Abstract
Venous thromboembolism (VTE) is a universal health hazard. Inherited and acquired risk factors increase the risk of VTE. We evaluated the relationship between factor V (G1691A, A1090G, and A1299G), prothrombin (PT G20210A), methylenetetrahydrofolate reductase (MTHFR C677T) mutations, plasminogen activator inhibitor 1 (PAI-1 -675) polymorphism, and VTE in Turkish population. In all, 80 patients with VTE and 104 controls were included. Heterozygous factor V Leiden (FVL) mutation was significantly higher among patients (P = .04) with allele frequency of 6.3% (P = .01). Heterozygous PT G20210A mutation was also significantly higher among patients (P = .001) with allele frequency of 6.9% (P = .003). MTHFR 677TT genotype was significantly higher in patients (P = .009) with allele frequency of 23.8% (P = .005). No significant difference was found in FV A1090G and FV A1299G mutation rate as well as PAI-1 genotypes and their allele frequencies (P > .05). Thus, frequencies of FV G1691A, PT G20210A, and MTHFR C677T mutations are higher in patients with VTE. FV A1090G, FV A1299G mutations, and PAI-1 gene polymorphisms may not be a risk factor for VTE in Turkish population.
Introduction
Venous thromboembolism (VTE) is a major health hazard throughout the world. It is a common disease with an annual incidence of 1 to 3 in 1000 individuals. 1 Pulmonary embolism (PE) along with deep venous thrombosis (DVT) is a major clinical manifestation of VTE. The incidence of PE among hospitalized patients is 0.23%, and the mortality rate of this entity without appropriate treatment is reported as 30%. 2
In recent years, a growing number of studies have supported VTE as a complex and multifactorial disease that results from the interaction between predisposition to both inherited and acquired risk factors. These factors tend to increase the risk of VTE transiently or all through the life. There has been considerable interest in identifying molecular risk factors that predispose to thrombosis. The disequilibrium in hemostatic system is the key mechanism that leads to all types of thrombosis. Point mutations and/or the single nucleotide polymorphisms in the genes encoding coagulation factors shift the delicate balance in hemostatic system toward thrombosis.3,4
A major theory delineating the pathogenesis of VTE, often called Virchow’s triad, proposes that VTE occurs as a result of alterations in blood flow (ie, stasis), vascular endothelial injury, and alterations in the constituents of the blood (ie, inherited or acquired hypercoagulable state). 5 The pathways of thrombin-stimulated fibrin clot formation and plasmin-induced clot lysis are linked and carefully regulated. Clotting process occurs in 4 phases: (1) initiation and formation of the platelet plug, (2) propagation of the clotting process by the coagulation cascade, (3) termination of clotting by antithrombotic control mechanisms, and (4) removal of the clot by fibrinolysis. 6
Inherited thrombophilia is a genetic tendency for VTE that usually presents in patients younger than 50 years of age and is often recurrent. The most frequent causes of an inherited hypercoagulable state are both factor V Leiden (FVL) and prothrombin (PT) gene mutations, which together account for 50% to 60% of cases. Defects in protein S, protein C, and antithrombin III (AT III) account for most of the remaining cases, whereas a rare cause is one of the various dysfibrinogenemias. 7
Protein C is a vitamin K-dependent glycoprotein, which inactivates factor Va and factor VIIIa. It combines with thrombomodulin to produce activated protein C (APC). Activated protein C then combines with protein S on the surface of a platelet. Activated protein C can then degrade factor Va and factor VIIIa. Protein C deficiency is present in approximately 2% to 5% of patients presenting with VTE. The risk of VTE in this population is roughly 7-fold over that of the general population. Approximately 40% of patients with VTE have one of the usual thrombotic risk factors, such as pregnancy, the postpartum state, hormonal therapy, surgery, or immobilization. 8
Protein S is a vitamin K-dependent anticoagulant protein. Its major function as a cofactor is to facilitate the action of APC on its substrates, activated factor V, (FVa) and activated factor VIII (FVIIIa). Protein S deficiencies are associated with VTE. 8 Recent studies have indicated that the prevalence of protein S deficiency is particularly high in the Japanese population. In several reported series of patients with VTE in the United States, protein S deficiency was seen in 1% to 7% of patients. The deficiency is rare in population surveys of Caucasians, at approximately 0.03%. However, Japanese patients with VTE have reported a frequency of approximately 12.7% protein S deficiency and similarly elevated population frequencies of approximately 0.63%. 9 Venous thromboembolism develops in 60% to 80% of patients who are heterozygous for protein S deficiency. The remaining patients are asymptomatic, and some heterozygous individuals never develop VTE. 10
