Abstract
Background:
Nucleolin has multiple functions within cell survival and proliferation pathways. Our previous studies have revealed that nucleolin can significantly reduce myocardial ischemia-reperfusion injury by promoting myocardial angiogenesis and reducing myocardial apoptosis. In this study, we attempted to determine the role of nucleolin in myocardial infarction (MI) injury recovery and the underlying mechanism.
Methods:
Male BALB/c mice aged 6–8 weeks were used to set up MI models by ligating the left anterior descending coronary artery. Nucleolin expression in the heart was downregulated by intramyocardial injection of a lentiviral vector expressing nucleolin-specific small interfering RNA. Macrophage infiltration and polarization were measured by real-time polymerase chain reaction, flow cytometry, and immunofluorescence. Cytokines were detected by enzyme-linked immunosorbent assay.
Results:
Nucleolin expression in myocardium after MI induction decreased a lot at early phase and elevated at late phase. Nucleolin knockdown impaired heart systolic and diastolic functions and decreased the survival rate after MI. Macrophage infiltration increased in the myocardium after MI. Most macrophages belonged to the M1 phenotype at early phase (2 days) and the M2 phenotype increased greatly at late phase after MI. Nucleolin knockdown in the myocardium led to a decrease in M2 macrophage polarization with no effect on macrophage infiltration after MI. Furthermore, Notch3 and STAT6, key regulators of M2 macrophage polarization, were upregulated by nucleolin in RAW 264.7 macrophages.
Conclusions:
Lack of nucleolin impaired heart function during recovery after MI by reducing M2 macrophage polarization. This finding probably points to a new therapeutic option for ischemic heart disease.
Introduction
Myocardial infarction (MI) contributes to global morbidity and mortality rates 1,2 a lot. Various complications develop in the myocardium owing to MI, e.g. apoptosis, metabolic changes, infiltration by multiple types of inflammatory cells, and an immune response, 3 leading to a severe adverse outcome. Myocardium recovery after MI depends on an orderly immune response, whereas macrophage infiltration plays a crucial role in the inflammatory process and proliferative process after MI. 4 After MI, excessive recruitment and sustained activation of immune cells, including macrophages, are detrimental. 5 On the contrary, moderate and well-regulated recruitment of macrophages could promote inflammation resolution, phagocytosis, angiogenesis, and infarct healing after MI. Generally, macrophages show plasticity functions under certain pathological conditions of MI. 6 Two main subpopulations of macrophages with distinct functions include classically activated macrophages (M1, proinflammatory phenotype) and alternatively activated macrophages (M2, anti-inflammatory phenotype). The therapeutic aim is usually to inhibit the proinflammatory actions of macrophages thereby avoiding an excessive inflammatory response after MI. Therefore, elucidation of the shift mechanisms of macrophage activation and polarization is important for the regulation of immune response, for tissue homeostasis, and for prevention and healing of pathological myocardial remodeling after MI.
Nucleolin (C23, NCL) is a 77-kDa phosphoprotein widely distributed in mammals, which was discovered by Busch and colleagues in 1973. 7 Decades of studies have shown that nucleolin has versatile functions within cell survival and proliferation pathways, including ribosome biogenesis, cell cycle and apoptosis, transcriptional regulation, and DNA replication and repair. 8 We have found in previous studies that nucleolin has cardioprotective effects and significantly reduce myocardial ischemia-reperfusion injury. So far, it is not clear the functions nucleolin performed in myocardial repair and myocardial remodeling after MI and how nucleolin works. Zhang et al 9 have found that the process of RAW 264.7 macrophage polarization induced by lipopolysaccharide (LPS) is accompanied by nucleolin downregulation. In the process of monocyte/macrophage differentiation, Jerke et al 10 have found that nucleolin bind to STAT1 specifically to regulate the expression of CD36, a macrophage scavenger receptor. These findings suggest that nucleolin may play an important regulatory role in macrophage polarization in myocardial repair. It is of great academic value and potential applications to investigate the involvement of nucleolin in macrophage polarization and the underlying mechanism.
Materials and Methods
Animal
Male BALB/c mice aged 6-8 weeks, were purchased from Central South University Experimental Animals Center. Animal handling and experiments were performed in accordance with the Guidelines of Animal Experimentation, Medical Ethics Committee of Xiangya Hospital, Central South University. Throughout the experiment, mice were maintained in suitable conditions of humidity, ambient temperature (23-24°C), and 12-hour light/dark cycle; mice were housed separately and were given free access to food and water.
