Abstract
We aimed to investigate the relationship between the extent of venous thromboembolism (VTE) and nonspecific inflammatory markers such as neutrophil to lymphocyte ratio (NLR) and high-sensitivity C-reactive protein (hs-CRP). We retrospectively enrolled 77 patients with VTE (distal deep vein thrombosis [DVT], n = 19; proximal DVT, n = 32; and pulmonary thromboembolism [PTE], n = 26) and 34 healthy controls. In the performed analysis of variance, the levels of white blood cell, NLR, and hs-CRP were clearly different among the groups (control, distal and proximal DVT, and PTE) (P < .001). Especially, a significant increase from the control group to the DVT and PTE was observed in the analysis made for NLR. In the performed receiver–operating characteristic curve analysis, area under curve (AUC) = 0.849 and P < .001 were detected for NLR > 1.84. For this value, the sensitivity and specificity were determined as 88.2% and 67.6%, respectively. The NLR is an inexpensive and a readily available marker that may be effective in determining the extent of VTE, and it is useful for risk stratification in patients with VTE.
Introduction
Venous thromboembolism (VTE) is classified into 2 main groups as deep vein thrombosis (DVT; proximal and distal) and pulmonary thromboembolism (PTE). 1 The course from distal DVT to proximal DVT and PTE is an important indicator of the extent of VTE. 2 There are published data suggesting that the inflammation has a role in not only arterial thrombosis but also the pathogenesis of venous thrombosis. 3–6 However, relatively little is known about the relationship between inflammation and venous thrombosis. High-sensitivity C-reactive protein (hs-CRP) is an acute-phase reactant and a marker of inflammation. 7 The hs-CRP is associated with the risk of arterial cardiovascular disease, but conflicting results have been reported on its role in VTE. 8 Although there are many studies pointing out that the level of hs-CRP increases in patients with VTE, 3–5 , 9–13 the relationship between the extent of VTE and the level of hs-CRP has ever been investigated. Neutrophil to lymphocyte ratio (NLR) as an indicator of systemic inflammation is an emerging marker for both cardiac and noncardiac disorders, and recent studies have demonstrated the prognostic value of NLR in coronary artery diseases (CADs). 14–16 Multiple inflammatory biomarkers were found to be associated with VTE including interleukin (IL)-6, IL-8, soluble platelet selectin (sP-selectin), monocyte chemotactic protein 1 (MCP1), tumor necrosis factor α (TNFα), and C-reactive protein (CRP). In particular, IL-6, IL-8, and CRP were elevated on the first day of admission in patients with DVT. 4 , 6 , 17 However, a study researching the relationship between VTE and its extent and NLR is not available in the literature.
Therefore, this study aimed to investigate the relationship between the extent of VTE and nonspecific inflammatory markers.
Methods
Study Population
We retrospectively enrolled 77 patients with VTE (distal DVT [n = 19]; proximal DVT [n = 32]; and PTE [n = 26]) and 34 healthy controls from January 2012 to December 2012. Patients with overt malignancies, rheumatic and inflammatory bowel diseases, antiphospholipid syndrome, and chronic renal or liver diseases were excluded. Exclusion criteria for both patients and controls also included pregnancy, delivery, surgery or known infectious disease, and use of anti-inflammatory medication within 4 weeks prior to blood collection. The study was approved by the local ethics committee on human research, and written informed consent was obtained from all the study participants. After written informed consent was obtained, a precise medical history was taken, with particular attention to questions about the presence of thrombotic risk factors at the onset of acute DVT.