Antithrombin III is a natural inhibitor of thrombin as well as of factor Xa, IXa, and XIIa. Antithrombin III deficiency appears to be a higher thrombotic risk than protein C and S deficiencies. It is not restricted to any particular ethnic group and has been found in many countries. 11
After the discovery of AT III, protein C, and S deficiencies, the factor V (G1691A, A1090G, and A1299G), methylenetetrahydrofolate reductase (MTHFR C677T), and prothrombin (PT G20210A) gene mutations has been reported respectively. 11
The recent discovery of hereditary conditions that affect coagulation have a major impact on our understanding of thrombosis, which is now viewed as a complex disease in which interactions between genetic and environmental components contribute to clinical phenotype. Therefore, prevalence of molecular risk factors for thrombosis varies greatly in different parts of the world, both in patients with thrombosis and in the general population. We felt it was prudent to study genetic risk factors for VTE in Turkish population as limited information exists on this subject.12–15 The objective of our study was to evaluate and confirm the relationship between the factor V, PT, MTHFR mutations, plasminogen activator inhibitor 1 (PAI-1 -675) polymorphisms, and PE and PE + DVT phenotype in Turkish population.
Materials and Methods
This study was approved by Baskent University Institutional Review Board and Ethics Committee (Project No: KA 02/42).
Study Population
Between years 2005-2009, 80 Turkish patients diagnosed with PE and PE + DVT in our institution and agreeing to participate were consecutively recruited in this prospective case control study.
The control group consisted of 104 healthy adults representing Turkish population. Individuals in this group met the inclusion criteria as being randomly selected, unrelated, apparently healthy individuals without family history of or any evidence for thrombosis, and no past history of myocardial infarction, DVT, PE, or stroke. They were either blood donors or patients visiting our hospital for screening or prophylaxis. Women, with prior history of abortions or other obstetric complications were also excluded. All patients and controls were of Turkish descents and were born in Turkey.
In the study group, PE was suspected by clinical presentation, laboratory data (
Medical records of the patients (study group) were reviewed for genetic and acquired risk factors; CXR reports,
Laboratory Studies
Blood samples were obtained from the antecubital vein from all patients as well as from the controls, either at the time of diagnosis before starting anticoagulant treatment or at the time of recruitment in the study, respectively. Where appropriate,
Gel Analysis and Genotyping
Genomic DNA was prepared by phenol–chloroform technique. 16 The primers used and the conditions for polymerase chain reaction (PCR) analysis were as described previously.12–22
Polymerase chain reaction of the exon 10 of the FV gene was performed according to the previously described method. Amplified DNA was digested with HindIII enzyme (Promega, Madison, Wisconsin) at 37°C and subjected to 2% agarose gel electrophoresis. 12 To analyze FVL (G1691A) mutation, the 241 bp digested PCR products were separated by electrophoresis in 12% polyacrylamide gel electrophoresis (PAGE). The ethidium bromide stained gel showed a nondigested band of 241 bp for G allele. In the presence of A allele, the PCR product 241 bp was cut into 2 fragments of 209 bp and 32 bp.
Factor V Cambridge (FVC; A1090G) mutation was detected by PCR reaction followed by digestion with BstNI (Promega) restriction enzyme as described before. 19 AA homozygote produced 2 fragments at 162 bp and 66 bp, the AG heterozygote produced 3 fragments at 228 bp, 162 bp, 66 bp, and the GG homozygote produced 1 fragment at 228 bp, which was not restricted with BstNI restriction enzyme due to the substitution of A by G.
Factor V A1299G mutation was performed according to the previously described method. 18 A 877 bp PCR product was cut with RsaI (Fermentas, Vilnius, Lithuania) for FV A1299G mutation. The uncut product 877 bp shows the presence of A allele. If the PCR product was cut into 2 fragments as 476 bp and 401 bp, it reveals the G allele.
The presence of 20210 G to A transition of the PT gene was determined by HindIII cleavage of 345 bp fragment amplified by PCR as described previously. 17 For the analysis of the PT G20210A mutation, a 345-bp PCR product digested with HindIII (Promega) enzyme. The 20210G allele lacks the HindIII site. However, the 20210A variant has 2 bands corresponding to both 322 bp and 23 bp.
