Abstract
Introduction
Venous thromboembolism (VTE) is a common and highly prevalent disease that affects an estimated 350 000 to 600 000 people in the United States every year and claims the lives of at least 100 000. 1 Obesity is one of the several conditions related to the development of VTE. Obesity is also a known risk factor for obstructive sleep apnea (OSA) and affects the disease severity. 2 The prevalence of OSA ranges between 2% and 10% of the general adult population in the United States 3 ; however, this is likely underestimated due to underdiagnosis of the disease. 4
Similar to VTE, several factors modify the risk of OSA such as age, race, and gender.5–9 Furthermore, OSA has been linked to many of cardiovascular diseases, including hypertension, ischemic heart disease, congestive heart failure (CHF), cerebrovascular accidents (CVAs), pulmonary hypertension, cardiac arrhythmias, end-stage renal disease, and overall cardiovascular mortality.10–12 Conversely, the link of OSA to VTE, another cardiovascular disease, is not well established. At least 2 published reports have suggested a potential relationship between OSA and VTE. However, both reports suffered from low sample size and design limitations,13,14 Due to the unclear association between OSA and VTE, we sought to study the potential relationship and estimate the prevalence of OSA among patients with VTE.
Methods
Design and Setting
A retrospective study was conducted at the University of Missouri Hospitals and Clinics from August 1999 to April 2009. This center is a tertiary university hospital and is equipped with 8 beds for polysomnography. The Institutional Review Board from the University of Missouri approved the study under the exempt category. The study was registered at clinicaltrials.gov (NCT01051297).
Patients
All adult patients (>18 years of age) with International Classification of Diseases, Ninth Revision (ICD-9) coding indicative of VTE were included. The initial search contained all of the following ICD-9 codes: 415.11, 415.12, 415.19, 453.4, 453.9, 453.41, 453.42, 671.33, and 671.44. The study investigators then reviewed the charts extensively to verify the clinical diagnosis of VTE. Venous thromboembolism was defined as the objective diagnosis of pulmonary embolism (PE) or deep vein thrombosis (DVT). Venous thromboembolism was confirmed by extremity venous Doppler ultrasonography, spiral computed tomography (CT) of the chest, high-probability ventilation−perfusion VQ scan, or pulmonary angiography. Only patients with confirmed VTE (as listed in past medical history or per available records of radiographic testing) were included in the study. Patients without objective confirmation of VTE were excluded. If the status of OSA or height and weight could not be ascertained, then such patients were also excluded.
Measurements
Patient charts were reviewed and the following data were collected: age, race, smoking status, and presence of diabetes mellitus, CHF, CVA, and coronary artery disease (CAD). Height and weight data, which allowed calculation of the body mass index (BMI), were also collected. Patients were classified into 5 groups according to the World Health Organization (WHO) classification15,16: Normal (BMI < 25.0), overweight (BMI 25.0-29.9), and obesity (BMI ≥ 30) was subdivided into grade 1 (BMI 30-34.9), grade 2 (BMI 35-39.9), and grade 3 (morbid, BMI ≥ 40). 15 Primary outcome for this study was the prevalence of OSA among this group of patients with VTE. Obstructive sleep apnea was defined as an apnea−hypopnea index (AHI) of ≥ 5 by overnight polysomnography or the presence of OSA in the patient’s medical history confirmed by a sleep study in an outside laboratory not available for our review.
Statistical Analysis
Data were entered into a database (Microsoft Excel, Microsoft Cooperation, Redmond, Washington, 2007). The cohort was then divided into groups according to presence or absence of OSA. The 2 groups (OSA and No OSA) were then compared. Continuous data were expressed as means or medians according to normality testing using Kolmogorov-Smirnov tests and compared using the student t or the Mann-Whitney tests, whichever applicable. Categorical variables were expressed as percentages and compared using the chi-square or Fisher exact tests. For statistical analysis, parametric or nonparametric tests were used according to normality testing. Furthermore, the prevalence of OSA was estimated according to classes of BMI.
Receiver operating characteristics (ROC) analysis was used to evaluate the performance of BMI in the prediction of OSA among patients with VTE. Area under the curve with 95% confidence interval was estimated. According to the curve, we defined the cutoff value with best combination of sensitivity and specificity. Statistical significance was defined as P <.05 and all tests were 2-sided.