The Animal Model of Acute MI (AMI)
The AMI model was established through ligation of the left anterior descending coronary artery. First, the mice were anesthetized with isoflurane and fixed on the operating table. Second, the left thoracic precordium was prepared by preoperative hair removal, then the skin was sterilized. Third, the heart was exposed through the third and fourth intercostal space on the left side of the sternum in a sterile environment. Fourth, left descending branch of the left coronary artery was ligated, followed by closure of the chest. Mouse ECG was performed before and after the operation. The elevation of the ST segment was noted after the ligation. At different time point after MI, the infarction area was collected for following experiments.
Hemodynamic Measurement
The mice were anesthetized with isoflurane and fixed on the operating table. Then the heart function +dP/dt and −dP/dt were measured by Powerlab system (AD Instruments, CA, USA) according to previously research. 11
Immunofluorescence Staining
The heart of a mouse after ligation was exposed, and the infarcted area was excised after perfusion and was fixed in 4% paraformaldehyde, then embedded in paraffin. The expression of F4/80 (1:100, Abcam) in the myocardium was determined by immunofluorescence staining as described previously. 12
Flow-Cytometric Analysis
Myocardial macrophages were isolated as described previously. 13 Freshly isolated macrophages were resuspended in FACS buffer, and then subjected to staining with the corresponding antibody for 20 to 30 min at 4°C. The antibodies used were anti-F4/80-FITC (Biolegend), anti-CD11c-APC (Biolegend), anti-CD206-PE (Biolegend). The cells were washed with PBS and resuspended in FACS buffer again. About 10000 cells were detected by flow cytometry and then analyzed in the Flowjo software.
Quantitative Reverse-Transcription Polymerase Chain Reaction (q-RT PCR)
Total-mRNAs of infarcted area in myocardium were extracted with the TRIzol Reagent (Invitrogen) according to the manufacturer’s instructions, followed by cDNA synthesis using the PrimeScript™ RT Reagent Kit with gDNA Eraser (TAKARA) according to the manufacturer’s instructions. q-RT PCR was carried out on an ABI 7500 real-time PCR System (Life Technologies Corp., Carlsbad, CA) with SYBR Premix Ex Taq™ (TakaRa). In the reaction, the following primers were used: GAPDH forward: 5′-GGTGAAGGTCGGTGTGAACG-3′ and reverse: 5′-CTCGCTCCTGGAAGATGGTG-3′; Arg-1 forward: 5′-AAGCCTGGTCTGCTGGAAAAA-3′ and reverse: 5′-CTGGTTGTCAGGGGAGTGTT-3′; IL-10 forward: 5′-ACCAGCTGGACAACATACTGC-3′ and reverse: 5′-TCACTCTTCACCTGCTCCACT-3′; NCL forward: 5′-GCCATGGAGATCAGATTAGTCA-3′ and reverse: 5′-TCTGCATCAGCTTCAGACTTAA-3′ .
Nucleolin Knockdown of Myocardium in Mice
The knockdown handing was performed as soon as the operation was finished, then a lentiviral vector containing the nucleolin RNA interference fragment (siNuc) or lentiviral vector containing control RNA interference fragment (siCon) was injected into the infarcted zone of myocardium. Healthy male BALB/c mice, aged 6-8 weeks, were randomized into 4 groups: (1) the sham + siCon group: the sham group mice were dealt with intramyocardial injection of siCon; (2) the sham + siNuc group: the sham group mice were challenged with intramyocardial injection of siNuc; (3) the MI + siCon group: hearts of mice were challenged with intramyocardial injection of siCon after MI surgery; (4) the MI + siNuc group: hearts of mice were subjected to intramyocardial injection of siNuc after MI surgery. 4 weeks after MI induction and these siCon or siNuc injection, we evaluated survival and the heart functions. Then heart tissues and serum of murine models were collected after measurements.
Cell Transfection
RAW264.7 cells were transfected with pcDNA3.1-Nuc (experimental group) or pcDNA3.1 (vector control) by Lipofectamine 2000™ (Invitrogen, USA) according to the manufacturer’s instructions and our previous research. Nucleolin ablation in RAW264.7 cells were conducted through transfecting with siNuc (experimental group) or siCon (negative control). siNuc (experimental group) transfection induced nucleolin ablation and siCon transfection were conducted in RAW264.7 cells.
Western Blot
Protein was extracted from the cells and tissues of infarcted area in myocardium as follows: the cell or tissue was lysed using RIPA buffer, the homogenate or cell lysate was centrifuged at 12000 g, at 4°C for 10 minutes, and the supernatant was collected. The protein concentration was checked by the BCA assay kit. The extracted protein was loaded onto a gel for SDS polyacrylamide gel electrophoresis followed by electrotransfer onto polyvinylidene difluoride (PVDF) membranes. The protein band on the PVDF membrane react to the primary antibody. After incubated with the corresponding second antibody, the band were detected by electrochemiluminescence (ECL) luminescence. Primary antibodies used were anti-nucleolin antibody (1:3000, Sigma), anti-GAPDH antibody (1:1000, Cell Signaling Technology), anti-Notch3 antibody (1:1000, Abcam), anti-STAT6 antibody (1:1000, Abcam).