MTHFR 677 C-T mutation was determined as previously described. 21 The primers generate a fragment of 198 bp. The substitution creates a HinfI (Promega) recognition sequence that digests the 198-bp fragment into 175- and 23-bp fragments. Polymerase chain reaction product was cut with Hinf I for MTHFR C677T mutation. The uncut product 198 bp shows the presence of C allele. If the PCR product was cut into 2 fragments as 175 and 23 bp, it reveals the T allele.
Plasminogen activator inhibitor 1 (PAI-1 -675) 4G/5G polymorphism was performed according to previously described method. 22 Amplified 98/99-bp product was digested with BseLI (Fermentas) at 55°C and subjected to 6% PAGE. The uncut product (99 bp) shows the presence of a 4G allele. If the PCR product was cut into 2 fragments as 77 bp and 22 bp, it revealed the 5G allele.
Statistical Analysis
The allele and genotype frequencies were calculated for all investigated gene variations by direct counting. Genotype and allele frequencies were analyzed by Pearson chi-square and Fisher exact test. P < .05 was considered statistically significant. Stepwise binary logistic regression analysis was used to analyze the risk factors. Statistical analyses were performed by SPSS software (Statistical Package for the Social Sciences, version 13.0, SSPS Inc, Chicago, Illinois).
Results
A total of 80 (44 men) patients with mean age 54.4 ± 18.4 years (age 17-40 years: 20; age 41-60: 25; age 61 and above: 35) were recruited in the study. A total of 104 individuals were recruited in the control group. The patients and the controls differ significantly neither by age nor by gender (P > .05). In all, 29 (36.2%) patients had both PE and DVT. There were 4 cases of recurrent PE and 8 of recurrent DVT. The acquired risk factors such as surgery (n = 13), trauma (n = 3), immobilization (n = 22), malignancy (n = 3), and oral contraceptives (n = 2) were identified in 55 patients.
The genotype and the allele distribution for all the genes in patients and controls were shown in Table 1 . Table 2 demonstrates the genetic mutations in patients with isolated PE and PE + DVT. Since we could not get informed consent for some of the molecular analysis, the number of studied participants was not equal for the mutations studied.
Genes and Alleles in All Patients and Controls

