Abstract
Introduction
Obstructive sleep apnea (OSA) is prevalent in patients with venous thromboembolism (VTE). We sought to evaluate the effectiveness and safety of rivaroxaban versus warfarin in VTE patients with comorbid OSA.
Methods
We performed retrospective cohort analyses using the Optum® de-identified Electronic Health Record data set (Optum® EHR) from November 1, 2011 to June 30, 2023. We included adults with OSA at baseline who newly initiated rivaroxaban or warfarin for an acute VTE and had ≥12 months of prior EHR activity. Patients who had nonvalvular atrial fibrillation, had a hip/knee replacement ≤35 days before the acute VTE, were pregnant, or had active cancer were excluded. Times to recurrent VTE and major bleeding were evaluated. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated using propensity score–overlap weighting proportional hazards regression.
Results
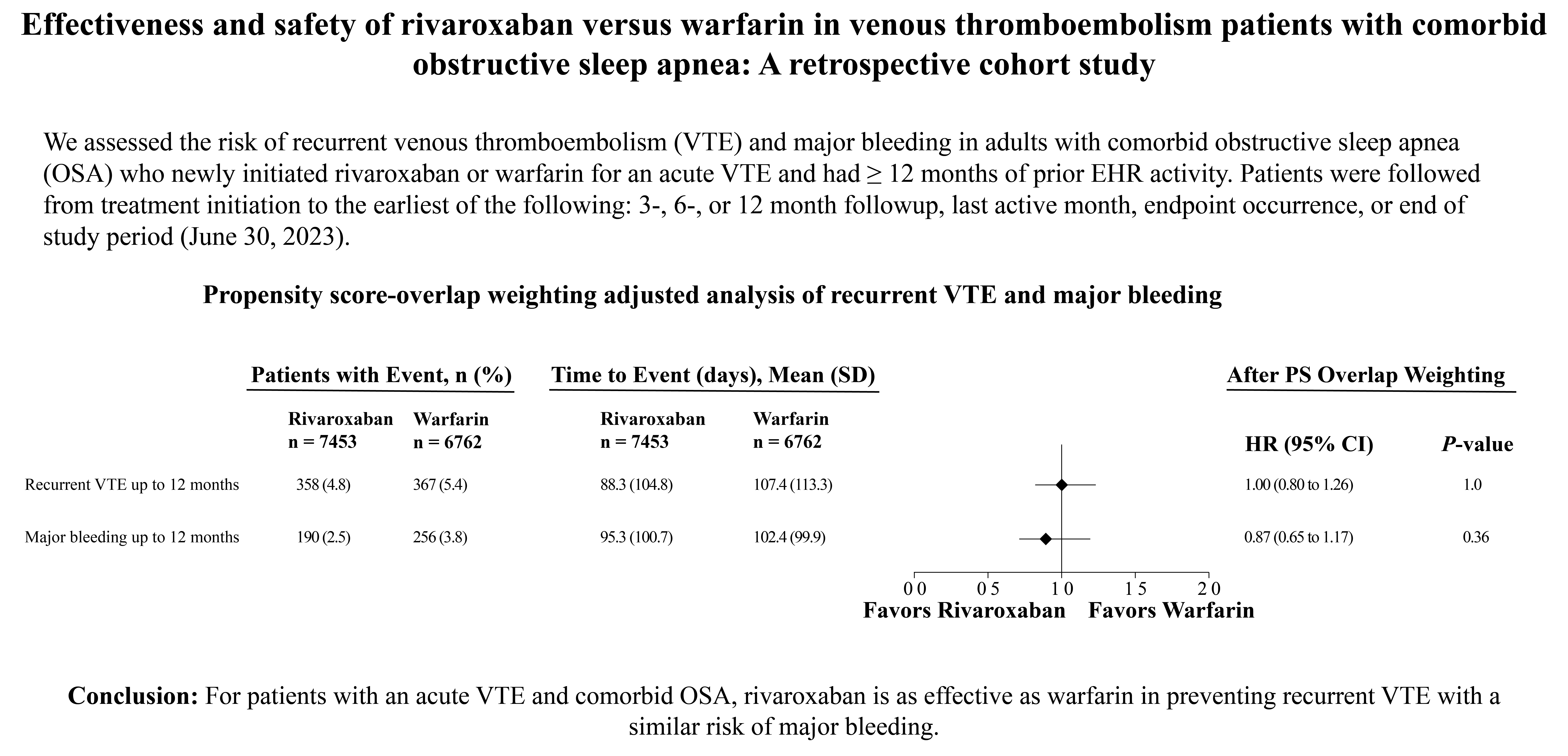
A total of 14,215 patients met the study criteria and were included in the analyses (rivaroxaban: 7453; warfarin: 6762). We observed similar risks of recurrent VTE (HR = 1.0, 95% CI: 0.80-1.26) and major bleeding (HR = 0.87, 95% CI: 0.65-1.17) through 12 months with rivaroxaban- compared to warfarin-treated patients.