Results
A total of 1239 patients were screened and 840 patients were included in the study. Figure 1 illustrates the flow diagram for study inclusion and exclusion. Of the 840 patients, 130 were also diagnosed with OSA, yielding a prevalence of 15.5% (13%-17.9%) of OSA in the cohort. Of the 840 patients, 619 (73.8%) had DVT with 524 (73.9%) in the No-OSA group and 95 (73.1%) in the OSA group (P = .843). In all, 530 (63.3%) of the patients had PE with 439 (61.9%) in the No-OSA and 91 (71.7%) in the OSA group (P = .047). In all, 309 (36.8%) had both DVT and PE with 253 (35.6%) in the No-OSA group and 56 (43.1%) in the OSA group (P = .106). A total of 453 (53.9%) patients were females and the median age was 55 (18-94).
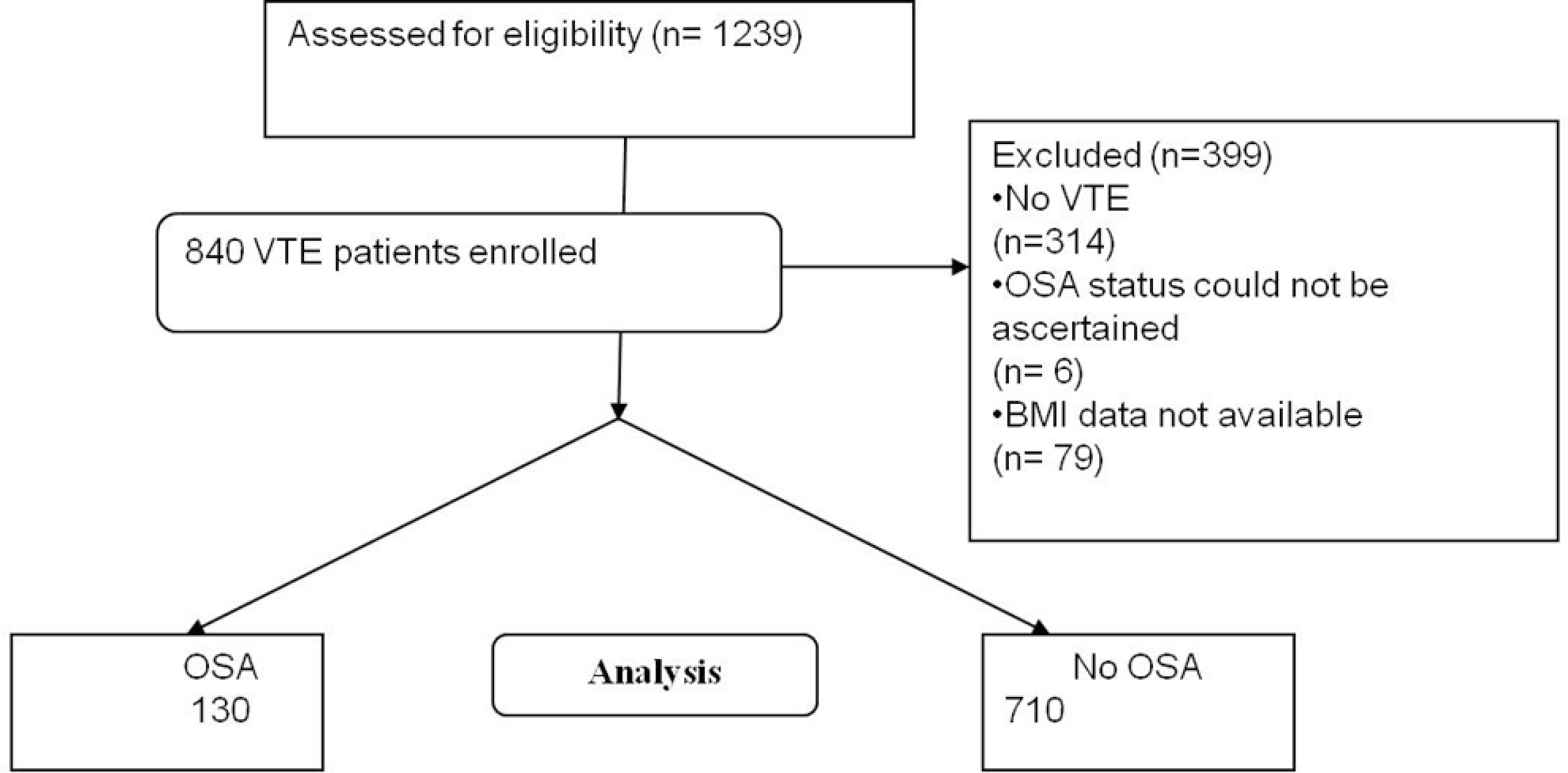
Flowchart for Study Population. OSA indicates obstructive sleep apnea; VTE, venous thromboembolic events; BMI, body mass index.
