Abstract
Introduction
Venous thromboembolism (VTE) is a major health hazard throughout the world. It is a common disease with an annual incidence of 1 to 3 in 1000 individuals. 1 Pulmonary embolism (PE) along with deep venous thrombosis (DVT) is a major clinical manifestation of VTE. The incidence of PE among hospitalized patients is 0.23%, and the mortality rate of this entity without appropriate treatment is reported 2 as 30%. A major theory delineating the pathogenesis of VTE, often called Virchow triad, proposes that VTE occurs as a result of alterations in blood flow (ie, stasis), vascular endothelial injury, and alterations in the constituents of the blood (ie, inherited or acquired hypercoagulable state). 3
Platelets are circulating, anucleate, disc-shaped cells, and their main role is to maintain the integrity of blood vessels through adequate hemostasis. 4 Circulating platelets may differ in size and hemostatic potential.4,5 Larger platelets contain more granules and produce greater amounts of vasoactive and prothrombotic factors, such as thromboxane A2, serotonin, and adenosine triphosphate; they aggregate more rapidly under the stimulus of agonists, such as adenosine diphosphate (ADP), collagen, and adrenaline, and finally, they express a greater number of adhesion molecules, such as P-selectin and glycoprotein IIb/IIIa.6,7 All this leads to greater hemostatic efficiency, in fact, increased mean platelet volume (MPV) values are associated with shortened bleeding times. 8 The main platelet parameters seem to be determined in megakaryocytes during platelet production. Actually, changes in platelet shape, and consequently in volume, may also occur at the sites of activation. In vitro, the contact with agonists, such as serotonin and ADP, causes important changes in shape, with the appearance of pseudopodia, in the activated platelets. 9
Mean platelet volume is a simple and easy method of assessing platelet function.10,11 Although it has been shown that platelet activation occurs in patients with DVT, to the best of our knowledge, no study has examined MPV in patients with DVT solely. In this study, we investigated the MPV in patients with DVT to evaluate platelet activation in this group of patients.
Patients and Methods
The study group consisted of 52 patients with newly diagnosed acute DVT (26 females and 26 males, mean age 56.2 ± 16.9 years). An age-, gender-, and body mass index (BMI)-matched control group was composed of 30 healthy volunteers (20 females and 10 males with a mean age 50.9 ± 17.1 years). In addition to the clinical presentation, all patients and control participants underwent physical examination and ultrasonograpic (US) examination for DVT. The patients were not taking any anticoagulant agent at the time of diagnosis. Hypertension was considered present if the systolic pressure was >140 mm Hg and/or diastolic pressure was >90 mm Hg or if the individual was taking antihypertensive medications. Hyperlipidemia was defined as total cholesterol >200 mg/dL. Exclusion criteria were ventricular systolic dysfunction (ejection fraction <50%), known coronary artery disease, trauma, a new previous surgical operation, atrial fibrillation, history of renal or hepatic disease, malignancy, diabetes mellitus, obesity, hematological disorders, acute or chronic infection, current use of lipid lowering and antiplatelet agents, and stroke. The study was approved by the local ethics committee and all patients gave their informed consent.
Biochemical Measurements
Blood samples were drawn from the antecubital vein by careful venipuncture using a 21-guage needle attached to sterile syringe without stasis at 8.00 to 10.00
Ultrasonographic Examination
In routine of clinical approach, US has become a widely accepted and a primary diagnostic procedure for the evaluation of patients with clinically suspected deep vein thrombosis. All Duplex Doppler ultrasonography (DUS) examinations were performed in a single department of radiology by the same experienced radiologist about vascular US. Deep vein thrombosis was determined DUS of the lower limb venous system using the same machine (GE Voluson 730 Expert; GE Medical Systems Kretztechnik GmbH & Co, Zipf, Austria).
The femoral vein examined in supine position, whereas examination of the popliteal vein was made in a prone position or lateral position. The veins were evaluated in the transverse and longitudinal planes using DUS with the linear array probe, at the frequency of 5.0 to 10.0 MHz. At first, we attempted to identify DVT using conventional gray scale US.