Enzyme-Linked Immunosorbent Assay (ELISA)
The murine serum was collected from the murine model. The blood was set at room temperature for 1 hour and then centrifuged at 1000 g for 15 min to collect serum. Then TGFβ and IL-10 (all purchased from Cusabio Biotech) were detected by ELISA kits according to the manufacturer’s instruments.
Statistics
All these results were statistically evaluated in GraphPad Prism 6.0 and SPSS software. These data were presented as the mean ± SEM. Unpaired Student’s t test was used to determine the significance of differences between the 2 groups, whereas the differences among 3 or more groups were evaluated by one-way analysis of variance (ANOVA) and the Student–Newman–Keuls test. The survival rate between different groups was compared by Kaplan-Meier analysis.
Results
Nucleolin Expression in Mouse Myocardium after MI
Our previous studies have shown that nucleolin has cardioprotective effects on myocardial ischemia-reperfusion injury. 14 However, whether nucleolin plays an important role during the recovery from MI is still unknown. Moreover, we measured nucleolin mRNA and protein expression levels in the infarcted zone of myocardium at different time points after MI induction. As shown in Figure 1A, on the first day after MI induction, nucleolin protein expression decreased and then increased gradually from the third day and reached a peak on the seventh day. The mRNA expression of nucleolin followed a similar pattern (Figure 1B). These in vivo results indicating that nucleolin expression changes over time.

Nucleolin expression in mice myocardium after MI. The infarcted area in myocardium was collected on days 0, 1, 3, 7, 14 and 28 after MI induction in mice. (A) The protein expression levels of nucleolin in myocardium after MI in mice. (B) Relative mRNA expression levels of nucleolin in myocardium after MI in mice. Total RNA was extracted and detected by quantitative real-time PCR. *P < .05 versus day 0, n = 5.
Nucleolin Knockdown Impairs Heart Function During the Recovery From MI
To further demonstrate the involvement of nucleolin in the recovery from MI, a murine model of myocardial nucleolin knockdown was established by intramyocardial injection of a lentiviral vector (siNuc or siCon). As depicted in Figure 2A, nucleolin expression in the myocardium was inhibited by siNuc 1 week after its intramyocardial injection. In addition, nucleolin upregulation induced by MI was attenuated by siNuc at 1 week after MI induction. A Powerlab system was applied to evaluate heart function 4 weeks after MI induction. As shown in Figure 2B, the maximal slope of systolic pressure decrement (+dP/dt) decreased markedly, and the nucleolin knockdown (siNuc) enhanced the decrease of +dP/dt. Similarly, the maximal slope of the diastolic pressure decrement (−dP/dt) decreased markedly, and the nucleolin knockdown (siNuc) enhanced the decrease of −dP/dt. Furthermore, the survival rate after MI among the mice was determined, and we found that the survival rate in the nucleolin knockdown group significantly reduced the survival rate in the mice 4 weeks after MI (Figure 2C). These results indicated that lack of nucleolin impaired heart function during recovery after MI nucleolin and decreased the survival rate of mice after MI. However, the underlying mechanism was still unclear.
