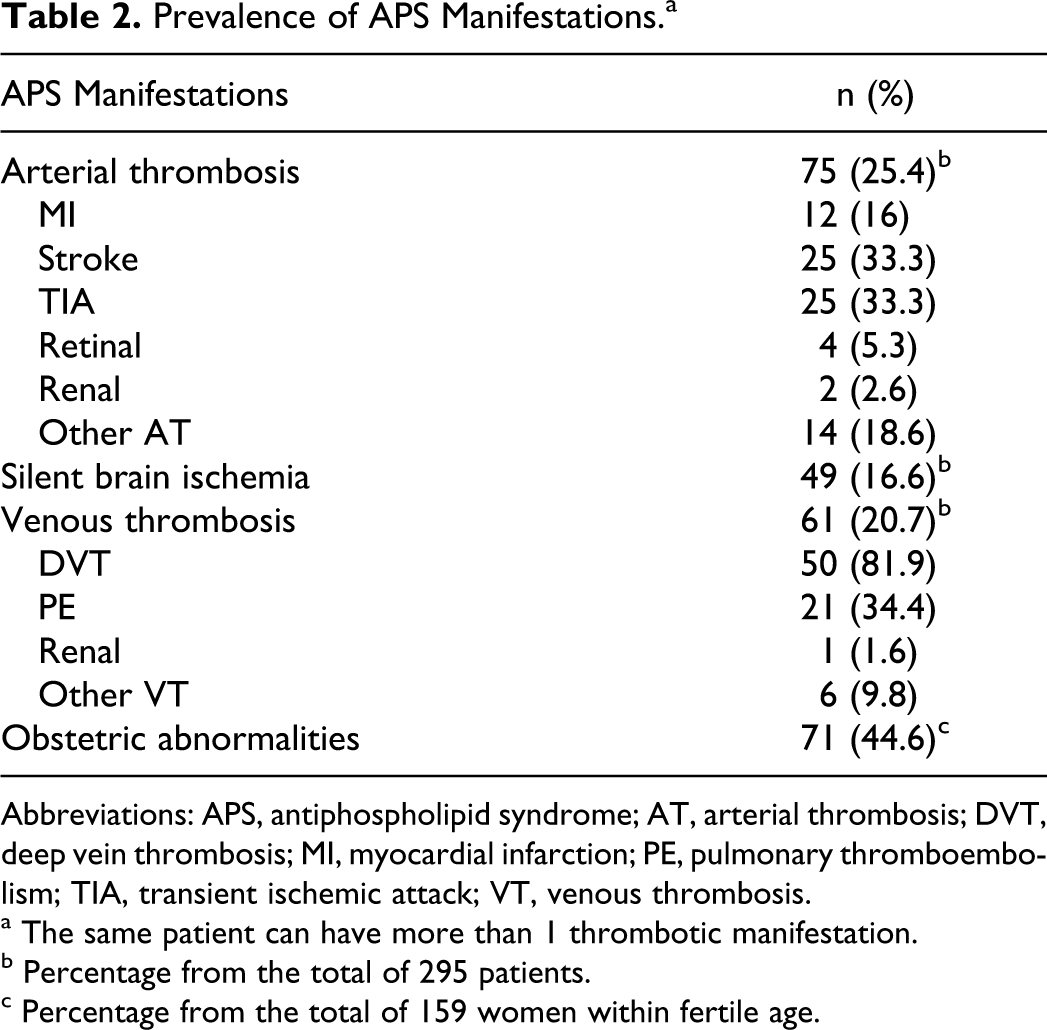
The prevalence of major APS manifestations in the study group was that from the total of 295 patients, 75 patients were presented with AT, 61 with VT, and 49 with SBI. From the 159 fertile women, 71 had obstetric abnormalities (Table 2).
Prevalence of APS Manifestations.a
Abbreviations: APS, antiphospholipid syndrome; AT, arterial thrombosis; DVT, deep vein thrombosis; MI, myocardial infarction; PE, pulmonary thromboembolism; TIA, transient ischemic attack; VT, venous thrombosis.
a The same patient can have more than 1 thrombotic manifestation.
b Percentage from the total of 295 patients.
c Percentage from the total of 159 women within fertile age.