When comparing the 2 groups (OSA, No-OSA), there was no difference in age, race, gender, or rates of CVA events (P > .05; see Table 1 for details). Compared to the control group (No-OSA), those who had OSA were more obese (83.8% vs 43.8%) and had higher rates of diabetes, CAD, and CHF (P values: <.0001, <.0001, .028, and <.0001, respectively).
Characteristics of the Study Patients and Those With or Without Obstructive Sleep Apnea
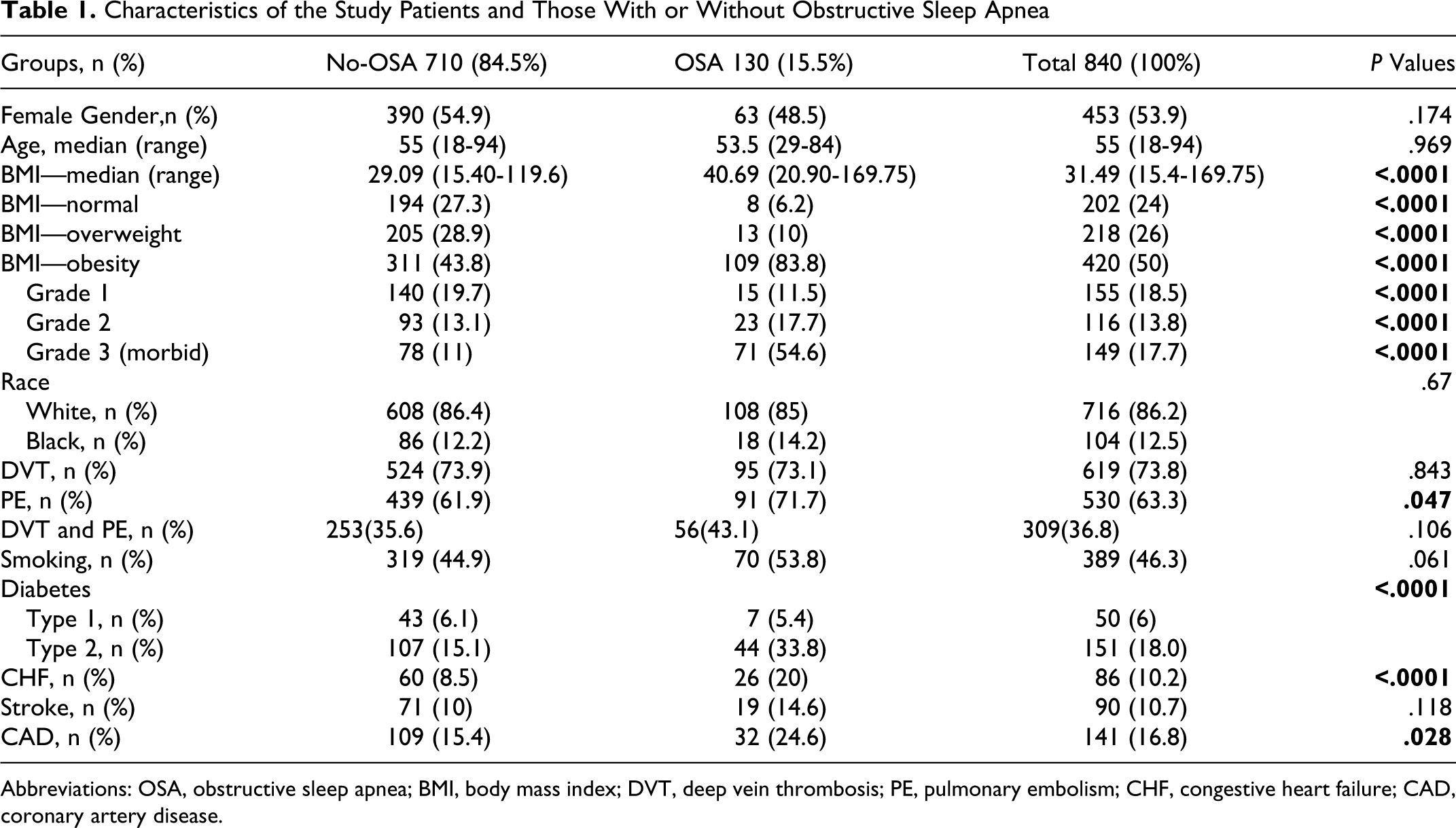
Abbreviations: OSA, obstructive sleep apnea; BMI, body mass index; DVT, deep vein thrombosis; PE, pulmonary embolism; CHF, congestive heart failure; CAD, coronary artery disease.