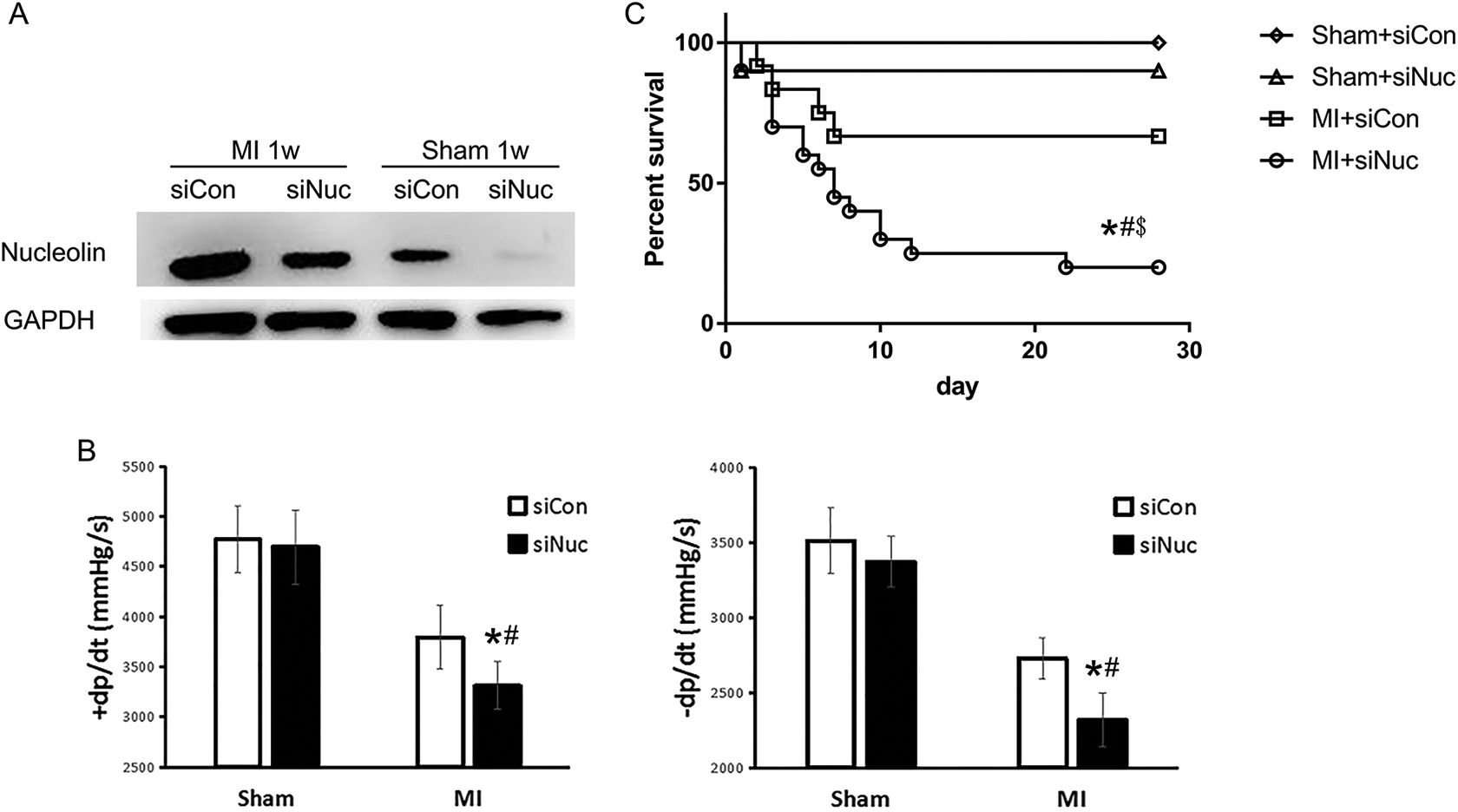
Nucleolin knockdown impaired cardiac function and decreased the survival rates after MI in mice. (A) The expression of nucleolin in the myocardium of mice after intramyocardial injection of siNuc or siCon. (B) The maximal slope of systolic pressure decrement (+dP/dt) and the maximal slope of the diastolic pressure decrement (−dP/dt) were observed in mice after intramyocardial injection of siNuc or siCon for 4 weeks. *P < .05, versus sham+siCon, n = 8; # P < .05, versus MI+siCon, n = 8. (C) The impact of low nucleolin expression on the survival rate of mice at 28 days after MI. Sham+siCon, n = 8; Sham+siNuc, n = 10; MI+siCon, n = 12; MI+siNuc, n = 20; *P < .05 versus MI+siCon, # P < .05 versus Sham+siCon, $ P < .05 versus Sham+siNuc.
Effect of the Nucleolin Knockdown on Macrophage Polarization in the Myocardium after MI
Researchers have found that macrophage infiltration is important for the inflammatory process and proliferative process after MI. Similarly, we found that macrophage infiltration increased significantly after MI induction (Figure 3A) according to immunofluorescence analyses. Flow cytometry showed that most macrophages in the myocardium belonged to the M1 phenotype (F4/80+CD11c+) at 2 days after MI induction. However, 5 days after MI induction, most of the macrophages in the myocardium were M2 phenotype (F4/80+CD206+; Figure 3B).

Macrophage infiltration and polarization in the myocardium after MI. (A) Immunofluorescence staining was performed to analyze the expression of F4/80 (red), a macrophage surface marker, in the myocardium at 5 days after MI. *P < .05 versus Sham, n = 5. (B) The percentages of M1 (F4/80+CD11c+) and M2 (F4/80+CD206+) macrophages among the macrophages isolated from the myocardium 2 days and 5 days after MI have been confirmed via flow cytometry. *P < .05 versus Sham in M1 (F4/80+CD11c+), n = 5; # P < .05 versus Sham in M2 (F4/80+CD206+), n = 5.
