Abstract
The choice of inodilator drug in the acute management of patients with pulmonary hypertension (PH) having right ventricular (RV) failure remains unsettled and challenging. Comprehensive experimental evaluations may provide further insight and fundamental translational research clues to support inodilator selection and clinical trial design. Our aim was to compare acute dose–response hemodynamic effects of inodilators dobutamine (DOB), milrinone (MIL), and levosimendan (LEV) in chronic experimental PH. Seven-week-old male Wistar rats were randomly injected with 60 mg·kg−1 monocrotaline (MCT) or vehicle (Ctrl, n = 7) and underwent systemic and pulmonary artery (PA) pressure and RV pressure–volume (PV) hemodynamic evaluation under halogenate anesthesia 24 to 30 days after injection. The MCT-injected animals (n = 7 each) randomly received dose–response infusions of DOB (1, 3, 6 and 12 μg·kg−1·min−1), MIL (MIL: 1, 3, 6 and 12 μg·kg−1·min−1), or LEV (0.3, 0.6, 1.2 and 2.4 μg·kg−1·min−1). Load-independent indexes were obtained by inferior vena cava occlusion at baseline and after the last dose. All inodilators increased RV ejection fraction, preload recruitable stroke work, and ventricular–vascular coupling without jeopardizing perfusion pressure. Dobutamine raised heart rate and PA pressure. Only LEV increased cardiac index and decreased PA elastance and pulmonary vascular resistance (PVR). Moreover, only LEV downward-shifted the end-diastolic PV relationship, thereby improving RV compliance. Adding sildenafil to LEV further decreased PVR. Levosimendan had beneficial acute systolic and diastolic functional effects in experimental chronic PH and RV afterload compared to DOB and MIL. It should be further tested in clinical trials enrolling patients with PH in the perioperative and critical care settings.
Introduction
Effective pharmacological management of pulmonary hypertension (PH) and right ventricular (RV) failure remains the unmet challenges in cardiac anesthesiology and critical care. 1,2 Although PH and RV failure may occur suddenly in a previously healthy heart such as in pulmonary thromboembolism or RV myocardial infarction, most cases involve progression of chronic disease. 3 Although the majority corresponds to patients with left heart and lung disease, improved therapy of pulmonary arterial hypertension (PAH) is steadily raising the number of patients with PAH presenting in the critical and perioperative settings. 2 Management is highly complex and requires upholding systemic perfusion pressure, lowering lung vascular tone, strict control of volemia and ventilation, and judicious use of inotropes. In a recent systematic review, Price et al highlighted that the quality of evidence is generally low for intensive care management of pulmonary vascular dysfunction. Based on the GRADE methods, they advised as strong recommendations that phosphodiesterase type 3 inhibitors (PDE3i) and inhaled pulmonary artery (PA) vasodilators should be used mostly after cardiac surgery, whereas the use of dobutamine (DOB) or levosimendan (LEV) was advised as weak recommendation. 1 These recommendations are constrained by the small number of available studies. Clinical or experimental trials comparing the acute effects of inodilators LEV, milrinone (MIL), and DOB in the setting of chronic PH are currently unavailable. A large randomized clinical trial (RCT) would be highly desirable to guide inodilator drug selection, 4 but currently none is ongoing. Conducting large RCTs on the acute effects of inodilators in chronic PH during the perioperative period and in critical illness is a huge task from patient enrollment to outcome analysis. Recently, a meta-analysis resourcing to Bayesian networks that included 46 trials and 2647 adult patients submitted to cardiac surgery suggested that among inodilators, only LEV was associated with reduced mortality. 5 Tightly controlled comprehensive experimental evaluations may provide further insight and fundamental clues to support inodilator selection. The aim of this work was to compare the head-to-head acute dose–response hemodynamic effects of inodilators LEV, DOB, and MIL in the experimental model of chronic monocrotaline (MCT)-induced PH. Additionally, we assessed the joint effect of LEV and the phosphodiesterase type 5 inhibitor sildenafil.
Methods
Animal Model
Seven-week-old male Wistar rats (Charles River Laboratories, Barcelona, Spain) were randomly assigned (6:1) to receive either 60 mg·kg−1 subcutaneous MCT (n = 42) or an equal volume of vehicle (Ctrl, n = 7). Hemodynamic evaluation was carried out 24 to 30 days after injection. Surviving MCT-injected rats randomly underwent cumulative dose–response assessment of the effects of inodilators DOB (DOB hydrochloride; Sigma-Aldrich Co, Sintra, Portugal), LEV (kindly provided by Orion corporation, Espoo, Finland), and MIL (Sigma-Aldrich Co, Sintra, Portugal; n = 7 each group). In another set of MCT-injected animals (n = 16), we tested combined LEV and sildenafil infusion (kindly provided by Pfizer Inc, NY, USA). Animals were kept in groups of 2 per cage under controlled environment with a 12-hour light–dark cycle and at 22°C room temperature. All animals received humane care. Experimental procedures were approved by the ethical committee of the Faculty of Medicine of Porto and were performed in accordance with the Portuguese law on animal welfare, the European Union (EU) Directive 2010/63/EU for animal experiments, and the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH publication no. 85-23, revised 2011).
