Abstract
Patients with pulmonary arterial hypertension (PAH) who are admitted to the hospital pose a challenge to the multidisciplinary healthcare team due to the complexity of the pathophysiology of their disease state and PAH-specific medication considerations. Pulmonary arterial hypertension is a progressive disease that may lead to death as a result of right ventricular (RV) failure. During acute on chronic RV failure it is critical to decrease the pulmonary vascular resistance with the goal of improving RV function and prognosis; therefore, aggressive PAH-treatment based on disease risk stratification is essential. Pulmonary arterial hypertension treatment for acute on chronic RV failure can be impacted by end-organ damage, hemodynamic instability, drug interactions, and PAH medications dosage and delivery. Sotatercept, a first in class activin signaling inhibitor that works on the bone morphogenetic protein/activin pathway is on track for Food and Drug Administration approval for the treatment of PAH based on results of recent trials in where the medication led to clinical and hemodynamic improvements, even when added to traditional PAH-specific therapies. The purpose of this review is to highlight important considerations when starting or continuing sotatercept in patients admitted to the hospital with PAH.
Keywords
Introduction
Pulmonary hypertension (PH) is a chronic and progressive cardiopulmonary disease defined by an elevated mean pulmonary artery pressure of > 20 mm Hg. 1 A pulmonary artery wedge pressure (PAWP) ≤ 15 mm Hg and an elevated peripheral vascular resistance (PVR) [>2 Wood units (WU)] are used to define precapillary PH, while an elevated PAWP (>15 mm Hg) and normal PVR (≤2 WU) support the diagnosis of postcapillary PH. 1 Patients with PH are further subclassified by the World Symposium on PH 2 into 5 groups based on etiology. Group 1, pulmonary arterial hypertension (PAH), is characterized by precapillary PH due to idiopathic, heritable, drug-induced, or associated with connective tissue or congenital heart disease. Group 2 PH is the most common type, characterized by postcapillary PH due to left heart disease. Group 3 PH is the second most common type, characterized by precapillary PH due to obstructive or restrictive lung disease and/or chronic hypoxemia. Group 4 PH, is characterized by precapillary PH due to chronic thromboembolic pulmonary disease, and group 5 PH is characterized by variable degrees of pre- and postcapillary PH due to miscellaneous and often multifactorial causes.1,2
Currently, targeted medication therapies approved by the Food and Drug Administration (FDA) are primarily available for patients with group 1 PAH with the exception of riociguat, which is also approved for use in patients with group 4 PH, and inhaled treprostinil which is also approved for use in patients with PH due to interstitial lung disease (a subset of patients with group 3 PH).1,3,4 Approved medications for PAH target unique pathways which increase nitric oxide, decrease the endothelin, or enhance the prostacyclin signaling pathways (Table 1). Given the unique mechanisms of action, the European Society of Cardiology (ESC) and European Respiratory Society (ERS) guidelines recommend combination therapy for management of PAH without significant comorbidities. 1 The first PAH medication, epoprostenol, was approved by the FDA in 1995, while the latest medication approval was selexipag in 2015. Despite the relatively large number of PAH medications available, patients with the disease continue to require frequent hospitalizations and experience a high mortality rate. The REVEAL registry demonstrated that the 5-year survival of patients previously diagnosed with PAH was 65.4% and 61.2% for newly diagnosed patients and declined as functional class (FC) worsened. 5 For PAH patients who are admitted to an ICU, the in-hospital mortality rate is 25% to 40%, and of those patients who survive the hospitalization, 10% to 25% of patients die within 6 months of discharge.6-8
Medications Approved for the Management of PAH.

Abbreviations: IV, intravenous; SC, subcutaneous; PAH, pulmonary arterial hypertension; cGMP, cyclic guanosine monophosphate; cAMP, cyclic adenosine monophosphate.
