Abstract
Antithrombotic drugs, which include antiplatelets and anticoagulants, are effective in prevention and treatment of many cardiovascular disorders such as acute coronary syndromes, stroke, and venous thromboembolism and are among the drugs most commonly prescribed worldwide. The advent of direct oral anticoagulants, which are safer alternatives to vitamin K antagonists and do not require laboratory monitoring, has revolutionized the treatment of nonvalvular atrial fibrillation and venous thromboembolism. The combination of oral anticoagulant and antiplatelet therapy is required in many conditions of great clinical impact such as the coexistence of atrial fibrillation and coronary artery disease, with indication to percutaneous coronary intervention. However, strategies that combine anticoagulant and antiplatelet therapies lead to a significant increase in bleeding rates and it is crucial to find the right combination in the single patient in order to optimize the ischemic and bleeding risk. The aim of this review is to explore the evidence and controversies regarding the optimal combination of anticoagulant and antiplatelet therapy through the consideration of past dogmas and new perspectives from recent clinical trials and to propose a tailored therapeutic approach, according to specific clinical scenarios and individual patient characteristics. In particular, we separately explored the clinical settings of stable and acute coronary syndromes and percutaneous revascularization in patients with atrial fibrillation.
Introduction
Antithrombotic drugs, which include antiplatelets and anticoagulants, are effective in prevention and treatment of many cardiovascular disorders such as acute coronary syndromes (ACSs), stroke, and venous thromboembolism (VTE) and are among the drugs most commonly prescribed worldwide.
Traditionally, the vitamin K antagonists (VKAs), notably warfarin, have been the cornerstone for the treatment of VTE and for the prophylaxis of stroke and systemic embolism in patients with atrial fibrillation (AF). The advent of direct oral anticoagulants (DOACs) has revolutionized the therapy of nonvalvular atrial fibrillation (NVAF) and the therapy of VTE. 1
On the other hand, to date, the optimal treatment of ischemic artery diseases is antiplatelet therapy: single antiplatelet therapy for secondary prevention of stable coronary artery disease (CAD) and dual antiplatelet therapy (DAPT), with aspirin and an inhibitor of the platelet P2Y12 receptor, for the treatment of patients with ACS and those undergoing PCI. 2
The combination of oral anticoagulant (OAC) and antiplatelet therapy is required in many conditions of great clinical impact. However, strategies that combine anticoagulant and antiplatelet therapies lead to a significant increase in bleeding rates. 3,4
For this reason, finding an optimum balance in antithrombotic regimen for each single patient with an indication for both treatments remains crucial to achieve a net clinical benefit.
The aim of this article is to explore the evidence and controversies regarding the optimal combination of anticoagulant and antiplatelet therapy through the consideration of past dogmas and new perspectives coming from recent clinical trials and to propose a tailored therapeutic approach, according to specific clinical scenarios and individual patient characteristics.
Evidence
Oral anticoagulant treatment is the gold standard for the prevention of thromboembolic events in AF. Indeed, international guidelines recommend that patients with CHA2DS2-VASc scores of 1 or more for men or 2 or more for women should be considered for OAC treatment, preferably DOACs. 5
Dual antiplatelet therapy is a highly effective treatment for the prevention of ischemic events across the whole spectrum of CAD. 2 The modern concept of DAPT dates back to 1998 when 2 pivotal studies the FANTASTIC (Full Anticoagulation Versus Aspirin and Ticlopidine) and the STARS (Stent Anti-thrombotic Regimen Study) trials, demonstrated that antiplatelet therapy after coronary stenting significantly reduced rates of bleeding and subacute stent occlusion compared with conventional anticoagulation. 6,7 Currently, DAPT is still the standard of care for patients undergoing PCI. 3 New-generation drug-eluting stents (DESs) are recommended in cases of high bleeding risk allowing a shorter duration of DAPT (1 or 6 months). 8
Even though both DAPT and OAC are antithrombotic treatments, each one has specific indications and cannot be used in place of the other. Indeed, after coronary artery stenting, DAPT is associated with a lower rate of cardiac events compared to OAC. Conversely, in NVAF patients, DOACs have been demonstrated to be superior to DAPT for the prevention of embolic events. 9,10
The coexistence of CAD and AF represents a challenging clinical scenario which requires evaluation of both bleeding and ischemic risk.
