Abstract
Background:
Little data is published describing the use of medications prescribed for pulmonary arterial hypertension (PAH) in patients receiving extracorporeal membrane oxygenation (ECMO). Even though many patients with PAH may require ECMO as a bridge to transplant or recovery, little is reported regarding the use of PAH medications in this setting.
Methods:
This retrospective case series summarizes the clinical experience of 8 patients with PAH receiving ECMO and reviews medication management in the setting of ECMO.
Results:
Eight PAH patients, 5 of whom were female, ranging in age from 21 to 61 years old, were initiated on ECMO. Veno-arterial (VA) ECMO was used in 4 patients, veno-venous (VV) ECMO and hybrid ECMO configurations in 2 patients respectively. Common indications for ECMO included cardiogenic shock, bridge to transplant, and cardiac arrest. All patients were on intravenous (IV) prostacyclin therapy at baseline. Refractory hypotension was noted in 7 patients of whom 5 patients required downtitration or discontinuation of baseline PAH therapies. Three patients had continuous inhaled epoprostenol added during their time on ECMO. In patients who were decannulated from ECMO, PAH therapies were typically resumed or titrated back to baseline dosages. One patient required no adjustment in PAH therapy while on ECMO. Two patients were not able to be decannulated from ECMO.
Conclusion:
The treatment of critically ill PAH patients is challenging given a variety of factors that could affect PAH drug concentrations. In particular, PAH patients on prostacyclin analogues placed on VA ECMO appear to have pronounced systemic vasodilation requiring vasopressors which is alleviated by temporarily reducing the intravenous prostacyclin dose. Patients should be closely monitored for potential need for rapid titrations in prostacyclin therapy to maintain hemodynamic stability.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is a chronic and progressive cardiopulmonary disease with a reported prevalence ranging from 5 to 50 cases per 1 million adults globally. 1 -5 With PAH being a progressive condition that leads to right heart failure and death, 6 it is likely that patients will require intensive care unit (ICU) level care at some point in their disease course. Some of these patients may be candidates for extracorporeal membrane oxygenation (ECMO) support, including candidates for lung transplantation as a bridge to transplant or in patients with a potentially reversible cause of right-sided heart failure as a bridge to recovery. 7,8
ICU admission for pulmonary hypertension (PH) is associated with poor survival after discharge. 9 The mortality rate for patients with PAH admitted to the ICU ranges from 25% to 40%, with 45% to 80% of patients dying from right ventricular (RV) failure. 6,10 -13 Retrospective data found that older age, PH due to lung disease and/or hypoxia, baseline right heart failure, degree of organ dysfunction, higher baseline Acute Physiology and Chronic Health Evaluation (APACHE) II scores, prostacyclin use, and need for cardiopulmonary resuscitation, mechanical ventilation, or dialysis are prognostic factors associated with greater ICU mortality for patients with PH. 9,14 -16
Although there is currently no curative pharmacologic agent, there are a number of medications approved by the Food and Drug Administration (FDA) for the treatment of PAH. These treatment agents target endothelin (ET), nitric oxide (NO), and prostacyclin (PGI2) pathways, which are implicated in the pathogenesis of the disease. 17,18 These disease-specific therapies have been shown to improve pulmonary hemodynamics, 6-minute walk distance (6MWD), and World Health Organization (WHO) functional class and delay disease progression. 19 The FDA approved medications for PAH are listed in Table 1. There is currently limited data evaluating the use of PAH medications in critically ill patients, and in particular, critically ill patients on ECMO.
Physiochemical and Pharmacokinetic Properties of Pulmonary Arterial Hypertension Medications.