The suboptimal outcomes for patients with PAH have motivated researchers to develop medications that act on different pathways, particularly agents that could reverse the pulmonary vascular damage caused by the disease (disease-modifying agents). In PAH, there is a reduction in the antiproliferative bone morphogenetic protein receptor (BMPR)-2 mediated signaling, leading to unchecked pro-proliferative activin signaling through activin receptor type 2A/B, that promotes cellular proliferation in the vascular wall.9,10 Activin-Smad 2/3 signaling is overactive in human PAH and higher levels of activin A and follistatin-like 3 are directly associated with worse transplant-free survival. 11 Sotatercept is a first in class activin signaling inhibitor (ASI). This activin receptor type IIA-Fc (ActRIIA-Fc) fusion protein binds and sequesters ligands of the transforming growth factor-β (TGF-β) superfamily, therefore, rebalancing the antiproliferative and pro-proliferative signals, reverting the PH-related damage in the pulmonary artery (PA) and right ventricle (RV) in preclinical models.9,10,12,13 Given its additional effect on reducing the TGF-β signaling pathway, sotatercept has also been studied in patients with hematologic disorders such as chemotherapy induced-anemia, multiple myeloma, myelodysplastic syndrome and end-stage kidney disease.14-17 Sotatercept is expected to come to market in the beginning of 2024. 18
Studies Evaluating Sotatercept in PAH
Phase 2 and phase 3 trials evaluating in PAH have been completed and there are several important trials ongoing (Table 2). Sotatercept was studied in PAH in the PULSAR study, a phase 2, randomized, double-blind, placebo-controlled trial enrolling 106 adult patients with PAH in World Health Organization (WHO) FC II or III, already receiving background PAH therapy. Patients received sotatercept at any of 2 doses (0.3 mg/kg or 0.7 mg/kg) or placebo every 3 weeks. Most patients were female (87%) with idiopathic PAH (58%) and on triple background PAH therapy at baseline (56%). The study met the primary endpoint, with a least-square mean reduction in PVR from baseline to week 24 of −1.8 WU (95% CI, −3.0 to −0.6; P = .003) when sotatercept at a dose of 0.3 mg/kg every 3 weeks was compared with placebo group, and of −3.0 WU (95% CI, −4.1 to −1.9; P < .001) when sotatercept at a dose of 0.7 mg/kg every 3 weeks was compared with placebo. In addition, the PULSAR study also showed improvements in exercise capacity, as measured by the 6-min walk distance (6MWD), and N-terminal-pro brain natriuretic peptide (NT-proBNP) levels. 9
Clinical Trials Evaluating Sotatercept.
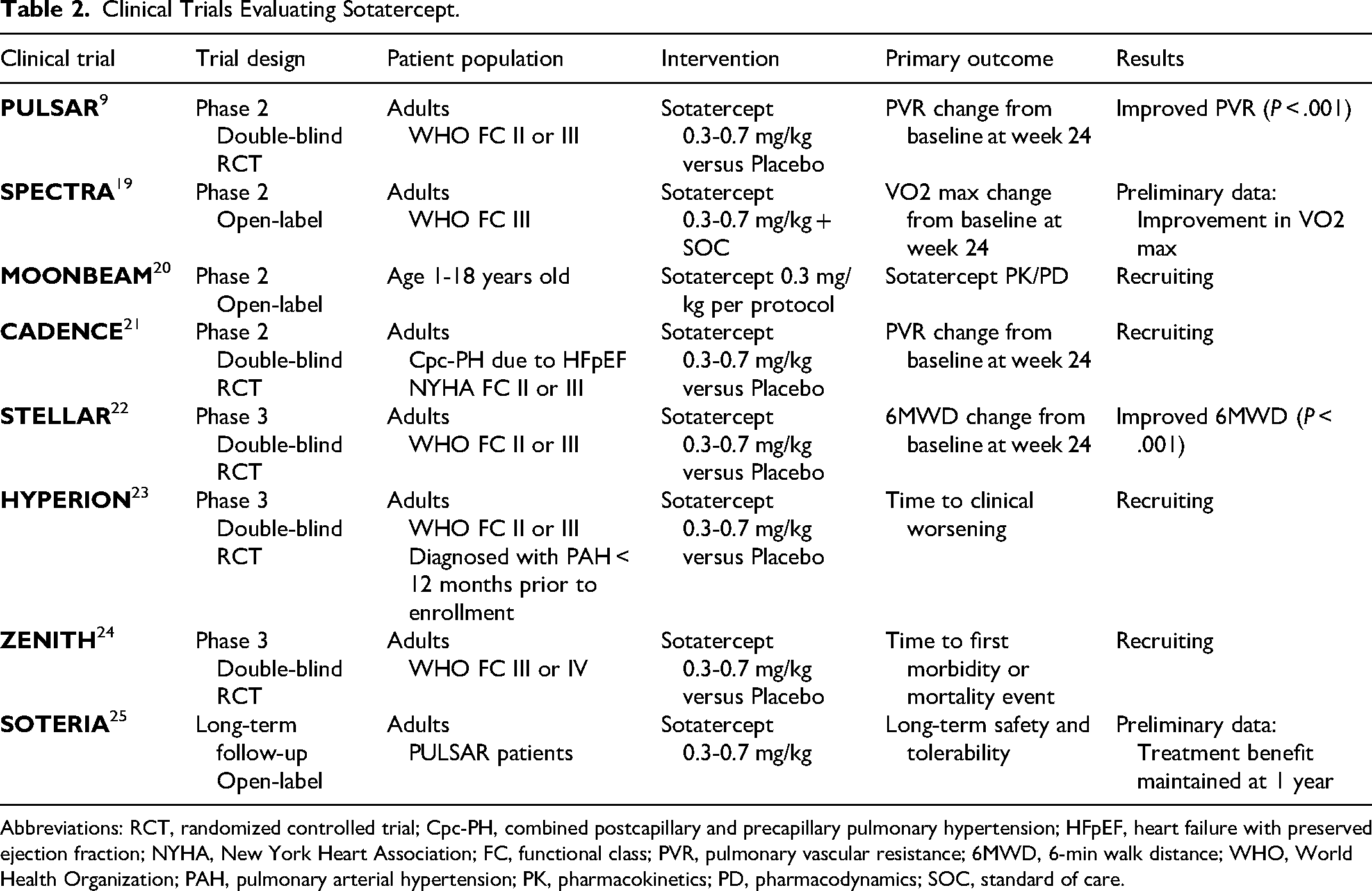
Abbreviations: RCT, randomized controlled trial; Cpc-PH, combined postcapillary and precapillary pulmonary hypertension; HFpEF, heart failure with preserved ejection fraction; NYHA, New York Heart Association; FC, functional class; PVR, pulmonary vascular resistance; 6MWD, 6-min walk distance; WHO, World Health Organization; PAH, pulmonary arterial hypertension; PK, pharmacokinetics; PD, pharmacodynamics; SOC, standard of care.
