Abstract
The objective of this study was to evaluate the heritability of neonatal arterial ischemic stroke (NAIS) in relation to family history of thromboembolic event, maternal diseases, and thrombophilia in both parents (F5G1691A, F2G20210A, and MTHFRC677 T mutations). Forty-two consecutive infants ≥36 weeks of gestation <28 days of life with acute symptomatic NAIS and their parents, as well as 129 controls, were prospectively recruited. Information on maternal data (age, body mass index, oral contraception, migraine, epilepsy, hypertension, and immune disease) and a 3-generation pedigree regarding myocardial infarction, pulmonary embolism, cerebrovascular event, and deep vein thrombosis were obtained. Thrombophilia and maternal diseases did not differ between cases and controls, except for the use of oral contraceptives (more frequent in mothers of controls). No differences were found regarding each studied antecedent of thromboembolic event in the families. The NAIS group showed a higher presence of positive family history among second-degree maternal relatives than did the control infants (odds ratio 4.10; 95% confidence interval 1.29-12.99). Our study does not support the hypothesis that common genetic thrombophilia or familial predisposition to thromboembolic events is associated with the occurrence of idiopathic NAIS.
Introduction
With an incidence of 1/2800 to 1/5000 live births, perinatal arterial stroke is the most frequent form of focal cerebral infarction in children and a major source of long-standing neurological sequelae, including not only hemiplegia but also impairments in language, cognition and behavior, and epilepsy. 1,2 Neonatal arterial ischemic stroke (NAIS) is the most common variety and presents acutely in the first days of life, typically with seizures. NAIS is defined as acute focal cerebral infarction in an arterial territory between birth and 28 days of life that is confirmed by neuroimaging. 1,2
However, its pathogenesis is far from settled, and multifactorial interactions related to both maternal–placental and fetal–neonatal disorders have been suggested. 1,3,4 Susceptibility to ischemic stroke may be influenced by genetic factors, including thrombophilia and constitutional determinants for cardiovascular disease. 1,2,4 –9 However, the role of familial predisposition to NAIS has not been examined in case-controlled studies. The aim of this multicenter case–control study was to evaluate the heritability of NAIS in relation to family history of thromboembolic event, maternal diseases, and genetic thrombophilia in both parents.
Methods
Patients and Control Population
From October 2006 to December 2012, 42 consecutive infants ≥36 weeks of gestation and <28 days of life with acute symptomatic NAIS and their parents were prospectively recruited in 3 university hospitals (Sant Joan de Dèu Hospital, Clinic Hospital, and Hospital of Burgos). Acute symptomatic NAIS was defined as (1) seizures, recurrent apnea, or acute neurological deficit and (1) magnetic resonance imaging (MRI) confirmation of acute ischemic lesion(s) within the territory of the main cerebral arteries. Standard and diffusion-weighted imaging sequences on MRI were performed within 7 days after symptoms, using a specific neonatal head coil and a General Electric 1.5-T signal. Infants with images of tissue loss, atrophy, or cysts and infants with major congenital anomalies, meningitis, sepsis, congenital infections, ECMO, and metabolic diseases were excluded.
One hundred twenty-nine controls were randomly recruited during the study period in the well-baby nurseries of the participating hospitals. Thrombophilia investigations were performed in the 85 first consecutive healthy neonates and their parents (case–control relation about 1:2) between 48 and 72 hours of life, together with standard metabolic screening tests.
Family History and Maternal Diseases
Information was obtained through a structured interview with both the parents. Medical charts from the infants and their mothers were systematically reviewed. The following maternal data were collected: age, weight and height to calculate body mass index (BMI), hypertension, spontaneous abortions, antiphospholipid syndrome, connective-tissue or autoimmune disease such as lupus or dermatomyositis, thrombocytosis, idiopathic thrombocytopenia, polycystic ovary syndrome, migraine, epilepsy, and ingestion of oral contraceptives (OCs). A 3-generation pedigree was obtained for each family regarding (1) myocardial infarction, (2) pulmonary embolism, (3) cerebrovascular event, and (4) deep vein thrombosis.
Laboratory Evaluation
Fathers of infants with NAIS and 85 of 129 fathers of healthy infants were tested for F5 G1691A (F5), F2 G20210A (F2), and MTHFR C677 T (MTHFR) genotypes. Regarding MTHFR C677 T genotype, only homozygosity was considered abnormal.