Figure 2 shows prevalence of OSA according to BMI categories. There was a clear trend of increasing prevalence of OSA based on BMI. Both the normal and overweight groups had comparable rates to general population, but the 3 obese (grade 1, grade 2, and grade 3—morbid) group had much higher rates with prevalence rates reaching 50% in the morbidly obese (grade 3) group.
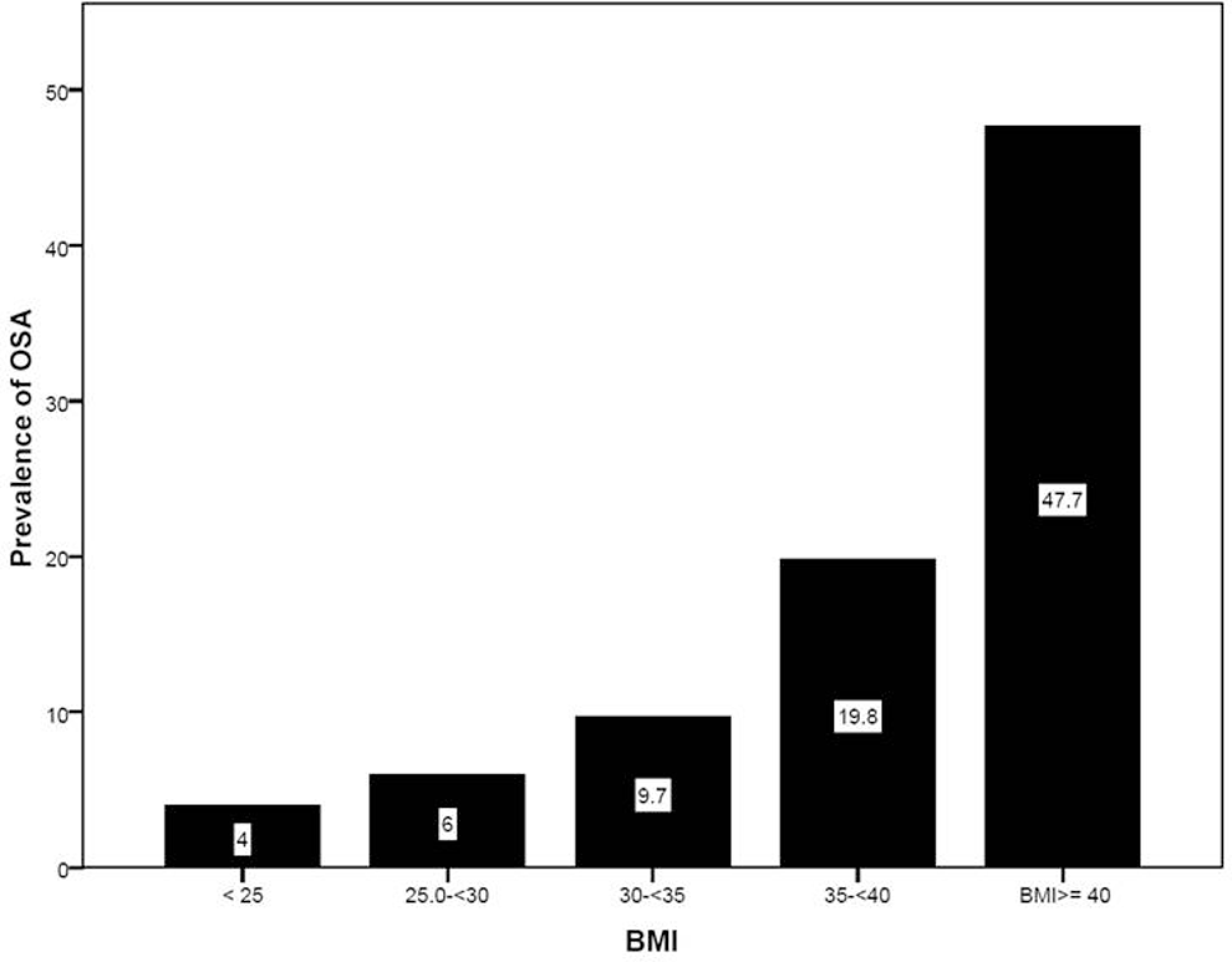
Prevalence of OSA among patients with VTE according to BMI categories. OSA indicates obstructive sleep apnea; BMI, body mass index.