Standard Antithrombotic Therapy in Stable CAD
In stable CAD, DAPT is not indicated unless a PCI has been performed. The latest European Society of Cadiology guidelines on myocardial revascularization (Figure 1) suggest that following elective stenting in stable CAD, patients should be treated with DAPT consisting of aspirin and clopidogrel for a 6-month period. This time span can be shortened to 3 months (IIa A) in high bleeding risk patients (eg, PRECISE-DAPT ≥25) or 1 month (IIb C) if bleeding risk is very high or, conversely, prolonged to 12 months in high ischemic risk or complex procedures in patients with a PRECISE-DAPT <25 (IIb A). 2,8

European Society of Cardiology guidelines recommendations for DAPT duration in patients undergoing PCI according to clinical presentation and bleeding risk. ACS indicates acute coronary syndrome; DAPT, dual antiplatelet therapy; HBR, high bleeding risk; stable CAD, stable coronary artery disease.
Standard Antithrombotic Therapy in ACS
In patients with ACS undergoing PCI, DAPT is recommended for 12 months. A meta-analysis including 4758 patients with ACS at high bleeding risk, compared 3 versus 6 versus 12 months of DAPT regimen, and demonstrated a nonstatistically significant increase in myocardial infarction (MI) or definite/probable stent thrombosis (ST) in the 6 months regimen versus the 12 months regimen. In the group treated for 3 months, the risk of MI or ST significantly increased. On the basis of these results, discontinuation of DAPT in high bleeding risk patients should be considered after 6 months (IIa B). 11
On the other hand, PEGASUS-TIMI 54 (Prevention of Cardiovascular Events in Patients With Prior Heart Attack Using Ticagrelor Compared to Placebo on a Background of Aspirin-Thrombolysis in Myocardial Infarction 54) trial investigated, in secondary prevention, a long-term DAPT strategy after ACS with ticagrelor 90 mg twice daily or 60 mg twice daily versus placebo on top of aspirin. The primary efficacy end point (cardiovascular death, MI, stroke) was significantly reduced in both arms. However, ischemic protection was achieved at the expense of an increased rate of Thrombolysis in Myocardial Infarction (TIMI) major bleeding. 12
Previously, the DAPT trial investigated the benefits and risks of a prolonged DAPT strategy over >12 months versus aspirin alone in patients with or without MI. The extended DAPT arm showed a significant reduction in ST, major adverse cardiovascular event (MACE), and recurrent MI with an increased risk of moderate or severe bleeding in the long-term DAPT arm. 13
Udell et al recently published a meta-analysis comprised of patients with previous MI from the PEGASUS, CHARISMA, PRODIGY, DES-LATE, ARTIC-Interruption and DAPT trials which evaluated the potential benefit of extended DAPT over 12 months versus aspirin alone. Patients treated with extended DAPT showed a reduced incidence of MACE with significantly increased major bleedings but not fatal bleeding. 14
Standard Antithrombotic Therapy in Patients With AF Undergoing PCI
About 7% of patients who undergo PCI need or will need lifelong OAC therapy. Of note, AF affects about 1% to 2% of the general population (6 million people in Europe) and 20% of those affected will undergo PCI. A combined antithrombotic therapy with a P2Y12 inhibitor in addition to aspirin and an anticoagulant agent, so-called, triple therapy (TT) is theoretically the best strategy to manage patients with AF and CAD undergoing PCI. Unfortunately, TT exposes patients to a 2- to 3-fold increase in bleeding risk.