F5 and F2 mutation were analyzed in genomic DNA extracted from leukocytes of the whole blood using Thrombo-type test based on the DNA Strip (QIAGEN, Hilden, Germany). This technology permits the combined characterization of position 1691 in the F5 gene and position 20210 in the prothrombin (F2) gene. The procedure is divided into 3 steps: DNA extraction from a patient sample (EDTA blood), a multiplex amplification with biotinylated primers, and a reverse hybridization. The C677 T polymorphism in the MTHFR gene was identified using the Hinfl cleavage of a 198-bp polymerase chain reaction-amplified product. Results for infants and their mothers were previously published. 10 In case enough blood was not available to perform all the tests, priority was given to F5 and F2 analysis.
Statistical Analysis
Dichotomous variables were compared by chi-square test or Fisher exact test depending on the number of samples analyzed in each group and expressed as odds ratios (ORs) with 95% confidence intervals (95% CI). Continuous variables were compared by the Mann-Whitney U test. A 2-tailed P < .05 was considered statistically significant. Our sample size of 42 patients was 80% powered to detect a 20% difference in the proportion of our primary variable of interest (positive family history) between infants with NAIS and controls (type 1 error P = .05). Statistical analysis was performed using SPSS for Windows version 21.0. The study was approved by the research ethics committees of the participating hospitals. Informed written consent was obtained from the parents.
Results
Demographic data of the 42 infants with maternal ethnicity were not significantly different, compared to the control group (Table 1).
Demographic Characteristics of 42 Mother–Infant Pairs With NAIS Compared to 129 Controls.a
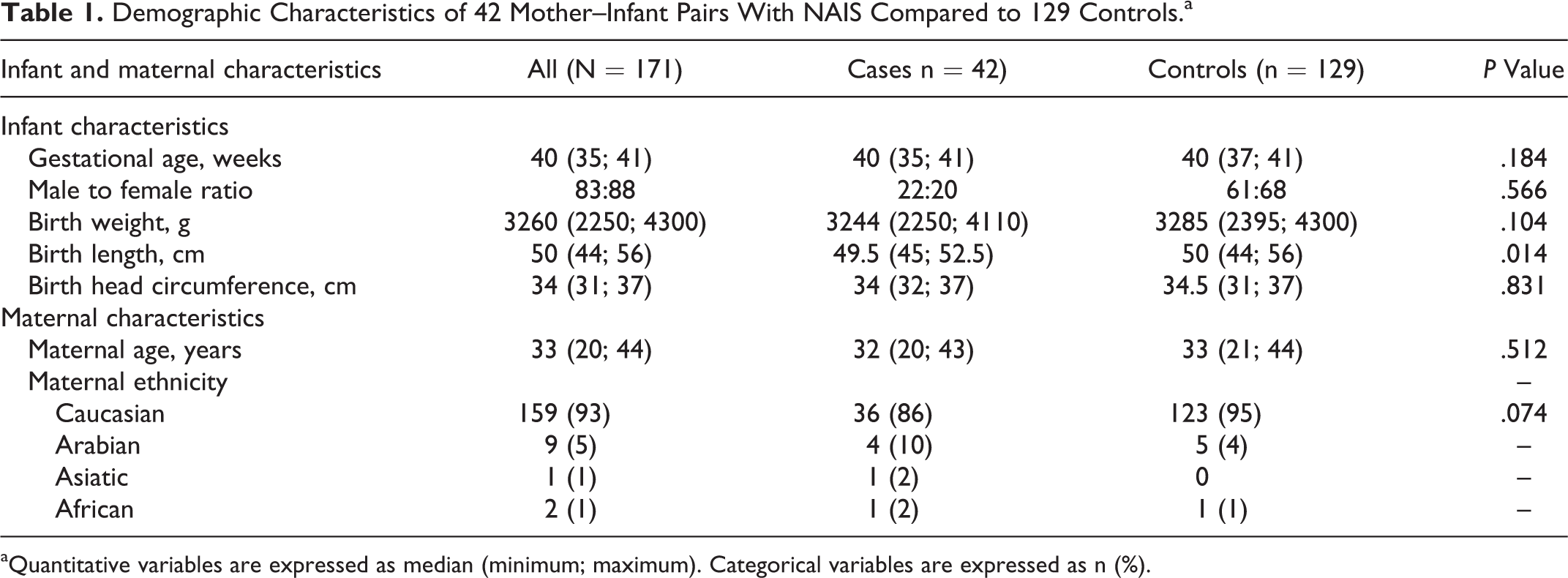
aQuantitative variables are expressed as median (minimum; maximum). Categorical variables are expressed as n (%).