Figure 3 depicts the ROC curve for BMI. The analysis had good performance with area under the curve (AUC) of 0.797 (95% confidence intervals [0.753-0.840], P < .0001). The best cutoff value was a BMI of 34, with sensitivity of 78% and specificity of 75%.
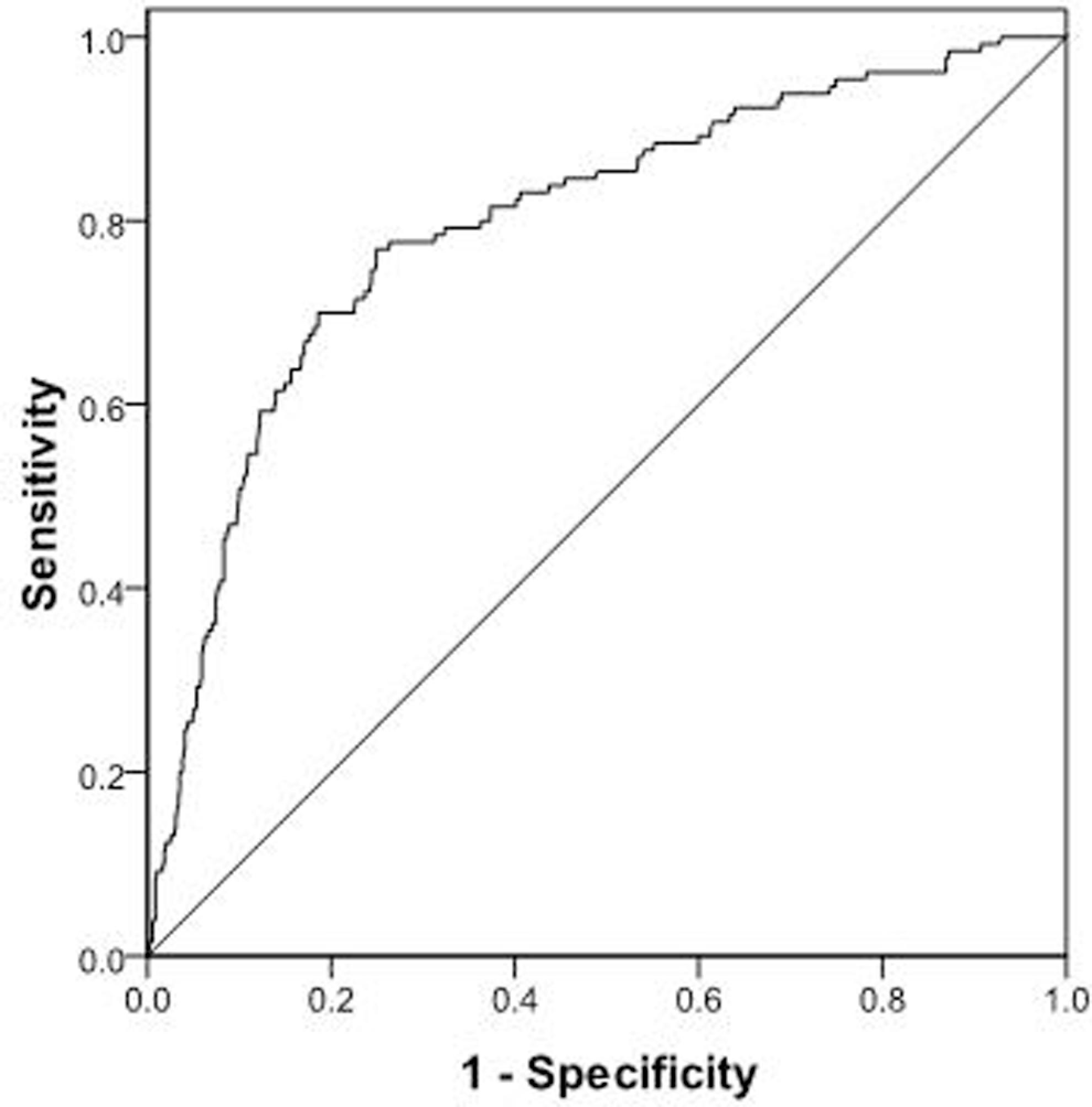
Receiver operating characteristics (ROC) curve for BMI predicting the status of OSA. Best cutoff value was found to be a BMI of 34, with a sensitivity of 78% and specificity of 75%. OSA indicates obstructive sleep apnea; BMI, body mass index.
Discussion
In this population of patients with confirmed VTE, the prevalence of OSA 15.5% (13%-17.9%) appears to be higher than the general population (2%-10%). 3 Our data also suggest that patients with prior diagnoses of OSA and VTE are more likely to be obese with higher rates of CAD, CHF, and diabetes. Interestingly, the OSA group had higher prevalence of PE (71.7% vs 61.9%; P = .047) but similar rates of DVT. This latter finding may, in part, explain the higher overall cardiovascular mortality in OSA patients noted by other studies. 10 Although OSA has been clearly linked to arterial thrombosis, this study suggests a link between OSA and the venous thrombotic disorders.
Several studies have established a pathophysiologic link between OSA and thrombosis. Obstructive sleep apnea has been shown to induce an inflammatory phenotype and a hypercoagulable state, consistent with Virchow triad. Patients with OSA have increases in plasma cytokines, adhesion molecules, leukocyte activation, and TNF-α. 17 Obstructive sleep apnea also leads to alterations in the levels of vasoactive substances, including increases in endothelin, 18 and aldosterone, 19 and decreases in nitric oxide. 20 Recent studies have established the association between OSA, endothelial dysfunction/apoptosis,21–24 and elevated C-reactive peptide levels. 25 In patients with OSA, microvascular endothelial function (tested by relaxation to acetylcholine) has been shown to be impaired. In addition, this impairment is reversed with proper OSA treatment. 