In the WOEST (What is the Optimal antiplatElet and anticoagulant therapy in patients with oral anticoagulation and coronary StenTing) study, 573 patients undergoing PCI and with an indication to OAC were randomized to dual antithrombotic therapy (DAT; consisting of clopidogrel and OAC) or TT (aspirin, clopidogrel, and OAC). Triple therapy was continued for 1 month after bare metal stent (BMS) and for 12 months after DES. Of note, among enrolled patients only 69% had AF and 27.5% underwent PCI for ACS. In the dual therapy arm, not only TIMI bleedings were significantly reduced as expected but also all-cause mortality was lower. Moreover, the composite end point of major bleedings, MI, target vessel revascularisation, ST, and stroke did not differ significantly between the 2 arms at 12 months of follow-up. Notably, the WOEST study was underpowered to detect statistically significant differences in ischemic end points between the 2 groups. 15
The ISAR-TRIPLE study randomized 614 patients to receive clopidogrel for a duration of either 6 weeks or 6 months, in addition to aspirin and OAC. In this trial, all patients underwent DES implantation and 32% had ACS on admission. At 9 months, there were not statistically significant differences in the rates of MI, ST, ischemic stroke, TIMI major bleeding, and death between the 2 strategies. 16
Overall, in patients undergoing PCI who require oral anticoagulation, a short period of TT of at least one month is recommend, regardless of the clinical setting (ACS or CAD) based on the balance between bleeding and ischemic risk. 5 In patients with NVAF treated with DOACs, there is no striking evidence for choosing one drug over another; however, the lowest effective dose for stroke prevention should be used. Notably, dabigatran is the only DOACs that has been investigated at a reduced dosage (110 mg twice daily) and for which noninferiority versus warfarin was demonstrated. 17 Of note, patients with DAPT were excluded in all phase III DOACs NVAF trials except in the RELY trial which enrolled 812 patients on triple antithrombotic therapy and has shown the lowest bleeding risk with dabigatran 110 mg twice a day. 18
The ischemic risk should be evaluated on the basis of the clinical setting (ACS or elective PCI), coronary anatomy, and procedural complexity. In a TT strategy, the use of clopidogrel is recommended because of the lack of evidence regarding the safety of ticagrelor and prasugrel.
Bleeding risk should be estimated by the HAS-BLED (Hypertension, Abnormal Renal/Liver Function, Stroke, Bleeding History or Predisposition, Labile INR, Elderly (>65 years), Drugs/Alcohol) or ABC (age, biomarkers [GDF-15, cTnt-hs, and hemoglobin], clinical history) score, taking into account that it is less predictable than ischemic risk and is very inconstant and dynamic. For this reason, it should be periodically reassessed during a follow-up program.
Considering these data, TT up to 6 months and dual therapy (OAC + aspirin or OAC + clopidogrel) for up to 12 months are recommend for patients at high ischemic risk.
If bleeding risk prevails, the TT can be shortened to only the first month with dual therapy thereafter. In patients with a very high bleeding risk, it should be considered to start with dual therapy for up to 12 months.
For all categories of patients, continuing antithrombotic treatment with OAC alone after 12 months may be considered. 2,5
New Perspective From Clinical Trials
Dual Pathway Antithrombotic Therapy in ACS
Thrombosis plays a crucial role in the pathogenesis of ACS and it is well known that it involves both the mechanism of coagulation and platelet aggregation. OACs can reduce the incidence of cardiovascular ischemic events after MI but they are associated with a significant higher risk of bleeding if compared to aspirin alone. Direct oral anticoagulants are relatively new molecules which have been shown to be at least noninferior to VKA in the prevention of stroke in patients affected by NVAF with an overall stronger safety profile. The addition of a DOAC to the standard antiplatelet therapy after an ACS could have a positive impact on cardiovascular outcomes with a concomitant higher but tolerable incidence of bleeding events (Table 1). One of the problems is the choice of an adequate dosage of the DOACs. Three trials were designed to answer this question: RE-DEEM, 19 APPRAISE, 20 and ATLAS ACS-TIMI 46, 22 respectively, tested increasing doses of dabigatran, apixaban, and rivaroxaban. All these studies showed that increasing doses of DOACs increases the incidence of hemorrhagic events. Specifically, the arms of APPRAISE with more than apixaban 10 mg/d were prematurely interrupted due to excess of bleeding events; conversely, RE-DEEM showed that only higher doses of dabigatran (110 mg twice a day and 150 mg twice a day) were able to significantly reduce the incidence of ischemic events. The APPRAISE-2 21 was a randomized, double-blinded, placebo-controlled clinical trial which recruited 7392 patients with a recent ACS and randomized them to placebo or apixaban 5 mg twice a day on top of standard medical therapy (including DAPT when clinically appropriate). The study was interrupted prematurely because of the occurrence of an excess of major bleeding events, including fatal bleeding and intracranial bleeding with apixaban, in absence of a counterbalancing reduction in adverse cardiovascular events.
Trials of DOACs in Patients With ACS Without AF.