Maternal Data and Family History
Regarding maternal data, no significant differences were found for each antecedent studied between the groups, except for the use of OC, which was more frequent in mothers of controls (OR: 0.38; 95% CI: 0.16-0.93, P = .033). When all maternal data were analyzed together, the occurrence of maternal risk factors was more frequent in controls than in cases with NAIS (OR: 0.46; 95% CI: 0.22-0.95, P = .034; Table 2).
Maternal Factors and Familial Antecedents of 42 Infants With Neonatal Arterial Ischemic Stroke Compared to 129 Controls.
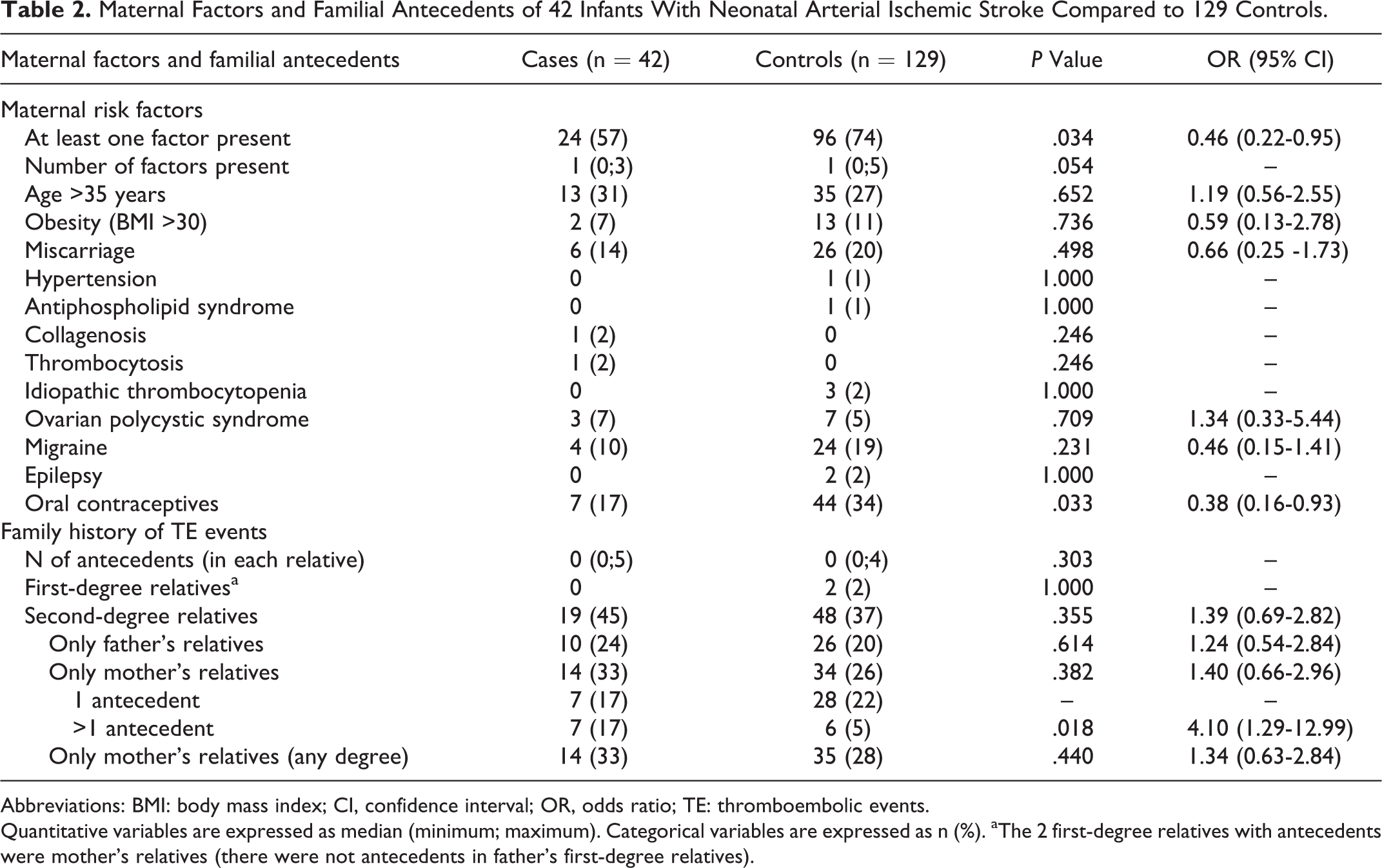
Abbreviations: BMI: body mass index; CI, confidence interval; OR, odds ratio; TE: thromboembolic events.
