Abstract
Soluble thrombomodulin (sTM) has been proposed as a potential marker of ischemic stroke. Results from previous studies remain controversial among different populations. We performed an analysis of plasma levels of sTM in Thai patients with acute ischemic stroke and determined whether sTM levels correlate with stroke subtypes, severity, and risk factors. Ninety-three patients and 76 controls were enrolled. Blood samples were obtained within 24 hours after stroke onset. Plasma sTM levels, measured using quantitative enzyme-linked immunosorbent assay, were significantly higher in patients than controls (P < .005), with the mean ±standard deviation (SD) levels of 3.08 ± 1.05 and 2.57 ± 1.15 ng/mL, respectively. Plasma levels of sTM in patients with cardioembolic subtype were significantly higher than in patients with other stroke subtypes, with the mean ± SD levels of 3.79 ± 1.26, 2.38 ± 0.68 (P < .009), and 2.38 ± 0.44 (P < .05) ng/mL for cardioembolism, large artery atherosclerosis, and small artery occlusion, respectively. Plasma sTM levels were not associated with stroke severity and risk factors of stroke; however, there was a slight relationship between high sTM levels and the presence of atrial fibrillation in the patient group. In conclusion, plasma sTM levels were increased in Thai patients with cardioembolic stroke and may be a potential marker during the acute phase.
Introduction
Thrombomodulin (TM) also known as CD141 antigen is an endothelial-specific type 1 membrane receptor that forms a complex with thrombin, leading to the activation of protein C. This results in the degradation of clotting factor Va and VIIIa and reduction of thrombin. Soluble thrombomodulin (sTM) has been proposed as a potential marker of endothelial injury.1,2 Increased plasma sTM levels have been reported among patients with various diseases, such as atherosclerosis, hypertension, coronary artery disease, and ischemic stroke.3–8 In contrast, other studies have found similar sTM levels between patients with acute ischemic stroke and those of controls.9,10 However, using oligonucleotide microarray, expression of TM gene was increased in peripheral blood mononuclear cells in patients with acute ischemic stroke. 11
In patients with acute ischemic stroke, high plasma sTM levels have been associated with hypertension, older age, high triglyceride levels, and low levels of high-density lipoprotein cholesterol, although the sTM levels were not increased when compared to controls.6,9 Some studies show no relationship between increased sTM levels and stroke subtype, stroke severity, infarct volume, and functional outcome,9,12 whereas other studies have shown that increased sTM levels in lacunar infarction are significantly higher than those in cardioembolic infarction, and the sTM levels are inversely correlated with stroke severity and good outcome. 10 Whether increased sTM concentration plays a role as a predictive or preventive factor for acute ischemic stroke remains controversial. The aim of this study was to investigate the relationship between plasma sTM levels and risk factors of ischemic stroke and sTM levels in Thai patients with different stroke subtypes.
Materials and Methods
Patients and Controls
The study contained 93 patients with acute ischemic stroke who were admitted to the stroke unit of the Department of Internal Medicine at Thammasat University within 24 hours after the onset of the symptoms during May 2009 and May 2010. A personal history including hypertension, diabetes mellitus, hyperlipidemia, coronary artery diseases, atrial fibrillations, previous stroke events, and smoking were obtained. Exclusion criteria consisted of cardiovascular instability, severe anemia (hemoglobin <8.0 g/dL), hemorrhagic diathesis, current infection, current severe allergic disorders, recent cardiovascular events (<6 months), and recent or concomitant deep venous thrombosis. Acute ischemic stroke was confirmed by cranial computed tomography or brain magnetic resonance imaging. Electrocardiography was performed for the detection of arrhythmias.
Acute stroke subtypes were classified as large artery atherosclerosis (LAA), small artery occlusion (SAO), cardioembolism (CE), stroke of other determined cause, and stroke of undetermined cause (UND), according to the Trial of Org10172 in Acute Stroke Treatment (TOAST) criteria. 13 Stroke severity at the presentation was assessed using the National Institute of Health Stroke Scale. 14 Blood was collected within 24 hours after admission and the exact date and time of blood sampling were noted.
Control individuals (n = 76) were recruited from the community elderly club supported by the Faculty of Medicine at Thammasat University. All controls met the same exclusion criteria as was used for the patients with stroke.