Treatment of PAH in the ICU
Best practice guidance recommends avoiding abrupt discontinuation of PAH medications due to risk for rebound PH and even death. 20 -24 However, PAH medication management is particularly challenging in critically ill patients due to alterations in medication pharmacokinetics due to end-organ damage, changes in volume of distribution, and patient factors impacting medication absorption, distribution, metabolism, and excretion. Many of the medications approved for use in PAH patients are oral therapies which may present a challenge in critically ill patients who do not always have oral access or adequate enteral absorption due to high vasopressor requirements or impairments impacting gastrointestinal function. 25,26
Other treatments which may be challenging to administer in critically ill patients are the inhaled PAH medications, treprostinil and iloprost, because these medications are administered via proprietary inhalation systems which are not compatible with mechanical ventilators. Nevertheless, there is some data on the successful nebulization of these PAH medications via a mechanical ventilator 27,28 but with concerns that low tidal volume ventilation, particularly while on ECMO, may limit the adequate delivery and hence efficacy of inhaled therapies. 29
Other inhaled medications that may be used for pulmonary vasodilation in critically ill patients includes inhaled nitric oxide and off-label inhaled epoprostenol. 30,31 Inhaled nitric oxide is a highly diffusible gas that when inhaled diffuses across the lung, reaching the smooth muscle cells of subjacent vessels causing selective pulmonary vasodilation. 32 Inhaled nitric oxide is theoretically an interesting therapy in patients with PAH while on ECMO since it can cause relaxation of the pulmonary vasculature, lower pulmonary vascular resistance, improve lung perfusion to well ventilated areas of the lung and hence improve oxygenation. However, only a minority of patients have a pronounced response to inhaled nitric oxide at least in the short term. 32,33 Continuous administration of inhaled nitric oxide can increase methemoglobin and nitric oxide oxidative products (NO2) which can cause airway hyperactivity and pulmonary edema; but overall clinical studies have found it safe. 34,35 One can postulate that selectively lowering the pulmonary vascular resistance would facilitate the use of VV ECMO and decrease recirculation, 36 with less clear impact in patients on VA ECMO, except during the process of weaning. However, the additional benefit of inhaled nitric oxide in adequately treated PAH patients is likely limited. 37 Data evaluating the use of inhaled nitric oxide during ECMO is lacking, with one large observational study demonstrating no clinical or financial benefit of using inhaled nitric oxide in pediatric patients requiring ECMO for pulmonary or cardiac failure. 38 Further study would be needed to determine the impact of using inhaled nitric oxide in patients with PAH during ECMO. Inhaled epoprostenol has demonstrated similar clinical outcomes compared to inhaled nitric oxide, with decreased associated cost, and thus is often used in place of inhaled nitric oxide. 39 The impact of inhaled epoprostenol during ECMO remains unclear and worth investigating.
Patients who are admitted to the ICU on intravenous (IV) prostacyclins should have additional IV access in place to reduce the risk of abrupt discontinuation of therapy in the event of line malfunction, which can be life threatening. 24,40 For patients receiving subcutaneous (SC) treprostinil, consideration should be made to transition the patient to IV administration due to concerns for impaired SC absorption in critically ill patients, especially those in shock with high vasopressor requirements. Patients should be closely monitored as they are transitioned from SC to IV treprostinil as lower doses may be needed. 41
Up to 20% of critically ill patients have liver injury or failure, 42 which could significantly impact metabolism of the PAH medications, as the majority are metabolized by the liver, specifically the CYP450 system. Although most PAH medications, with the exception of epoprostenol and iloprost, require dose adjustments in the setting of liver injury, there is very little guidance on how and when to modify them. 26 Depending on the ICU population studied and the definition used for acute kidney injury (AKI), its incidence ranges from 15% to 70% in critically ill patients. 43 Despite the higher incidence of AKI, its impact on PAH treatments is less pronounced since only tadalafil, riociguat, and epoprostenol are renally eliminated. Patients on these therapies should be monitored in the event dose adjustments are needed. 26 Additionally, augmented cardiac output resulting in increased drug clearance and volume resuscitation leading to increased volume of distribution are common in critically ill patients and may affect PAH drug concentration. 25
PAH Treatment in Patients on ECMO