Diagnosis of VTE
Distal and proximal DVT were diagnosed using peripheral vascular duplex ultrasonography. Proximal DVT was defined as thrombosis at the level of the popliteal veins or above. Distal DVT was defined as thrombosis occurring within the calf veins. Duplex examinations were performed by 2 experienced vascular technicians using Mindray DC-T6 (Shenzhen, China) with a 7-MHz probe. The examination was performed with the patient in a reverse Trendelenburg position at 45°, the knees flexed, and the feet resting on a foot support. The common femoral vein, the femoral vein (proximal and middle), the great saphenous vein (proximal, middle, and distal), the popliteal vein, the short saphenous vein, the posterior and anterior tibial veins, and the peroneal and gastrocnemial veins were examined. The diagnosis of PTE was confirmed in all cases by computed tomography. The VTE was defined as idiopathic if the event occurred in the absence of a known triggering condition, such as surgery, trauma, fracture, immobilization, pregnancy, puerperium, and the use of oral contraceptive. The VTE was defined as secondary if it occurred in the presence of at least 1 of the previous conditions.
Analysis of Blood Samples
After an overnight fasting of 12 hours, peripheral venous blood samples were drawn at the first day of diagnosis. Total and differential leukocyte counts were measured as part of the automated complete blood count using a Coulter LH 780 Hematology Analyzer (Beckman Coulter Inc, Miami, Florida). The hs-CRP and other biochemical parameters were determined by standard laboratory tests. The NLR was obtained by dividing the total count of neutrophils by lymphocytes count.
Statistics
Continuous variables as average ± coefficient of variation and categorical variables as percentage were shown. The difference between not normally distributed continuous variables was tested by Mann-Whitney U test or Kruskal-Wallis test, while the difference between normally distributed continuous variables was tested by independent samples or by analysis of variance (ANOVA). For the difference between the categorical variables, chi-square test or the appropriate one of Fisher exact tests was used. Receiver–operating characteristic (ROC) analyses were used to compare the performance and prognostic power of the NLR, hs-CRP, and white blood cell (WBC) for VTE. The predictive validities were quantified as the area under the ROC curves (c-statistics) and the comparisons of c-statistics were performed by MedCalc statistic software (version 11.3.8.0, Mariakerke, Belgium). A P value of <.05 was considered significant in all statistical analyses. All of the statistical analyses were performed by using SPSS 11.5 (SPSS Inc, Chicago, Illinois).