Blood Sampling
The venous blood samples were obtained following nontraumatic venipuncture within 24 hours after stroke onset. All samples from patients and control individuals were collected in tubes containing sodium citrate. Plasma was removed following centrifugation for 15 minutes at 1000g at 8°C, subdivided and frozen at −80°C until analysis.
Measurement of Plasma Thrombomodulin
Plasma concentrations of sTM were measured using a commercial monoclonal-based, quantitative enzyme-linked immunosorbent assay kit according to the manufacturer’s instructions (R&D Systems Inc, Minneapolis, Minnesota). A monoclonal antibody specific for human TM had been pre-coated onto a microplate. Standards (recombinant human TM) and samples were pipetted into the wells and any TM present was bound by the immobilized antibody. After washing away any unbound substances, a horseradish peroxidase-conjugated monoclonal antibody specific for TM was added to the wells. Following a wash to remove any unbound antibody-enzyme reagent, a substrate solution was added to the wells and color developed in proportion to the amount of TM bound in the initial step. The color development was stopped and the intensity of the color was measured with a microplate reader (Molecular Devices Inc, Sunnyvale, California).
Statistical Analysis
The data were analyzed with the SPSS software package (version 17.0 for Windows, SPSS, Chicago, Illinois). Continuous data were expressed as mean ± standard deviation (SD) or median with interquartile range ([IQR] 25th and 75th percentiles). The statistical significance of the difference between 2 groups was analyzed with Mann-Whitney test for quantitative variables and the χ2 test for frequency variables. Thrombomodulin of patients with different subtypes was analyzed with analysis of variance. The strength of the correlation was determined using the Spearman rank correlation coefficient. Values at a level of P < .05 were considered statistically significant.
Results
We studied 93 patients (48 females and 55 males). The baseline characteristics of the stroke and control individuals are given in Table 1 . There were significant differences (P < .05) in the prevalence of hypertension (17.2% vs 31.4%) and hyperlipidemia (11.8% vs 25.7%) between patient and control groups, whereas