Abstract
This study aimed to explore the association between mean platelet volume (MPV) and preoperative deep vein thrombosis (DVT) in older patients with hip fracture. A total of 352 consecutive older patients with hip fracture were included from January 2014 to December 2020. MPV values were measured on admission, and color Doppler ultrasonography was performed for DVT screening before the planned surgery. The receiver operating characteristic (ROC) curve was used to establish the optimal cut-off value for the prediction of DVT. Univariate and multivariate logistic regression analysis were used to examine the association between factors and DVT. The overall prevalence of preoperative DVT was 15.1%, and patients with DVT had a lower value of MPV than non-DVT patients (11.6 ± 1.2 fL vs 12.3 ± 1.4 fL, P < .01). The cut-off point according to the ROC curve for MPV was 13.3 fL, and multivariate logistic regression analysis showed that MPV level < 13.3 fL was significantly associated with an increased risk of DVT (OR = 4.857, 95% CI: 1.091-21.617, P = .038), and with every 1.0 fL decrease in MPV, the risk increased by 27.7% (OR = 1.277, 95% CI: 1.001-1.629, P = .047). Our findings indicate that a low MPV level is associated with DVT in older patients with hip fracture. As MPV is a simple indicator that can be calculated from the blood routine test, it may be a potential biomarker of DVT with the combination of other tests, further studies are needed to confirm these results.
Introduction
Venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE), is a common and potentially fatal disease. 1 Despite the development of prevention efforts, the incidence of VTE remains high, contributing to a substantial mortality rate, recurrence risk and global health burden.2–4 As is well known, timely and accurate diagnosis of VTE followed by treatment is crucial to prevent serious complications. 1 In clinical practice, D-dimer testing is a most commonly used to rule out or confirm VTE events, but its diagnostic value is still limited due to a lack of specificity. 5 Therefore, it is essential to identify effective diagnostic biomarkers for VTE.
Thrombus formation is a complex process, in which platelet function play an essential role in both venous and arterial thrombosis.6,7 Mean platelet volume (MPV), the most commonly used measure of platelet size, is regarded as a potential marker of platelet activity.8,9 Increasing evidences indicated that MPV was significantly associated with VTE events.10–14 However, MPV is influenced by age, 15 and few studies have evaluated this relationship in older adults. 16
With an aging worldwide population, hip fractures represent a global public health issue. 17 In addition to high morbidity and mortality, lower extremity DVT is a common complication in hip fracture patients, and reported incidence rates as high as 11.1-35.0%.18–22 Likewise, the incidence of asymptomatic and symptomatic PE was not low, reaching up to 18.9% and 0.2%, respectively.23,24 After the development of VTE, length of stay and total cost increased approximately two-fold, 25 and significantly decreased patients’ survival rate. 24
However, to our knowledge, little research has investigated the association between MPV and thrombosis in older patients with hip fracture. On the basis of the hip fracture database that established by our team, 26 we aimed to explore the relationship between MPV and preoperative DVT in these patients.
Materials and Methods
This was a retrospective case-control study based on our hip fracture database, which has been described in detail in the previous study. 26 Briefly, the inclusion criteria of this database were: (1) confirmed diagnosis of hip fracture, and not pathological fracture; (2) age 60 years and over; (3) caused by low-energy fall; (4) fresh fracture less than 3 weeks. From January 1, 2014 to December 31, 2020, a total of 1044 patients have been consecutively enrolled into the hip fracture database. In this study, patients with the following were excluded: (1) no DVT screening by Doppler ultrasonography; (2) postoperative DVT; (3) previous VTE before the fracture; (4) without MPV value. The study protocol was approved by the Institutional Ethics Committee at Deyang People's Hospital (approval number 2021-04-019-K01), and conducted in accordance with the principles of the Declaration of Helsinki. The need for informed consent was waived because all data were collected retrospectively and anonymously without unique patient identifiers.