Conclusion
For patients with acute VTE and comorbid OSA, rivaroxaban is as effective as warfarin in preventing recurrent VTE, with a similar risk of major bleeding.
Graphical Abstract
Key Points
OSA is prevalent in patients with VTE and is a major contributor to the healthcare burden in this patient population worldwide.
There is a paucity of data comparing the effectiveness and safety of rivaroxaban versus warfarin for managing acute VTE in patients with comorbid OSA.
This retrospective cohort study used EHR data to assess the effectiveness and safety of rivaroxaban versus warfarin among VTE patients with comorbid OSA.
Among 14,215 patients identified during the study period (November 1, 2011 to June 30, 2023), we found similar risks of recurrent VTE and major bleeding among patients treated with rivaroxaban compared to warfarin. The findings were consistent across multiple subgroups.
Findings from this study suggest that rivaroxaban, compared to warfarin, is an effective and safe option for preventing recurrent VTE in patients with OSA.
Introduction
Obstructive sleep apnea (OSA) is a sleep-related breathing disorder characterized by repeat episodes of partial or complete closure of the upper airway during sleep, leading to intermittent hypoxia and sleep fragmentation. It is a common disorder with increasing prevalence among middle-aged adults worldwide and affects approximately 34% of men and 17% of women in the United States (US).1–3 OSA is associated with an increased incidence of thromboembolic, cardiovascular, and cerebrovascular events, and is a major contributor to rising healthcare utilization and costs.4–6
Venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and pulmonary embolism (PE), is a frequent and potentially fatal vascular disease with an estimated annual incidence of 1 to 2 cases per 1000 person-years in the US population. 7 While acute VTE can be effectively treated with anticoagulant therapy, there is a risk of recurrence after treatment discontinuation, which can reach 10% in the first year. 8 A meta-analysis found that after discontinuing anticoagulant therapy, the cumulative risk of recurrent VTE was 16% at 2 years, 25% at 5 years, and 36% at 10 years. 9
OSA and VTE share several risk factors, including age, obesity, hypertension, and prolonged physical inactivity. Growing evidence suggests that OSA is an independent risk factor for VTE and VTE recurrence.10–15 Emerging research indicates a complex bidirectional relationship between OSA and VTE.13,16,17 On the one hand, the intermittent hypoxia caused by OSA may induce a persistent prothrombotic state and increase the risk of developing VTE and VTE recurrence.12,13,16 On the other hand, acute PE, the most serious complication of VTE, may present more severely in patients with OSA and require higher doses of anticoagulant to manage.18–20
Direct-acting oral anticoagulants (DOACs) including rivaroxaban, an oral factor Xa inhibitor, are recommended as first line oral anticoagulants for managing acute VTE. 21 A recent meta-analysis shows that rivaroxaban is associated with lower incidence of VTE and lower risks of bleeding in the overall VTE population. 22 However, to date, no study has assessed the effectiveness and safety of rivaroxaban compared to warfarin, a vitamin K antagonist, for managing VTE in patients with concomitant OSA. In this study, we sought to evaluate the effectiveness and safety of rivaroxaban versus warfarin in patients with an acute VTE and comorbid OSA.