Then, the effects of the nucleolin knockdown on macrophage infiltration and polarization were studied in the myocardium after MI. As presented in Figure 4A, during the recovery of the myocardium after MI induction, macrophage infiltration increased and the nucleolin knockdown had no significant effect on macrophage infiltration. Then, the percentage of M2 macrophages in the myocardium was analyzed by flow cytometry. As illustrated in Figure 4B, 5 days after MI induction in the mice, the percentage of M2 macrophages in the myocardium in the siCon group increased. By contrast, the percentage of M2 macrophages in the myocardium in the siNuc group decreased markedly as compared to the siCon group. Furthermore, TGFβ and IL-10 in serum, markers of M2 macrophages, were measured by ELISA. We found that serum levels of TGFβ and IL-10 increased in siCon mice after 5 days of MI compared to Sham group and decreased in MI mice between the siNuc group and the siCon group (Figure 4C). These results suggested that the nucleolin knockdown inhibited the M2 macrophage polarization with no effect on macrophage infiltration during the recovery of myocardium after MI.

Effects of the nucleolin knockdown on the polarization of myocardial macrophages after 5 days of MI in mice. (A) Analysis of the percentage of macrophages (F4/80+) 5d after MI in the myocardium by flow cytometry. *P < .05 versus Sham+siCon, n = 5. (B) Analysis of the percentage of M2 macrophages (F4/80+CD206+) in myocardium by flow cytometry. *P < .05 versus Sham+siCon, n = 5; # P < .05, versus MI+siCon group, n = 5. (C) The levels of serum TGF-β and IL-10 after 5 days of MI in mice. *P < .05, versus Sham+siCon, n = 5; # P < .05, versus MI+siCon group, n = 5.
Nucleolin Ablation Inhibits M2 Macrophage Polarization in RAW 264.7 Cells
To elucidate the underlying mechanism by which nucleolin regulates macrophage polarization during recovery from MI, M1 and M2 macrophages were isolated from the murine myocardium 5 days after MI, and then nucleolin expression was analyzed. The results revealed that nucleolin expression (Figure 5A) in M2-type macrophages increased markedly as compared with M1-type macrophages and RAW 264.7 macrophage cell line. These results implied a close relation between nucleolin expression levels and macrophage polarization in vitro.
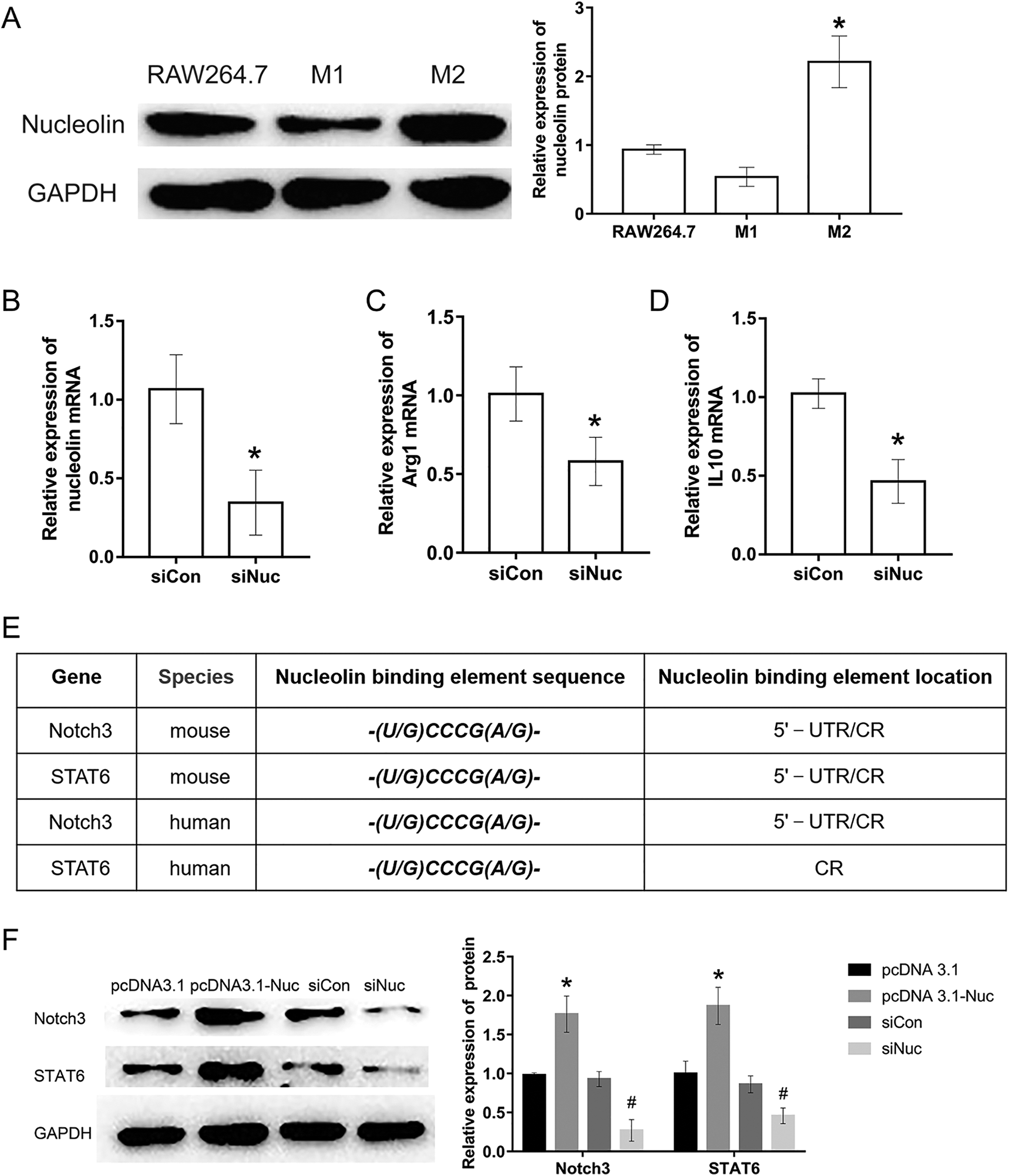