Sotatercept was most recently studied in the multicenter, double-blind, phase 3 STELLAR trial which enrolled 163 patients with PAH in WHO FC II or III, on stable background therapy. Patients were randomized to a starting dose of sotatercept 0.3 mg/kg, with a target dose of 0.7 mg/kg, every 3 weeks versus placebo. Similar to the PULSAR trial, the majority of patients were female (79.3%) with idiopathic PAH (58.5%) and on triple background PAH therapy (61.3%). The study met its primary endpoint as patients on sotatercept experienced an improvement in 6MWD at 24 weeks compared to placebo, with an estimated Hodges-Lehmann difference between groups of 40.8 m (95% CI, 27.5-54.1; P < .001). In addition, patients who received sotatercept in the STELLAR trial also had improvements in PVR, WHO FC, and NT-proBNP levels. 22
A post hoc analysis of the STELLAR trial evaluated data from right heart catheterization and echocardiography. Sotatercept treatment led to significant improvements in PA pressures, PA compliance, PA–RV coupling, and right heart function. 26 In an exploratory analysis of the PULSAR open-label extension (OLE) study, investigators evaluated the effect of sotatercept on RV function using echocardiography. During the PULSAR-OLE study, participants who were continued on sotatercept therapy maintained the improvements noted in the PULSAR study including a better RV end-systolic area index, RV-end diastolic area index, RV–PA coupling (estimated by the tricuspid annular plane systolic excursion (TAPSE)/pulmonary arterial systolic pressure (PASP) ratio), PASP, and right atrial pressures at 12-, 18-, and 24-month follow-up. TAPSE improved at months 18 and 24 while the estimated cardiac output (CO) and left ventricle (LV) function remained unchanged. 27
Additional long-term follow-up of sotatercept was evaluated in the SOTERIA trial which included patients who completed a prior sotatercept study (SPECTRA, PULSAR, ZENITH, HYPERON, and STELLAR) without drug discontinuation. Treatment effects were maintained at 1-year with 37% of the patients achieving a low-risk score by the ERS/ESC criteria. Interestingly, of the patients on a parenteral prostacyclin, 14.3% (22/154) had a decrease in dose with 2.2% (9/154) experiencing a reduction of 10% or more. 28 The PULSAR-OLE observed that clinical efficacy was maintained in the patients who continued on sotatercept therapy and the placebo-crossed group showed significant improvement in PVR, 6WMD and FC. Remarkably, at 18 and 24 months, both the continued-sotatercept group and the placebo-crossed group had a similar magnitude of improvement in primary and secondary endpoints; showing that patients initially randomized to placebo caught up to those initially randomized to sotatercept treatment. 29
In a subgroup of PULSAR and STELLAR patients (n = 35), arterial blood gases and lung diffusion capacity for carbon monoxide (DLCO) were obtained before and at week 24 of sotatercept treatment. At 24 weeks, DLCO had increased by 4% in the sotatercept group compared to a 4% decrease in the placebo group, and PaO2 increased by 3 mm Hg in the sotatercept group compared to a 2 mm Hg decrease in the placebo group. No significant changes were noted in the PaCO2, SaO2, and alveolar-arterial oxygen gradient in this preliminary study. 30
A biomarker analysis of the PULSAR study showed that activin A and NT-proBNP rapidly decreased in sotatercept treated patients compared to placebo with similar BMPR2 mRNA expression over time. Interestingly, changes in PVR, 6MWD, and NT-proBNP were similar regardless of BMPR2 mutation at baseline (26.6% of patients had ≥ 1 variation in BMPR2). 31 Although these results are exciting, more research is needed before implementing PAH-specific therapies based on subphenotypes and gene expression.