In the AT group, the most frequent thrombosis were Stroke and Transient Ischemic Attack (TIA, which is defined as a cerebrovascular event resulting in brief neurological dysfunction that persists less than 24 hours), both present in 25 patients. The most frequent VT was lower limb DVT present in 50 patients and PE in 21 patients. We found that 1 patient may have more than 1 type of thrombosis, previously described as natural history of the disease. 4
Presence of aPS/PT
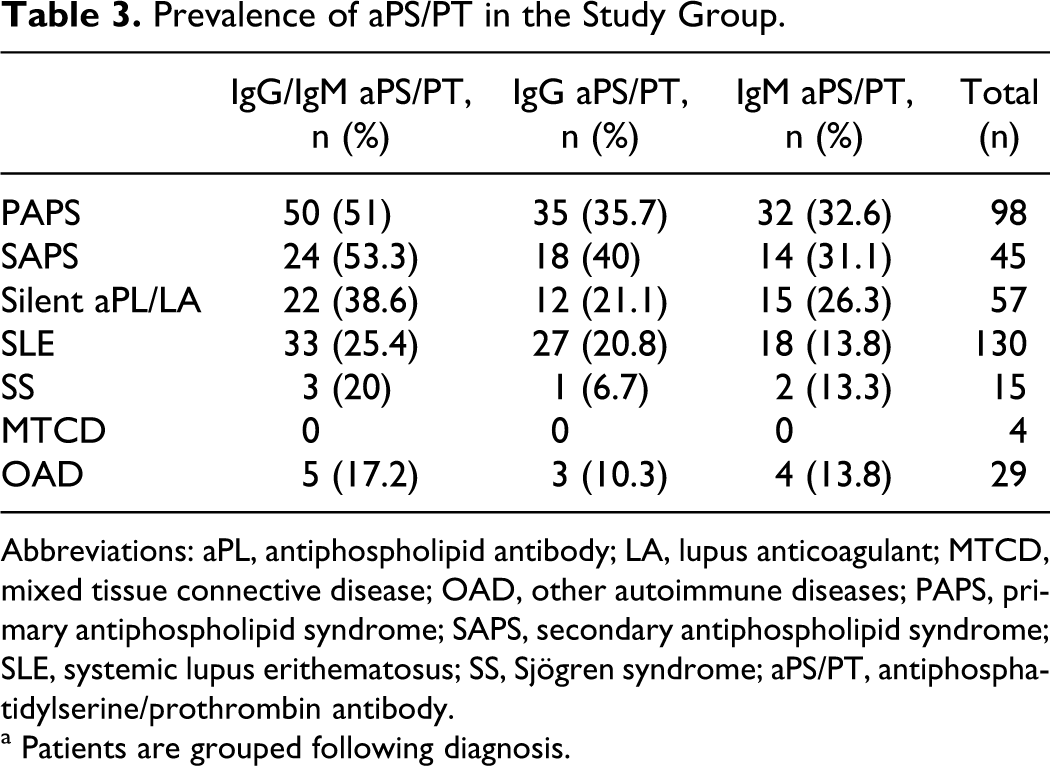
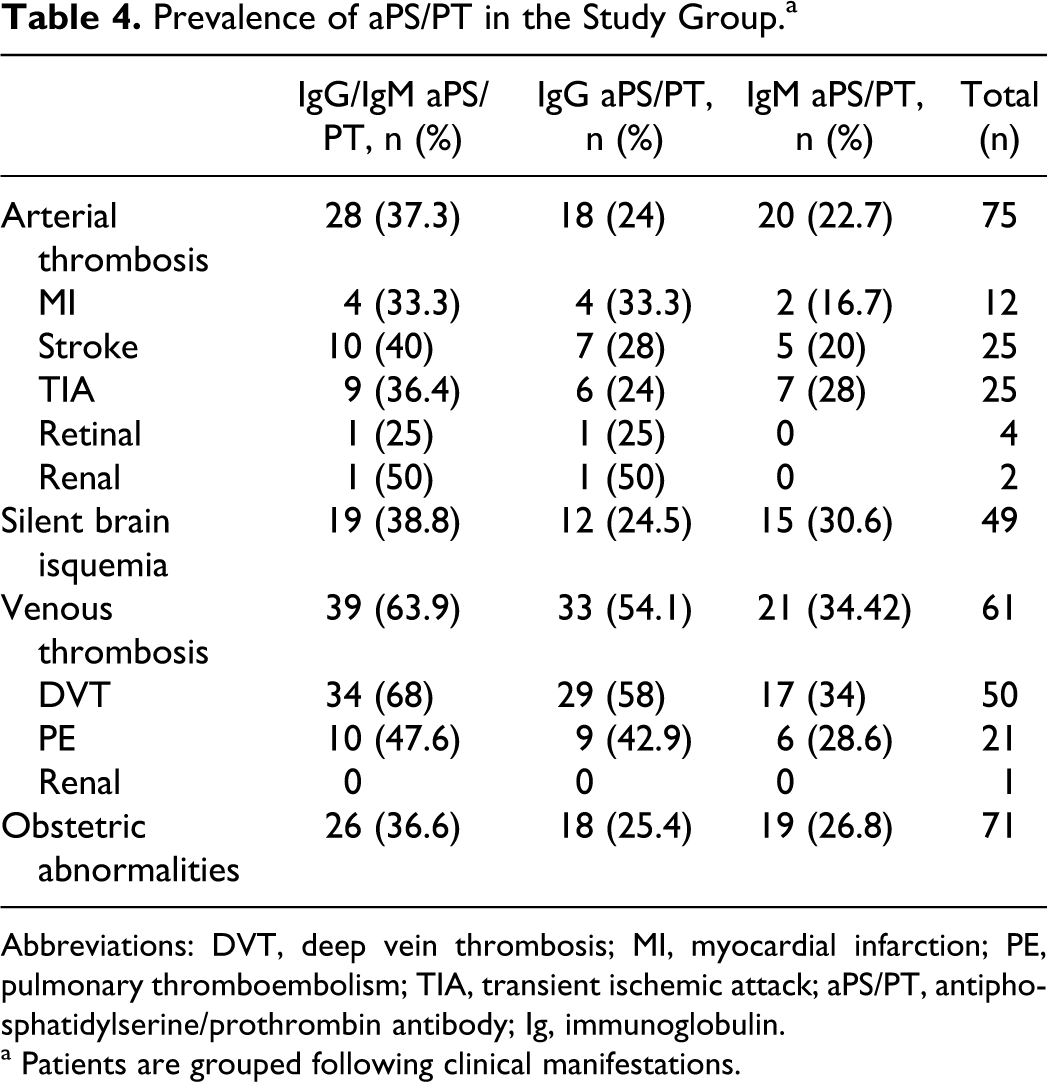
In the study group, 200 patients (67.8%) were positive for at least one aPL (aCL, aβ2GPI, and aPS/PT) and/or for the activity of LA. Among them, 96 patients (32.5%) had positive IgG and/or IgM aPS/PT (from now on referred to as IgG/IgM aPS/PT). In 30 patients, only IgG aPS/PT was found; in 33 patients, only IgM aPS/PT and in 33 patients, both IgG and IgM aPS/PT isotypes were simultaneously detected. Tables 3 and 4 show the presence of aPS/PT in our cohort, grouped by diagnostics and clinical manifestations of APS. The highest aPS/PT prevalence was encountered in APS, with a similar percentage in PAPS and SAPS. Both IgG and IgM aPS/PT were