Results
A total of 77 patients with VTE matching on age and sex (age average 53.7 ± 10.2 years and 70.6% male) and 34 healthy controls (age average 54.1 ± 8.8 years and 73.5% male) were included in the study. Baseline clinical characteristics are summarized in Table 1.
The General Clinical Features of the Study Group.
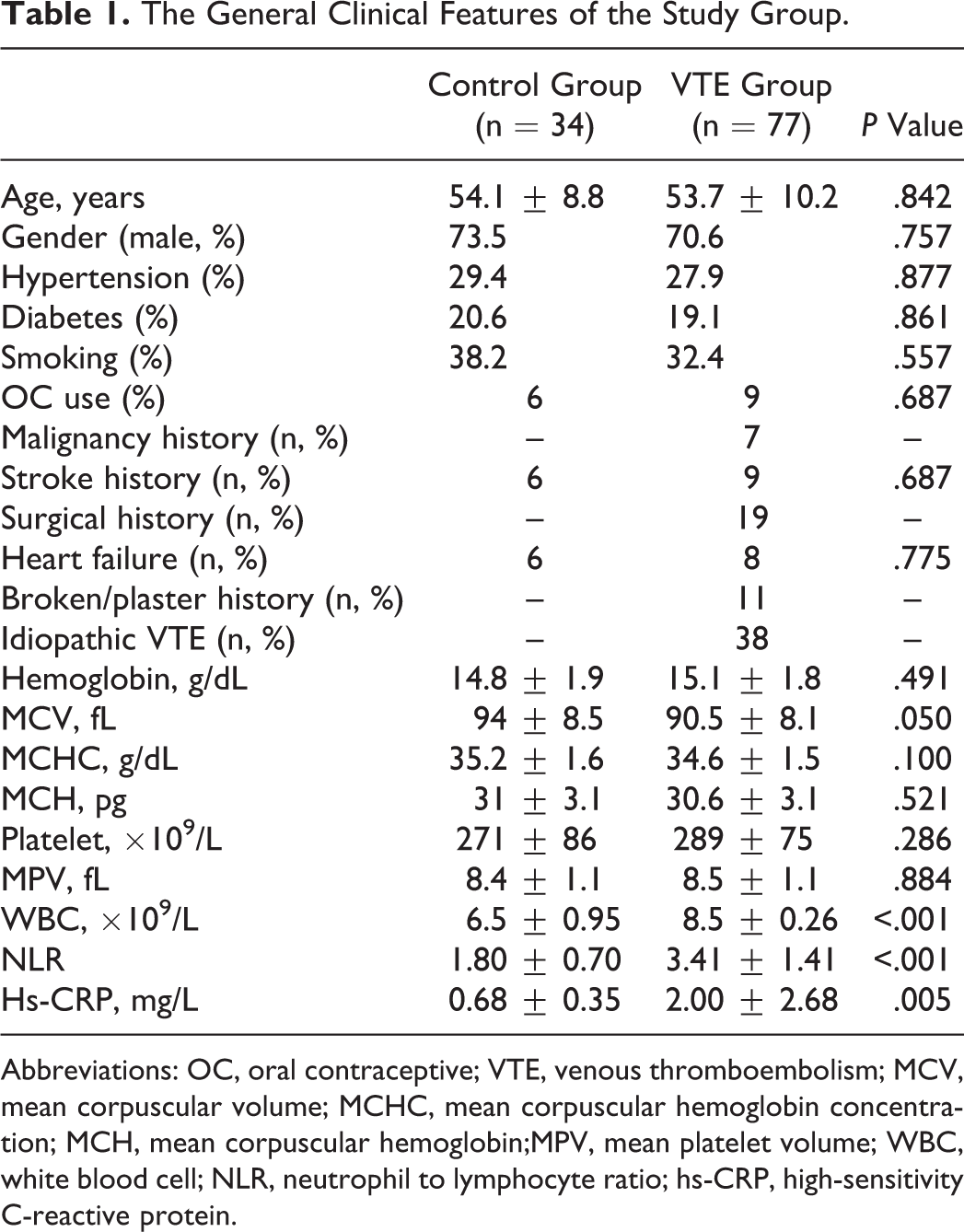
Abbreviations: OC, oral contraceptive; VTE, venous thromboembolism; MCV, mean corpuscular volume; MCHC, mean corpuscular hemoglobin concentration; MCH, mean corpuscular hemoglobin;MPV, mean platelet volume; WBC, white blood cell; NLR, neutrophil to lymphocyte ratio; hs-CRP, high-sensitivity C-reactive protein.
It was detected in patients with VTE that WBC (8.5 ± 0.26 vs 6.5 ± 0.95, P < .001), NLR (3.41 ± 1.41 vs 1.80 ± 0.70, P < .001), and hs-CRP (2.00 ± 2.68 vs 0.68 ± 0.35, P = .005) levels were significantly higher in comparison to the healthy control group. According to the analysis made by ANOVA, the levels of WBC, NLR, and hs-CRP were clearly different among the groups (control, distal and proximal DVT, and PTE) (P < .001). According to the ANOVA test with least significant difference as a post hoc test performed to find the source of the difference between the groups, only hs-CRP was found high in the PTE group in comparison with the other groups, and no difference was detected between the other groups. A significant increase from the control group to DVT and PTE was observed in the analysis made for NLR. In the analysis for WBC, although there was no difference between the control group and the distal DVT, it was found significantly higher in the proximal DVT and PTE (Table 2). The ROC curve analysis was performed in order to find the best cutoff values of these variables in predicting VTE. Area under curve (AUC) : 0.849 and P < .001 were detected for NLR >1.84. For this value, sensitivity and specificity were determined as 88.2% and 67.6%, respectively. The AUC :0.741 and P < .001 were detected for hs-CRP >1.17. For this value, sensitivity and specificity were determined as 45.6% and 94.1%, repectively. For WBC >7.7, AUC = 0.761 and P < .001 were detected. For this value, sensitivity and specificity were determined as 57.4% and91.2%, respectively (Table 3 and Figure 1). The ROC curve AUC values of each of the 3 variables were compared. Although there was no significant difference between hs-CRP and WBC (P = .742), it was observed that there was a clear difference between both NLR and WBC (P = .078), and NLR and hs-CRP (P = .044).