Mechanical support may be considered in patients with PAH with right heart failure refractory to medical treatment, either as a bridge to lung or combined lung/heart transplantation or bridge to recovery, in those with a potentially reversible condition (such as treatment naïve PAH patients or those with a reversible cause, like sepsis or arrhythmia, for acute on chronic RV failure). 7 There are 2 main types of ECMO circuits, veno-venous (VV), in which blood is drained and returned in the venous circulation, and veno-arterial (VA), in which blood is drained from the venous circulation and returned to the arterial circulation, bypassing the pulmonary circulation at different degrees. The more common method to emergently support or unload the RV is to employ VA ECMO (peripherally, percutaneously inserted) bypassing the pulmonary circulation, decompressing the RV and thus limiting pressure/volume overload. However more creative and labor intensive options requiring cardiotomy/thoracotomy may be considered in the correct clinical context. For example, the pulmonary artery (PA)-left atrium (LA) Novalung system (pumpless) allows the high PA pressures to drive flow from the PA toward the Novalung (artificial oxygenator) and to the LA, utilizing vascular grafts anastomosed to the large vessels. Similarly, the Oxy-RVAD system employs a drainage cannulation system that siphons blood from the femoral vein with the reinfusion cannula anastamosed to the main pulmonary artery. 44,45 In VA ECMO venous access (drainage cannula) is commonly placed in the femoral veins and/or internal jugular vein and the reinfusion cannula is placed in the arterial circulation. Retrograde flow commonly reaches the arch of the aorta and is a function of the ECMO flow and the residual contractility of the native heart. Severe complications related to ECMO use are possible, including but not limited to cannulation related complications, cardiac thrombosis, coronary or cerebral hypoxia, ECMO circuit thrombosis, sepsis, and metabolic derangements. 46
In addition to alterations in pharmacokinetics influencing the efficacy and safety of medications in critically ill patients, the use of ECMO may further impact the pharmacokinetics and pharmacodynamics of PAH medications. The ECMO circuit typically consists of tubing, a membrane oxygenator, and a centrifugal pump, which are critical components that may affect PAH drug concentrations through alterations in the volume of distribution and drug sequestration within the circuit. Inconsistent outcomes related to observed differences in drug sequestration in ECMO circuits may be related to variability in components of the studied ECMO circuits such as tubing and membrane oxygenator material, tubing length, and priming solution. 47
Drug properties that influence how a medication is impacted by ECMO include lipophilicity, protein binding, degree of ionization at physiologic pH, and molecular weight. 48,49 A physiochemical property of medications that can help describe its degree of lipophilicity is the octanol-water partition coefficient or log P, with log P values <1 indicating a medication is more hydrophilic and log P values >2 being more lipophilic. Increased lipophilicity results in increased permeability, thus, lipophilic medications are more likely to be sequestered in the ECMO circuit, consequently reducing drug concentrations. 48 -50 Hydrophilic drugs can also be reduced by the increase in volume of distribution occurring at the initiation of ECMO. 48,49
Protein binding also plays a large role in predicting how a medication will be impacted by ECMO. Modern ECMO circuits are frequently bonded with albumin. Highly protein bound drugs (typically >70%) are more likely to be sequestered in the ECMO circuit, thereby decreasing drug levels. 48,49 Furthermore, the degree of ionization and molecular weight are also thought to play a role, but there is insufficient data to describe their potential impact.
Table 1 includes the physiochemical properties of PAH medications to aid in determining the degree by which an ECMO circuit may impact the drug levels of these medications. Given that PAH medications are lipophilic and highly protein bound, we would expect drug concentrations to be significantly impacted by the ECMO circuit. Unfortunately, data evaluating these medications in ECMO are scarce, especially in adult patients. A study evaluating the plasma concentration of sildenafil in 11 neonates recently decannulated from ECMO found high variability. 51 A study evaluating treatment with treprostinil in 5 neonates on ECMO found that doses ranging from 20 to 58 ng/kg/min resulted in concentrations ranging from 0.99 to 4.39 ng/ml and clinical improvement. 52
Given the limited data on the impact of ECMO on PAH medications, information from medications with similar physiochemical properties could be extrapolated. For instance, many sedative and analgesic medications commonly administered to patients on ECMO are lipophilic and highly protein bound. In ex-vivo studies using adult ECMO circuits, less than 15% of fentanyl, propofol, dexmedetomidine, and midazolam concentrations were detectable at 24 hours, whereas there was no decrease in morphine concentrations since morphine is hydrophilic and less protein bound than the other sedatives studied. 53 -55 The impact of the ECMO circuit on sedatives and analgesics has been more variable in in-vivo studies. A retrospective single-center study of 29 patients receiving VV or VA ECMO found that patients had escalating sedative and analgesia requirements throughout their ECMO course, 56 whereas another retrospective study of 32 VA and VV ECMO patients found no escalation in sedation or analgesia throughout the ECMO course, 57 reflecting the pronounced variability in the available literature.