Side Effect Profile of Sotatercept
Although the clinical efficacy of sotatercept in PAH trials is compelling, there were a few notable associated adverse effects that warrant consideration. In the PULSAR trial, serious adverse effects occurred in 6% of sotatercept treated patients randomized to a dose of 0.3 mg/kg every 3 weeks, and 24% of sotatercept treated patients receiving a dose of 0.7 mg/kg every 3 weeks. The most common serious adverse effects were increases in hemoglobin (3% in 0.3 mg/kg group vs 17% in 0.7 mg/kg group vs 0% in placebo group) and thrombocytopenia (6% in 0.3 mg/kg group vs 12% in 0.7 mg/kg group vs 0% in placebo group). Three patients were withdrawn from the trial due to hemoglobin increases and one sotatercept (0.7 mg/kg) patient experienced cardiac arrest. 9 In the STELLAR trial, serious adverse events occurred in 14.1% of sotatercept-treated patients and in 22.5% of the placebo group. The most common adverse events in patients receiving sotatercept included increased hemoglobin, thrombocytopenia, hypertension, epistaxis, and dizziness. 22 The adverse effects and hemoglobin increases noted in the PULSAR and STELLAR trials are in line with data evaluating sotatercept in other disease states.14-17
In the preliminary data from SOTERIA, a long-term, open-label, follow-up study of sotatercept (median exposure 462 days), treatment-emergent adverse effects (TEAE) occurred in 37% (150/409) of patients with 1.5% (6/409) TEAE leading to drug discontinuation. Telangiectasias were noted in 22.7% (93/409) of patients without any serious adverse effect or discontinuation due to this occurrence. Epistaxis was noted in 12% (50/409) of patients, with 4 patients in whom the bleeding was directly attributed to telangiectasia. Thrombocytopenia was reported in 3.7% (15/409) of patients of whom only 2 patients were thought to be attributed to sotatercept treatment. 28
The PULSAR-OLE also assessed long-term safety at 18 and 24 months. Serious TEAE were reported in 32 (30.8%) patients of which 5 patients had events (pyrexia, red blood cell increase, systemic lupus erythematosus, ischemic stroke, pleural effusion, and PH) considered to be related to sotatercept treatment. Ten patients (9.6%) had TEAE leading to drug discontinuation and 3 patients died, but none of these deaths were attributed to sotatercept. A total of 15 (14.4%) patients experienced increased hemoglobin, particularly in those receiving sotatercept at 0.7 mg/kg every 3 weeks. Telangiectasias occurred in 11 patients and on average developed after 1.5 years of sotatercept treatment. Headache, diarrhea, and nasopharyngitis were predominantly reported during first 6 months of treatment in approximately 15% of patients, with rates decreasing over 7 to 12 months. Sotatercept dose reductions due to TEAEs were required in 41 patients, but 22 of these patients were able to tolerate a dose increase again. 29 Although long-term follow-up of sotatercept is limited, it appears to be well-tolerated with limited adverse effects. More research is needed to evaluate the long-term implications of the telangiectasis that develop under sotatercept treatment.