Hemodynamic Evaluation
Upon sedation and analgesia with 100 µg·kg−1 and 5 mg·kg−1 intraperitoneal fentanyl and midazolam, respectively, and anesthesia with sevoflurane inhalation (8% and 2.5%-3% for induction and maintenance, respectively), animals were endotracheally intubated (14 G) and monitored with electrocardiogram (Animal Bio Amp, FE136; ADInstruments, Oxford, UK), peripheral oximetry (MouseSTA—Pulse Oximeter & Heart Rate Monitor, Physiosuite; Kent Scientific, CT, USA), and capnography and monitored for respiratory rate (RR), minute ventilation (CapnoScan—End-Tidal CO2 Monitor, Physiosuite; Kent Scientific, CT, USA), and central temperature. Animal temperature was automatically controlled on a heating pad (RightTemp—Temperature Monitor & Homeothermic Controller, Physiosuite; Kent Scientific, CT, USA). Fluid replacement with 64 mL·kg−1·h−1 warmed Ringer lactate (NE-1000; New Era Pump Systems, NY, USA) was established through the right dorsal foot vein (24 G). Mechanical ventilation was instituted with 100% O2 and positive end-expiratory pressure (PEEP) held at 5 cm H2O in controlled pressure ventilation mode with inspiratory pressure set at 12 cm H2O above PEEP and an inspiration to expiration ratio of 1:2. The RR was adjusted to maintain normocapnia. A left thoracotomy was performed in the right lateral decubitus. A flow probe was placed in the ascending aorta (2.5PS; Transonic, NY, USA), a 3-0 silk lace was positioned around the inferior vena cava (IVC), and pressure–volume (PV) catheters were inserted through the apex along the RV long axis and into the ascending aorta (SPR-869 and SPR-847, respectively; Millar Instruments, TX, USA). Another small-gauge catheter (PV-1035; Millar Instruments, TX, USA) was inserted into the main PA through the RV infundibulum. Purse string sutures were used to secure the catheters and minimize bleeding at the site of insertion. Baseline (B) recordings were obtained after a stabilization period of 30 minutes. The IVC occlusion recordings were also obtained to derive load-independent indexes of contractility and compliance. After randomization, cumulative dose–response effects of inodilators were assessed by continuous infusion at 4 increasing doses (D1-4). Doses were 1, 3, 6, and 12 μg·kg−1·min−1 for MIL and DOB and 0.3, 0.6, 1.2, and 2.4 μg·kg−1·min−1 for LEV. Recordings at each step-up dose were obtained upon stable effect of at least 10 minutes. The IVC occlusion recordings were repeated after the final dose. In another set of MCT-injected animals, we performed a 2.4 μg·kg−1·min−1 LEV infusion and then added a 42.5 μg·kg−1·min−1 sildenafil infusion. Recordings were performed upon stable effect at baseline, after LEV, and after joint LEV and sildenafil infusion. Fearing systemic vasodilation from vasodilator drug combination, a 0.1 μg·kg−1·min−1 norepinephrine infusion was started immediately upon venous cannulation. Otherwise procedures were as previously described. All acquisitions were performed with ventilation suspended at end expiration. Signals were continuously acquired (MPVS 300; Millar Instruments, TX, USA) and digitized at 1000 Hz (ML880 PowerLab 16/30; ADInstruments, Oxford, UK). Volume signal was corrected for field inhomogeneity by slope factor α as derived from gold standard flow meter cardiac output measurement (TS420; Transonic, NY, USA) and subtraction of parallel conductance as assessed by 40 μL 10% hypertonic saline injection. Volumes were indexed to body surface area (BSA) as defined by 9.1 × body weight (BW, in g)2/3 to account for differences between Ctrl and MCT. Time constant of isovolumic relaxation was derived by monoexponential fitting with 0 asymptote (τexp). Load-independent indexes of cardiac performance were derived from IVC occlusion acquisitions by linear and exponential fitting of the end-systolic and end-diastolic PV relationships (ESPVR and EDPVR). The correction term in quadratic model fits to the ESPVR was very close to 0, supporting the linear fit. Pulmonary and systemic vascular resistance indexes (PVRI and SVRI, respectively) were obtained neglecting central venous pressure and left atrial pressure by dividing mean pulmonary and systemic arterial pressures by cardiac index (CI).
Statistical Analysis
Baseline hemodynamic features were assessed by 1-way analysis of variance (ANOVA) with least square means contrasts to compare vehicle-injected and MCT-injected rats or Kruskal-Wallis nonparametric test whenever deviation either from normality of residuals or from homogeneity of variances was observed according to Shapiro-Wilk statistic or Levene test, respectively. Analysis of covariance was applied to ESPVR and EDPVR to account for the role of multiple parameters. Log transforms were used whenever necessary to meet assumptions. Dose–response effects on hemodynamics were assessed by general linear model (GLM) with dose and group as within-subject and between-subject variables, respectively. The effects of dose or dose × group interactions were assessed by multivariate analysis with Pillai trace. When significance was found, further analysis was performed by univariate tests with Bonferroni critical value correction. Cumulative effects of LEV and sildenafil were assessed with 1-way repeated-measures ANOVA. Huynh-Feldt correction with adjusted df was used whenever sphericity assumption was violated as assessed by Mauchly test. Hemodynamic data from IVC occlusions at baseline and after the last dose were also compared with GLM. Absolute changes in intercept (pressure at volume 0) and scaling constant were included as covariates in ESPVR and EDPVR analysis, respectively. Data are mean ± standard error of the mean. Significance was set at 2-tailed, P < .05.