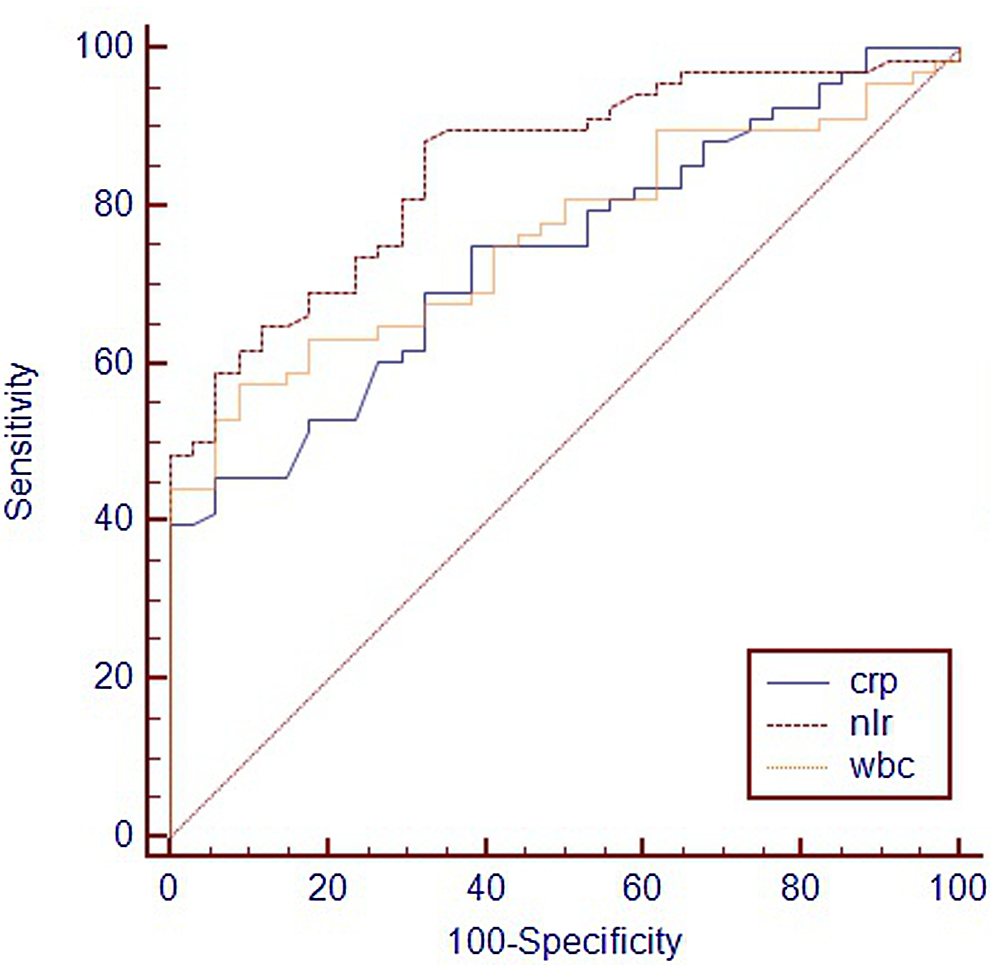
Comparison of receiver–operating characteristic curve for neutrophil to lymphocyte ratio, white blood cell, and high-sensitivity C-reactive protein in prediction of venous thromboembolism.
The Distribution of NLR, WBC and hs-CRP Values Between the Control Group and the Subgroups of VTE.