Quantitative variables are expressed as median (minimum; maximum). Categorical variables are expressed as n (%). aThe 2 first-degree relatives with antecedents were mother’s relatives (there were not antecedents in father’s first-degree relatives).
No differences were found between the 2 groups regarding each studied antecedent of thromboembolic event in the families (Table 3) nor were differences found about family history in first-degree relatives. The NAIS group showed a higher presence of positive family history regarding second-degree maternal relatives (OR: 4.10; 95% CI: 1.29-12.99, P = .018; Table 2).
Familial History of Thromboembolic Events of 42 Cases Compared to 129 Controls.a
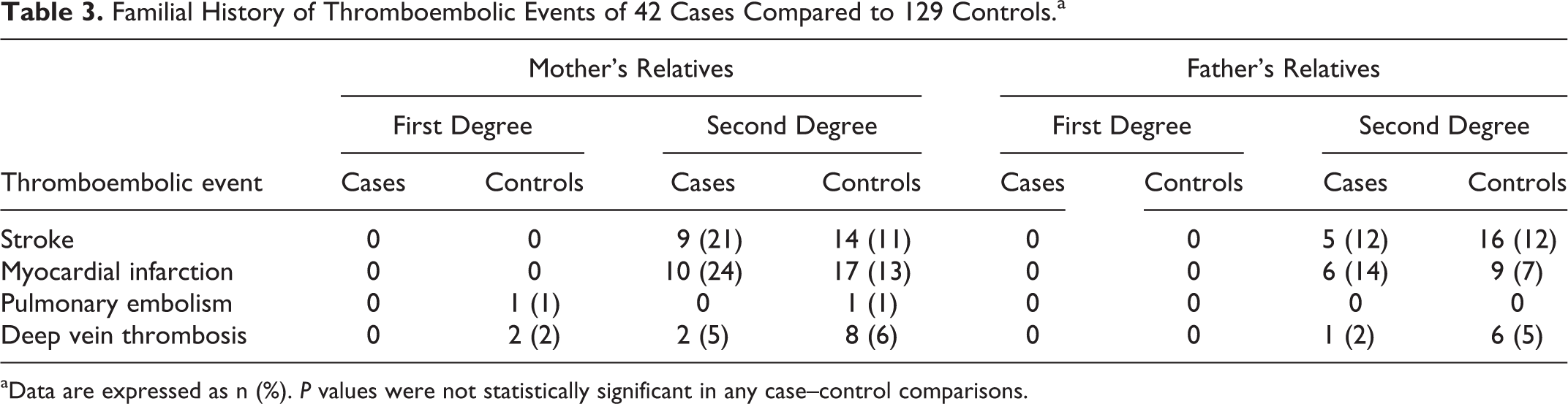
aData are expressed as n (%). P values were not statistically significant in any case–control comparisons.
Thrombophilia Results
The prevalence of the 3 mutations tested in parents did not differ significantly between the 2 groups (Table 4). There were no cases of combined mutations in any mother or father of the infants with NAIS. Combined mutations in mother–father pairs were found in only 2 of 85 in the control group (F5-F2 mutation in one case and F5-MTHFR in the other one).