Methods
Study Design and Data Source
This was a retrospective cohort study using the Optum® de-identified Electronic Health Record data set (Optum® EHR) from November 1, 2011 to June 30, 2023. Optum® EHR contains longitudinal patient-level medical records for more than 91 million patients treated at more than 700 hospitals and 7000 clinics across the US. The dataset includes information on insured and uninsured patients of all ages, providing a representative sample of US patients experiencing a VTE. It also includes records of prescriptions, patient-reported over-the-counter medications, laboratory results, vital signs, anthropometrics, other clinical observations, diagnosis codes (International Classification of Diseases, Ninth and Tenth Revisions, Clinical Modification [ICD-9-CM and ICD-10-CM], and procedure codes (ICD-9, ICD-10, Current Procedural Terminology-4, Healthcare Common Procedure Coding System, and Revenue codes). This study and the use of the dataset were determined by the New England Institutional Review Board (IRB) to not constitute research involving human subjects and therefore exempt from IRB oversight.
Cohort Selection
This study included adult patients (≥18 years of age) who were admitted to a hospital, or had an emergency department (ED) or observation unit visit for an acute DVT and/or PE (index event) during the patient intake period (November 1, 2012, to June 30, 2023); received rivaroxaban or warfarin as the first oral anticoagulant (OAC) within 7 days of the VTE event (index date); were active in the dataset for ≥12 months before the index event (baseline period to confirm rivaroxaban or warfarin as the first OAC and to evaluate baseline patient characteristics); were diagnosed with OSA prior to or on the index date; and had ≥1 provider visit during the 12-month baseline period. The study excluded patients who used an OAC (per written or electronic prescription or patient self-report) during the baseline period, had nonvalvular atrial fibrillation, had hip or knee replacement within 35 days before the index date, were pregnant, or had active cancer (defined as metastatic cancer or any cancer diagnosis plus systemic cancer treatment in the 6 months prior to the index date). Patients with prescription records for both rivaroxaban and warfarin on the index date were also excluded.
The identification of OSA was based on the presence of ICD-9-CM and/or ICD-10-CM billing codes of 327.20 (organic sleep apnea, unspecified), 327.23 (obstructive sleep apnea [adult, pediatric]), 327.29 (other organic sleep apnea), 780.51 (insomnia with sleep apnea, unspecified), 780.53 (hypersomnia with sleep apnea, unspecified), 780.57 (unspecified sleep apnea), G47.30 (sleep apnea, unspecified), G47.33 (obstructive sleep apnea [adult, pediatric]), and G47.39 (other sleep apnea) using all available data prior to or on the index date. While some of these codes may have included central sleep apnea, it is likely that these codes are associated predominantly with OSA in most cases. Therefore, we did not restrict case selection by excluding codes that do not differentiate between central and obstructive sleep apnea. This coding mechanism has been shown to have a positive predictive value (PPV) of >90% for identifying OSA (sensitivity of 77.7% and specificity of 99.5%), 23 with additional evidence suggesting that the PPV improves in the presence of comorbidities that are common in patients with VTE. 24
Confounder Adjustment and Handling of Missing Data
To adjust for potential confounding between rivaroxaban and warfarin cohorts during the baseline period and on the index date, we calculated propensity scores based on multivariable logistic regression. 25 The multivariable logistic regression model included all commonly used variables in the baseline characteristic table and accepted risk factors for differential OAC exposure, including demographics, comorbidities, laboratory results, body mass index (BMI), and concurrent outpatient medication use. Other variables included covariates specific to OSA, including the presence of ≥1 code for a sleep study, need for positive airway pressure (PAP) treatment, and surgical treatment for OSA (Table 1). To ensure a comprehensive evaluation, all candidate variables were included in the model, regardless of statistical significance. The presence of comorbidities was determined based on billing codes and/or supporting laboratory and observation data. Comorbidities were identified by ICD diagnosis codes, and medications were identified using National Drug Codes and drug names. The absence of such data was assumed to represent the absence of the disease or medication use. Estimated glomerular filtration rate and BMI values were imputed using the median value of the nonmissing values. Generated propensity scores were used to weight patients for analysis using an overlap weighting (OLW) approach described by Thomas and colleagues, 26 which assigns weights to patients that are proportional to their probability of belonging to the alternative treatment cohort. Specifically, patients in the rivaroxaban cohort were weighted by the probability of receiving warfarin (ie, 1 minus the propensity score), and patients in the warfarin cohort were weighted by the probability of receiving rivaroxaban (ie, propensity score).