After admission, all patients were routinely assessed for both bleeding and thromboembolism risk. For patients without contraindications, enoxaparin (4000 AxaIU/0.4 mL, once per day; Changzhou Qianhong Bio-pharma Co., Ltd China) was subcutaneously administered for preventing VTE. The anticoagulant therapy was discontinued 12 h prior to surgery and restarted 12 h after surgery. Meanwhile, a plantar arteriovenous pump (20 min, twice per day; DSM-4S, Daesung Co., Ltd Korea) was used to promote blood reflux. Before the planned surgery, patients underwent color Doppler ultrasonography (iU 22, Philips, The Netherlands) for DVT screening, which were conducted by an experienced radiologist, and the results were reviewed by another senior radiologist. Briefly, the following veins of bilateral extremities were scanned: iliac vein, common femoral vein, superficial femoral vein, popliteal vein, anterior tibial vein, posterior tibial vein, fibular vein and calf muscle vein. The diagnosis of DVT was based on the Robinov group's criteria. 27 If confirmed diagnosis of DVT, a therapeutic dose of enoxaparin (100 IU/kg, twice per day) was administered immediately. Furthermore, the inferior vena cava (IVC) filter was implanted when necessary.
The following data were collected from the hip fracture database, including demographics [age, sex, body mass index (BMI), and smoking], comorbidities (hypertension, diabetes, anemia, pulmonary disease, atrial fibrillation, renal disease, liver disease and tumor), fracture information (time from injury to hospital, hip fracture type), and ultrasonography result (with/without DVT, thrombosis location). BMI was calculated as weight in kilograms divided by height in meters squared. Smoking status was categorized as current smoking, no current smoking or unknown. Time from injury to hospital (days) was defined as the time interval from injury date to admission date. According to the location of thrombosis, DVT was then classified into three types: (1) proximal type (iliac vein, common femoral vein, superficial femoral vein, popliteal vein); (2) distal type (fibular vein, tibial vein and calf muscle vein); (3) mixed type (both proximal and distal thrombosis).
On admission, whole blood samples were collected into Na2-ethylenediaminetetraacetic acid (EDTA) tubes, and MPV values were measured within 2 hours by using an automatic hematology analyzer (Sysmex XN2000, Kobe, Japan). At our institution, the reference range of MPV is 7.6-13.3 fL. At the same time, other admission laboratory data were obtained, including neutrophil [reference range: (1.8-6.3) × 109/L], hemoglobin (reference range: 11.5-15.0 g/dL), platelet [reference range: (101-320) × 109/L], thromboplastin time (PT; reference range: 9.7-14.2 s), activated partial thromboplastin time (APTT; reference range: 24.8-33.8 s), fibrinogen (reference range: 2.0-4.0 g/L), and D-dimer (reference range: 0.08-0.55 mg/L).
Statistical Analysis
According to the ultrasonography results, patients were divided into DVT group and non-DVT group. For continuous variables, the Shapiro-Wilk test was used to assess the normal distribution, and then expressed as mean ± standard deviation (SD) or median (interquartile range, IQR). Differences between mean or median were evaluated by Student's t-test or Wilcoxon rank-sum test, according to the distribution. Categorical variables were represented as counts (percentages), and compared with chi-square test. When continuous variables reached statistical significance, the receiver operating characteristic (ROC) curve was conducted to determine the optimal cut-off value, including time from injury to admission, platelet, MPV and albumin. To examine the association between MPV and DVT, several logistic regression analyzes were performed: model 1: crude, not adjusted; model 2: minimally adjusted for age, sex, BMI and smoking; model 3: additionally adjusted for time from injury to admission, hip fracture type, hemoglobin, platelet and albumin. Subsequently, MPV was also entered into these models as a continuous variable (per 1.0 fL decrease) instead of a categorical variable. Odds ratio (OR) and 95% confidence interval (CI) were calculated. All reported P values are two-sided, and P < .05 were considered statistically significant. All analyzes were performed using JMP Pro software (version 13.2.1; SAS Institute Inc., Cary, NC, USA).
