Abstract
Background:
Direct oral anticoagulants (DOACs) have been found to be similar or superior to warfarin in reducing ischemic stroke and intracranial hemorrhage (ICH) in patients with atrial fibrillation (AF). We sought to examine the anticoagulation prescription patterns in community since the advent of DOACs and also evaluate the outcomes in terms of gastrointestinal (GI) bleeding, ischemic stroke, and ICH in real-world patients with AF receiving anticoagulation.
Methods:
This is a retrospective study comprising patients who were newly diagnosed with nonvalvular AF and were prescribed anticoagulants for stroke prevention. Prescription pattern of the anticoagulants based on CHA2DS2Vasc score was studied. Clinical outcomes of GI bleeding, ischemic stroke, and ICH were analyzed using a multivariate logistic regression model.
Results:
Of the 2362 patients with AF on anticoagulation, 44.7% were prescribed DOACs. Patients with CHA2DS2VASc score of ≥3 received a prescription for warfarin more often than DOACs (P < .001). Multivariate logistic regression analysis revealed that the incidence of GI bleed (odds ratio [OR]: 0.91, 95% confidence interval [CI]: 0.62-1.35, P = .66) and stroke (OR: 0.77, 95% CI: 0.57-1.05, P = .10) was similar between warfarin and DOAC users. However, there was a trend toward lower ICH in the DOAC group (OR: 0.60, 95% CI: 0.36-1.01, P = .06).
Conclusions:
Prescription rate of DOACs for nonvalvular AF has increased significantly, with apixaban being the most commonly used agent. Patients with higher CHA2DS2-VASc score (≥3) are prescribed DOACs less often than warfarin. The reason for this discrepancy is unclear. Given the favorable risk–benefit profile of DOACs, further studies are needed to identify factors that determine anticoagulant selection in patients with AF with high thromboembolic risk.
Introduction
Atrial fibrillation (AF) is an important risk factor for stroke and accounts for approximately half of all ischemic strokes. Anticoagulation is the mainstay of therapy for stroke reduction in patients with AF. The vitamin K antagonist warfarin reduces the risk of embolic stroke associated with AF by almost two-thirds and has been in use for many decades. 1 Over the past decade, several direct oral anticoagulants (DOACs) have been developed that inhibit thrombin (dabigatran) or activated factor Xa (rivaroxaban, apixaban and edoxaban) and have been shown to be noninferior to warfarin in stroke reduction in patients with nonvalvular AF. A meta-analysis of the major DOAC trials showed a decreased incidence of ischemic stroke, intracranial hemorrhage (ICH), and mortality but increased risk of gastrointestinal (GI) bleed without an increase in overall major bleeding when compared to warfarin. 2 Fewer drug–food interactions and lack of a need for frequent monitoring have made DOACs an attractive alternative to warfarin.
Bulk of the clinical evidence of efficacy with the use of DOACs in nonvalvular AF was obtained from 4 major, large sample size, phase III, randomized controlled trials (RELY [dabigatran], ROCKET-AF [rivaroxaban], ARISTOTLE [apixaban], and ENGAGE-TIMI 48 [edoxaban]) that compared DOACs with warfarin. 3 -6 These large-scale trials were designed to evaluate the main treatment effect, that is, reduction in stroke or systemic embolic events. 2 Overall time in therapeutic range for warfarin users in these trials was in the range of 65%, which may not be representative of the general population where time in therapeutic range for warfarin is frequently suboptimal. 2 We sought to examine anticoagulation prescription patterns and clinical outcomes of GI bleeding, ischemic stroke, and ICH in a real-world all-comer population of patients with nonvalvular AF.