no significant differences were found with regard to age, prevalence of diabetes, coronary heart disease, atrial fibrillation, previous ischemic stroke, or smoking in the 2 groups (all P values > .1 except for smoking, P < .09). Among the patients with stroke, LAA was the most prevalent cause of stroke, followed by CE and small artery occlusion.
Baseline Characteristics of Patients and Controls
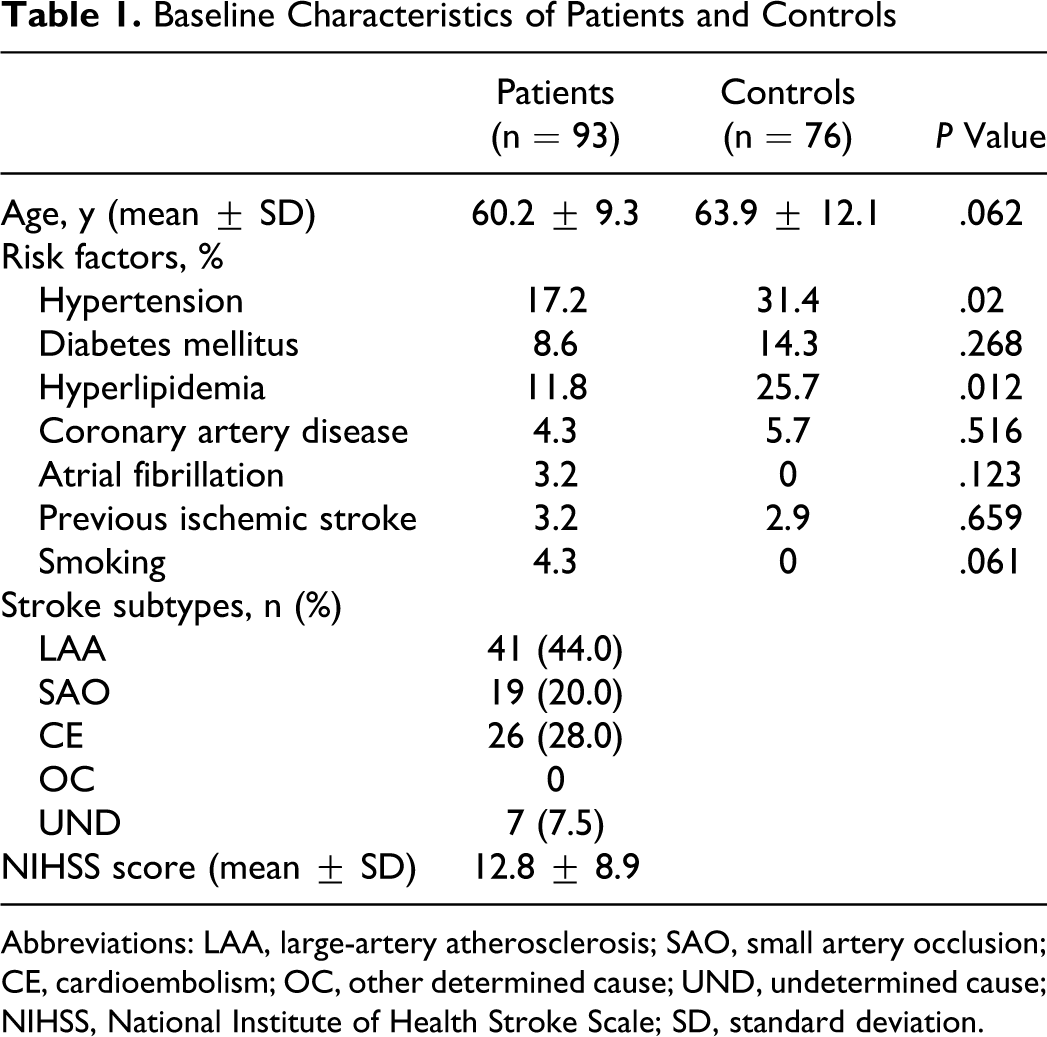
Abbreviations: LAA, large-artery atherosclerosis; SAO, small artery occlusion; CE, cardioembolism; OC, other determined cause; UND, undetermined cause; NIHSS, National Institute of Health Stroke Scale; SD, standard deviation.
Plasma sTM levels in patients were significantly higher than those of controls (P < .005), with the mean ± SD levels of 3.08 ± 1.05 and 2.57 ± 1.15 ng/mL, respectively (Figure 1 ). Using the TOAST criteria, plasma levels of sTM in patients with CE subtype were significantly higher than in patients with other stroke subtypes (CE vs LAA, P < .009; CE vs SAO subtype, P < .05), with the mean ± SD levels of 3.79 ± 1.26, 2.38 ± 0.68, and 2.38 ± 0.44 ng/mL for CE, LAA, and SAO, respectively. The