Effects of the nucleolin on M2 macrophage polarization in RAW264.7 cells. (A) Nucleolin expression levels in RAW264.7 cell, M1 and M2 macrophages isolated from mouse myocardium 5 days after MI. *P < .05, versus RAW264.7, n = 3. (B-D) NCL, Arg-1 and IL-10 mRNA levels in RAW264.7 macrophages. RAW264.7 macrophages were transfected with nucleolin RNA interference vector for 48 h. Cells were treated with IL-4 (20 ng/mL) for 24 h in C and D. *P < .05 versus siCon, n = 5. (E) Bioinformatics analysis showed that multiple nucleolin binding elements were located in the 5′-UTR and CR of Notch3 and STAT6 mRNAs both in human and mouse. (F) Nucleolin regulated the expression of Notch3 and STAT6. Western blot was used to detect the expression of Notch3 and STAT6 in RAW264.7 macrophages transfected with nucleolin full-length expression vector (pcDNA3.1-Nuc) or siNuc for 48 h. *P < .05 versus pcDNA3.1 group, n = 3; # P < .05 versus siCon group, n = 3.
To further examine the participation of nucleolin in macrophage polarization in vitro, the nucleolin siRNA vector (siNuc) was used. We found that siNuc significantly inhibited the mRNA level of nucleolin (Figure 5B) in RAW 264.7 macrophages after transfection for 48 hours. Then, the impact of nucleolin ablation on M2 macrophage polarization of RAW 264.7 cells was studied. As depicted in Figure 5C-D, the mRNA levels of Arg1 and IL-10, which are known to be markers of M2 macrophage polarization, 15 increased in control RAW 264.7 macrophages transfected with siCon for 48 hours and treated with IL-4 (20 ng/mL) for 24 h. In contrast, nucleolin ablation inhibited the upregulation of Arg1 and IL-10 induced by IL-4 (20 ng/mL). These results suggested that nucleolin could modulate M2 macrophage polarization.
Notch signaling molecules and STAT6 performed key functions in M2 macrophage polarization. 16,17 To further elucidate the molecular mechanism of nucleolin in M2 macrophage polarization regulation, we carried on bioinformatics analysis, and found that there are multiple nucleolin binding elements (-(U/G)CCCG(A/G)-) in the 5′-noncoding region (5′-UTR) and coding region (CR) of Notch3 and STAT6 mRNAs (Figure 5E). These results suggested that nucleolin may contribute to macrophage polarization via binding to Notch3 and STAT6 mRNAs and regulating the expression of Notch3 and STAT6. Then, Notch3 and STAT6 expression were measured in vitro when nucleolin expression was changed by the overexpression plasmid or by siRNA in RAW 264.7 macrophage cells. As shown in Figure 5F, nucleolin overexpression upregulated Notch3 and STAT6, whereas nucleolin ablation down regulated Notch3 and STAT6. These data suggested that nucleolin promotes M2 macrophage polarization by upregulating Notch3 and/or STAT6.