Abbreviations: ACS, acute coronary syndrome; AF, atrial fibrillation; ASA, acetylsalicylic acid; CRN, clinically relevant nonmajor; DAPT, dual antiplatelet therapy; DM, diabetes mellitus; DOACs, direct-acting oral anticoagulants; FU, follow up; HR, hazard ratio; ISTH, International Society of Thrombosis and Hemostasis; NSTEMI, non-ST-elevation myocardial infarction; PCI, percutaneous coronary intervention; STEMI, ST-elevation myocardial infarction; TIMI, Thrombolysis in Myocardial Infarction; UA, unstable angina; VKA, vitamin k antagonist.
ATLAS ACS2-TIMI 51 was a randomized, double-blinded, placebo-controlled clinical trial which recruited 15 526 patients with a recent ACS to receive rivaroxaban 2.5 mg twice a day or 5 mg twice a day or placebo for a mean of 13 months and up to 31 months on top standard medical therapy (including DAPT). Rivaroxaban, both 2.5 and 5 mg twice a day, significantly reduced the primary efficacy end point (death from cardiovascular causes, MI, or stroke) as compared to placebo, at the price of an increase in major bleeding not related to CABG and intracranial hemorrhage. Rivaroxaban 2.5 mg twice a day also reduced the rates of death from cardiovascular causes and from any causes. 23 In both APPRAISE 2 and ATLAS ACS2-TIMI 51, the vast majority of patients were concomitantly treated with aspirin and a P2Y12-receptor antagonist (81% in APPRAISE 2, >90% ATLAS ACS2-TIMI 51); the prevalent P2Y12 receptor antagonist was clopidogrel. The main difference between these trials is the dosage of the DOACs: in APPRAISE-2, it was the same dosage approved for the prevention of ischemic stroke in NVAF; in ATLAS ACS2-TIMI 51, the 2 dosages tested were, respectively, half and a quarter of the daily dose tested for ischemic stroke prevention in NVAF. It is surprising that the reduction in mortality is evident just for the lowest dose of rivaroxaban: one possible explanation could be that the slightly higher number of nonfatal bleeding events recorded in the 5 mg twice a day arm could have affected mortality. Based on the evidence from ATLAS ACS2-TIMI 51, it has been suggested to consider the addition of rivaroxaban 2.5 mg twice a day to aspirin and clopidogrel for approximately 1 year in patients with ACS with no prior TIA/stroke and at high ischemic risk (level of evidence IIb B). 5,8
More recently, the phase 2 trial GEMINI-ACS-1 randomized 3037 patients with ACS (49% STEMI; 40% NSTEMI; 87% PCI; 67.3% DES) to receive rivaroxaban 2.5 mg twice a day plus a P2Y12 inhibitor or aspirin 100 mg plus a P2Y12 inhibitor. The aim of the trial was to test the safety of the substitution of aspirin with low-dose rivaroxaban in association with a P2Y12 inhibitor, including ticagrelor (56% of patients); GEMINI-ACS-1 showed that the 2 therapeutic strategies entail a similar bleeding risk. 24 Moreover, this trial suggests that dropping aspirin and adding vascular, low dose of rivaroxaban to a P2Y12 inhibitor has, respectively, a similar and a better safety profile compared to DAPT and triple antithrombotic therapy (DAPT + low-dose rivaroxaban as tested in ATLAS ACS2-TIMI 51). Large and adequately powered trials would be required to demonstrate the efficacy of dual pathway antithrombotic therapy without aspirin in ACS.