Abbreviations: FV, factor V; MTHFR, methylenetetrahydropholate reductase; PAI, plasminogen activator inhibitor; PT, prothrombin.
Genetic Analysis in Patients With PE + DVT and Isolated PE
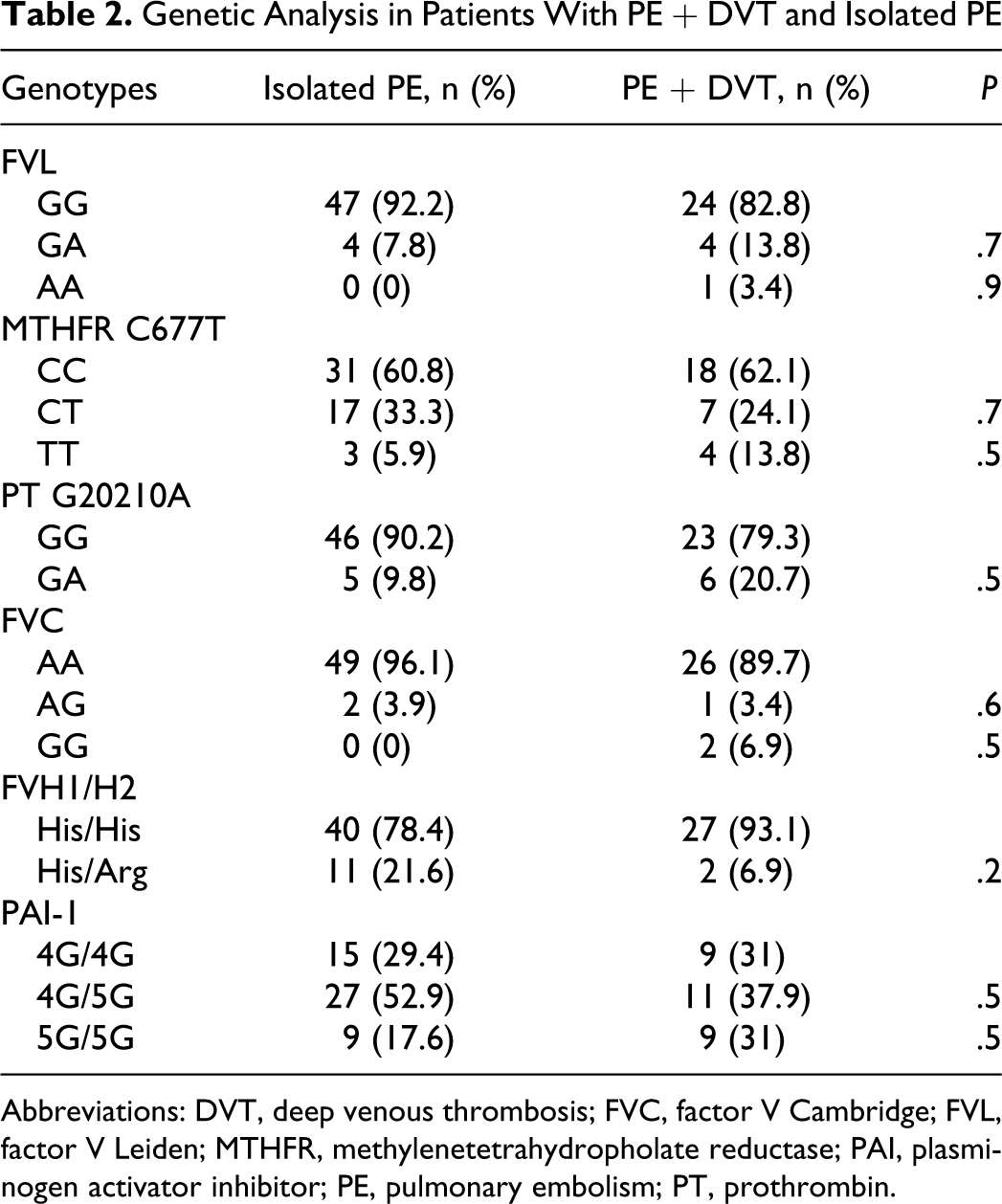
Abbreviations: DVT, deep venous thrombosis; FVC, factor V Cambridge; FVL, factor V Leiden; MTHFR, methylenetetrahydropholate reductase; PAI, plasminogen activator inhibitor; PE, pulmonary embolism; PT, prothrombin.
There was no significant association between the presence or absence of acquired risk factors and the genetic mutations (P > .05).
Heterozygous FVL mutation was significantly higher in patients than the control group (P = .04; OR = 5.3; CI [1.1-25.9]. The allele frequency of FVL was 6.3% (P = .01; OR = 6.4; CI [1.4-29.6]). Factor V Leiden mutation was found as 3.9% and 10.3% in patients with isolated PE and PE + DVT, respectively.
Heterozygous PT gene mutation was found to be significantly higher than the control group (P = .001). The allele frequency of PT G20210A was 6.9% (P = .003). Prothrombin gene mutation was found as 4.9% and 10.3% in patients with isolated PE and PE + DVT, respectively.
Homozygous MTHFR gene mutation was seen significantly high in patients than the control group (P = .009). The allele frequency of MTHFR 677T was 23.8% in patient group (P = .005; OR = 2.2; CI [1.2-3.7]). All mutations for MTHFR gene were 20.5% and 25.8% in patients with isolated PE and PE + DVT, respectively, (P = .5). In patients with homozygous MTHFR mutation, PE was seen significantly higher in patients with acquired risk factors than patients without (P = .02).
When we compare the patient and the control groups, no significant difference was found in FV A1090G and FV A1299G mutation rate, as well as PAI-1 genotypes and their allele frequencies (P > .05).
Extent of PE and Genetic Mutations
In all, 13.8% of the patients had massive, 12.5% had submassive, and 73.8% had nonmassive PE. There were no significant difference between the extent of PE and any of the genetic mutations (P > .05).
Deep Vein Thrombosis and Gene Mutations
There were no significant differences between the patients with or without DVT in terms of any of the genetic alterations (P > .05; Table 2).
Homozygous FVL and MTHFR mutations were seen in higher percentage in the patients with DVT than the isolated patients with PE but the difference was not significant (P > .05; Table 2).