Results
A total of 352 consecutive older patients with hip fracture were included in this study. Patient characteristics are summarized in Table 1. The mean age was 79.0 ± 8.6 years, 228 (64.8%) were female, and 198 (56.3%) were intertrochanteric fracture. Among them, 53 patients had DVT, and the incidence of DVTwas 15.1%. Regarding the DVT localization, 37 cases were distal DVT, 11 cases were proximal DVT, and 5 cases were mixed DVT. Almost 70% of the DVTs (37/53) were diagnosed within 3 days after admission, and no patient developed pulmonary embolism. In addition, most DVTs (31/53, 58.5%)
Demographic characteristics and laboratory data of patients with DVT and without DVT

Abbreviations: DVT, deep vein thrombosis.
occurred in the fractured extremity (ipsilateral DVT), 13 (24.5%) in the non-fractured extremity (contralateral DVT), and 9 (17.0%) in the bilateral extremities (bilateral DVT). When compared with non-DVT group, patients with DVT had a longer time from injury to admission, and higher proportion of intertrochanteric fracture (P < .05). However, there were no significant differences between the two groups with regard to age, sex, BMI, current smoking and comorbidities.
Laboratory findings on admission showed that the median platelet count was 154.0 × 109/L [IQR: (120.0-200.3) × 109/L], and mean MPV level was 12.2 ± 1.4 fL. MPV and platelet concentration were inversely correlated (Spearman r = −0.505, P < .001). When compared with non-DVT group, platelet count was found to be higher in patients with DVT, whereas the hemoglobin, MPV, and albumin levels were significantly lower (P < .05). Also, both distal and proximal DVT exhibited a lower MPV level, as well as in patients with ipsilateral or contralateral DVT (Figure 1), whereas no significant difference was observed between patients with mixed DVT (P = .585) or bilateral DVT (P = .404). Moreover, DVT group had a slight increase in fibrinogen and D-dimer, but these differences failed to reach statistical significance.