Given the limited understanding of how PAH medications interact with the ECMO circuit and the additional pharmacokinetic challenges related to critically ill patients, a multi-disciplinary team including critical care pharmacists, ECMO-trained intensivists, PAH-specialized physicians and critical care nurses should align efforts to carefully balance efficacy and safety. If possible, medications should be titrated to effect with therapeutic drug monitoring and medications with shorter durations of action may be preferred to better facilitate titration. 48 PAH patients on IV prostacyclins may require rapid dose adjustments while on the ECMO circuit, depending on hemodynamics and side effects.
As stated earlier, due to differences in configuration, VV and VA ECMO circuits will each bypass the pulmonary circulation to a different extent. Therefore, the amount of flow through the native pulmonary circulation varies based on the ECMO configuration. Consequently, IV prostacyclin therapy may bypass the lung and be directly infused in the arterial circulation potentially causing systemic vasodilation. As ECMO flow is started or weaned, alterations in PAH therapies dosage may be needed. Therefore, providers should be extra cautious of medication levels when drug concentrations and volume of distribution can be significantly impacted, such as when ECMO is initiated or flows are adjusted, when elements of the circuit are replaced, and at the time of decannulation. Below, we describe the management of PAH medications in a series of 8 patients requiring ECMO support at Cleveland Clinic.
Patient Cases
Eight patients with an age range of 21 to 61 years, five of whom were female, were managed for PAH while requiring ECMO support (Table 2). Patients who were known to have required ECMO support were identified from an internal database of PAH patients at Cleveland Clinic main campus. VA ECMO was the most commonly utilized configuration while 2 patients utilized VV ECMO, and 2 patients utilized hybrid ECMO configurations. Common indications for ECMO included cardiogenic shock, bridge to solid organ transplant, and cardiac arrest. All patients were on parenteral prostacyclin therapy at baseline, with 5 patients also on oral PAH therapy prior to ECMO cannulation.
Characteristics of Patients Receiving Pulmonary Arterial Hypertension Medication Therapies While on Extracorporeal Membrane Oxygenation.

Abbreviations: ECMO, extracorporeal membrane oxygenation; VA, veno-arterial; VV, veno-venous; PO, oral; PAH, pulmonary arterial hypertension; IV, intravenous; NE, norepinephrine; AVP, vasopressin; PE, phenylephrine; DIC, disseminated intravascular coagulopathy; BID, twice daily; TID, 3 times daily; EPI, epinephrine; CTEPH, chronic thromboembolic pulmonary hypertension.
In our cohort, refractory hypotension requiring vasopressors was noted in 7 patients. In 5 patients, hypotension was managed via downtitration or discontinuation of baseline PAH therapies, which resulted in an improvement in vasopressor requirements. Three patients had continuous inhaled epoprostenol added during their time on ECMO, typically during the period when parenteral prostacyclin therapy was downtitrated or discontinued. Importantly, in patients who were able to be decannulated from ECMO, PAH therapies were typically resumed or titrated back to baseline dosages prior to ECMO decannulation. Only one patient required no adjustment in PAH therapy while on ECMO.