Results
By the time of hemodynamic evaluation (24-30 days after injection), 50% of MCT-injected animals had died. This is expected as MCT-induced PH is an experimental model of severe, rapidly progressive PH and ultimately RV failure. Indeed, when animals were evaluated, they showed signs of impending RV failure such as labored breathing, ruffled hair, reduced activity, and weight loss. Despite this, there were no animal losses during hemodynamic evaluation. Both BW and BSA were decreased in all MCT-injected groups compared to Ctrl (Table 1). Despite some heterogeneity, no baseline differences were observed between LEV, DOB, or MIL-treated MCT-injected rats. All showed increased PA pressures, PVRI and PA arterial elastance (Eai), low ejection fraction (EF) and reduced CI, a trend toward RV dilation as assessed by end-diastolic volumes (EDVi), elevated end-diastolic pressure (EDP), upward shifted EDPVR, and delayed relaxation as assessed by τexp compared with Ctrl (Table 1). Despite this, peak rate of pressure rise (dP/dtmax) tended to be higher, and end-systolic elastance (Eesi) was significantly elevated. Preload recruitable stroke work (PRSW), which is a geometry and chamber size-independent index of contractility, was preserved (Table 1).
Baseline Hemodynamics.a

Abbreviations: αi, scaling factor for exponential fitting of the end-diastolic pressure–volume relationship; βi, chamber stiffness constant for indexed volumes; BSA, body surface area; CI, cardiac index; dP/dtmax, peak rate of pressure rise; EDP, end-diastolic pressure; EDVi, indexed end-diastolic volume; EF, ejection fraction; Eesi, end-systolic elastance for indexed volumes; HR, heart rate; MBP, mean blood pressure; MPAP, mean pulmonary artery (PA) pressure; PA Eai, PA effective arterial elastance for indexed volumes; PVRI, pulmonary vascular resistance index; V0i, volume axis intercept of the end-systolic pressure-volume relationship for indexed volumes; PRSW, preload recruitable stroke work; VVC, ventricular-vascular coupling; τexp, time constant of isovolumetric relaxation by monoexponential fitting.
aBaseline hemodynamic features of monocrotaline (MCT)-injected pulmonary hypertensive rats treated with dobutamine (DOB), levosimendan (LEV), and milrinone (MIL) and their healthy vehicle-injected controls (Ctrl).
b P < .005 versus MCT, n = 7 each group.
As regards dose–response effects of inodilators in MCT-injected rats, DOB increased heart rate (Figure 1A) and systolic PA pressure at doses higher than D2 (Figure 2A), whereas LEV increased CI and decreased PA Eai at D4 (Figure 1). Similar results were obtained for PVRI, which significantly decreased only at D4 of LEV (P < .05, group × dose interaction). Although there was a trend toward decreased mean PA pressure with increasing LEV doses (Figure 2B), this was not significant (P = .18). None of the inodilators decreased mean blood pressure at the tested dose ranges (Figure 1D). There was an overall increase in RV EF, dP/dtmax, and stroke work index (SWI) as main effect of all inodilators (Table 2). There was a trend for interaction with LEV and MIL eliciting higher increases in SWI than DOB (P = .13) and LEV higher increases in EF (P = .12). All inodilators enhanced RV relaxation as assessed by decreased τexp (Table 2). The RV end-diastolic elastance was significantly decreased in MCT-LEV as assessed by lower EDP and higher EDVi at D4, whereas EDVi was significantly decreased in DOB (Figure 3). Load-independent indexes of systolic and diastolic function derived from IVC occlusions obtained at baseline and after the last dose in each MCT-injected group revealed increased Eesi in DOB and MIL and decreased Eesi in LEV (Figure 4), while there was an overall increase in PRSW and improved ventricular–vascular coupling (VVC) in all groups at D4 (Figure 5). The EDPVR was downward-shifted in LEV and upward-shifted in DOB. No significant differences were observed for MIL (Figure 4).