26 Regarding hypercoagulability, von Kanel et al has shown that patients with OSA have elevations in prothrombotic factors. These include increased plasma fibrinogen, reductions in fibrinolytic capacity, activation of clotting factors, and heightened platelet activity.27,28
Our findings are consistent with previous studies that have also noted a relationship between OSA and VTE. Ambrosetti et al have shown through a limited sample size study (N = 89) that patients with OSA appear to be at increased risk of VTE. 13 Of 89 patients with OSA, 2 developed proximal DVTs and 1 of those patients developed PE over a 3-year follow-up. Another study by Arnulf et al showed that in patients (N = 68) admitted to the hospital with a VTEs, 82.4% (56 of 68) had an AHI ≥ 5 and 63% (43/68) had an AHI > 15. 14 Of note, the patients studied in the Arnulf cohort were from a respiratory clinic population.
Obesity appears to be a common factor between OSA and VTE. In our OSA group, 83.8% of the patients were obese with a median BMI of 40.69 (versus 43.8% and a BMI of 29.09 in the No-OSA cohort), though the causal relationship is unclear. Obesity has been shown to effect hemostasis by many of the same mechanisms as OSA. As summarized by Stein and Goldman, obesity has been shown to effect fibrinolysis, plasminogen activator inhibitor 1 levels, and platelet aggregability. 29
Obese patients had a higher prevalence of OSA. The morbidly obese group (BMI ≥ 40) had a prevalence rate of about 50%, indicating that one half of the patients in this group had OSA. Receiver operating characteristics analysis showed that a BMI of 34 was the best cutoff value. Our data suggest that sleep evaluation may be necessary in those patients with prior VTE and obesity grade 1, 2, or 3 as these 3 groups had much higher prevalence of OSA. In the other 2 groups (normal and overweight), OSA was comparable to general population where we suggest sleep evaluation based on other suggestive signs and symptoms.