Baseline Patient Characteristics.
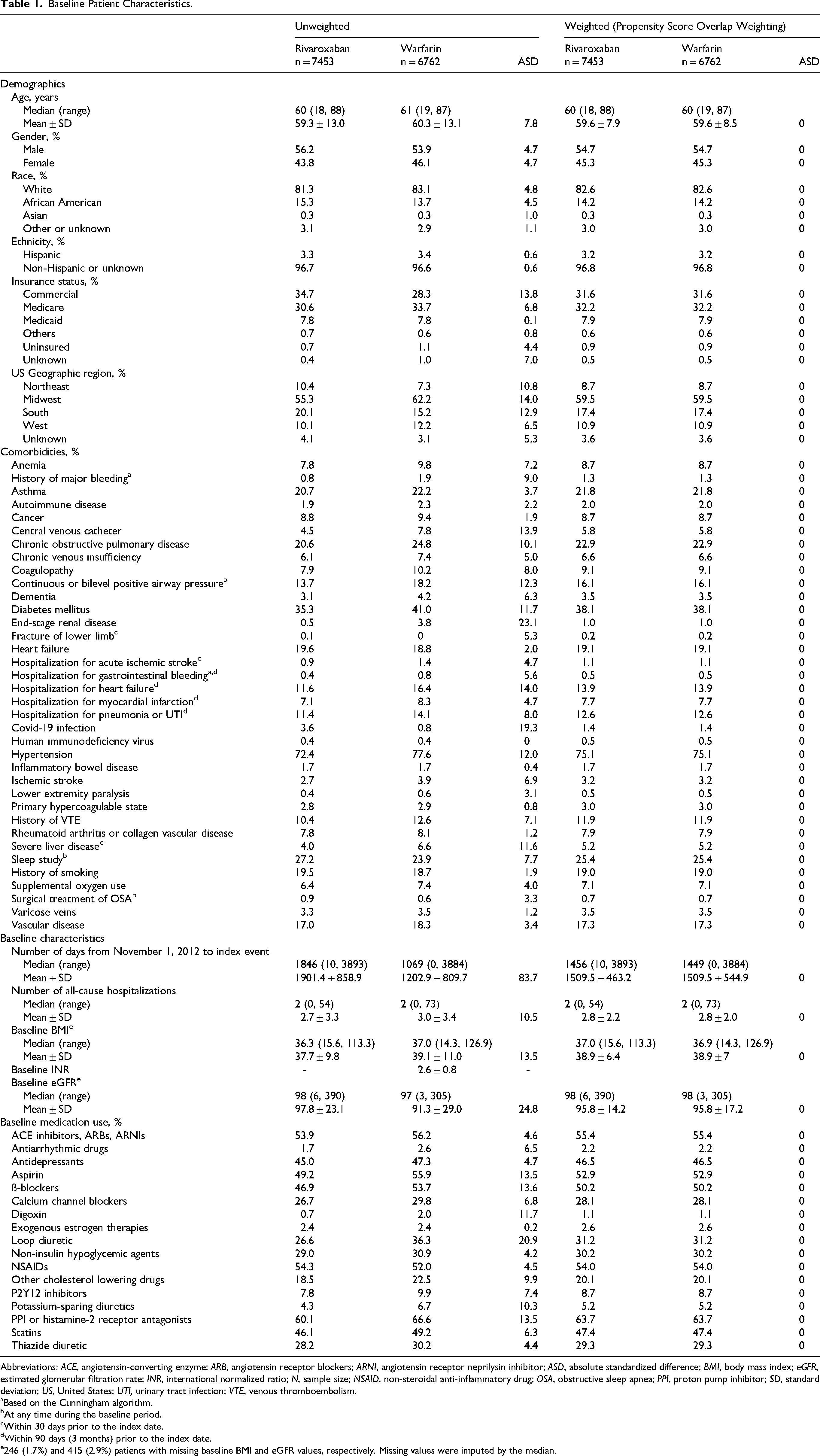
Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blockers; ARNI, angiotensin receptor neprilysin inhibitor; ASD, absolute standardized difference; BMI, body mass index; eGFR, estimated glomerular filtration rate; INR, international normalized ratio; N, sample size; NSAID, non-steroidal anti-inflammatory drug; OSA, obstructive sleep apnea; PPI, proton pump inhibitor; SD, standard deviation; US, United States; UTI, urinary tract infection; VTE, venous thromboembolism.