median levels (IQR) of sTM for CE, LAA, SAO, and UND were 3.67 (2.46-4.73) ng/mL (n = 26), 2.14 (1.85-3.28) ng/mL (n = 41), 2.25 (2.01-2.82) ng/mL (n = 19), and 2.04 ng/mL (n = 7), respectively (Figure 2 ).
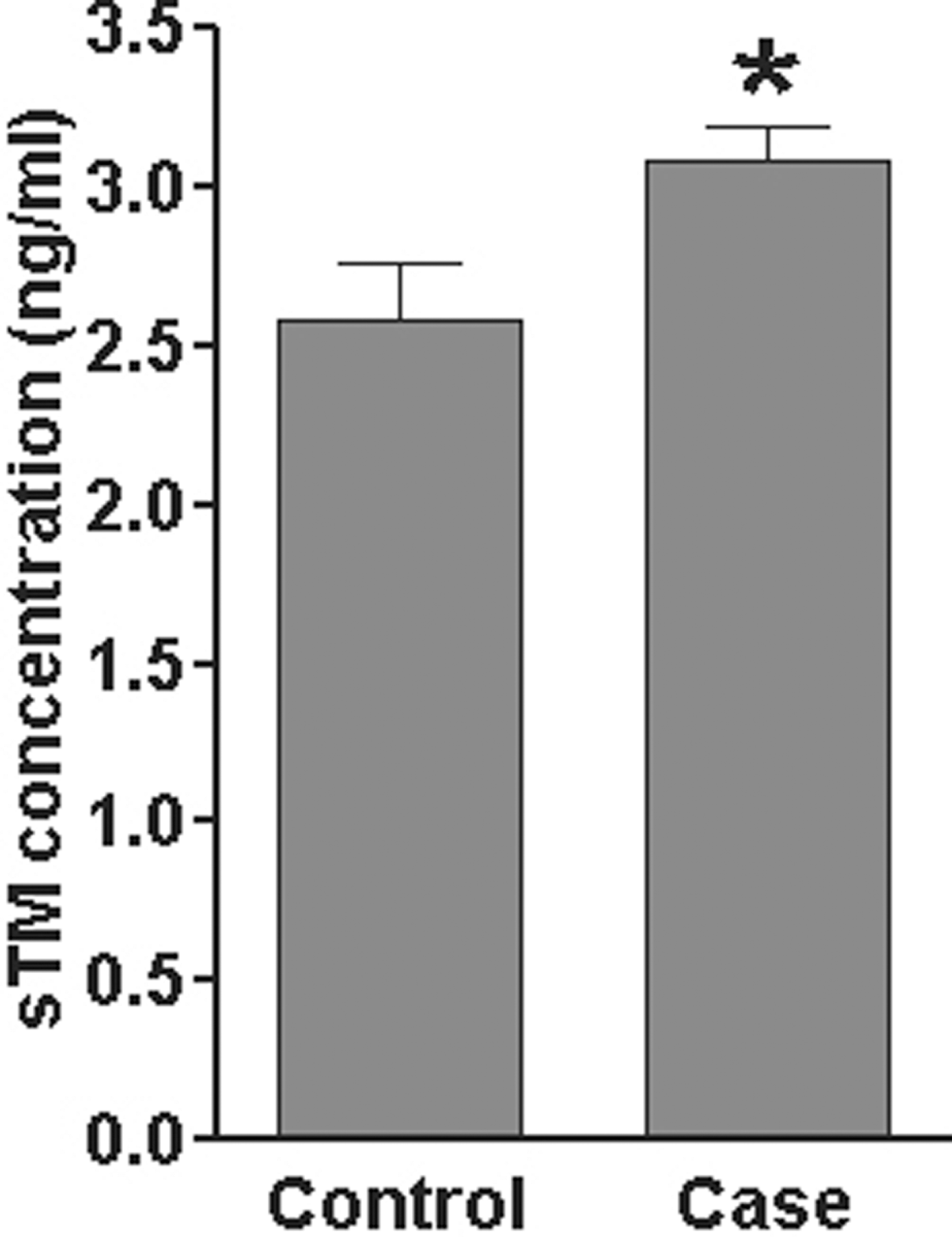
The mean plasma levels of thrombomodulin in controls and patients with acute ischemic stroke. Plasma sTM levels in patients are significantly higher than those of controls. *P = .003. sTM indicates soluble thrombomodulin.
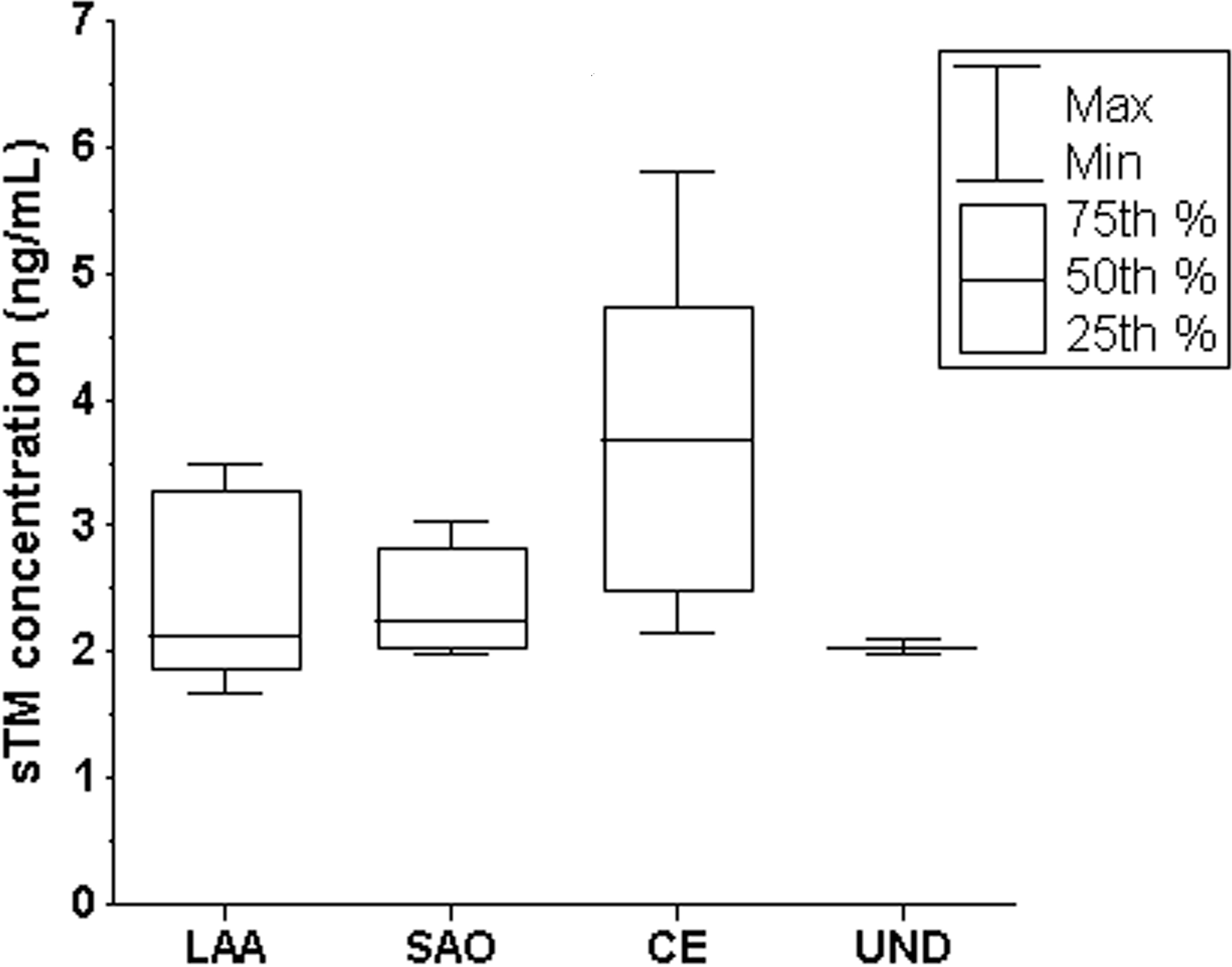
Box and whisker plot of the plasma levels of thrombomodulin in patients with acute ischemic stroke with different stroke subtypes. LAA indicates large artery atherosclerosis; SAO, small artery occlusion; CE, cardioembolism; UND, stroke of undetermined cause.
We found no relationship between sTM concentration and stroke risk factors in either patients or controls (Table 2 ). However, there was a slight relationship between high sTM levels and the presence of atrial fibrillation (P < .09) in the patient group. There was no relationship between increased sTM levels and stroke severity (Spearman correlation coefficient= −.076; P = .5).