Discussion
As documented in our case series, VA ECMO can be considered a salvage measure in select PAH patients. Blood pressure augmentation by VA ECMO (centrifugal pump) is non pulsatile and therefore is best characterized by mean arterial pressure (MAP). 58 Ideal MAP targets during VA ECMO are controversial but a MAP ≥65 mmHg is often an accepted goal. 59 Hypotension necessitating vasoactive medications may occur during ECMO. Etiology is frequently multifactorial and includes sedation, volume depletion and sepsis. In addition, VA ECMO support increases the afterload of the left ventricle as a result of the retrograde blood flow, which could impact hemodynamics particularly in left ventricular failure. 60 Furthermore, as blood interacts with the extracorporeal circuit, the inflammatory response is compounded by a low cardiac output state and tissue hypoperfusion. During ECMO initiation, there is a rapid upsurge in levels of pro-inflammatory cytokines, 61,62 followed by activation of the complement system, 63 resulting in leukocyte activation. 64 A systemic inflammatory response syndrome analogous to sepsis manifests as vascular dysregulation with ensuing vasoplegia and capillary leak syndrome. 65
Recirculation, explicitly seen in VV ECMO, results in the oxygenated blood being reinfused back into the drainage cannula, without contributing to systemic oxygenation. 66 Cannula configuration, pump speed and/or blood flow, cannula size, and changes in intrathoracic/intraabdominal pressure are contributory factors to recirculation. We postulate that a comparable phenomenon is the most likely culprit of the vasoplegia noted in our patients. Close approximation of the drainage cannula of the VA circuit to the infusion port of the catheter delivering the prostacyclin therapy poses a therapeutic challenge (Figure 1). An unaccounted amount of the prostacyclin infused could be inadvertently siphoned off and injected into the systemic arterial circulation with pronounced hemodynamic consequences. This phenomenon could be more pronounced at ECMO initiation, when there is rapid efflux of blood into the ECMO circuit and simultaneous injection of a relatively large bolus dose of prostacyclin therapy into the arterial circulation.
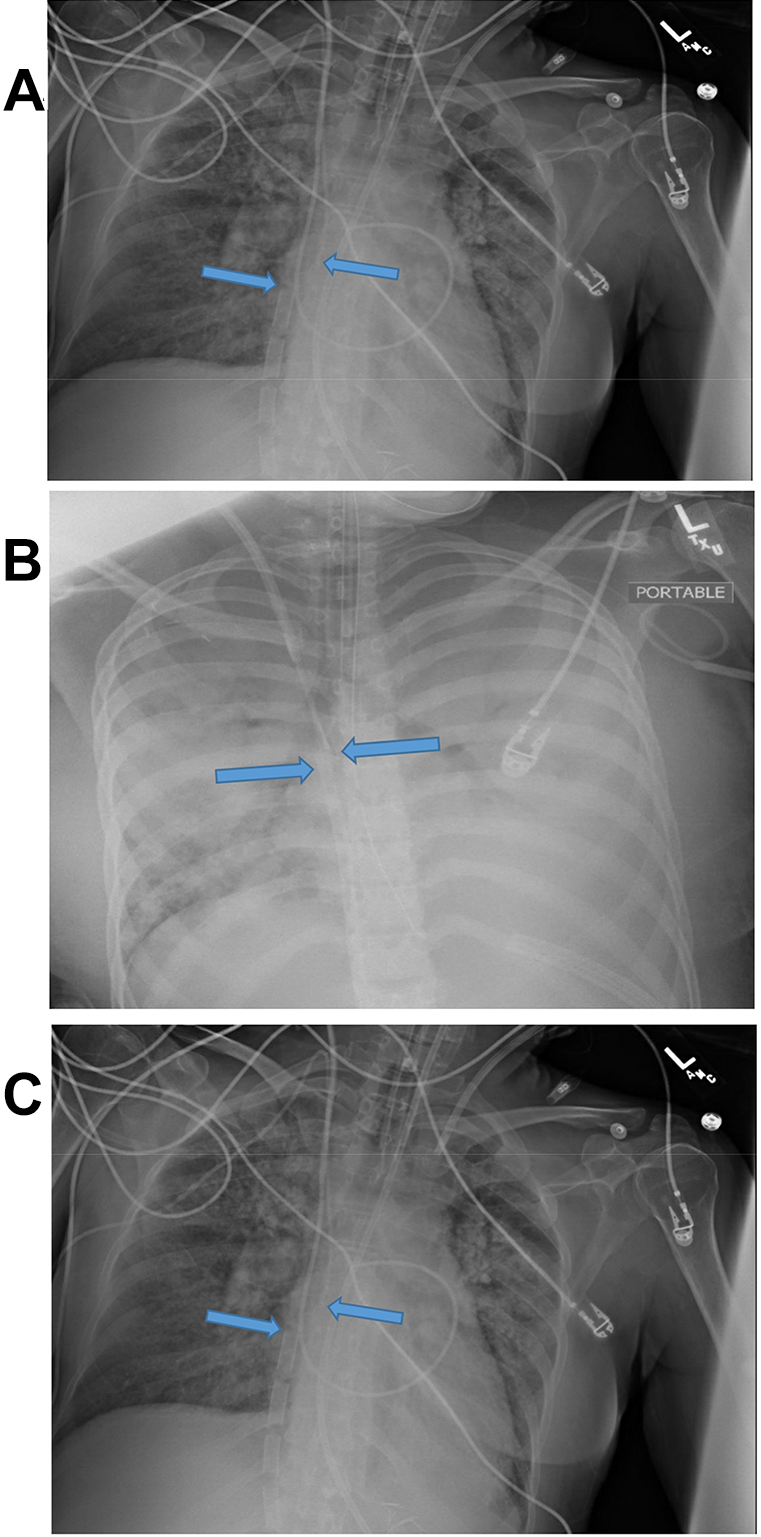
Extracorporeal circuit configuration and pulmonary arterial hypertension medication delivery. Chest X-rays demonstrating potential interaction between extracorporeal circuit configuration and intravenous access used for administration of intravenous prostacyclin therapy. A, Patient 1—Receiving treprostinil IV 147 ng/kg/min prior to VA ECMO cannulation. Required downtitration over a 6 hour period on ECMO day 1 due to hypotension requiring vasopressor support. Arrows are pointing