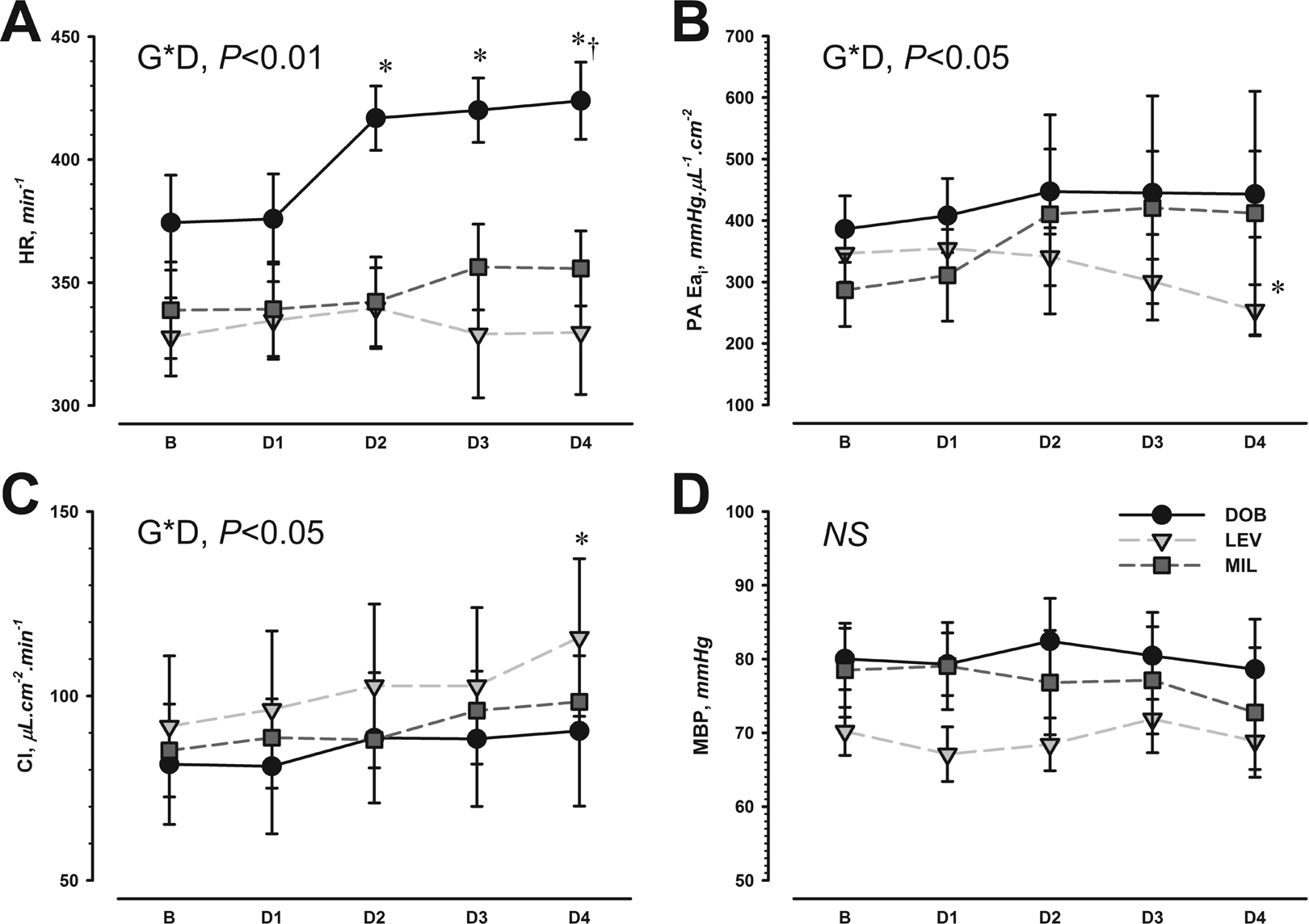
Dose–response effect of dobutamine (DOB), levosimendan (LEV), and milrinone (MIL) on heart rate (HR; A), pulmonary artery effective arterial elastance for indexed volumes (PA Eai; B), cardiac index (CI; C), and mean blood pressure (MBP; D) in monocrotaline-induced pulmonary hypertensive rats. Baseline (B) and 4 cumulative doses (D1-4) are presented; specific doses for each drug are given in the Methods section. Significant results for interaction between group and dose (G × D) are shown, as well as a univariate decomposition of significance. *P < .05 versus B, †P < .05 versus other groups at the corresponding dose, n = 7 each group.

Dose–response effect of dobutamine (A), levosimendan (B), and milrinone (C) on systolic, mean, and diastolic pulmonary artery pressures (sPAP, mPAP, and dPAP, respectively). Baseline (B) and 4 cumulative doses (D1-4) are presented; specific doses for each drug are given in the Methods section. Significant results for interaction between group and dose (G × D) are shown, as well as adjusted univariate decomposition of significance. *P < .05 versus B, n = 7 each group.
Dose–Response Right Ventricular Hemodynamics.a

Abbreviations: D, dose; DOB, dobutamine; dP/dtmax, peak rate of pressure rise; EF, ejection fraction; G, group; I, interaction; LEV, levosimendan; MCT, monocrotaline; MIL, milrinone; ME, main effect; SWI, stroke work index; τexp, time constant of isovolumetric relaxation by monoexponential fitting.
aDose–response effects on right ventricular performance of DOB, LEV, and MIL in MCT-injected pulmonary hypertensive rats. Baseline (B) and increasing doses (D1-4) are represented. For detailed dosing on each drug, see Methods section. The ME and I of G ×D are presented in the rightmost columns, n = 7 each group.

Dose–response effect of dobutamine (DOB), levosimendan (LEV), and milrinone (MIL) in right ventricular (RV) end-diastolic pressure (EDP) and volume (EDV). Baseline (B, large circles), cumulative doses (small circles), and final dose (D4, triangles) are represented along with arrows that indicate increasing dosages. Significant results for interaction between group and dose (G × D) are shown, as well as adjusted univariate decomposition of significance. *P < .05 versus B in EDP, †P < .05 vs B in EDV, n = 7 each group.