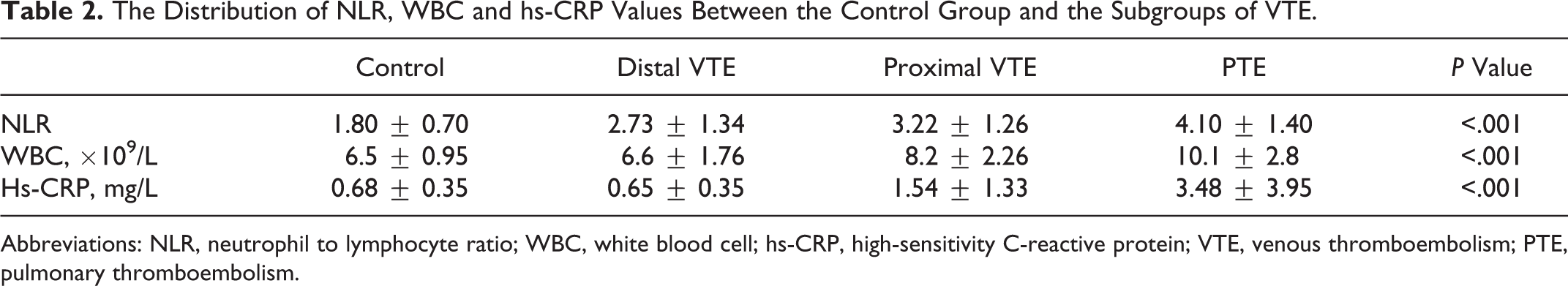
Abbreviations: NLR, neutrophil to lymphocyte ratio; WBC, white blood cell; hs-CRP, high-sensitivity C-reactive protein; VTE, venous thromboembolism; PTE, pulmonary thromboembolism.
Comparison ROC Curve for NLR, WBC and hs-CRP in Prediction of VTE.
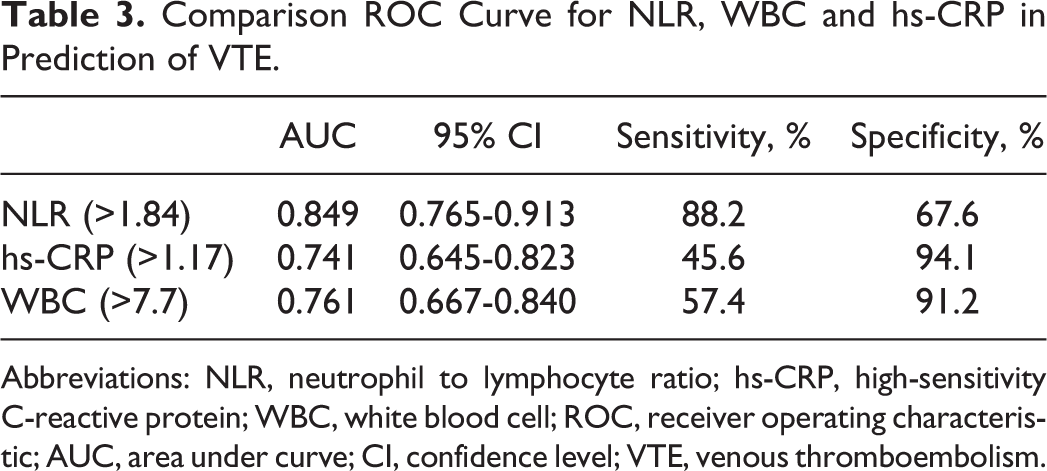
Abbreviations: NLR, neutrophil to lymphocyte ratio; hs-CRP, high-sensitivity C-reactive protein; WBC, white blood cell; ROC, receiver operating characteristic; AUC, area under curve; CI, confidence level; VTE, venous thromboembolism.
Discussion
To our knowledge, our study is the first in the literature researching the relationship between the extent of VTE and NLR and hs-CRP. We used subgroup evaluations (distal DVT, proximal DVT, and PTE) as the indication of the extent of VTE. The results of our study demonstrated that NLR significantly increased in the case of VTE. In the evaluations of the subgroups, NLR was significantly higher in common VTE cases such as PTE and proximal DVT.