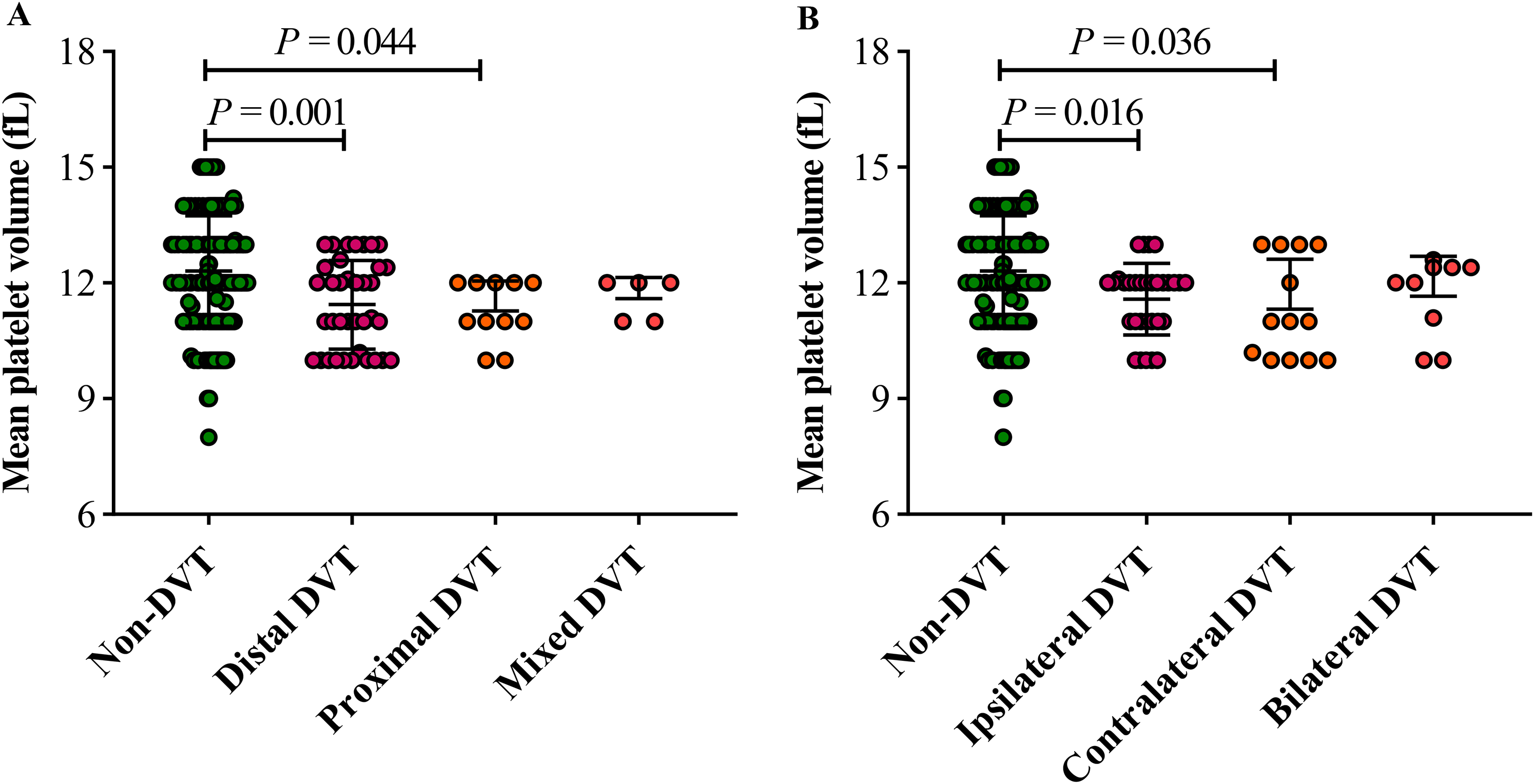
Comparison of mean platelet volume between deep vein thrombosis (DVT) and control patients.
According to the ROC curves for predicting DVT (Figure 2), the optimal cut-off values were 2.0 days for time from injury to admission [sensitivity: 43.4%, specificity: 73.6%, area under curve (AUC): 0.585, 95% CI: 0.469-0.660], 10.8 g/dL for hemoglobin (sensitivity 64.2%, specificity 60.2%, AUC: 0.590, 95% CI: 0.499-0.667), 130.0 × 109/L for platelet (sensitivity 86.8%, specificity 33.4%, AUC: 0.595, 95% CI: 0.518-0.663), 13.3 fL for MPV (sensitivity 98.1%, specificity 77.6%, AUC: 0.631, 95% CI: 0.554-0.703), and 43.0 g/L for albumin (sensitivity 88.7%, specificity 28.1%, AUC: 0.592, 95% CI: 0.510-0.667).

Receiver operating characteristic (ROC) curve of time from injury to admission, platelet, mean platelet volume and albumin to predict deep vein thrombosis.
As shown in Table 2, the univariate logistic regression analysis showed that time from injury to admission, hip fracture type, and hemoglobin, platelet and albumin levels were significantly associated with DVT in older patients with hip fracture (all P < .05). After adjusting for these potential confounding factors, MPV level < 13.3 fL was significantly associated with an increased risk of DVT (OR = 4.857, 95% CI: 1.091-21.617, P = .038). Likewise, with every 1.0 fL decrease in MPV, the risk increased by 27.7% (OR = 1.277, 95% CI: 1.001-1.629, P = .047, Table 3).
Univariate logistic regression analysis of factors associated with DVT

Abbreviations: DVT, deep vein thrombosis; OR, Odds ratio; CI, confidence interval.
Multivariate logistic regression analysis for the association between MPV and DVT

Abbreviations: MPV, mean platelet volume; DVT, deep vein thrombosis; OR, Odds ratio; CI, confidence interval.
not adjusted.
minimally adjusted for age, sex, BMI and smoking.
additionally adjusted for model 2 and time from injury to admission, hip fracture type, hemoglobin, platelet and albumin.