We used various US techniques such as compression ultrasonography, color Doppler Flow Imaging, and spectral Doppler for diagnosis of DVT. Real-time images of the deeps veins (common femoral, deep femoral, femoral, popliteal, and soleal veins), the superficial veins (great saphenous, and small saphenous veins with accessories), perforator veins, saphenofemoral junction, and saphenopopliteal junction were directly visualized and subsequently assessed for their compressibility in the transverse and longitudinal planes. After confirming the common femoral vein, the longitudinal plane examination was made upward until iliac veins. If an abnormal finding was present, the iliac veins and inferior vena cava were obtained.
Noncompressibility or partial compressibility of the vein, no flow signal, the absence of spontaneous flow, and the absence of respiration-modulated phasicity of the evaluated veins were taken as the criteria for DVT. The valvular regurgitation was assessed using the Valsalva maneuver or manual compression.
Statistical Analysis
Data were analyzed with the SPSS software version 10.0 for Windows. Continuous variables from the study groups were reported as mean
Results
The clinical and laboratory findings of the study and control groups were summarized in Table 1 . There were no statistically significant differences between the 2 groups with respect to age, gender, systolic and diastolic blood pressures, and levels of glucose, creatinine, and hemoglobin (Hb). Mean duration of symptoms was 6 ± 4 days in both groups. In all study patients, DVT was limited and above the knee. Mean platelet volume was significantly higher among patients with DVT when compared with control group (8.6 ± 0.8 vs 7.7 ± 0.9 fL, respectively; P < .001). White blood cell count was significantly higher among patients with DVT when compared with control group (8.7 ± 2.8 vs 6.6 ± 1.9 × 109/L, respectively; P < .001). However, white blood cell counts in 2 groups were in normal physiological limits. Platelet count was significantly higher among patients with DVT when compared with control group (221.5 ± 58.5 vs 195.0 ± 36.9 ×109/L, respectively; <.02). The correlation analysis indicated that MPV was only negatively correlated with Hb levels (P = .03, r = −.22). There was no correlation between MPV and other variables. In linear regression analysis, when MPV was taken as independent variable with other study variables which are potential confounders (age, gender, BMI, smoking, etc), MPV was independently negatively correlated with Hb levels (P = .03, β = −.23).
Comparison of Clinical and Laboratory Findings of the Patients With Deep Vein Thrombosis and Control Participants
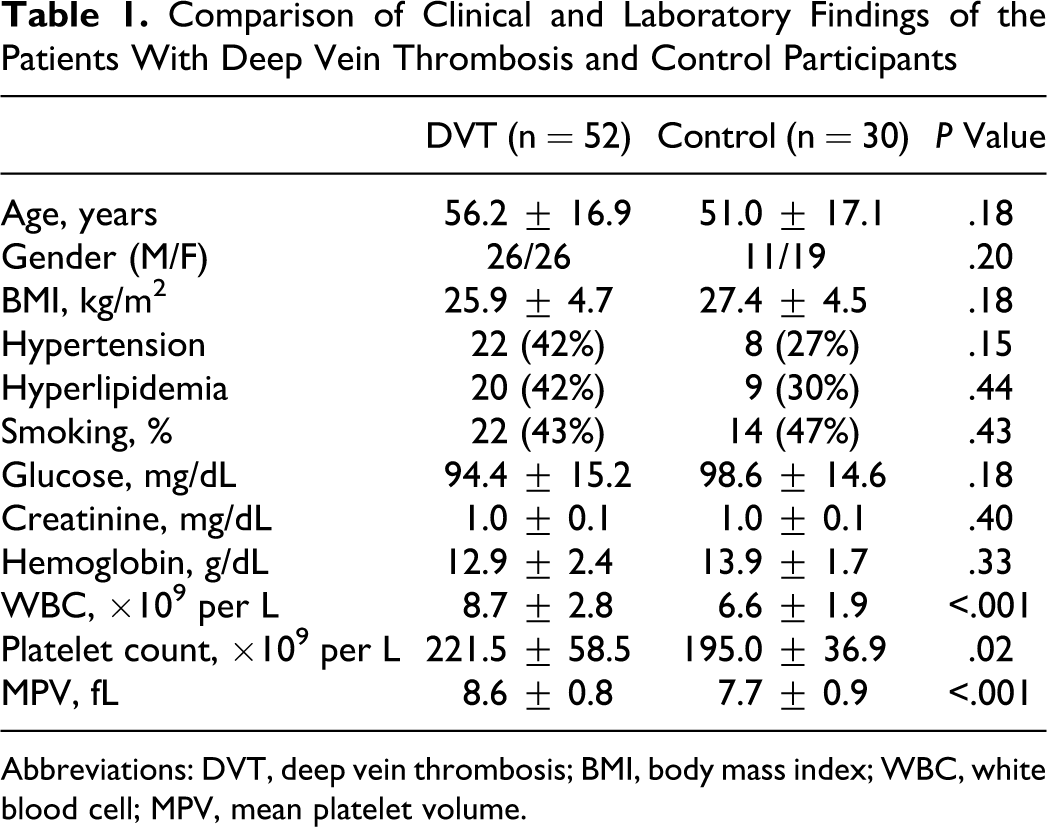
Abbreviations: DVT, deep vein thrombosis; BMI, body mass index; WBC, white blood cell; MPV, mean platelet volume.