Discussion
Our previous studies have revealed that nucleolin protect the myocardium against myocardial ischemia-reperfusion injury. It is also known that nucleolin attenuated apoptosis induced by oxidative stress in cardiomyocytes by regulating Hsp70 degradation. 18 Monte has identified (by proteomics) the regulatory role nucleolin plays in the growth and cellular plasticity of murine and zebrafish cardiomyocytes, 19 in agreement with the finding that proliferation inhibition and apoptosis promotion in C2C12 cells were accompanied with nucleolin downregulation. 20 In human heart disease, nucleolin was also strongly involved in cell proliferation and apoptosis. It has been reported that the prevalence of the anti-nucleolin antibody was 2.0% among healthy people but was 17.1% among heart transplant recipients. These results indicate that apoptosis is elevated in human umbilical vein endothelial cells, and endothelial cell proliferation is inhibited in both recipients and murine allogeneic transplantation models. 21,22 Additionally, nucleolin expression was higher in ischemic cardiomyopathy or dilated cardiomyopathy and also correlated with heart function and left ventricular parameters in ischemic cardiomyopathy. 23 Accordingly, nucleolin is a critical molecule involved in heart function and cell proliferation. In the present study, we found that nucleolin expression was down-regulated after MI, and then increased gradually to promote myocardial repair. Nucleolin knockdown by siRNA after MI impaired the protective effect of nucleolin in heart. These data suggested that nucleolin may promote the recovery of the myocardium after MI.
It has been discovered that M1 macrophage infiltrated the ischemic myocardium in the first 1–3 days after MI and played a variety of roles by facilitating inflammation, phagocytizing necrotic cells and tissues, degrading the extracellular matrix, and exerting other effects. 24 Nevertheless, M2 macrophages were mainly recruited to the infarct area 4–14 days after MI, leading to relief of inflammation, vascular endothelial growth factor (VEGF) secretion, angiogenesis promotion, and collagen formation, thereby contributing to the healing of the infarcted myocardium. 25,26 Studies have shown that after MI, macrophage polarization was steered by infarcted myocardium-derived dendritic cells toward M2 polarization, thereby improving myocardial remodeling and left ventricular systolic function recovery. 27 Recent research also indicated that human embryonic stem cell derived-cardiovascular progenitor cells activate STAT6 through paracrine action, leading to M2 macrophage polarization, which attenuated the deterioration of myocardial function and reduced the damage of myocardial scar formation. 28 It is also reported that adipose-derived stem cells mitigated cardiac fibrosis 4 weeks after MI in rats via a shift of the macrophage phenotype. 29 Macrophage polarization not only participates in myocardial remodeling and heart function but also is involved in myocardial inflammation. Macrophages were recruited to the infarcted zone in an infarcted myocardium, and then differentiated into different polarization types of macrophages, including the proinflammatory macrophage M1 polarization type and anti-inflammatory macrophage M2 polarization type. 30 Niacin promoted inflammation resolution in a timely manner in the infarcted heart by elevating M2 polarization. 31 Calpastatin overexpression in mice affected the infiltration of the infarcted zone by M2 macrophages and CD4+ T cells, thereby leading to impaired scar healing, elevating the probability of ventricular rupture and decreasing the survival rate of patients with MI. 32 When M1 macrophage-related molecule transcription factor interferon-regulatory factor 5 (IRF5) was downregulated, inflammation resolution and wound healing after MI were promoted and heart failure post-MI was attenuated. 33 Similarly, we found that macrophages had a proinflammatory M1 phenotype early after MI, and M2 macrophage numbers increased greatly later after MI. This result is consistent with the recovery process after MI.
Whether nucleolin improved heart function during the recovery from MI through modulating macrophage polarization is not very clear before. Researchers have found that nucleolin expression on the cell surface was elevated in M2 macrophages in non–small cell lung cancer. 34 We found that a nucleolin knockdown may inhibit the M2 polarization significantly in the late stage of recovery after MI. The infarcted myocardium has been hypothesized to induce elevation of M2 macrophage numbers in the late phase after MI. This phenomenon could explain the higher nucleolin expression level in the myocardium in the late phase of recovery, indicating that nucleolin was closely related to M2 macrophage polarization and myocardium recovery after MI. Additionally, our previous research has revealed that nucleolin could preserve macrophages from foam cell formation induced by oxidized low-density lipoprotein during atherosclerosis. Other researchers also have found that nucleolin could elevate granulocyte-macrophage colony-forming units in mobilized peripheral-blood–derived hematopoietic stem/progenitor cells. 35 Thus, nucleolin could regulate macrophage polarization in the myocardium after MI.