Inferior vena cava occlusion pressure–volume loops and corresponding end-systolic and end-diastolic pressure volume relationships (ESPVR and EDPVR, respectively) at baseline (gray lines) and after the last dose (black lines) of dobutamine (A), Levosimendan (B), and milrinone (C). Loops are representative of all animals and were plotted by resampling and interpolation to obtain 20 pressure–volume points in every animal. Significant results for interaction between group and timing (G × T) are shown for both ESPVR and EDPVR as well as univariate decomposition of significance. *P < .05 versus B, n = 7 each group.
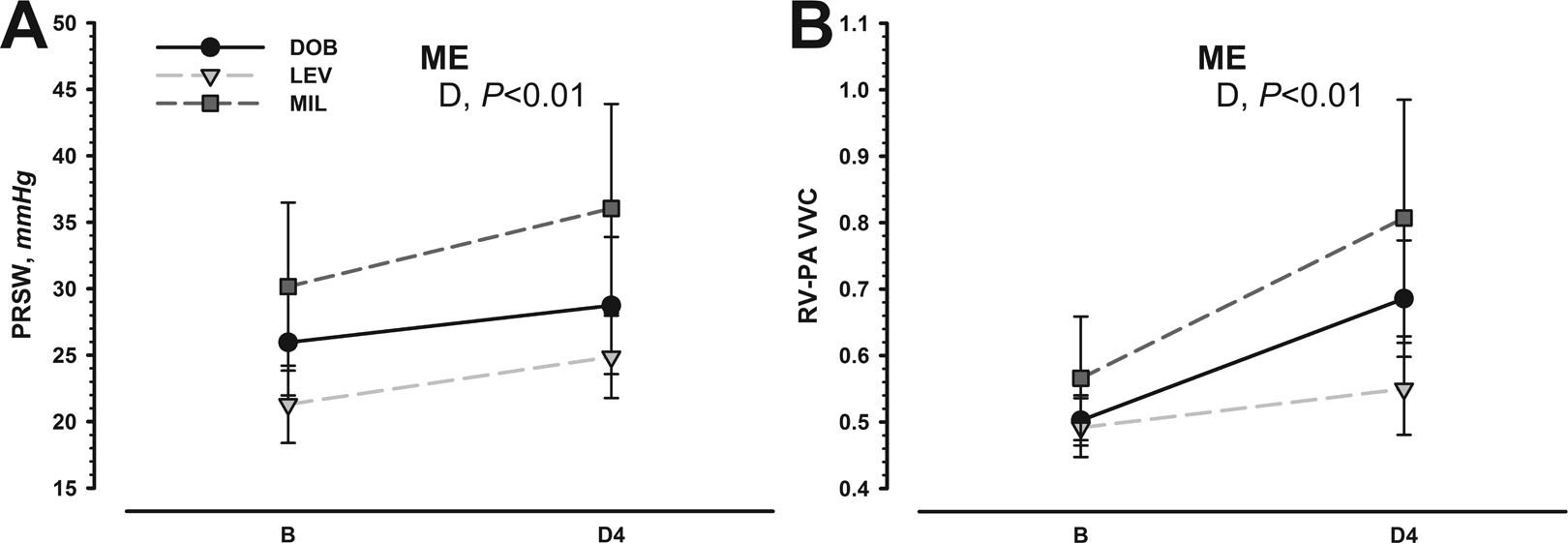
Preload recruitable stroke work (PRSW; A) and right ventricular pulmonary artery ventricular–vascular coupling (RV-PA VVC; B) at baseline (B) and after the last dose (D4) of dobutamine (DOB), levosimendan (LEV), and Milrinone (MIL). Main effects (ME) of drug (D) are given. No differences were observed for the different groups, n = 7 each group.
Based on the hemodynamic advantages of LEV, compared to MIL and DOB (it was the only inodilator able to significantly increase CI, decrease Eai, and improve compliance and diastolic function), in a separate group of MCT-injected rats, we tested combined therapy with pulmonary arterial vasodilator sildenafil. Representative PA pressure tracings and RV PV loops are depicted in Figure 6. This group of animals was sicker as assessed by lower CI and higher PA pressures. Despite relatively unchanged PA pressures (Figure 6A) upon LEV infusion, we observed a marked increase in CI (Figure 6C), which accounted for decreased PVRI (Table 3) and decreased PA Eai (Figure 6C). As described for the first animal group, LEV enhanced relaxation and improved compliance as assessed by shorter τexp and downward-shifted the EDPVR, respectively, but contrastingly, in this sicker group, we observed a noticeable increase in contractility judged by PRSW and Eesi elevation (Table 3 and Figure 6B). The Eesi elevation and less steep PA Eai together determined improved RV VVC (Figure 6C). Adding sildenafil to LEV (LEV+SIL) decreased PA pressures (Figure 6A) and further decreased PVRI (Table 3) without lowering CI. Still, overall contractile performance was decreased in LEV+SIL compared to LEV alone (Table 3 and Figure 6B). The fall in Eesi therefore jeopardized RV VVC (Figure 6C).