The Extent of DVT
In DVT group, 21 (72.4%) of the patients had unilateral DVT. Only 12% of patients with any gene mutations had bilateral DVT and only homozygous MTHFR gene mutation was significantly increased in patients with bilateral DVT than the unilateral ones (37.5% vs 9.5%; P = .03; Table 3).
Extent of DVT and the Genetic Analyses
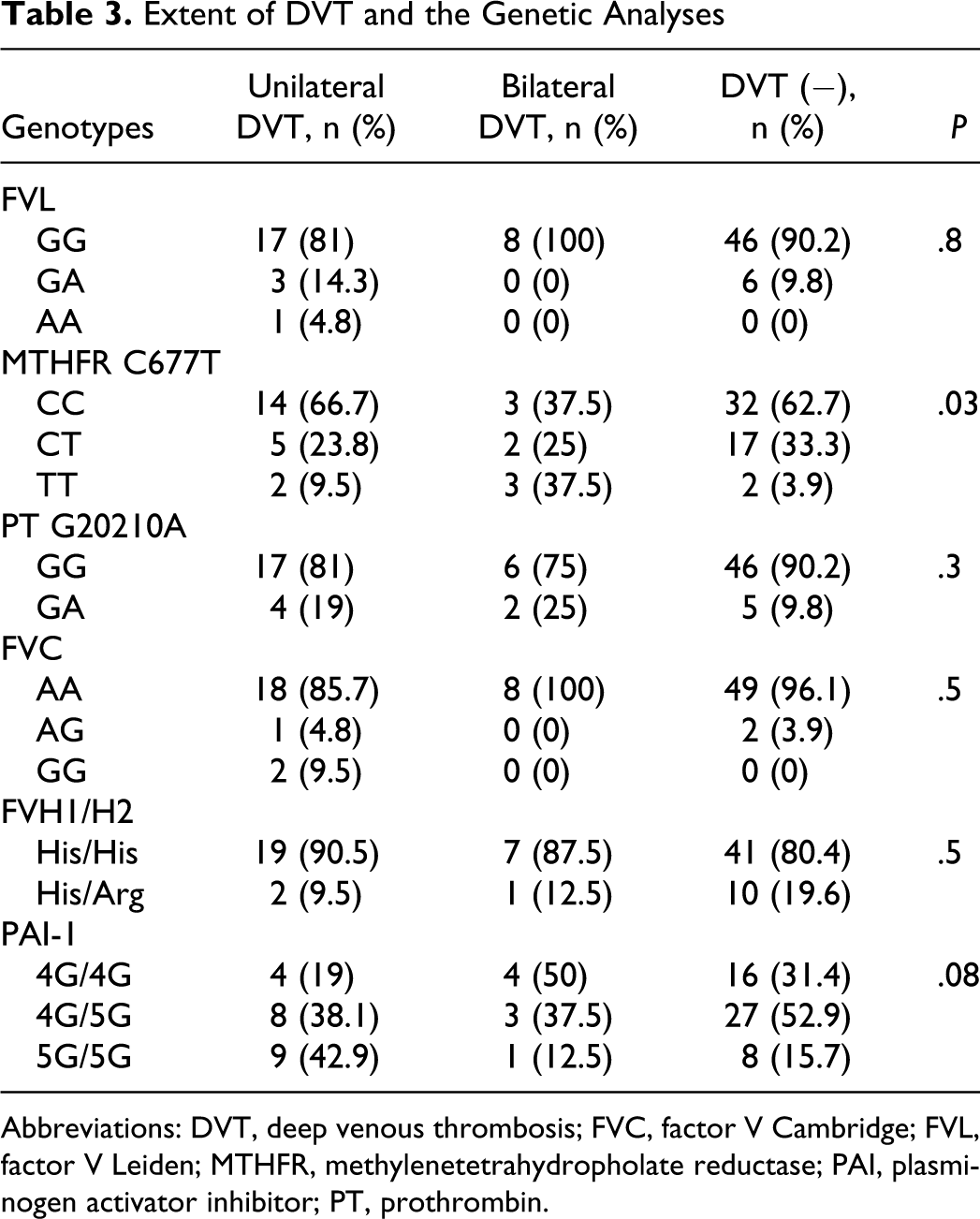
Abbreviations: DVT, deep venous thrombosis; FVC, factor V Cambridge; FVL, factor V Leiden; MTHFR, methylenetetrahydropholate reductase; PAI, plasminogen activator inhibitor; PT, prothrombin.