Discussion
Increasing evidences support that an increase in platelet size is often associated with increased platelet reactivity, shortened bleeding time, and increased platelet aggregation,8,9 but remain debated in the formation of venous thrombosis. A population-based prospective study included 25 923 patients, and 445 VTE events were occurred during a mean follow-up period of 10.8 years. when compared with patients with MPV < 8.5 fL, patients with MPV ≥ 9.5 fL had a 1.3-fold (95% CI: 1.0-1.7) higher risk of total VTE and a 1.5-fold (95% CI: 1.1-2.3) higher risk of unprovoked VTE after adjusting for age, sex, smoking, BMI, and platelet count. 12 Consistently, a systematic review and meta-analysis enrolled 18 studies containing 5012 patients, and concluded that MPV was significantly higher in patients with VTE (mean difference 0.69 fL, 95% CI: 0.39-0.98), and the diagnostic accuracy of MPV for VTE was 0.66 (95% CI: 0.58-0.77). 16 However, another large-scale population-based study explored this relationship in 11 084 inhabitants, and concluded that a high MPV was not a risk factor for both arterial and venous thrombosis. 28 Also, four of 18 studies had no difference in MPV between VTE patients and controls in the previously mentioned meta-analysis. 16
In the present study, the mean MPV level was 12.2 fL, which was consistent with a study conducted in China (mean 11.8 fL), 29 but higher than studies conducted in Denmark (mean 11.1 fL), 30 Brazil (mean 10.3 fL), 31 and Korean (mean 8.13 fL). 32 This difference is possibly due to sex, age, and ethnicity, as well as the very poor standardization of the methodologies used for MPV measurement. 33 In addition, a recent study found that traumatic hemorrhage enhanced platelet mobilization from its storage pool, and led to larger platelets. 34 This may be another reason for the higher MPV in our study.
To our knowledge, this is the first study to explore the association between MPV and DVT in older patients with hip fracture. In contrast to the aforementioned studies,12,16,28 patients with DVT exhibited a lower MPV level, even in distal or proximal DVT, as well as ipsilateral or contralateral DVT. Even so, this finding was in line with several previous studies.7,11,13,14 According to the ROC curve for the prediction of DVT, the cut-off value for MPV was 13.3 fL with 98.1% sensitivity, and 77.6% specificity. In the crude logistic regression model, we found an inverse association between MPV and the risk of DVT, regardless of whether it was analyzed as a categorical variable (< 13.3 fL) or a continuous variable (per 1.0 fL decrease). Meanwhile, the univariate logistic regression analysis revealed that time from injury to admission, hip fracture type, and hemoglobin, platelet and albumin levels were significantly associated with DVT. Even after adjusting for these potential confounding factors, MPV level < 13.3 fL was significantly associated with an increased risk of DVT (OR = 4.857, 95% CI: 1.091-21.617), and with every 1.0 fL decrease in MPV, the risk increased by 27.7% (OR = 1.277, 95% CI: 1.001-1.629). Consistent with our findings, Lippi et al. 13 found that a decreased MPV (< 10.8 fL) was associated with an increased risk of diagnosing VTE [relative risk (RR) = 1.18, 95% CI: 1.09-1.28], as well as of diagnosing DVT (RR = 1.19, 95% CI: 1.07-1.31) and isolated PE (RR = 1.17, 95% CI: 1.04-1.30) in the emergency department. Another prospective, observational cohort study also reported that high MPV (≥ 10.8 fL) was associated with a significantly decreased risk of VTE in cancer patients (HR = 0.59, 95% CI: 0.37-0.95). 14
Underlying pathophysiological mechanisms of low MPV and increased risk of VTE are still unknown. As explained by Lippi et al., 13 the delay from venous thrombus formation to VTE diagnosis may be associated with a progressive normalization of platelet size. This interpretation was supported by a recent study that both immature platelet count and absolute platelet count increased, whereas immature platelet fraction decreased significantly in the first two weeks after diagnosis of VTE. 35 Thereby, the reduced MPV in patients with VTE may be attributable to the delayed diagnosis. In our study, nearly 70% of the DVTs were diagnosed within 3 days after admission, but the time of initial thrombus formation was still not clear. Except for this, Riedl et al. 14 suggested that small platelets have a lower MPV, and might exhibit a stronger prothrombotic tendency in cancer patients than large platelets. Stefan et al. 36 designed a protocol to separate large and small platelets, and found that large platelets represented a more faster adhesion to collagen after stimulation with thrombin protease-activated receptor-1 (PAR1) activating peptide and collagen, but in contrast small platelets expressed more P-selectin upon stimulation with adenosine diphosphate (ADP), which is a key surface adhesion molecule for the interaction of platelets with leukocytes in the formation of thrombosis. 37 Moreover, functions of large and small platelets were highly dependent on extracellular Ca2+ availability. Briefly, platelet aggregation was stronger in large platelets under Ca2+-poor conditions but comparable to small platelets when extracellular Ca2+ was available. 38 In our previous study, serum calcium disorder was common in older patients with hip fracture. 39 This may be another reason explaining our findings, and the influence of platelet size on platelet activity and risk of VTE would be needed to investigate further.