Thrombophilia Results for the 3 Mutations Tested in Both Parents and Combination of Risk Factors Between 42 Infants With Neonatal Arterial Stroke Compared to 85 Controls.a
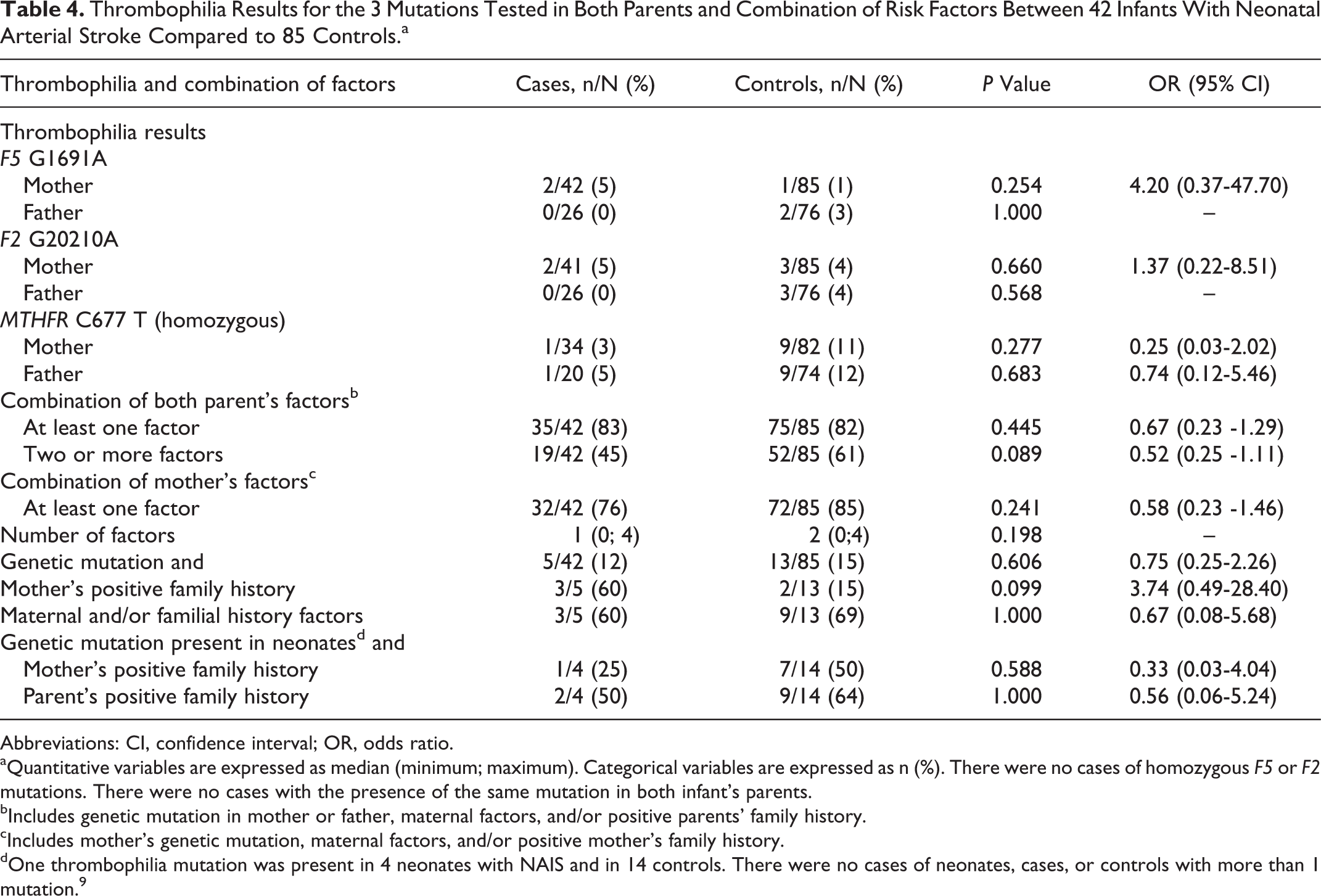
Abbreviations: CI, confidence interval; OR, odds ratio.
aQuantitative variables are expressed as median (minimum; maximum). Categorical variables are expressed as n (%). There were no cases of homozygous F5 or F2 mutations. There were no cases with the presence of the same mutation in both infant’s parents.
bIncludes genetic mutation in mother or father, maternal factors, and/or positive parents’ family history.
cIncludes mother’s genetic mutation, maternal factors, and/or positive mother’s family history.
dOne thrombophilia mutation was present in 4 neonates with NAIS and in 14 controls. There were no cases of neonates, cases, or controls with more than 1 mutation. 9
Combination of Risk Factors
When considering all risk factors studied (maternal data, family history, and thrombophilia), no differences were found between the groups, nor were there differences when considering only the presence of factors related to the mother (Table 4). Focusing on the group of infants who had a genetic mutation (4/42 cases, 14/85 controls), neither additional risk factors in the mother nor in both parents were associated with NAIS. Considering only mothers with genetic mutations (5/42 cases, 13/85 controls), there was a trend toward the coexistence of maternal relatives with a history of thromboembolic events in the NAIS group compared to the controls (OR 3.74; 95% CI 0.49-28.40; Table 4).
Discussion
This is the first case-controlled study of NAIS to examine family history of thromboembolic events and common thrombophilia test in both parents. Importantly, cases were prospectively recruited according to carefully defined perinatal stroke criteria, avoiding the recruitment of heterogeneous populations such as preterm infants and presumed perinatal ischemic stroke, 7,9,11 and all cases were confirmed with MRI. The results of this study suggest that there are no known relevant hereditary factors that predispose infants to idiopathic NAIS.