Methods
Study Design and Data Source
We conducted a retrospective study of all patients with a new diagnosis of nonvalvular AF. Data for the study were provided by the Arkansas Clinical Data Repository (AR-CDR) maintained by the Department of Biomedical Informatics in the College of Medicine at the University of Arkansas for Medical Sciences (UAMS). The AR-CDR is approved to operate as an enterprise data resource to support research across UAMS. Data in the AR-CDR are extracted from UAMS electronic medical record (EMR), tumor registry, billing, and cancer genomic data and comprises of encounters since May 2014. The patient sample includes all outpatient and inpatient visits across any university health system facility across the state. Data on demographics, comorbidities, hospital admissions, discharge disposition, medication, medical history, and procedure information are readily available from AR-CDR. Mortality data were obtained by matching individual patient code with the state Social Security database. In addition, a natural language processing algorithm was used to extract unstructured data related to social and family history of patient. The data were extracted to readable files using Microsoft SQL Data tool. Using this data set, we identified all patients with a new diagnosis of AF (International Classification of Diseases, Ninth Revision [ICD-9] code 427.3) from May 2014 to May 2017. These patients were followed up till April 2018. The study was approved by the local institutional review board.
Data Collection
Adults greater than 18 years of age, with newly diagnosed AF who were prescribed either warfarin or DOAC for stroke prevention, were included in the study. Patients with an existing diagnosis of AF or AF in the setting of valvular heart disease (prosthetic heart valve, mitral stenosis, rheumatic heart disease, history of mitral valve repair) in the past were excluded. In addition, patients with nonvalvular AF who were not on chronic anticoagulation, who were on anticoagulation with agents other than those studied such as enoxaparin, and those who received prescriptions for both warfarin and DOAC during the study period were excluded. Patients were divided into warfarin group and DOAC group. The DOAC group consisted of patients who received prescription for rivaroxaban, apixaban, or dabigatran during the study period. No patients were prescribed edoxaban. Data obtained from each patient record included demographic information such as age, sex, race, body mass index, comorbidities categorized by the Elixhauser comorbidity criteria, and medication use information. CHA2DS2-VASc score for each patient was calculated through EMR and was validated by chart review. Prescription pattern in relation to CHA2DS2-VASc score was assessed by identifying the percentage of patients who received prescription for warfarin and DOAC in each score group. Outcome information collected included new occurrence of ischemic stroke (ICD-9 434), GI bleed (ICD-9 578.9), and ICH (ICD-9 430, 431,432.1, 432.0, 432.9).
Statistical Analysis
Baseline characteristics were described as percentages for categorical variables and mean and standard deviation for continuous variables. An unadjusted comparison was made using χ2 test or Wilcoxon rank sum test (categorical variables) and t test (continuous variables) as appropriate. Logistic regression models were constructed with the outcome variables, including ischemic stroke, GI bleed, and ICH as dependent variables. Independent variables that were significant on univariate analysis at P < .25 in a stepwise forward selection method were included in the multivariate analysis. In addition, the variables that were thought to have an important influence on the outcome were also included in the model. Goodness of fit was assessed using Pearson goodness-of-fit test and area under the curve of the receiver operating characteristic of logistic regression. Data analysis was performed using StataCorp 2015 (Release 14.2, StataCorp LP, College Station, Texas).
Results
Patient Characteristics
During the study period, there were 4818 adults with a new diagnosis of nonvalvular AF. After applying the exclusion criteria described previously, a total of 2362 patients were included in the study (Figure 1). Among the 2362 patients included in the analysis, 1306 (55.3%) were prescribed warfarin and 1056 (44.7%) were prescribed one of the DOACs.