to drainage cannula and vascular cannula. B, Patient 3—Receiving epoprostenol IV 67 ng/kg/min prior to VA ECMO cannulation. Required discontinuation of epoprostenol due to ongoing hemorrhage. Arrows are pointing to right and left femoral reinfusion. C, Patient 5—Receiving epoprostenol IV 4 ng/kg/min prior to VA ECMO cannulation. Required uptitration of epoprostenol after vasopressor requirements stabilized due to right ventricular failure. Arrows are pointing to right femoral drainage cannula and left femoral artery return.
Our hypothesis could be potentially tested by measuring prostacyclin levels at different sites of the ECMO circuit; however, this is challenging since (a) epoprostenol has a short half-life and only the metabolite is usually measured and (b) treprostinil levels are measured using a proprietary assay. 67 It is interesting that in 5 of our patients, the dose of PAH medications was rapidly decreased; however, after their shock improved the PAH medications were resumed and maintained at prior or higher doses. Though other reasons (i.e., sepsis) for the persistent vasoplegia on VA ECMO cannot be discounted, microbiological cultures were negative in all of these patients. The negative blood cultures and the rapid response to alterations in PAH medications (discontinued or down-titrated) is suggestive of a direct effect of PAH medications, particularly prostacyclin analogues on the systemic circulation giving credence to our hypothesis. Our hypothesis is further supported by findings from other single center retrospective experiences, which have reported the need to down-titrate PAH-specific therapies to minimize systemic effects, particularly in patients on VA ECMO, who are at greater risk of medication diversion into the arterial circulation. 68,69
Conclusions
The treatment of PAH patients during their ICU stay is challenging given the multiplicity of factors that could affect PAH drug concentrations. In particular, PAH patients on prostacyclin analogues placed on VA ECMO appear to have pronounced systemic vasodilation requiring vasopressors which is alleviated by temporarily reducing the intravenous prostacyclin dose. Research is needed to better understand this finding and develop protocols to better manage this hemodynamic condition.
Footnotes
Authors’ Note
Site where work was completed: Cleveland Clinic, Cleveland, Ohio.
Author Contributions
All authors participated in the conception, design, writing and critical revision of the manuscript for important intellectual content and final approval of the manuscript submitted. Adriano R. Tonelli is the guarantor of the paper, taking responsibility for the integrity of the work, from inception to published article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