statistically more frequent in APS than in the rest of the autoimmune pathologies (P < .001 between SLE and APS). Among the clinical manifestations, aPS/PT was more frequent in VT, with the highest prevalence in DVT. The aPS/PT was present in about the same number of patients with AT and with obstetric abnormalities (Table 4). The prevalence of other aPL in the group of patients with APS was 40% for IgG/IgM aCL, 33.9% for IgG/IgM aβ2GPI, and 44.1% for the activity of LA.
Prevalence of aPS/PT in the Study Group.
Abbreviations: aPL, antiphospholipid antibody; LA, lupus anticoagulant; MTCD, mixed tissue connective disease; OAD, other autoimmune diseases; PAPS, primary antiphospholipid syndrome; SAPS, secondary antiphospholipid syndrome; SLE, systemic lupus erithematosus; SS, Sjögren syndrome; aPS/PT, antiphosphatidylserine/prothrombin antibody.
a Patients are grouped following diagnosis.
Prevalence of aPS/PT in the Study Group.a

Abbreviations: DVT, deep vein thrombosis; MI, myocardial infarction; PE, pulmonary thromboembolism; TIA, transient ischemic attack; aPS/PT, antiphosphatidylserine/prothrombin antibody; Ig, immunoglobulin.
a Patients are grouped following clinical manifestations.