Dual Antithrombotic Pathway in Stable CAD
Recently, the efficacy of DOACs in secondary cardiovascular prevention was tested. COMPASS was a double-blind, double-dummy trial which recruited 27 395 patients with stable CAD (>90%) and/or peripheral artery disease (27%) and randomized them to receive rivaroxaban 2.5 mg twice a day plus aspirin 100 mg, rivaroxaban 5 mg twice a day alone, or aspirin 100 mg alone. The primary efficacy end point was the composite of death, stroke, or MI. The trial was prematurely interrupted when just 50% of planned events occurred, because a consistent difference in the primary efficacy end point was observed in favor of rivaroxaban 2.5 mg twice a day plus aspirin. In this group, the rate of bleeding was higher by 70% but the net clinical benefit outcome was lower by 20%. Moreover, there was no significant difference in intracranial or fatal bleeding between the rivaroxaban plus aspirin arm and the aspirin arm. The arm treated with rivaroxaban 5 mg twice a day alone, compared to the arm with aspirin alone, showed a higher rate of bleeding without significant benefit on efficacy outcome. 25 The possible clinical implications of these results are significant but in the future management of stable CAD other possible drug combinations have to be considered, for example, the long-term use of reduced doses of DAPT as suggested by the PEGASUS-TIMI 54 trial. 12
Dual Antithrombotic Therapy in AF Patients Undergoing PCI
The rise in the incidence of bleeding in patients undergoing TT is evident just within 30 days from its initiation. 17 Similar conclusions came from the WOEST trial 15 in which the patients treated with VKA and clopidogrel showed fewer bleeding complications and no increase in the rate of thrombotic events. Despite this initial evidence, ST is a serious, though uncommon, complication of PCI and current guidelines and consensus documents still suggest, in patients with AF who need to undergo PCI to start with TT for at least one month and up to 6 months if the patient is not at high bleeding risk after PCI. 2,5,8
Two trials have recently investigated the association of DOACs with a P2Y12 inhibitor in patient with NVAF undergoing PCI, 26, 27 essentially testing the hypothesis of DAT dropping aspirin (Table 2). Triple antithrombotic therapy, DAPT and a very low dose of rivaroxaban (2.5 mg twice daily), has been studied only in the PIONEER AF-PCI trial. This treatment has been found to be associated with a lower incidence of bleeding than standard therapy with VKA but higher risk of stroke at 6 months of follow-up and was not approved for this indication.
The Design and End Points of the Main Trials Investigating the Employment of DOACs in Patients With AF With Indication to PCI.
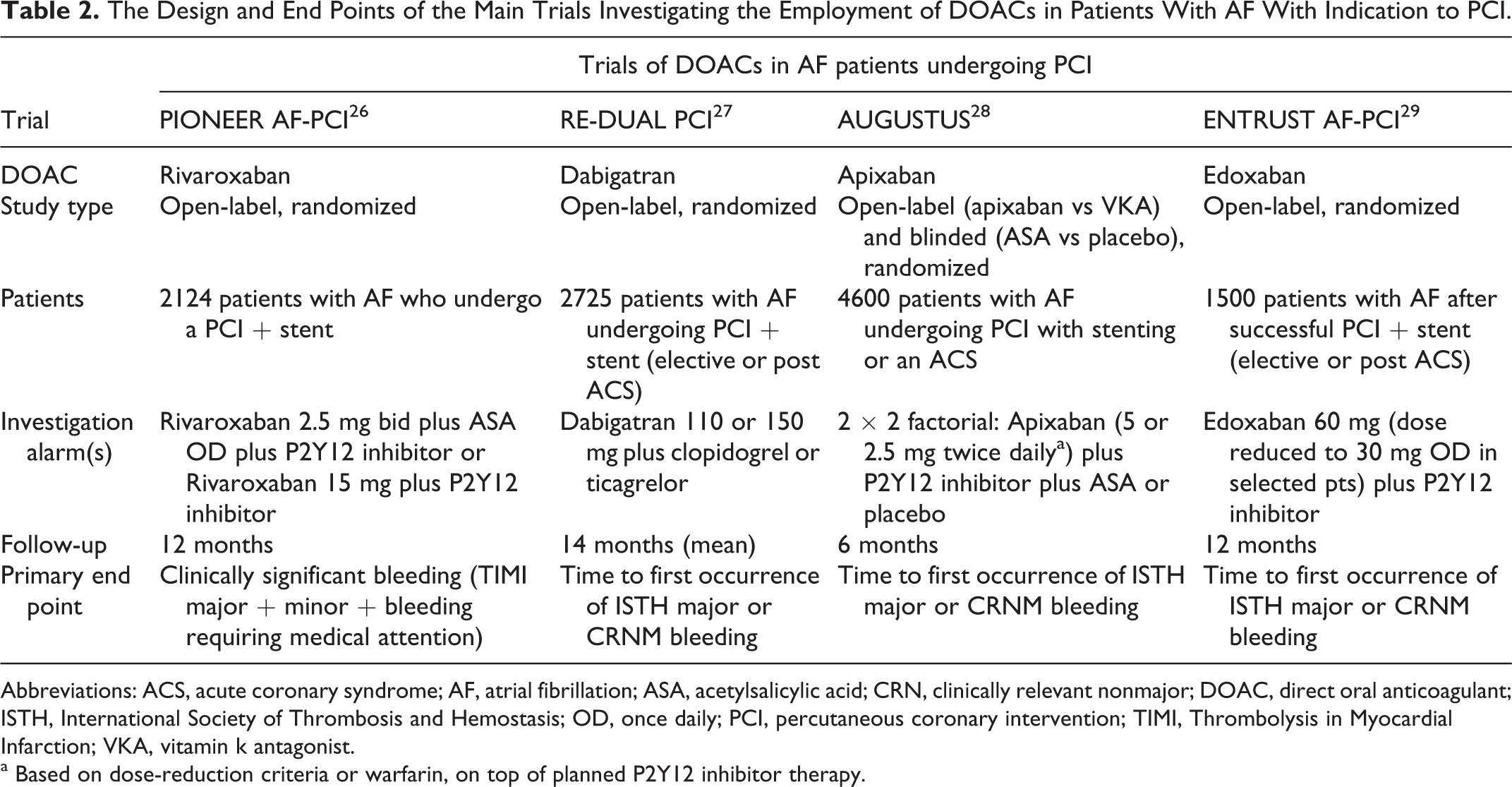
Abbreviations: ACS, acute coronary syndrome; AF, atrial fibrillation; ASA, acetylsalicylic acid; CRN, clinically relevant nonmajor; DOAC, direct oral anticoagulant; ISTH, International Society of Thrombosis and Hemostasis; OD, once daily; PCI, percutaneous coronary intervention; TIMI, Thrombolysis in Myocardial Infarction; VKA, vitamin k antagonist.
a Based on dose-reduction criteria or warfarin, on top of planned P2Y12 inhibitor therapy.