Initiation/Continuation of Sotatercept in the Inpatient Setting
As PAH is a progressive disease, it is likely that at some point patients will require hospitalization due to acute on chronic RV failure. These patients may require initiation of sotatercept, continuation, or a dose increase. Initiation and continuation of sotatercept in the inpatient setting will have its own challenges. New patients will need to be carefully assessed to ensure they are candidates for sotatercept therapy based on type of PH, severity of disease, potential drug adverse effects, and financial considerations. Hospitals will need to have sotatercept available on formulary, nurses will require training for administration and monitoring, and protocols will need to be developed for appropriate use, dispensing and administration of the drug. If patients are to be initiated or uptitrated on sotatercept in the inpatient setting, close monitoring should be in place to evaluate for side effects and potential adjustments of background PAH-specific treatments. 28
Pulmonary Arterial Hypertension in the Intensive Care Unit
A registry study evaluating ICU admissions of patients with PAH found that right heart failure (53.3%) was the leading cause of ICU admission, followed by infection (17.4%) and arrhythmias (11%). The 1-year mortality rate for this population was 39% and risk factors for mortality included hyponatremia, elevated NT-proBNP, bilirubin, and lactate, and need for vasopressors/inotropes, mechanical ventilation, and renal replacement therapy. 32 Patients included in the ASPIRE registry who had PAH and a critical care admission were evaluated to determine mortality rates and predictors of in-hospital mortality. The in-hospital survival rate for patients with PAH was 71%. Independent predictors of in-hospital mortality included higher Acute Physiology and Chronic Health Evaluation II scores, age, and lactate, as well as lower oxygen saturation, sodium, and platelets. Only 50% of patients receiving renal replacement therapy and 11% of patients requiring mechanical ventilation were discharged alive. 33 Despite the high mortality rates associated with PAH patients admitted to an ICU, the treatment strategies for these patients are limited. Critically ill patients with PAH should be managed by a multidisciplinary team, including physicians, advanced practice providers, pharmacists, respiratory therapists, and nurses familiar with PAH and PAH-specific medications to ensure the best possible care. The implementation of institution-specific guidelines and protocols, highlighting best practices regarding the management of critically ill patients with PAH, may help improve the care provided to these complex patients.
Management of Acute on Chronic RV Failure due to PAH
Patients with PAH who present with acute decompensation to the ICU often require a multimodal treatment algorithm to maintain hemodynamic stability and manage RV failure. Any reversible causes of acute decompensation should be identified and appropriately treated. Fluid balance must be carefully managed, as both hypovolemia and hypervolemia can have damaging effects to PAH patients further worsening RV dysfunction and CO.34-36 As volume status is optimized, treatment strategies to improve CO, decrease PVR, and resolve hypotension should be implemented. Inotropes may be added to help improve CO and potentially decrease PVR. Dobutamine remains the mainstay of therapy in patients with PAH and RV failure and may be preferred over milrinone due to its proarrhythmic effects and risk of accumulation in the presence of renal dysfunction.34,35,37
If PAH patients are still experiencing hypotension despite optimizing volume and adding inotrope therapy, a vasopressor should be added to ensure an adequate perfusion pressure. Norepinephrine is preferred in patients with PAH due to its ability to improve systemic perfusion, CO and LV afterload, but its benefit may be dose-dependent due to risk of tachycardia at higher doses. Phenylephrine should be avoided due to risk for decreased CO and increased PVR, thereby worsening RV function.37-39 The use of vasopressin as an adjunct to norepinephrine remains controversial as vasopressin may theoretically worsen pulmonary vascular constriction in patients with RV failure. 40