Based on the Cunningham algorithm.
At any time during the baseline period.
Within 30 days prior to the index date.
Within 90 days (3 months) prior to the index date.
246 (1.7%) and 415 (2.9%) patients with missing baseline BMI and eGFR values, respectively. Missing values were imputed by the median.
This study used propensity score–overlap weighting as the primary method for confounder adjustment because it allows for all eligible patients to be included in the analysis. By assigning greater weight to patients whose treatment cannot be predicted and less weight to patients with extreme propensity scores (approaching 0.0 or 1.0), it prevents outliers from dominating the analysis and reducing precision, leading to the exact balance of all variables included in the multivariable logistic regression model used to derive the propensity score, and results in absolute standardized differences (ASDs) of 0 for each covariate.
Outcomes
The primary effectiveness endpoint was time to the first recurrent VTE through 12 months after the index date. The primary safety endpoint was time to the first major bleeding event, defined using the validated Cunningham algorithm, through 12 months after the index date. 27 The Cunningham algorithm is a validated method for identifying bleeding-related hospitalizations and is commonly used in real-world EHR- and claims-based studies of oral anticoagulant use. Recurrent VTE was defined as a subsequent hospitalization with a primary ICD-9-CM or ICD-10-CM diagnosis code for DVT or PE. 28 Secondary effectiveness and safety endpoints included time to recurrent VTE and time to major bleeding through 3 or 6 months after the index date, as well as time to the first intracranial hemorrhage (ICH) and major extracranial bleeding through 3, 6, and 12 months post index. Extracranial bleeding included all major bleedings from the Cunningham algorithm that did not occur in the head.
Statistical Analysis
Baseline patient characteristics were analyzed using descriptive statistics. Categorical variables were reported as percentages and continuous variables as means and standard deviations (SDs). Outcomes were analyzed using an intent-to-treat approach. Hazard ratios (HRs) with 95% confidence intervals (CIs) were calculated using propensity score–overlap weighted Cox proportional hazards regression models. The proportional hazard assumption was tested based on Schoenfeld residuals. Patients were followed until outcome occurrence, end of EHR activity, or end of the study period. For primary outcomes, we performed a sensitivity analysis using stabilized inverse probability of treatment weighting (sIPTW), which also weights patients based on their propensity score but includes an added step to stabilize the weights by multiplying them by the marginal probability of receiving their actual treatment. Like OLW, sIPTW intends to reduce the impact of extreme weights. Unweighted descriptive results were reported as medians and ranges and subsequently transformed into means and SDs to meet the requirements of OLW and sIPTW.
Additionally, we conducted subgroup analyses stratifying patients by age (≥65 years or <65 years), sex, BMI (≥30 kg/m2 or <30 kg/m2), prior history of VTE, hospitalization for heart failure in the prior 3 months, hospitalization for myocardial infarction in the prior 3 months, and use of continuous or bilevel PAP therapy. Propensity score models and overlap weighting were rerun for each subgroup analysis that used the same variables as the main analyses.
All database management and statistical analyses were performed using SAS Enterprise Guide Version 7.15 (SAS Institute, Cary, North Carolina). A P-value < .05 was considered statistically significant unless otherwise noted. To reduce the chances of obtaining false-positive results because of multiple hypothesis testing, we calculated Bonferroni-corrected P values for heterogeneity across subgroups.
Results
A total of 22,840 patients met the inclusion criteria during the patient intake period (Figure 1). After applying exclusion criteria, 14,215 patients were included in the study, with 7453 patients in the rivaroxaban cohort and 6762 in the warfarin cohort. Unweighted and weighted baseline characteristics of included patients are summarized in Table 1. The mean age of patients in both cohorts was approximately 60 years. After propensity score–overlap weighting, all baseline patient characteristics of the rivaroxaban and warfarin cohorts were identical (ASD = 0). Overall, 16.1% of patients received continuous or bilevel PAP treatment, 7.1% used supplemental oxygen, 25.4% had a sleep study, and 0.7% underwent a surgical procedure to treat OSA during the baseline period.