PIONEER AF-PCI was a randomized open-label trial that enrolled 2124 patients with NVAF who had undergone PCI with stenting; the patients were randomized to receive rivaroxaban 15 mg every day plus a P2Y12 inhibitor for 12 months (group 1), rivaroxaban 2.5 mg twice a day plus DAPT for 1, 6, or 12 month (group 2) or a VKA plus DAPT for 1, 6, or 12 months (group 3). Patients with a history of stroke or transient ischemic attack were excluded. The primary end point was a composite of major and minor bleeding according to TIMI criteria or bleeding requiring medical attention. The 2 groups treated with rivaroxaban were associated with a lower risk of bleeding across all subgroups (duration of DAPT). The RE-DUAL PCI was a randomized, open-label trial which recruited 2725 patients with NVAF who had just undergone PCI and randomized them to VKA plus DAPT for 1 to 3 months, according to the presence of a BMS or a DES, or dabigatran (110 mg twice a day or 150 mg twice a day) plus a P2Y12 inhibitor. The primary end point was the occurrence of major or clinically relevant nonmajor bleeding events as defined by the International Society of Thrombosis and Hemostasis. The rate of bleeding events was significantly lower in dual therapy group (both with dabigatran 110 mg twice a day and with dabigatran 150 mg twice a day). In both trials, there were no differences between the groups as far as the efficacy end points (thromboembolic events) are concerned; however, neither PIONEER AF-PCI nor RE-DUAL PCI was adequately powered to answer this specific question, in particular in patients with ACS which were about half of the overall population. In both studies, the P2Y12 inhibitor employed was clopidogrel in the vast majority of patients; this is why current guidelines do not suggest to prescribe prasugrel or ticagrelor in association with DOACs or VKA. Conversely, the study differed in the proportion of patients treated with a DES (>80% in RE-DUAL PCI vs round 65% in PIONEER AF-PCI). Of note, RE-DUAL PCI enrolled patients with previous stroke or TIA, whereas in PIONEER AF-PCI they were excluded per protocol. Furthermore, the most striking difference between the 2 studies is the dosage of the DOACs: in RE-DUAL PCI 2 doses of dabigatran were tested whose efficacy had already been demonstrated in the context of stroke prevention in NVAF patients, 18 whereas in PIONEER AF-PCI the doses of rivaroxaban tested have not been shown to be protective against stroke in NVAF and are not approved with this indication alone. In accordance with the results of RE-DUAL PCI and PIONEER, the European Society of Cardiology Guidelines on DAPT of 2017 suggests to treat patients with an indication to oral anticoagulation undergoing a PCI with clopidogrel alone plus the OAC ab initio, if the patient is considered to be at high bleeding risk (class IIa). 2 Consistently, the European Heart Rhythm Association Practical Guide on the use of non-VKA OACs in patients with NVAF 30 favors an extreme individualization, suggesting even the use of DAPT alone in patients with high bleeding risk with a low CHA2DS2-VASC. Lastly, the 2018 Joint European consensus document on the management of antithrombotic therapy in NVAF patients presenting with ACS and/or undergoing percutaneous cardiovascular interventions 31 recommends the prescription of DOACs in the context of triple or dual therapy because of their better safety profile in terms of bleeding; the dosage of the DOACs should not be reduced in TT or in association with an antiplatelet drug, unless indicated for other characteristics of the patient; the only exception is for rivaroxaban, which should be prescribed at the dosage of 15 mg every day.