Relationship Between sTM Concentration and Risk Factors Among Patients and Controls
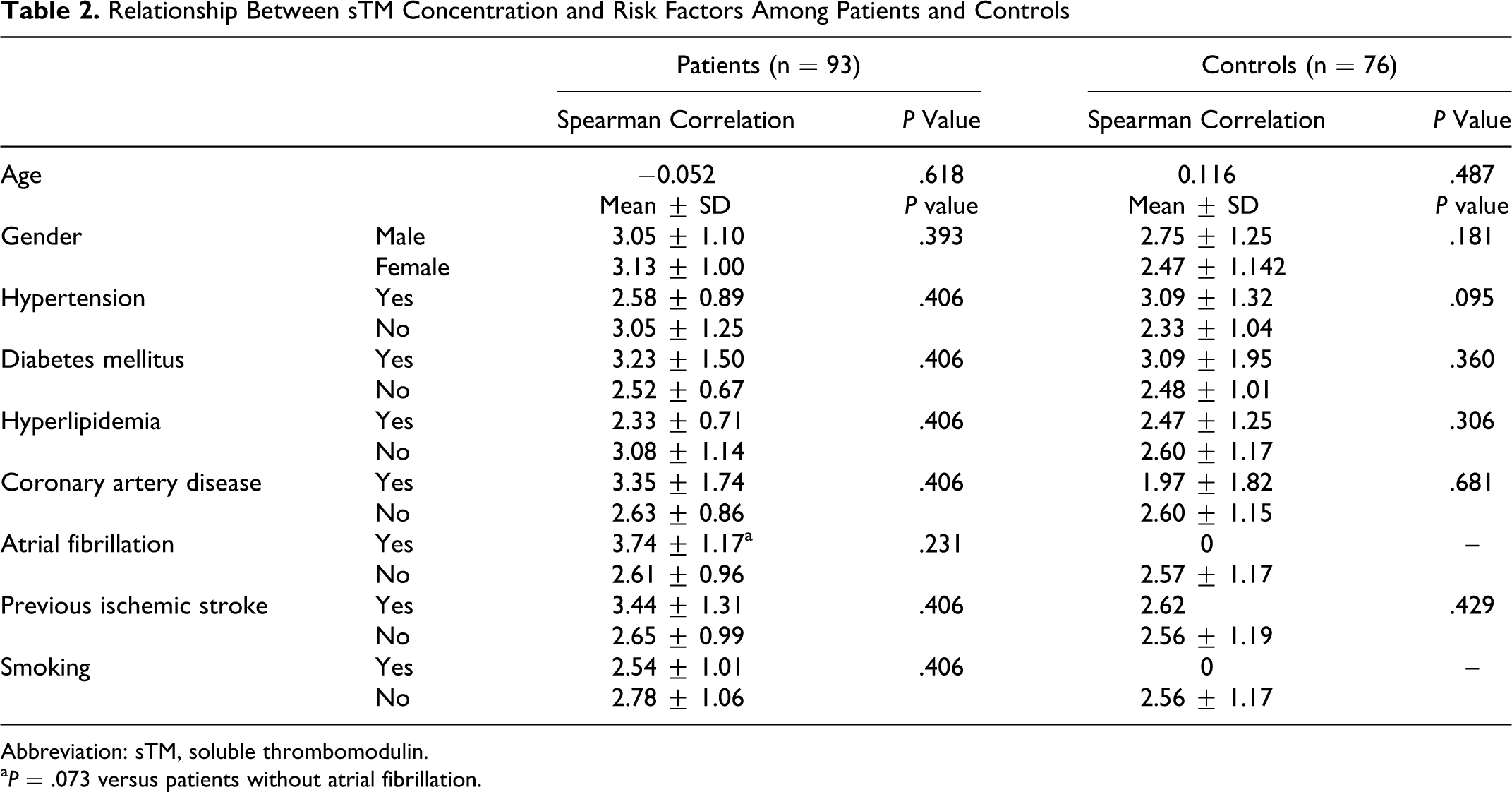
Abbreviation: sTM, soluble thrombomodulin.
a P = .073 versus patients without atrial fibrillation.
Discussion
Peripheral markers of brain injury may help in the diagnosis and management of acute ischemic stroke. Our findings showed that mean plasma levels of sTM were increased in patients with acute ischemic stroke and that patients with acute cardioembolic stroke had, compared to other stroke subtypes, significantly higher mean plasma levels of sTM. Thrombomodulin is a specific endothelial cell receptor that forms a 1:1 stoichiometric complex with thrombin. This complex is involved in the conversion of protein C to the activated form. Once evolved, the activated protein C triggers its downstream pathway, leading to the reduction of the amount of generated thrombin. Through this pathway, it is possible that TM may be released from injured endothelial cells, during acute ischemic attack, to prevent ongoing thrombosis. Thus, plasma sTM is a potential marker of endothelial injury in the brain. The present study supports the relationship between increased plasma sTM levels and acute ischemic stroke. Olivot et al compared patients with stroke to hospitalized nonneurological individuals, who might have conditions that also caused increases in sTM levels, and the result did not show significant differences in sTM levels between patients and controls. 9 The sTM levels increase in various diseases, including pulmonary embolism, respiratory distress syndrome, acute hepatic insufficiency, chronic renal insufficiency, diabetic vasculopathy, systemic erythematosus, and hematologic and nonhematologic malignancies.1,15–17 Zhang et al recruited controls from hospitalized patients with more stringent criteria, and their results showed a significant increase in sTM levels in patients compared to those in controls. 6
Another factor that might affect the levels of sTM is the phase of stroke, acute or chronic. In concordance with Zhang et al, 6 our present study showed a significant increase in sTM levels in patients with stroke during the acute phase of the insult (<48 hours after attack), compared with controls, whereas others did not.7,9,10 No relationship between sTM levels and the time from stroke to blood sampling was found in either acute or chronic phases of stroke,8,9 although sTM levels tend to progressively increase in the chronic phase.7,10 Little is known about the protein kinetics of sTM in the acute phase of stroke. The response to an acute injury of endothelial cells may be different for an acute injury in preexisting atherosclerotic vessels such as in patients with hypertension, diabetes mellitus, and hyperlipidemia and that in presumably normal vessels such as in patients without risk factors for atherosclerosis.