Family History
A family history of vascular disease is an independent risk factor for some types of arterial stroke in young adults, 12,13 but little is known regarding pediatric arterial stroke. Series that included heterogeneous populations of NAIS, including preterm babies 7 or different types of stroke, 14 have reported a positive family history in 0% to 42% of the infants studied. 6 –8,14,15 However, we have not found any previous prospective case–control study designed to investigate the presence of familial history of thromboembolic event in NAIS. In fact, although we found positive family history in 45% of cases with stroke, this finding was similar in the control population (37%). We did not find any antecedent of thrombosis in first-degree relatives of infants with NAIS, but there was a greater presence of positive family history among second-degree maternal relatives of the NAIS group. As the degree of relatedness correlates with the percentage of shared genes, our results may support the hypothesis that there is not a relevant genetic triggering factor associated with NAIS nor a significant effect of shared familiar environments.
The interpretation of this finding is subject to several caveats. First, the presence or absence of thromboembolic events and stroke in family members was based on parents’ recall without verification of medical charts, except for the obstetrical records. Second, since the incidence rates for stroke and vascular events rise with increasing age, the age of parents at recruitment could be a limitation to finding a positive family history in first-degree relatives. 13
Maternal Data
Although some case-controlled studies have shown associations with some obstetrical risk factors, 2,4,6 previous pregnancy-related disorders have scarcely been studied. We did not find an increased prevalence of maternal diseases related to thrombosis associated with an increased risk of NAIS. Most of the studied factors tended to be more prevalent in the control group, albeit no significant differences were found, except for the use of OC. Certain types of OCs seem to have favorable effects on lipid metabolism and vascular tone, while others have potentially detrimental effects and increase the risk of heart disease and stroke. Compared to combined OC, progestin-only OC seem to be associated with decreased heart disease and stroke incidence. 16 Unfortunately, we did not record the type of OC.
Thrombophilia
It has been suggested that maternal thrombophilia may lead to perinatal stroke due to thrombus formation at the placenta. 2,7,9 Some studies, including ours, have found no association 10,13 while others have reported association for the MTHFR 7 and F5 mutations. 9 Some authors have suggested studying fathers as well, as their thrombophilia profile could contribute to the fetal status 4 ; however, we did not find any association between the thrombophilia profile of the fathers with NAIS and controls. One of the weaknesses of this study is that thrombophilia data from 20% of the fathers were lacking, although our rates of parental thrombophilia are comparable to general population in our country. 17,18
This lack of association between parental thrombophilia and NAIS is in line with recent studies that indicate neonatal thrombophilia may not be related to perinatal arterial stroke. 10,19
Combination of Factors
The most widely accepted hypothesis is that arterial occlusion is more likely to result from multifactorial interactions. 2 Therefore, our aim was to study whether the addition of potential hereditary risk factors could contribute to NAIS. We analyzed whether family history is of further value in predicting the risk of NAIS, once a genetic risk factor has been identified in the neonate or in the mother. In our study, however, positive family history of thromboembolic events, maternal risk factors, and the presence of thrombophilia in the newborn or in the mother were not more prevalent in the NAIS group.
In conclusion, although NAIS is probably a complex heterogeneous multifactorial disorder that might result from the interaction of multiple genes with environmental risk factors, this study does not support the hypothesis that common mutations or familial predisposition to thromboembolic events are associated with the occurrence of idiopathic NAIS. Our data question whether it is reasonable to perform thrombophilia screens in infants who have an episode of NAIS, and to their parents, even if there are antecedents of thromboembolic event in their relatives. However, newer techniques such as genome-wide association studies or next-generation sequencing might give insight into the pathophysiology of this disorder.
Footnotes
Acknowledgments
The authors thank Dr Cilla for her helpful comments on the manuscript and Sara Calvo for providing statistical advice.
Author’s Contribution
J. Arnaez and A. Garcia-Alix contributed to the conception and design of the study. J. Arnaez, G. Arca, A. Martín-Ancel, T. Agut, and A. Garcia-Alix contributed to the acquisition and interpretation of data. J. Arnaez analyzed the data. J. Arnaez and A. Garcia-Alix wrote the paper, and G. Arca, A. Martín-Ancel, and T. Agut revised it critically.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant PI08/1366 from the Instituto de Salud Carlos III cofunded by the European Regional Development Fund.