Clinical Association of aPS/PT
The presence of aPS/PT was significantly associated with at least 1 APS major clinical manifestation. Sensitivity and specificity of IgG/IgM aPS/PT for APS diagnosis were similar to ab2GPI (Table 5). The activity of LA was the most sensitive and the less specific parameter, as it was expected. Specificity of IgG and IgM isotypes for all aPL was similar, indicating that no false reactivity of IgM is detected in our assays.
Sensibility and Specificity of aPL and LA for APS Diagnosis.
Abbreviations: SE, sensitivity; SP, specificity; aPL, antiphospholipid antibody; LA, lupus anticoagulant; APS, antiphospholipid syndrome; Ig, immunoglobulin; aPS/PT, antiphosphatidylserine/prothrombin antibody.
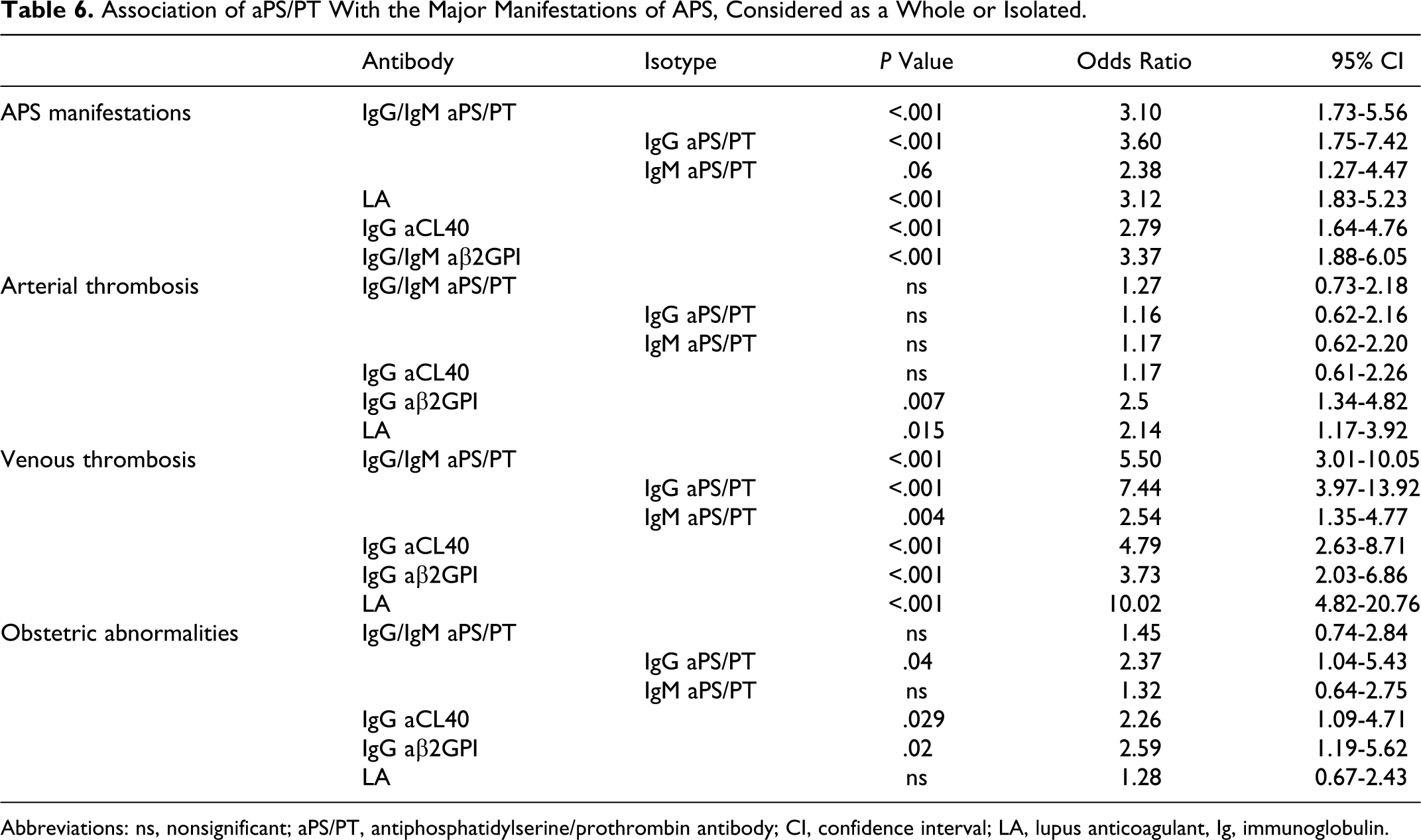
The presence of IgG/IgM aPS/PT was a risk factor for VT (OR 5.5 95% CI 3.01-10.05, P < .001) and the association of IgG aPS/PT was significant for obstetric abnormalities (OR 2.37, 95% CI 1.04-5.43; Table 6). No association was found between the presence of aPS/PT and AT (considered as a group or as a specific arterial thrombotic manifestations: MI, stroke, TIA, retinal artery thrombosis, and renal artery thrombosis).