Although the medications approved for use in PAH have many adverse effects including hypotension, pulmonary arterial vasodilators are the main treatment for RV failure due to PAH, due to their ability to decrease RV afterload. In fact, abrupt discontinuation of PAH-specific therapies can be life-threatening.41-44 In the setting of acute decompensation, patients who are on parenteral prostacyclins at baseline may benefit from uptitration of their prostacyclin infusion rate. Consideration to add parenteral prostacyclins in treatment naive patients who are candidates for parenteral therapy should be made. These patients can also be supported by inhaled therapies (inhaled epoprostenol, iloprost, or nitric oxide) if they cannot tolerate parenteral prostacyclins or are not candidates for long-term parenteral therapy.37,45-47 The benefit of adding or uptitrating oral PAH medications in the setting of acute decompensation is limited due to their long half-lives, time to reach steady state, and impaired absorption in the setting of shock.37,48
Sotatercept Considerations
Timing
Until more data become available, sotatercept will likely be initially used as an add-on therapy in PAH patients with WHO FC II and III on background PAH therapy. If patients present to the hospital, while on sotatercept, it should be continued according to their outpatient administration schedule. In a pharmacokinetic study of healthy women, the mean terminal half-life of a subcutaneous sotatercept at a dose of 0.3 mg/kg and 1 mg/kg was 23.3 ± 4.5 days and 20.9 ± 7.7 days, respectively. This study evaluated a dosing interval of every 28 days and measured increase in hemoglobin, which had returned to baseline by day 29. 49 These data suggest that sotatercept dosing should not be delayed by greater than 1 week, particularly in patients on low-dose sotatercept, as the effects are likely not sustained beyond 28 days.
For patients admitted with an acute decompensation while on sotatercept therapy, clinicians should consider a dose increase to 0.7 mg/kg if the patient is on 0.3 mg/kg every 3 weeks. Currently no data exist to guide clinicians on whether a supplemental dose of sotatercept should be given or if the 0.7 mg/kg dose should be administered earlier. This decision will require a careful evaluation of risk/benefits, including tolerability of the medication and expected benefits.
For treatment naive patients, it remains unclear when and in what order to start sotatercept if patients are hospitalized with acute on chronic RV failure. Current PH guidelines recommend triple PAH regimen including prostacyclin analogues for patients hospitalized for acute on chronic RV failure at moderate high or high risk of 1-year mortality. 1 One could postulate that initiation of sotatercept would be appropriate given the additional benefits shown in clinical studies when added to triple background therapy9,22 and the need to be aggressive to obtain a rapid response. However, since sotatercept is not a vasodilator but a disease modifying agent, it is expected that it would take longer than pulmonary vasodilators to provide a substantial effect.
Based on pharmacokinetic data from healthy volunteers, the median time to maximum sotatercept concentration with 0.3 mg/kg and 1 mg/kg doses were 7 (7-14) and 7 (7-27) days, respectively. 49 Given these data, it is unlikely that sotatercept would provide rapid benefits to patients with PAH during an acute decompensation. Both the PULSAR and STELLAR trials reported beneficial clinical and hemodynamic effects at 6 months.9,22 An extension study also showed a sustained effect over time,28,29 but there is no information on how long it takes for sotatercept to produce a significant hemodynamic impact. Ongoing trials evaluating sotatercept in FC III and IV may help better answer this question.19-24
Administration
Sotatercept has advantages and disadvantages in its mechanism of delivery. Subcutaneous administration may be associated with discomfort at the injection site, and may require reconstitution prior to administration.9,22 The ability to administer medications subcutaneously in other disease states has allowed for greater patient satisfaction through both tolerability and improved quality of life, by permitting for at-home administration instead of a required outpatient visit. 50 Subcutaneous administration may also be advantageous compared to parenteral prostacyclins which require the medication to be prepared using sterile technique before administration through bulky and complicated infusion pumps.51-53
For patients in acute decompensation and shock, particularly those requiring vasopressor therapy, the absorption of subcutaneous medications is impaired as blood flow is shunted away from subcutaneous tissue, thus potentially impacting the efficacy and safety of sotatercept. 48 Although intravenous sotatercept has not been studied in PAH, the pharmacokinetic effects of both the intravenous and subcutaneous routes were compared to placebo in a study of patients with end-stage renal disease (ESRD) demonstrating similar pharmacokinetic and pharmacodynamic effects. 17 It is unclear if these findings can be extrapolated in the future to patients with PAH, especially given the small sample size.