Discussion
In the present study, we examined MPV, an indicator of platelet activation in patients with newly diagnosed acute DVT. We mainly showed that MPV was significantly higher in patients with DVT than control participants.
Human platelets are anucleated cells derived from megakaryocytes and they are involved in many pathophysiological processes, including hemostasis and thrombosis, thrombus retraction, vessel constriction and repair, inflammation including promotion of atherosclerosis, host defense, and even tumor growth/metastasis. Platelet size, measured as MPV, is a marker of its function and is positively associated with indicators of platelet activity, including aggregation and release of thromboxane A2, platelet factor 4, and β-thromboglobulin.6,12,13 The MPV is considered a marker and determinant of platelet function since larger platelets are hemostatically more reactive than platelets with normal size. It was well documented that platelet activation has an important role in atherothrombosis. 11 Increased MPV, a simple marker of platelet activation,6,8 was found to predict fatal outcome in acute coronary syndromes and was related to the severity of acute ischemic cerebrovascular events.13–17 Recently, platelet activation has also been reported in VTE.18,19 We studied MPV, an indicator of platelet activation, in patients with DVT. As a difference, we compared this patient group with control group. Our study confirms the hypothesis of MPV elevation in patients with DVT.
Hypercoagulability, venous stasis, and endothelial injury are important factors involved in the pathogenesis of VTE. Since the first 2 factors facilitate activation of the coagulation pathway, it is generally accepted that venous thrombi, known as red thrombi, are mainly composed of erythrocytes, with a large amount of fibrin. 20 However, they also contain a few platelets.
Platelets have central role in pathophysiology of atherothrombotic cardiovascular diseases. Increased MPV has been shown in patients with cardiovascular diseases and in patient groups with known coronary artery disease risk factors 11 Moreover, increased MPV has also prognostic role in cardiovascular diseases, for example, it is associated with higher mortality following myocardial infarction.11,21 These findings raise the hypothesis of the potential importance of MPV in the underlying pathophysiology of atherothrombotic cardiovascular disease. Recently, it has been suggested that there are common pathophysiological links between VTE and atherothrombosis.22–24 A significantly higher incidence of acute coronary syndromes and stroke was reported in patients after an episode of VTE, suggesting that arterial thromboembolism and VTE may share common risk factors.
Previous studies have shown platelet activation in patients with VTE. Increased levels of P-selectin have been shown in patients with VTE, 25 and a higher level of circulating P-selectin was associated with increased risk of recurrent VTE in patients with first unprovoked VTE. 26 Altogether, these studies support the hypothesis that platelet activation may also play a role in the pathogenesis of VTE. Recently, increased levels of MPV were identified as a predictor for VTE in particular, VTE of unprovoked origin. 18 Both PE and DVT are 2 clinical presentations of VTE and share the same predisposing factors. In most cases, PE is a consequence of DVT.
There are some limitations of this study that should be concerned. The small number of patients was the limitation of the study. We included the patients with newly diagnosed acute DVT. Because of that our sample size was small. Another limitation of this study is that the analysis was based on a simple baseline determination that may not reflect the patient status over long periods.
In conclusion, we have shown that MPV was significantly elevated in patients with DVT compared to control participant. This study shows that platelet activation has an important role in pathogenesis of DVT. Further prospective studies are mandatory to establish the pathophysiological and clinical significance of increased MPV in patients with DVT.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