What is described above represents the European perspective for which TT still remains the main strategy that should be stopped at hospital discharge, after 1 month, or 3 to 6 months depending on the balance between the individual thrombotic and bleeding profile, whereas DAT should be reserved ab initio only for patients with very high bleeding risk, in particular in the setting of ACS.
Otherwise, the North American perspective suggests DAT with OAC and clopidogrel as the default strategy immediately after hospital discharge for most patients, whereas TT up to 1 month should be reserved only for patients at a high ischemic/thrombotic risk and low bleeding risk. 32,33
Recently AUGUSTUS, a multicenter, prospective study with a 2-by-2 factorial design, randomized 4614 patients with NVAF and ACS and/or undergoing PCI, planned for P2Y12 inhibitor for at least 12 months, to take apixaban or a VKA and to take aspirin or combining placebo for 6 months. The primary outcome consisted of major or clinically relevant nonmajor bleeding. Clopidogrel was used in 92.6% of the patients, whereas prasugrel and ticagrelor therapy was less frequently adopted (1.1% and 6.2%, respectively). This study allowed to compare directly DOACs to VKA and aspirin regimen to placebo asserting the safety and the efficacy of apixaban at a dose recommended for NVAF patients as demonstrated by a lower incidence of both bleeding and the composite of death and hospitalization in patients treated with apixaban than in VKA arm. It was also demonstrated that therapeutic strategy without aspirin had more benefit on the incidence of bleeding than placebo. 28
Conclusion
Managing patients with AF who also need antiplatelet therapy for MI and/or PCI is an increasingly common and controversial issue in clinical practice. In order to choose an ideal antithrombotic drug, dosage, and their combination, it is necessary to consider the thrombotic and bleeding risk of each single patient. Growing evidence suggests that DOACs should be preferred over VKAs due to their more favorablerisk/benefit profile which has also been demonstrated in association with antiplatelet therapy. Furthermore, the overall duration of the combined therapy of OACs and antiplatelets should be shortened as much as possible, in accordance with international guidelines.
Supplemental Material
Supplemental Material, Colivicchi_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, Colivicchi_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, De_Luca_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, De_Luca_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Di_Fusco_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, Di_Fusco_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Gabrielli_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, Gabrielli_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Gelsomino_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, Gelsomino_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Giubilato_coi_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, Giubilato_coi_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Gulizia_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, Gulizia_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Iorio_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, Iorio_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Leone_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, Leone_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Luca_coi_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, Luca_coi_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Poli_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, Poli_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Supplemental Material
Supplemental Material, Rao_disclosure - The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead
Supplemental Material, Rao_disclosure for The Combination of Oral Anticoagulant and Antiplatelet Therapies: Stay One Step Ahead by Fabiana Lucà, Simona Giubilato, Stefania Angela Di Fusco, Angelo Leone, Stefano Poli, Carmelo Massimiliano Rao, Annamaria Iorio, Sandro Gelsomino, Domenico Gabrielli, Furio Colivicchi, Leonardo De Luca and Michele Massimo Gulizia in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Author Contributions
All authors contributed substantially to the writing of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