Location of DVT
In all, 20.6% of the patients had proximal, 37.9% of them had distal, and 41.3% of them had both proximal and distal DVT. No significant difference was found between the location of the DVT and any of the genetic mutations (P > .05; Table 4).
Location of DVT and the Genetic Mutations
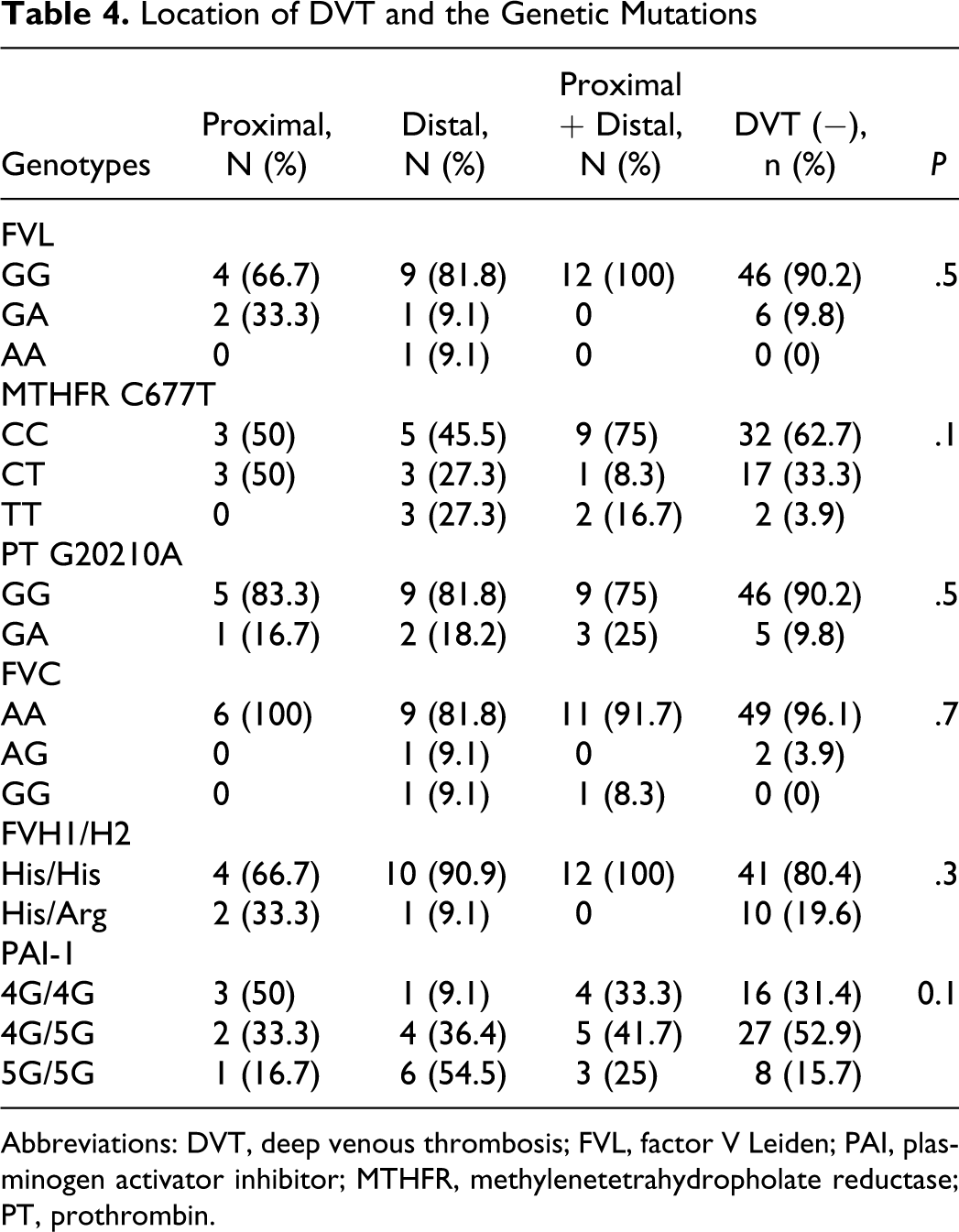
Abbreviations: DVT, deep venous thrombosis; FVL, factor V Leiden; PAI, plasminogen activator inhibitor; MTHFR, methylenetetrahydropholate reductase; PT, prothrombin.