Recent research revealed that the Notch signal mediated by RBP-J could mediate the polarization of M2 macrophages by regulating SOCS3 expression. 12,16,34 Therefore, the Notch signal played a crucial role in the commitment to M1 or M2 polarization among macrophages, and a deficient Notch pathway may lead to M2-like tumor-associated macrophages (TAMs). 16 In addition, the JAK/STAT6 pathway has also been found to be a key signaling cascade in M2 polarization during atherosclerosis. 36 Researchers have found that nucleolin could bind to STAT1 to form a complex during monocyte or macrophage differentiation. 10 Bioinformatics analysis showed that there were multiple nucleolin binding elements in Notch3 and STAT6 mRNAs. It is possible that nucleolin contributed to macrophage polarization via binding to Notch3 and STAT6 mRNAs and regulating the expression of Notch3 and STAT6. Here, we demonstrated that RAW 264.7 cells transfected with pcDNA3.1-Nuc showed elevated expression of STAT6 and Notch3, which were key molecules in the regulation of the M2 macrophage polarization pathway. When nucleolin was downregulated in the RAW 264.7 cells transfected with siNuc, the expression of STAT6 and Notch3 decreased likewise.
Our previous researches have shown that H2O2 preconditioning and myocardial ischemic preconditioning can up-regulate the nucleolin expression which mediated an important myocardial protection. 37 Nucleolin expression is up-regulated, and plays a pro-angiogenic role during the recovery of heat-denatured dermis and its mechanism is probably dependent on production of VEGF. 38,39 Hydroxysafflor Yellow A increased angiogenesis and promoted nucleolin levels both in mRNA and protein in the infarcted myocardium of AMI mice. 11 At the same time, several researches focused on interfering nucleolin expression or function by pharmacological methods have been shown. An antitumor agent oridonin can target nucleolin to affect its role in mRNA stabilization activity, protein synthesis and cell cycle progression. 40 N6L (IPP-204106), which is in Phase I/IIa Study, can bind to the nucleolin and block its angiogenesis in mouse models of retinopathies. 41 Nucleolin has been identified as a novel target for treatment. 40,41 Our studies have shown that nucleolin could protect myocardium from myocardial ischemic injury, indicating that it is a promising pharmacological target for treatment.
Conclusion
In conclusion, our studies demonstrated that nucleolin overexpression promoted macrophages to polarize toward the M2 type both in vitro and in vivo, whereas nucleolin knockdown had an opposite impact on the macrophage polarization and without significant effect on macrophage infiltration. The underlying mechanism of nucleolin on macrophage polarization was upregulating STAT6 and Notch3 in the myocardium after infarction, thereby causing macrophages to polarize toward the M2 type. The latter promoted tissue repair and survival and inflammation dissipation after MI and constituted a new target for post-MI treatment.
Supplemental Material
Supplemental Material, sj-docx-1-cpt-10.1177_1074248421989570 - Nucleolin Improves Heart Function During Recovery From Myocardial Infarction by Modulating Macrophage Polarization
Supplemental Material, sj-docx-1-cpt-10.1177_1074248421989570 for Nucleolin Improves Heart Function During Recovery From Myocardial Infarction by Modulating Macrophage Polarization by Yuting Tang, Xiaofang Lin, Cheng Chen, Zhongyi Tong, Hui Sun, Yuanbin Li, Pengfei Liang and Bimei Jiang in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, sj-tif-1-cpt-10.1177_1074248421989570 - Nucleolin Improves Heart Function During Recovery From Myocardial Infarction by Modulating Macrophage Polarization
Supplemental Material, sj-tif-1-cpt-10.1177_1074248421989570 for Nucleolin Improves Heart Function During Recovery From Myocardial Infarction by Modulating Macrophage Polarization by Yuting Tang, Xiaofang Lin, Cheng Chen, Zhongyi Tong, Hui Sun, Yuanbin Li, Pengfei Liang and Bimei Jiang in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, sj-tif-2-cpt-10.1177_1074248421989570 - Nucleolin Improves Heart Function During Recovery From Myocardial Infarction by Modulating Macrophage Polarization
Supplemental Material, sj-tif-2-cpt-10.1177_1074248421989570 for Nucleolin Improves Heart Function During Recovery From Myocardial Infarction by Modulating Macrophage Polarization by Yuting Tang, Xiaofang Lin, Cheng Chen, Zhongyi Tong, Hui Sun, Yuanbin Li, Pengfei Liang and Bimei Jiang in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
Yuting Tang and Xiaofang Lin contributed equally to this study. Bimei Jiang conceived the project and experiments and provided funds. Yuting Tang and Xiaofang Lin performed the experiments, collected the data, and analyzed the data. Cheng Chen, Xiaofang Lin, Zhongyi Tong, Hui Sun, Yuanbin Li, and Pengfei Liang provided reagents, materials, analysis tools, and data analysis. Bimei Jiang, Yuting Tang, and Pengfei Liang contributed to writing the manuscript. All authors approved of the manuscript to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81770306, 81971820, 81974287), the Province Natural Science Foundation of Hunan (2018JJ2547), the Fundamental Research Funds of Central South University for postgraduate students (2018zzts832, 2020zzts228), and the Open Sharing Fund for the Large-scale Instruments and Equipments of Central South University (CSUZC201945).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