Venous thromboembolism is found to be the frequent cause for morbidity and mortality. 18 Although many acquired reasons such as trauma, surgical intervention, and malignancy were defined, it is thought that some of VTE cases stem from hereditary tendency. 19 , 20 Prandoni et al 21 reported that patients with atherosclerosis may be at increased risk of venous thrombosis. On the other hand, evidence suggests the involvement of inflammation not only in the pathophysiology of arterial thrombosis but also in DVT 4 , 5 An established relation exists between inflammatory status and the prothrombotic state in the published data. 22 , 23 Inflammation may interfere with various stages of hemostasis, either through the activation of coagulation or through the inhibition of fibrinolysis and anticoagulant pathways. The main mediators of inflammation-induced activation of coagulation are proinflammatory cytokines. Cytokines, mainly IL-6, are also inducers of hepatic production of acute-phase proteins, such as CRP. 6 The CRP is independently associated with incident cardiovascular disease and may be of value in the identification and stratification of individuals at risk of cardiovascular events. 24 In contrast, the role of CRP in VTE is unclear. A recent review found weak evidence for a causal role of CRP in VTE etiology. 25 A population-based study reports a 1.6-fold increased risk of VTE among patients in the elevated level of hs-CRP. 26 These findings are contradictory to 2 large prospective studies, showing no association between serum CRP levels and the risk of VTE. 8 , 27 The Justification for the Use of Statins in Primary Prevention: An Intervention Trial Evaluating Rosuvastatin (JUPITER) trial showed a significant decrease in DVT in patients with nonhyperlipidemia, with elevated CRP levels, treated with rosuvastatin. 28 The protective effect of statins could be due to their nonlipid effects that include not only antithrombotic properties but also anti-inflammatory properties. 29 Although VTE is not characterized as a chronic systemic inflammatory disease, an inflammatory response, regardless of its origin, could lead to hypercoagulability and increase the risk of VTE. The presence of a systemic inflammatory response can be determined by both the expression of hs-CRP 30 and an elevation in NLR. 31 There are many studies in the literature demonstrating that the hs-CRP level increases in patients with VTE. Elevated level of hs-CRP in patients with VTE was detected in our study in accordance with the previous data.
In the literature, although there are many studies researching the relationship between the multiple inflammatory biomarkers, such as IL-6, IL-8, sP-selectin, MCP1, TNFα, and CRP, 4 , 6 , 17 and VTE, a study researching the relationship between NLR as an inflammatory biomarker and VTE is not yet available. Therefore, our study has the feature of being first. The NLR is the sign of balance between neutrophil and lymphocyte levels in the body. Several studies have shown a relationship between NLR and CAD. It has been shown that NLR was an independent predictor of adverse cardiac events in patients with stable CAD 14 and acute coronary syndrome. 32 Kaya et al 33 have shown that NLR is associated with severity of coronary atherosclerosis. In the present study, neutrophils and the NLR were significantly greater in the patients with VTE, and the lymphocyte count was significantly lower. This observation may be explained by 2 distinct mechanisms, neutrophilia as a reflection of systemic inflammatory status and consequently increased the risk of VTE and lymphopenia as a reflection of the acute stress presented by VTE.
The VTE can be studied in 3 main groups as distal DVT, proximal DVT, and PTE. As well, these 3 groups (from distal DVT to PTE) are the indicators of the extent of VTE. In all, 25% of the symptomatic distal DVT may tend to be proximal and appear as proximal DVT. Similarly, 50% of the proximal DVT may spread and appear as PTE. The course from distal DVT to proximal DVT and PTE is an important indicator of the extent of VTE. In our study, we detected that NLR similarly increased as the extent of VTE increased.
Conclusion
The NLR increases in VTE. The NLR increases as the extent of VTE increases. These findings suggest that the inflammatory process might have an important role in the prothrombotic state in patients with VTE. Also, NLR may be effective in determining the extent of VTE. Unlike many other inflammatory markers and bioassays, NLR is an inexpensive and a readily available marker that may be useful for risk stratification in patients with VTE.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