Recurrent VTE and the Genetic Mutations
Among the 4 patients with recurrent PE, 2 patients were found to have heterozygous PT gene and in 1 with heterozygous FVL mutations. Only in 2 of 8 patients with recurrent DVT, heterozygous PT gene mutation was observed.
Discussion
The point mutations and/or the single nucleotide polymorphisms in the genes encoding coagulation factors varies greatly in different parts of the world, both in patients with thrombosis and in the general population. We felt it was prudent to further evaluate the relationship between genetic mutations and polymorphisms with VTE in Turkish population, as limited information exists on this subject in the literature.12–15
Beside the acquired risk factors, high incidence of the genetic predispositions in patients with PE is intriguing. Thus, a patient with VTE even with acquired major risk factors should also be evaluated for molecular risk factors.
Prior to 1993, the evaluation of inherited thrombophilia was limited to plasma-based assays for protein C, protein S, or antithrombin deficiency, which together today are found in less than 10% of the patients with VTE. 4 Dahlbäck et al 23 described a new and a common thrombophilia due to a mutation of FV, in which glutamine replaces arginine, so cleavage of FV by APC is inhibited. Factor V Leiden is the most common inherited cause of thrombophilia, being present in the heterozygous form in 12% to 20% of patients with incidental VTE and in approximately 40% to 50% of those with recurrent or familial VTE. 24
The prevalence of FVL mutation is high among Europeans (4%–15%) but is rarely seen in Asians, Americans, or the individuals from Far East.25–27
In Turkish population, the frequency of FVL mutation has been reported between 9% to 10.4%.12,28–32 Recent studies from Turkish population revealed that the prevalence of FVL mutations in patients with PE was not significantly different from that in the general population.33,34 On the contrary, other studies have demonstrated a increased prevalence of FVL mutation, between 18.8% and 21% in patients with PE.35,36
In regard to PE, the prevalence of FVL mutation has been found to be lower in patients with isolated PE (4.8%–9%) compared to those with PE + DVT (12.6%-24%).37–41 A study from Turkish population has also demonstrated a higher prevalence of FVL mutation in patients with PE + DVT (29.7%) than the isolated PE (15%) ones. 42 In the current study, FVL allele frequency was found as 7.8% and 10.3% in isolated patients with PE and in patients with PE + DVT, respectively, which was similar to the other published Turkish studies.
The second most frequent genetic risk factor for VTE is the PT G20210A mutation. The G to A substitution of the 3′-untranslated region of the PT gene is associated with elevated plasma PT levels and has been found in approximately 6% of Caucasians with VTE. 17 The prevalence of this mutation is 2.3% in healthy controls, 6.2% to 18% in patients worldwide. Heterozygous form of this mutation has been associated with a 3- to 9-fold increase in the risk of VTE.7,17,43 Although many studies indicate that PT G20210A mutation is an independent risk factor for VTE in Caucasians, the prevalence of this mutation is very low among Asians and Africans, indicating that it could not be considered a risk factor for VTE in these populations. 44 In healthy Turkish population, its prevalence is 2.6%. 45 In patients with PE, the mutation rate was found to be 7.7% and 6.3% in 2 different studies from Turkey.35,36 The PT G20210A mutation is found in 6% to 16% of patients with unselected DVT.7,17,46 Although FVL has been found more strongly associated with the occurrence of DVT than with PE, PT gene mutation was found to be evenly distributed in patients with DVT and PE. 46 Patients with DVT had a PT gene mutation rate of 6.5% in the southeast of Turkey. 45 In the current study, PT A allele frequency was found as 9.8% and 20.7% in patients with PE and PE + DVT, respectively, which was consistent with the previous reports from Turkey.30,45
Conflicting results have been reported on the contribution of hyperhomocystenemia related to the homozygous form of C677T point mutation in the MTHFR gene to PE development.47,48 The relevant point mutation of MTHFR has been suggested as a risk factor for venous and arterial thrombosis, particularly in combination with deficiency of folic acid, vitamin B12, and B6. 48 In a recent study, it has been concluded that there is no evidence of association between MTHFR gene mutation and the risk of VTE. 48 The frequency of the MTHFR C677T mutation has been reported to be 34.9% for our population. 13 Akar et al 13 found the C677T mutation rate between 38.4% and 48.5% in Turkish patients with PE.13,29 We found C677T mutation rate as 20.5% and 25.8% in patients with PE and PE + DVT, respectively. In the current study, 31 patients had MTHFR gene mutation, 14 of 31 had additional gene mutations such as FVL, PT G20210A, and FV 1299 His-Arg. We suspect that the presence of additional genetic mutations to MTHFR C677T may increase the risk of VTE, as previously reported. 13
The highly conserved FV gene haplotype was marked as R2 geneotype, an A to G alteration at position 4070 in exon 13 that predicts the His 1299 Arg substitutions. 49 R2 haplotype was reported in populations from Somali, Southern Indians, Italians, and Greek Cypriots with a frequency of 0.075.18,49 Akar et al 14 determined that 8.5% of the healthy controls and 10.1% of the patients with VTE carry this mutation. Akar et al also concluded that FV 1299 His-Arg mutation is prevalent in Turkish population and does not have any effect on the occurrence of VTE. In the current study, FV 1299 His-Arg mutation was reported as 16.3% in all patients. To our knowledge, this is the second study that showed FV 1299 His-Arg mutation is prevalent in our population and does not have any effect on the occurrence of VTE similar to Akar’s observations.