Patient Selection Criteria. Abbreviations: DVT, deep vein thrombosis; EHR, electronic health record; ED, emergency department; NVAF, non-valvular atrial fibrillation; OAC, oral anticoagulant; OSA, obstructive sleep apnea; PE, pulmonary embolism.
The mean ± SD follow-up period was 3.7 ± 2.5 years (3.2 ± 2.3 years for the rivaroxaban cohort and 4.2 ± 2.6 years for the warfarin cohort). Between the rivaroxaban and warfarin cohorts, propensity score–overlap weighted proportional hazards regression showed similar risks of recurrent VTE (HR = 1.0, 95% CI: 0.80-1.26, P = 1.0) and major bleeding (HR = 0.87, 95% CI: 0.65-1.17, P = .36) up to 12 months after the index event (Figure 2). Similar risks of recurrent VTE and major bleeding were also observed for the two cohorts across the subgroups evaluated (Supplemental Table 1) and in the secondary outcomes (Figure 2). Sensitivity analysis utilizing sIPTW found that rivaroxaban was associated with a reduced risk of major bleeding up to 12 months post index (HR = 0.81, 95% CI: 0.67-0.97, P = .02) and up to 6 months (HR = 0.81, 95% CI: 0.66-1.00, P = .05), and a reduced risk of ICH up to 6 months (HR = 0.20, 95% CI: 0.05-0.73, P = .01) and 12 months post index (HR = 0.33, 95% CI: 0.12-0.87, P = .02) (Table 2).

Primary and Secondary Outcome Results. Abbreviations: CI, confidence interval; HR, hazard ratio; ICH, intracranial hemorrhage; PS, propensity score; VTE, venous thromboemobolism.
Results of the Sensitivity Analysis Using Stabilized Inverse Probability of Treatment Weighting.

Abbreviations: CI, confidence interval; HR, hazard ratio; ICH, intracranial hemorrhage; SD, standard deviation; sIPTW, stabilized inverse probability of treatment weighting; PS-OLW, propensity score–overlap weighting; VTE, venous thromboembolism.
Includes all major bleedings from the Cunningham algorithm that did not occur in the head.
Rounded from 0.0475 to 2 decimal places.
Sample sizes for the sensitivity analyses: rivaroxaban, 7473; warfarin: 6837.
Discussion
In this retrospective cohort study, we observed that rivaroxaban and warfarin had similar effectiveness in preventing recurrent VTE and were associated with comparable bleeding risks in patients with OSA who initiated rivaroxaban or warfarin for acute VTE, when propensity score overlapping weighting was applied. However, upon sIPTW sensitivity analyses, rivaroxaban appeared to have significantly reduced risks of major bleeding and ICH compared to warfarin.
To our knowledge, this is the first study evaluating the comparative effectiveness and safety of rivaroxaban versus warfarin for managing acute VTE and preventing recurrent VTE in patients with concomitant OSA. The findings of this study are consistent with another recent cohort analysis evaluating the effectiveness and safety of rivaroxaban versus warfarin in patients with non-valvular atrial fibrillation (NVAF) and concomitant OSA. 29 In these patients, rivaroxaban had a similar risk of stroke or systemic embolism but was associated with a significantly reduced risk of bleeding-related hospitalizations and reductions in intracranial and extracranial bleeding in patients with NVAF and concomitant OSA. 29
The findings of this study also align with the results of the pooled analysis of the EINSTEIN-DVT and PE randomized studies, which demonstrated similar efficacy but a lower rate of major bleeding for rivaroxaban compared to enoxaparin plus warfarin or acenocoumarol in patients with VTE (HR = 0.54, 95% CI: 0.37-0.79; P = .002). 30 A recent meta-analysis of 230,320 patients (74,018 treated with rivaroxaban and 156,302 with warfarin) also reported a lower incidence of VTE (risk ratio [RR] = 0.71, 95% CI: 0.61-0.84, P < .0001) and reduced risks of major (RR = 0.84, 95% CI: 0.77-0.91, P < .0001) and non-major bleeding (RR = 0.55, 95% CI: 0.41-0.74, P < .0001) with rivaroxaban. 22 In this study, extracranial bleeding, including gastrointestinal bleeding, was similar across treatment groups in both the main and sensitivity analyses. Collectively, these findings support rivaroxaban as a safe and effective anticoagulant option.