Association of aPS/PT With the Major Manifestations of APS, Considered as a Whole or Isolated.
Abbreviations: ns, nonsignificant; aPS/PT, antiphosphatidylserine/prothrombin antibody; CI, confidence interval; LA, lupus anticoagulant, Ig, immunoglobulin.
Even more so, when patients with isolated TIA events were excluded from the AT group, 4 the lack of any statistically significant correlation maintained. No association was found between aPS/PT and SBI. In fact, none of the studied aPL or LA is associated with SBI. The presence of LA, IgG aCL40, and IgG aβ2GPI was a higher risk factor for VT than AT (Table 6).
In order to further study the strength of the association of IgG/IgM aPS/PT with VT, a logistic regression model was created as following: a first step of univariate analysis including all studied prothrombotic risk factors (Table 1), the presence of aPL (aCL, aβ2GPI, and aPS/PT) and LA, to determine those with statistical significance. All aPL and LA correlated with VT, as well as prolonged immobility, obesity, and male gender. In a second step, a multivariate analysis including all variables associated with VT. The variables closely associated with VT, independent of other risk factors, were IgG/IgM aPS/PT (OR 2.32, B 0.842, SE 0.432, P = .05), LA (OR 4.76, B 1.561, SE 0.499, P = .002), and male gender (OR 2.462, B 0.901, SE 0.371, P = .015), with an ROC curve area of 0.785 (95% CI 0.721-0.850).
Relationship of aPS/PT With LA
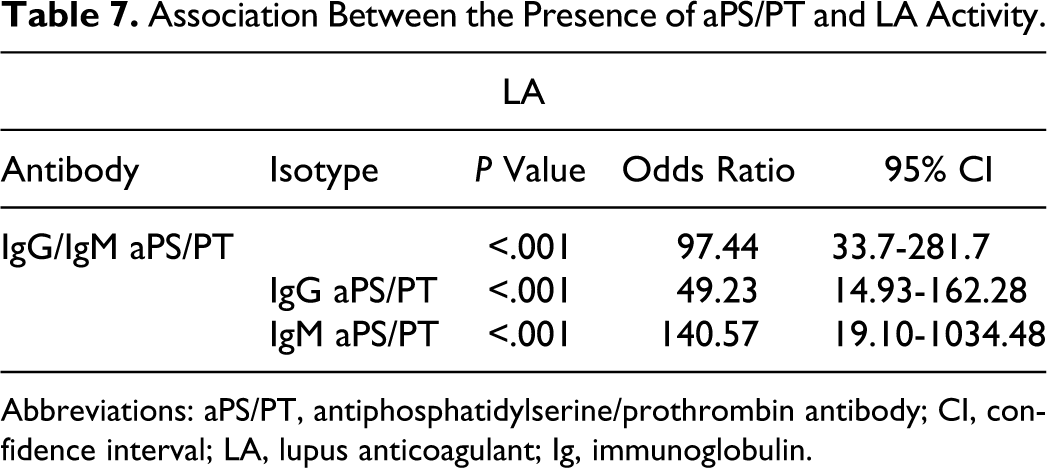
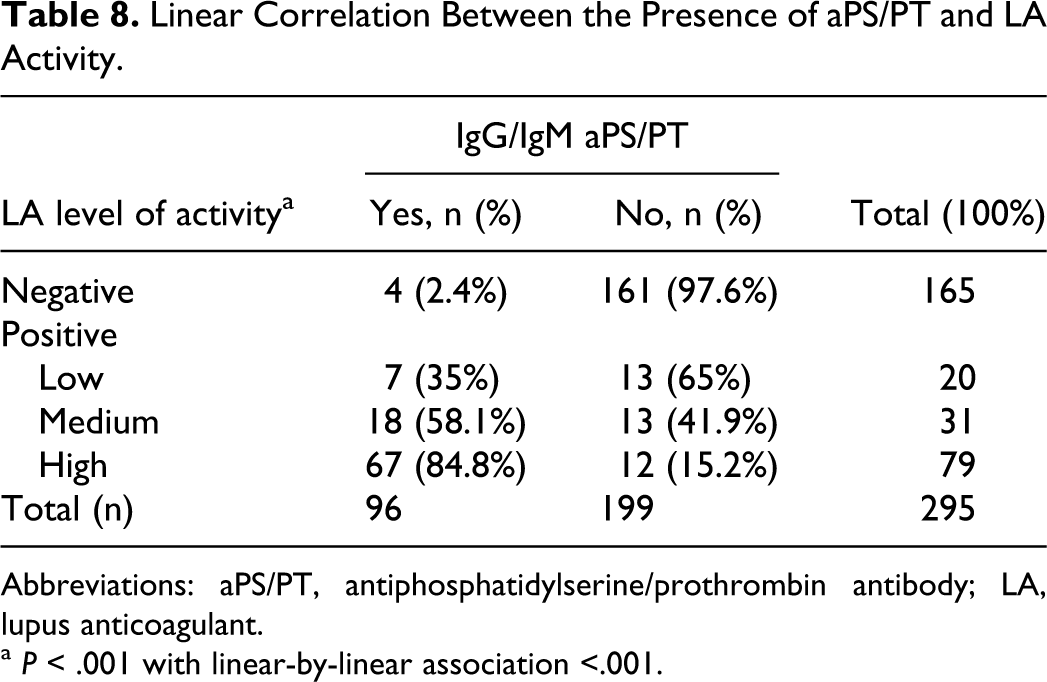