Plasminogen activator inhibitor 1 is the primary inhibitor of plasminogen activation in plasma, inactivating both tissue plasminogen activator and urokinase plasminogen activator. 50 Its high levels could lead to a hyperfibrinolytic state, which may cause a thrombotic tendency and levels of this substance depend on genetic factors. 4G/5G insertion/deletion polymorphism in the promoter region (-675) of the PAI-1 gene located on chromosome 7 is one of the most frequently studied polymorphisms of the gene. In some studies, it has been suggested that the 4G allele was associated with high PAI-1 levels in which its presence could be a predictor of increased thrombotic risk.51,52 On the contrary, Ridker et al as well as Stegnar et al found no association with PAI-1 genotype among patients with VTE and the controls.53,54 The concurrence of PAI-1 gene polymorphism and other thrombophilic factors has been also investigated in recent years. More than being a solitary risk factor for VTE, PAI-1 gene polymorphism, especially 4G allele, was found to be acting as an enhancer to many other genetic risk factors such as PT G20210A mutation, 55 protein S 56 deficiency, FVL mutation, 57 where different forms of VTE studied. In a meta-analyses by Tsantes et al 58 concluded that the presence of 4G allele might significantly increase the thrombotic risk in patients with inherited thrombophilia and to a lesser degree in cases without known risk factors.
Few studies have been reported from Turkey regarding this gene polymorphism. Akar et al found no association between PAI-1 4G/5G genotype and venous thrombosis, whereas the 4G allele appeared to increase the risk of venous thrombosis in the presence of concurrent inheritance of heterozygous FVL mutation. 15 Oguzulgen et al found 4G/5G polymorphism rate as 44.1% in PE patients and 55.9% in control group. They found no association between PAI-1 polymorphism and its concomitant presence with FVL and PT gene mutations in patients with PE. 36 The current study confirms the lack of association between PAI-1 4G/5G genotype and the occurrence of VTE, as in previous reports from our country.15,36
Factor V Cambridge is a transversion of G to C in nucleotide 1091 in exon 7 of the FV gene and results in the replacement of Arg306 with Thr. 19 Its association with the risk of VTE is still unclear. In the study of Williamson et al, 19 in 585 patients with PE, FVC mutation could be found only in 1 patient who also had APC resistance. A study by Franco et al 59 showed no FVC mutation in 104 patients with PE. No study surfaced in terms of “FVC mutation in Turkish population” in our literature search. In the current study, FVC mutation rate was found as 1.9% and 8.6% in patients with PE and PE + DVT, respectively, and 4.5% in control group, suggesting a high tendency for PE + DVT.
In conclusion, our results suggest that the frequencies of FV G1691A, PT G20210A, and MTHFR C677T mutations are higher in patients with VTE than those in our healthy population. Factor V A1090G, FV A1299G mutations, and PAI-1 gene polymorphisms may not be considered a risk factor for VTE in Turkish population. Moreover, our prominent finding was the high frequency of FV A1090G mutation in our healthy population. However, larger multicenter follow-up studies are required to determine whether these genetic mutations are risk factors for VTE in Turkish population.
Footnotes
Acknowledgment
We are grateful to Ayşenur Öztürk, PhD, and Canan Yazıcı, PhD, for their help in preparation of this article.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
This study was supported by Baskent University Research Fund (Project no: KA 02/42).