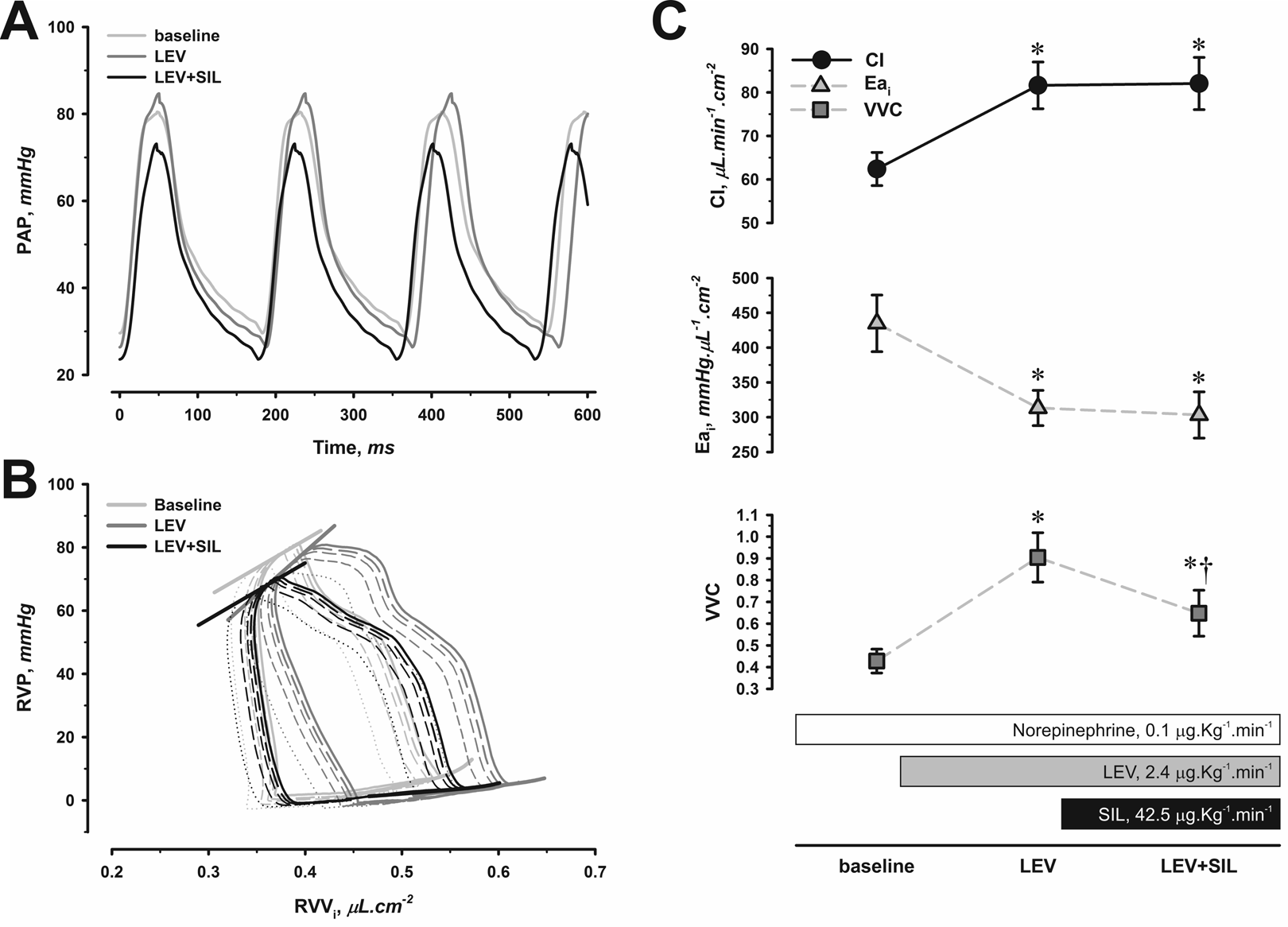
Representative pulmonary artery pressure (PAP; A) and right ventricular pressure (RVP)-indexed volume (RVVi) tracings (B) and changes in cardiac index (CI), indexed effective arterial pulmonary artery elastance (Eai), and RV ventricular–vascular coupling (VVC) from baseline to levosimendan (LEV) and then joint levosimendan and sildenafil (LEV+SIL) infusion (C). Detailed analysis of RV Loops can be found in Table 3. *P < .05 versus baseline, †P < .05 versus LEV, n = 8.
Hemodynamics With Combined Levosimendan and Sildenafil Infusions.a

Abbreviations: αi, scaling factor for exponential fitting of the end-diastolic pressure-volume relationship; βi, chamber stiffness constant for indexed volumes; dP/dtmax, peak rate of pressure rise; EDP, end-diastolic pressure; EDVi, indexed end-diastolic volume; Eesi, end-systolic elastance for indexed volumes; EF, ejection fraction; HR, heart rate; LEV, levosimendan; PRSW, preload recruitable stroke work; PVRI, pulmonary vascular resistance index; SIL, sildenafil; SVRI, systemic vascular resistance index; V0i, volume axis intercept of the end-systolic pressure–volume relationship for indexed volumes; τexp, time constant of isovolumetric relaxation by monoexponential fitting.
aHemodynamic features of monocrotaline (MCT)-injected pulmonary hypertensive rats at baseline and after infusion of 2.4 μg·kg−1·min−1 LEV and then combined infusion of LEV and 42.5 μg·kg−1·min−1 SIL (LEV+SIL).
b P < .05 versus baseline.
c P < .05 versus LEV, n = 8.