Metabolism and Clearance
Critically ill patients are at a greater risk of liver failure due to sepsis, shock liver, and drug induced liver injury with the number of reported cases in the United States ranging from 2000 to 3000 cases per year. Acute kidney injury in the ICU is even more common, with a reported incidence of 15% to 70% of ICU admissions, depending on criteria used and ICU population studied.54,55 There are limited data describing the metabolism of sotatercept, but it is thought to be unaffected by renal or hepatic impairment. Patients with acute liver failure and glomerular filtration rate < 30 ml/min/1.73m2 were excluded from the PULSAR and STELLAR trials.17,49 When studied in patients with ESRD, sotatercept was nondialyzable and eliminated slowly with no clinically significant differences in pharmacokinetics. 17 Given the limited data, patients initiated on sotatercept in the setting of acute hepatic or renal failure should be closely monitored for adverse effects, and risks/benefits of the medication carefully considered.
Drug Interactions
Data regarding drug interactions with sotatercept are not published. Given its mechanism of action as an ActRIIA-Fc fusion protein it is likely to have fewer drug interactions compared to other PAH medications which commonly interact with other drugs due to their metabolism through the CYP450 system. 37
Adverse Effects
As previously mentioned, the most common serious adverse effects associated with sotatercept administration include increased hemoglobin, thrombocytopenia, hypertension, epistaxis, and headache.9,22 Consideration of these adverse effects should be made prior to initiating sotatercept particularly in critically ill patients so as not to worsen existing hemodynamic instability or hematologic abnormalities which are common. 56 Patients who present to the ICU on a stable outpatient sotatercept regimen should also be monitored for adverse effects and consideration to delay administration due to hemodynamics or elevated hemoglobin or thrombocytopenia should be made. Patients who present with angioectasias, major cardiovascular event, hemorrhagic stroke, gastrointestinal or other major bleeding should also be evaluated for risk/benefit of continuing scheduled sotatercept. Given the selected populations included in the studies, it remains unclear if sotatercept may worsen vascular malformations such as gastrointestinal angioectasias that may occur with increased age or in different diseases such as heritable hemorrhagic telangiectasia, scleroderma and cirrhosis. 57 Telangiectasia as a result of other medications including calcium channel blockers, particularly amlodipine, lithium, venlafaxine, isotretinoin, and topical corticosteroids often results in cessation of the medication.58-61
Logistics and Cost
It is unknown what the cost of sotatercept therapy will be when approved for use by the FDA and how it will be distributed. This could potentially limit the ability of hospitals to carry sotatercept on formulary. The PK data for sotatercept indicate that the effects of sotatercept are negligible beyond 28 days from administration, 49 therefore, PAH centers should evaluate adding this medication to formulary to avoid abrupt discontinuation and rebound PAH. The storage and stability of sotatercept is also not published but will likely need to be factored into decisions on how to best operationalize preparation, storage, and delivery of the drug to inpatients.
If patients are initiated on sotatercept or uptitrated during a hospitalization, arrangements must be made to ensure there are no interruptions in therapy after discharge. Place of administration is not yet clear and options may include administration at an infusion center or at home either supported by a nurse on-site or virtually, versus independent self-administration. It is certainly possible that administration may vary over time, for instance, it may start at an infusion center and if tolerated transition to a nurse supervised delivery and then self-administration. There are different challenges with all these approaches that involve patient preference/convenience, availability of an infusion center, specialty pharmacy support, and insurance benefits and requirements.
Conclusion
The management of patients with PAH who are hospitalized is certainly challenging. Pulmonary arterial hypertension patients who present with acute decompensation are at risk for RV failure and death and therefore must be carefully managed to reverse the inciting event and optimize fluid balance, hemodynamics, and RV function. Sotatercept, an ASI used to treat PAH, is being fast-tracked for FDA approval based on clinical and hemodynamic improvement even when added to traditional PAH therapies. As with other new therapies, there will be challenges in the implementation of this treatment, particularly as it pertains to the adequate timing of initiation in the inpatient setting and management post-discharge.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