However, several limitations in this study should be noted. First, this was a retrospective study and a selection bias may exist. We included patients consecutively, which would have reduced a potential for selection bias. At the same time, we were unable to obtain some important variables affecting the MPV measurement and DVT risk, such as the recent use of antiplatelet agents before the fracture. Second, the sample size of enrolled patients was small, and all the patients came from one orthopedic center. For this reason, the confidence intervals in our study were quite wide, therefore limiting the credibility of the results. In spite of this, we analyzed MPV variable continuously and categorically by using multivariate logistic regression models, and found that both of the results were statistically significant, indicating that the association was relatively stable. In the future, prospective studies with large sample sizes are needed to confirm our findings. Third, all the DVT patients were diagnosed using ultrasonography, which may have a lower accuracy than venography. Fourth, the AUC of 0.631 for MPV was not sufficient for clinical VTE diagnosis. Further research can be conducted combining MPV with clinical findings or other laboratory markers, thereby improving the diagnostic accuracy. 16 Finally, the multivariate analysis revealed an association between MPV and DVT, rather than causative relationship. To date, whether the increased platelet size is the cause or the consequence of thrombosis remains unknown. 33
Conclusions
In conclusion, a low MPV level is associated with DVT in older patients with hip fracture. As MPV is a simple indicator that can be calculated from the blood routine test, it may be a potential biomarker of DVT with the combination of other tests, further studies are needed to confirm these results.
Footnotes
Abbreviations
Acknowledgements
We would like to thank several nurses from the Department of Orthopedics in Deyang People's Hospital, for the help with the data inspection.
Authors’ Contribution
Zhicong Wang: Conceptualization, Methodology, Formal analysis, Software, Visualization, Funding acquisition, Writing-original draft. Xi Chen: Methodology, Formal analysis, Writing-review & editing, Project administration. Jijun Wu: Methodology, Formal analysis, Writing-review & editing, Project administration. Qing Zhou: Conceptualization, Investigation, Supervision, Validation. Hailong Liu: Conceptualization, Investigation, Supervision, Validation. Yuxuan Wu: Conceptualization, Funding acquisition, Investigation, Supervision, Validation. Shuping Liu: Conceptualization, Writing-review & editing, Supervision, Validation. Yuehong Liu: Conceptualization, Writing-review & editing, Supervision, Validation.
Availability of Data and Materials
The data used during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The study protocol was approved by the Institutional Ethics Committee at Deyang People's Hospital (approval number 2021-04-019-K01), and conducted in accordance with the principles of the Declaration of Helsinki. The need for informed consent was waived because all data were collected retrospectively and anonymously without unique patient identifiers.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sichuan Science and Technology Program (No. 2021JDR0337) and Deyang Science and Technology Bureau Project (No. 2019SZ118, No. 2019SZ125).