Discussion
To our knowledge, this is the first study to compare the acute effects of inodilators LEV, DOB, and MIL head-to-head in chronic experimental PH. We demonstrate that in the studied dose ranges, all inodilators increased RV EF, SWI, PRSW, and PA-RV coupling in MCT-induced PH without jeopardizing systemic perfusion pressure, but only LEV was able to increase CI and decrease PA Eai. Moreover, although all inodilators similarly enhanced RV relaxation, only LEV improved end-diastolic elastance and downward-shifted the EDPVR compared with DOB and MIL. Adding sildenafil to LEV further decreased PVRI.
Equipotent dose ranges of drugs were chosen based on previous literature reports. 6 –10 We did not administer an initial loading dose of MIL and LEV to avoid sudden hemodynamic changes and enable a farer dose–response in comparison with DOB, but this demanded correction of doses to higher values. Overall, all inodilators increased RV EF, dP/dtmax, SWI, and PRSW denoting positive inotropy, and all improved VVC as well. Enhanced relaxation was also a common effect. Within the dose ranges, selected no significant systemic hypotension was observed. Nevertheless, important differences were observed between the drugs. The β-agonist DOB elicited more tachycardia and increased systolic PA pressure while impairing diastolic compliance, effects that may be potentially hazardous in patients with PH. Although there was a trend for tachycardia with the PDE3i MIL, this was not significant and both PA pressures and diastolic compliance were unchanged. Levosimendan distinctly raised CI, while it decreased PA Eai compared with other inodilators. Indeed, a trend for reduced PA pressures was only observed for LEV. Most importantly, LEV improved RV diastolic compliance, which may provide leverage for using preload reserve in PH. This distinctive and newly described feature may be highly relevant, given the recent evidence that impaired RV compliance substantially contributes to worse prognosis in PH. 11 Ca2+-sensitizer LEV binds to the N-terminal domain of troponin C (Tn C) with an affinity proportional to Ca2+ concentration. At high Ca2+ concentrations, it stabilizes the Ca2+–Tn C complex, inhibiting the effects of troponin I and thus effectively enhances and prolongs actin–myosin cross-bridge association without changing cross-bridge cycling. Due to this mechanism of action, and contrary to the effects of conventional inotropes such as β-adrenergic agonists or PDE3i, LEV increases contractility without significantly raising intracellular Ca2+, cyclic adenosine monophosphate (cAMP) concentrations, or myocardial O2 consumption. Because at low Ca2+ levels it does not alter cross-bridge dynamics, LEV does not impair relaxation and actually improves left ventricular (LV) diastolic function. 12 It also dilates systemic and coronary arteries by opening ATP-dependent K+ channels. 13 Although it may not vasodilate the healthy lung circulation, 14 it dilates lung vessels in PH. 15 Indeed, chronic oral LEV administration was shown to prevent lung vascular remodeling and RV hypertrophy in MCT-induced PH, 16 and acute beneficial effects have also been reported. 17 Besides its classical effects, LEV has anti-inflammatory and antiapoptotic actions 18 and it downregulates nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB) transcription factor and prevents deleterious inflammatory nitric oxide (NO) production. 19 We have previously described that even short-term acute infusions of the dual endothelin 1 receptor antagonist tezosentan have important effects on inflammatory NO production, 20 suggesting that similar mechanisms may partly underlie LEV’s effects.
The inability of both DOB and MIL to significantly decrease PA Eai and significantly increase CI may seem surprising. However, we must recall that the inodilator designation derives mainly from their effects in the LV and systemic circulation. Also, hemodynamic effects of inodilators in PH due to left heart or chronic lung disease can be markedly different from those in PAH, given the distinctive underlying pathophysiological features. 21 Indeed, patients with PAH challenged with Trendelenburg positioning and DOB infusion show marked increases in PA pressures with only slight increases in cardiac output mainly attributable to increased heart rate and not stroke volume, 22 which led to the proposal of DOB stress as a provocative test to assess exercise capacity and impaired RV functional reserve in PAH. 23 Our results lend further support to the view that DOB may not be the best choice for acute management of patients with PAH. As for MIL, previous studies have mostly assessed its effects in combination with lung arteries vasodilators and in the setting of acute PH or chronic PH due to left heart disease. 24,25 Of note, in a canine model of MCT-induced PH, Chen et al also found no significant reduction in PA pressures with MIL, supporting our findings. 26 Our results suggest that enhanced RV contractility with both DOB and MIL offsets their lung vasodilating actions, preventing the decrease in PA pressure, whereas stronger lung arteries vasodilation by LEV clearly reduced PA Eai and PVRI. Nevertheless, we must emphasize that we evaluated hemodynamic effects of monotherapy. The addition of lung artery vasodilators such as inhaled NO or sildenafil may change the response to both DOB and MIL, allowing additional increases in CI and improving hemodynamic response. 24 Indeed, combined therapy is the mainstay of therapy in severe PAH. 21 We therefore assessed combined actions of an inodilator with a PA vasodilator. Because of doubts concerning the effect of inhaled NO in the MCT model, 27 and the existence of a single report with the dual endothelin-1 antagonist tezosentan (from our group), 20 we selected sildenafil, a short-acting phosphodiesterase type 5 inhibitor that has been extensively used in MCT-induced PH even in acute infusion 28 as pulmonary arterial vasodilator. Given the stronger effects of LEV compared to DOB and MIL, we assayed combined therapy by adding a sildenafil infusion to LEV. Interestingly, we found no additional benefits, besides a reduction in PA pressure and a further decrease in PVRI. This could be due to the fact that this group of animals had severe end-stage PAH with no RV inotropic reserve. Indeed, in a small sample of patients with group III PH and RV failure already on DOB, the lack of response to sildenafil was related to low inotropic reserve. 29 On the other hand, it could also be due to the fact that sildenafil is mainly a pulmonary arterial vasodilator devoid of RV myocardial actions as recognized in an experimental comparison with PDE3i MIL. 30 Importantly, adding sildenafil to LEV did not significantly decrease SVRI.