The association between the presence of IgG/IgM aPS/PT and the activity of LA was highly significant (Table 7). Of the 96 patients, 93 patients with IgG/IgM aPS/PT had LA simultaneously (95.38% coincidence for IgG aPS/PT and 98.4% for IgM aPS/PT). Moreover, the prevalence of aPS/PT was higher in the patients with medium or high levels of activity of LA compared with patients with low levels of activity of LA (P < .001 with linear-by-linear association; Table 8). Using Spearman rho nonparametric test, a correlation between the level of activity of LA and IgG aPS/PT (rho 0.594) and IgM aPS/PT (rho 0.645) titters could be demonstrated, as shown in Figure 1. For the rest of the aPL, only the IgG isotype had rho correlation coefficients >0.5 with an activity of LA (IgG aβ2GPI rho, 0.511 and IgG aCL rho, 0.554).
Association Between the Presence of aPS/PT and LA Activity.
Abbreviations: aPS/PT, antiphosphatidylserine/prothrombin antibody; CI, confidence interval; LA, lupus anticoagulant; Ig, immunoglobulin.
Linear Correlation Between the Presence of aPS/PT and LA Activity.
Abbreviations: aPS/PT, antiphosphatidylserine/prothrombin antibody; LA, lupus anticoagulant.
a P < .001 with linear-by-linear association <.001.
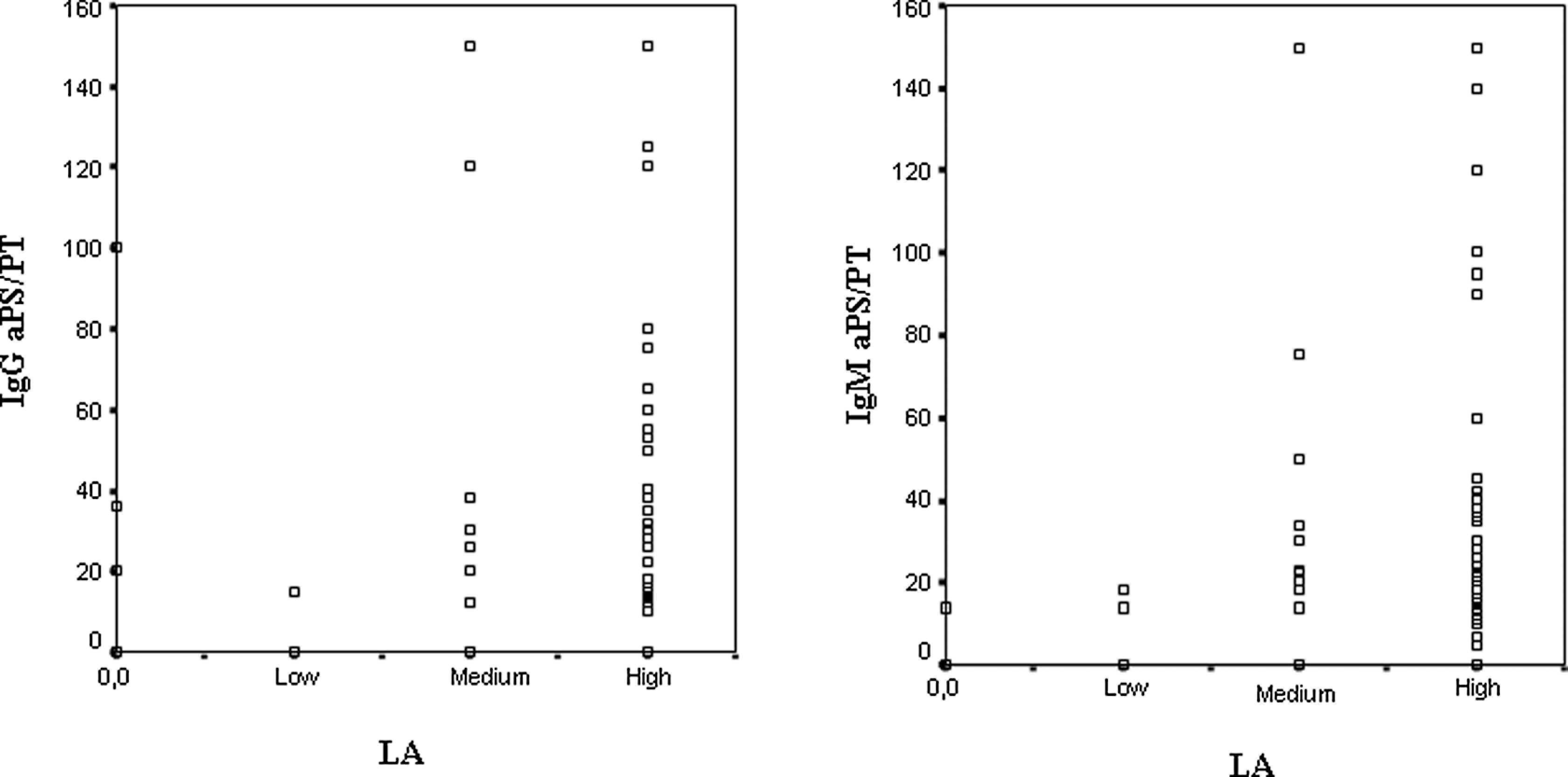
Correlation between the level of activity of LA and the presence of aPS/PT. aPS/PT indicates anti-phosphatidylserine/prothrombin antibodies; LA, lupus anticoagulant.
Discussion