Unlike a previous study, 10 we found that Thai patients with cardioembolic stroke are more likely to have higher sTM levels than those with other stroke subtypes. Together with this finding, we also found a modest relationship between high sTM levels and the presence of atrial fibrillation (P < .09) in the patient group (Table 2). Prevalence of LAA in our study (44%, n = 93) was higher than that of Nomura et al (35%, n = 83), 10 in which the prevalence of SAO (42%) was highest. Prevalence of risk factors for atherosclerosis in our CE subtype was relatively low but not significantly different to that of other subtypes (data not shown). The plausible explanation for our finding is that the vascular condition in the CE subtype may be better than that of other subtypes, making it more responsive to acute endothelial injury. In addition to sTM as shown in our study, recent studies showed that patients with acute cardioembolic stroke had significantly higher levels of tumor necrosis factor-α, interleukin (IL)-6, and IL-1β, compared to other subtypes,18,19 suggesting the potential role of serum markers of inflammation or thrombosis in the CE subtype.
Studies in animals showed significant regional differences in the amount of TM messenger RNA (mRNA) in brain capillaries (ie, cortical > cerebellar > pontine), and interestingly, TM mRNA and activity were undetectable in the brain parenchyma, suggesting that increased TM levels in patients with stroke may associate with lesions involving cortical regions, such as in cardioembolic stroke. 20 In addition, studies showed that the activation of protein C by TM in brain microcirculation was markedly impaired by major stroke risk factors in rats due to downregulation of TM, supporting our explanation that preexisting atherosclerotic vessels may respond poorly to acute endothelial injury. 21 Moreover, brain-specific protein C activation has been demonstrated in humans during carotid occlusion, suggesting a protective role of TM and/or activated protein C during cerebrovascular occlusion. 22 Studies in patients with chronic atrial fibrillation also showed that sTM levels were higher in patients than in controls with sinus rhythm, 23 which is relevant to our finding. The persistent hypercoagulable state, without diurnal variation in patients with chronic atrial fibrillation, may contribute to the high risk of cardioembolic stroke in these patients.
Interpretations of increased sTM levels should be taken with caution, particularly in patients with stroke with risk factors of atherosclerosis because the increased level might result from nonstroke complications of risk factors such as diabetes mellitus. Significantly increased sTM levels can be found in diabetic patients with proteinuria and/or proliferative retinopathy. 24 Most of the previous sTM studies in stroke showed a high prevalence of diabetes mellitus in patients, when compared to controls,6,8,10 which is in contrast to our present study, and particularly in the study that showed significantly increased sTM levels in lacunar infarction, the prevalence of diabetes mellitus in lacunar stroke subtype was as high as 37% (11% for controls, 26% for atherothrombotic stroke, and 19% for cardioembolic stroke). 10 Similar to diabetes, plasma levels of sTM have also been showed to be increased in hypertensive patients and those with atherosclerosis-related peripheral artery disease.3,4
In conclusion, our results demonstrated a significant association between plasma sTM levels and cardioembolic stroke in Thai patients and no association between sTM levels and risk factors for atherosclerosis and stroke severity. The significance of these results should be investigated with larger sample sizes with more stringent inclusion and exclusion criteria regarding risk factors for atherosclerosis for selection of patient and control individuals to further confirm our findings.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by the Commission of Higher Education and the Thailand Research Fund (Grant MRG5180018 to PD).