OSA is a recognized risk factor for VTE, 12 with intermediate hypoxia triggering oxidative stress and increasing pro-inflammatory mediators, leading to alterations to the blood coagulation system. 31 Rivaroxaban's Factor Xa inhibition has been shown to prevent oxidative stress associated with OSA-induced intermittent hypoxia. 31 Although our study did not find a reduction in recurrent VTE with rivaroxaban, current guidelines recommend DOACs, including rivaroxaban, over vitamin K antagonists for VTE treatment, except in cancer-associated VTE or antiphospholipid syndrome. 32 Given its efficacy, ease of use, and potential mechanistic benefits in OSA, the evidence suggests that rivaroxaban may be a viable anticoagulant option for this population.
This study has several strengths worth noting. First, the Optum® EHR database used in this study includes patients from different geographical areas in the US and captures commercially insured, Medicare, Medicaid, and uninsured patients. As a result, the study population is likely representative of the real-world population of patients with VTE treated with rivaroxaban or warfarin across the US. Second, the database uses clinical data as opposed to relying solely on billing codes, which are prone to incomplete data capture. Additionally, both prescribed and self-reported medication use are tracked in the database, allowing for assessment of over-the-counter medication use (eg, aspirin, proton pump inhibitors, St. John's wort). Furthermore, propensity scores were estimated based on commonly used variables and accepted risk factors for differential OAC exposure including demographics, comorbidities, and concurrent outpatient co-medications identified during the baseline period to adjust for potential confounding.
This study also has limitations. First, Optum® EHR claims databases are limited by sampling bias and misclassification, which can impact the internal validity of database analyses. Next, while the dataset used in this study provides data on prescribed or self-reported medications, it does not contain information about prescription medication claims. The lack of prescription claims data prevents the evaluation of persistence and treatment duration of therapy, making it difficult to accurately assess anticoagulant adherence and continuity. Given that an EHR entry to initiate an OAC does not guarantee that a patient took the prescribed medication or as in claims datasets, confirms that the patient picked up their OAC prescription from the pharmacy, what we observed in this study was a prescriber's intent-to-treat with an OAC. Additionally, residual confounding caused by unmeasured factors, missing data, or coding-related issues may be present even after propensity score–overlap weighting. Lastly, not all institutions contribute data to the Optum® EHR repository. As a result, follow-up events of patients who received care at institutions outside of the Optum® EHR system may have been missed.
Conclusions
Among patients with an acute VTE and comorbid OSA, rivaroxaban showed similar effectiveness to warfarin in preventing recurrent VTE and had a comparable risk of major bleeding.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251384278 - Supplemental material for Effectiveness and Safety of Rivaroxaban Versus Warfarin in Venous Thromboembolism Patients with Comorbid Obstructive Sleep Apnea: A Retrospective Cohort Study
Supplemental material, sj-docx-1-cat-10.1177_10760296251384278 for Effectiveness and Safety of Rivaroxaban Versus Warfarin in Venous Thromboembolism Patients with Comorbid Obstructive Sleep Apnea: A Retrospective Cohort Study by Craig I. Coleman, PharmD, Nitesh Sood, MD, Cindy Chen, PhD, Alicia K. Campbell, PharmD, and Veronica Ashton, MPH in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
Editorial and medical writing support was provided by Yanni Wang of Cobbs Creek Healthcare, LLC and was funded by Johnson & Johnson. We thank Lee Zarzabal from SimulStat Incorporated for programming support.
Ethical Approval
This study and the use of the dataset were determined by the New England Institutional Review Board (IRB) to not constitute research involving human subjects and therefore exempt from IRB oversight.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Johnson & Johnson, Titusville, New Jersey, USA.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CIC has received grant funding and consultancy fees from Johnson & Johnson and Bayer AG. NS has received consultancy fees from Johnson & Johnson, Medtronic, Inc., Abbott, and Bristol Myers Squib. CC, AKC, and VA are employees of Johnson & Johnson, the study sponsor and hold stock in Johnson & Johnson.
Data Availability
Beside the main data results, additional data are available in the supplemental materials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