Our study has limitations. The retrospective design limits our conclusions with the inherent weakness included in this design. The high exclusion rate of 25% is due to ICD-9 coding which labeled patients as having VTE but objective evidence was not present upon extensive review of medical records.
International Classification of Diseases, Ninth Revision coding was initially used to identify patients although complete chart reviews were performed to confirm included data. We found that many patients that were labeled by ICD-9 code as VTE did not actually have the disease. We did not add 2 codes (451.1 and 451.19); however we have included many others to give us a large enough sample size. We believe that the independent actual chart review decreased the impact of this limitation.
Though unexpected, we consider this as strength for our study as it required data to be confirmed for every patient. The diagnosis of VTE was not independently adjudicated which may limit our findings
Another limitation is that we could not consistently ascertain whether OSA preceded VTE or vice versa; therefore, we expressed the percentage of patients with OSA as a prevalence rather than incidence. In addition, we did not have access to all the sleep studies as many of them were performed in outside laboratories. In these instances, we chose not to classify the patient as sleep apnea unless we found objective evidence in the chart that the patient carried this disorder. We found objective evidence for a performed sleep study among 743 (88.5%) of the cohort. The remainder of the patients had clear data about presence or absence of OSA. We excluded patients that were described as high risk or potentially having OSA, without evidence of sleep study. Many patients may have had OSA without a clear medical record or proper testing and diagnosis; these were not included in our study. These uncertainties may have led to underestimation or overestimation of the prevalence of OSA in this cohort. We only included symptomatic proximal DVTs. We relied completely on the assessment done per the health care providers and there was no adjudication done by our investigators.
Our study also has much strength. This is the largest cohort in the literature that investigates the relationship between OSA and VTE. Furthermore, the previous 2 other reports had 157 patients combined.13,14 We confirmed the diagnoses of VTE and OSA and also studied baseline characteristics to discern if the relationship is independent or confounded by obesity, a common factor between these 2 disorders.
Conclusion
Patients with VTE seem to have a higher prevalence of OSA 15.5% (13%-17.9%), as compared to the general population. Diabetes, CAD, and CHF also appear to be more prevalent in patients with both VTE and OSA as compared to patients with VTE alone. Of particular interest, in a cohort of patients with VTE, we found similar prevalence of DVT but increased prevalence of PE in patients with OSA. Although OSA has been clearly linked to arterial thrombosis, this study suggests a link between OSA and venous thrombotic disorders. Obesity continues to be one of the confounding variables.
Due to the high prevalence of obesity and OSA, further studies of the development of VTE in patients with OSA would be helpful to clarify the causal relationship between these diseases. Establishing a time-line between the onset of OSA and development of VTEs may be helpful in elucidating the relationship. Nevertheless, health care workers should be aware of this association as both of these 2 diseases are associated with high morbidity and mortality.
Footnotes
Acknowledgments
Author contributions: James P. Bosanquet contributed to concept, study design, data collection, interpretation of results, manuscript draft and critical review. Brett C. Bade contributed to study design, data collection, interpretation of results, manuscript draft, and critical review. Maryam F. Zia contributed to data collection and manuscript critical review. Ammar Karo contributed to data collection and manuscript critical review. Oudai Hassan contributed to data collection and manuscript critical review. Brian T. Hess contributed to data collection and manuscript critical review. Ousama Dabbagh contributed to concept, study design, analysis, manuscript draft, critical review, and overall supervision.
James P. Bosanquet has no disclosures. Brett C. Bade has no disclosures. Maryam F. Zia has no disclosures. Ammar Karo has no disclosures. Oudai Hassan has no disclosures. Brian T. Hess has no disclosures. Ousama Dabbagh has the following disclosures: Research money granted to the University of Missouri from Pfizer and BMS for clinical studies enrollment; speaker bureau: Sanofi–Aventis.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