The presence of antiprothrombin antibodies and their association with APS were evaluated in recent years with contradictory outcomes,5,14 but most publications studying the clinical significance of aPT-A and aPS/PT encountered that the antibodies detected via PS correlated better with the clinical symptoms of APS.12,15,16 Currently, it is known that aPT-A and aPS/PT are 2 different antiprothrombin antibody subpopulations with partial identity10,11 and because our study is focused only on aPS/PT, in the present article, we only comment on previous studies performed with this antibody. Moreover, having preliminary results suggesting that aPS/PT may confirm an isolated activity of LA, we have investigated whether aPS/PT detection may be used as a second-level assay, next to the exploration of aCL and ab2GPI, to diagnose APS.
Our study included patients with clinical manifestations compatible with APS or with known APS-related autoimmune diseases (SLE, SS, and MCTD). More studies on the presence of aPS/PT have been performed in patients with SLE14–19 than in patients with OADs.12,20,21 In order to eliminate the possible confounding factors for clinical APS, we systematically looked for clinical and laboratory prothrombotic risk factors.
One third of our cohort had elevated levels of aPS/PT, being the prevalence in patients with APS (51% in PASP and 53% in SAPS) twice as high as in patients with SLE (25.4%), with statistical significance. Similar results have been reported by others authors (Amengual et al 2 : 39% in PAPS, 47% is SAPS, 10% isolated SLE; Bertollacini et al 22 : 54% in SAPS, 16% only SLE; Nojima et al 18 : 38% or 43.4% in SLE; Bertollacini et al 15 : 31% in SLE).