Regarding diastolic performance, previous works in lipopolysaccharide-treated rabbits also revealed an enhanced ability of LEV to improve diastolic function when compared to either DOB or MIL. 10 Acute changes in Tn I phosphorylation may partly mediate beneficial effects in diastolic function. 31 We have recently demonstrated that diastolic dysfunction rather than systolic performance heralds RV failure in MCT-induced PH, 32 and therefore, the beneficial effects of LEV on diastolic function compared to DOB and MIL may be highly relevant. Our experimental results support the conclusions of the recent meta-analysis evaluating the role of inodilators on mortality after adult cardiac surgery that neither MIL nor DOB seems to improve survival, whereas LEV does. 5 Current reports of clinical use of LEV in patients with PAH are scarce. Cavusoglu et al reported clinical deterioration in 2 cases, 33 while Martyniuk et al conversely reported clinical improvement in 9 patients. 34
Some limitations must be pointed out to our work. We intentionally evaluated only surviving animals with advanced stage PH and impending RV failure because this, and not compensated heart failure, was the scenario that best translated to the clinical setting under scrutiny. However, this raises the concern of survival bias. Nevertheless, we must underscore that randomization took place immediately before the intervention, therefore minimizing survival bias. Still, results cannot be extrapolated to earlier stages of the disease. We did not feel necessary to collect data from a vehicle-infused MCT-injected group because our hemodynamic preparations are usually stable for hours and also because each therapy group served as its own control, as the dose–response was compared with baseline. Ideally, we should have tested all drugs in a single set of animals with a crossover design to minimize the influence of random animal factors. Indeed, we did find heterogeneity within groups regarding disease severity. Nevertheless, a crossover design would have led to unusually long experiments and increased risk of animal loss or experimental preparation deterioration. Moreover, crossover effects from the previous drug could confound results. Indeed, although current evidence suggests that LEV is not readily metabolized to any circulating metabolite in rats, 35 it has long-acting metabolites in humans, OR-1855 and OR-1896. The MCT-induced PH is mainly a model of PAH, and therefore, our results are not generalizable to models in which PH is secondary to left heart or lung disease. Furthermore, our findings would be more robust if validation had been carried out in another PAH model such as the hypoxia + vascular endothelial growth factor 1 antagonist Sugen 5416 and in both genders. It is recognized that female rats fare better in MCT-induced PH, and no single model can entirely reproduce the pathophysiological features of PAH. Therefore, particularly in chronic therapy assessment, results should be validated in separate animal models and in both genders. 36 Because our aim was to conceptually compare acute cardiovascular effects of inodilators in severe PAH, we restricted our observations to a single model and to the male gender. Moreover, repeating all observations in other models and in females would prohibitively raise the number of animals and experimental groups. Still, this warrants a word of caution regarding generalization to female gender or other animal models of PH. Evaluation of LV performance was restricted to systemic arterial pressures. Systemic perfusion pressure was well preserved in all animals throughout the experimental protocol, controlling for 1 major confounder of RV performance. Nevertheless, subtle changes in LV performance and compliance were not assessed, and the backward effects on lung circulation were not accounted for. We only acquired baseline reference data for the group of control animals. Although it would have been informative, it was not the aim of the work to assess hemodynamic responses to inodilators in the healthy heart which have been previously reported. 37
Our results suggest beneficial acute systolic and diastolic functional effects of LEV in experimental chronic PH and RV afterload compared with DOB and MIL. They also suggest that LEV improves RV VVC and CI without further improvement with the addition of sildenafil, although this combination further decreased PVRI. Overall, they support further clinical trials with LEV in patients with PH, particularly in class 1 PH, in the perioperative and critical care setting.
Footnotes
Author Contributions
M. Tavares-Silva and M. Alaa have contributed equally to this article. M. Tavares-Silva, M. Alaa, and S. Leite contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. J. Oliveira-Pinto contributed to design, contributed to acquisition, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. L. Lopes contributed to acquisition, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. A. Leite-Moreira contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. A. Lourenço contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Portuguese Foundation for Science and Technology to the Cardiovascular Research Centre of the Faculty of Medicine of Porto (EXCL/BIM-MEC/0055/2012, PEST-C/SAU/UI 0051/2014 and PTDC/DTP-PIC/4104/2014) and by Portuguese Foundation for Science and Technology Grant SFRH/BD/110404/2015 (to S. Leite).