The association of the presence of aPS/PT (IgG/IgM or IgG) with clinical manifestations of APS was statistically significant. Relative risk for presenting APS symptoms conferred by the presence of IgG aPS/PT was comparable or even higher than the risk associated with the presence of LA, aCL, or ab2GPI. Similar data were reported by Atsumi et al, 12 Ieko et al 20 , and Amengual et al. 6
The presence of IgG/IgM aPS/PT, specially the IgG isotype, had a strong association with VT, higher than the presence of aCL or ab2GPI in concordance to previously published results.15,18 In the univariate analysis, VT was associated with the presence of aPS/PT, LA, aCL, and aβ2GPI among the antibodies and with immobility and obesity among the prothrombotic risk factors. The multivariate logistic regression for VT confirmed aPS/PT as an independent risk factor for such an event. The other independent variables associated with aPS/PT in a risk profile for VT were the activity of LA and male gender. Therefore, the prothrombotic information given by aPS/PT determination is not entirely redundant with the activity of LA, a result that confirms the findings of Nojima et al.14,18,19
The association of aPS/PT with AT was not significant, either for IgG/IgM or for the isolated isotypes. The presence of IgG aβ2GPI or LA was statistically associated with AT; nevertheless, the relative risk of AT associated with these antibodies was markedly lower than for VT. Our results, discordant with the ones obtained by other authors,15,17 might be explained by the differences in study populations (APS centered vs SLE centered). Notably, SBI did not associate with either of the aPL studied.
The novelty in our results, as compared to the findings of other authors, relies in the association of the IgG aPS/PT with obstetric abnormalities, with a relative risk similar to the one depicted by IgG aCL and IgG ab2GPI. This association was not demonstrated by LA, although the activity of LA was present in 48.4% of women with obstetric abnormalities and in 36.4% of women without such pathology. Characteristics in study group selection (APS patients) and data processing (including all obstetric abnormalities as a group of manifestations) make our results difficult to compare with those obtained by other authors. In patients with SLE, Bertolaccini et al encountered both aPS and PT isotypes associated with fetal death but not with miscarriages or prematurity. 15 Two articles from the group of Yamada et al included women with recurrent abortions 23 or pregnant women prospectively studied for pregnancy morbidity. 24 They did not find aPS/PT to be statistically associated with obstetric abnormalities despite a prevalence as high as 50% of IgG aPS/PT among premature deliveries.23,24 Another article, from Tsusumi et al, 25 excluding patients with overt thrombotic events, did not find aPS/PT to be present in women with recurrent miscarriages.
Our results confirm the strong correlation found by all the other studies between aPS/PT and LA activity; so close in fact, that aPS/PT was able to act as a confirmatory test for LA activity.2,11,12,18,19,26 This association was much stronger than that between LA and aCL or ab2GPI antibodies. A dose-dependent relationship has been shown between the concentration of aPS/PT and the level of activity of LA in agreement with the hypothesis formulated by Sakai et al, 11 which involve aPS/PT in the increased thrombin/fibrin generation of APS patients with the activity of LA. In fact, these authors showed that a monoclonal antibody with immunological properties of autoimmune aPS/PT had the activity of LA and increased generation of thrombin in a dose-dependent manner.
The accurate detection of activity of LA, despite internationally accepted guidelines and many efforts to improve the standardization of the assays, is still not fully achieved. Furthermore, LA determination is a laborious technique that requires a careful and sequential series of steps. The fact that the presence of aPS/PT strongly correlates with the activity of LA is a very useful feature of this antibody to be used as a confirmatory assay for APS-associated LA.6,12,17,26 Moreover, the aPS/PT assay is easy to reproduce and standardize and may be used to confirm if not substitute the determination of LA, mainly in small centers where hematimetric techniques may not be performed.
Recently, a task force of scientists dedicated to APS research 27 acknowledged that “noncriteria” aPL, like aPS/PT, may have clinical significance and contribute to a better recognition of APS, but more studies are necessary. We feel that the present study follows the recommendation of the task force by confirming previous studies and also bringing new data.
In summary, we propose the inclusion of the presence of aPS/PT antibodies determination in the laboratory criteria for APS classification, joining the opinion of other authors,2,17,18,20,23 as they are highly specific for APS diagnosis and are independently associated with VT and with obstetric abnormalities.
Footnotes
Authors' Note
A.V. performed clinical and laboratory research, designed the statistical analysis, and wrote the article; A.G. designed the study, the patient database, performed clinical research, and supervised the writing of the article; M.V.C. performed lupus anticoagulant determinations and reviewed the article; F.A. performed aPL determinations; J.D. performed the statistical analysis; P.L. performed clinical research; D.P.-S. participate in study design, analyzed and supervised laboratory data and wrote the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