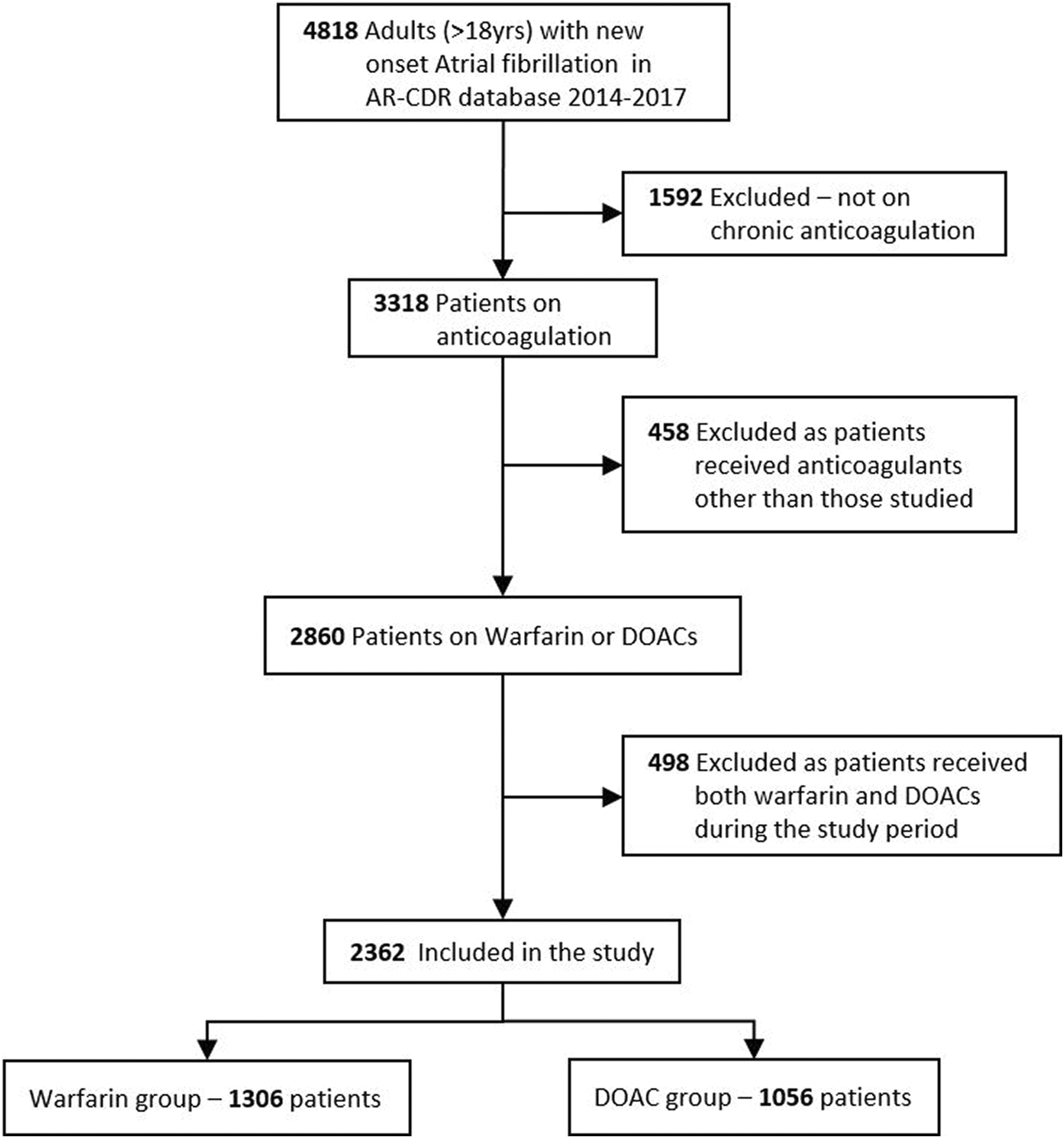
Study design depicting process of patient selection for the study.
Baseline characteristics of the study population are summarized in Table 1. In unadjusted analysis, patients prescribed warfarin were slightly older (median age 76 vs 75, P = .04) and had a higher prevalence of hypertension (67.8% vs 63.5%, P = .03), diabetes (30.7% vs 24.0%, P < .001), and congestive heart failure (41.0% vs 28.2%, P < .001), whereas the DOAC group had a higher proportion of Caucasians (80.7% vs 72.7%, P < .001). Median follow-up duration of patients was 491 days (interquartile range: 144–957 days). Follow-up duration was greater among patients in the warfarin group compared to that in the DOAC group (536 days vs 465 days; P = .002).
Descriptive Characteristics of Patients With AF in Warfarin Versus DOAC Groups.

Abbreviations: AF, atrial fibrillation; DOAC, direct oral anticoagulant.
aReference group: warfarin.
Prescription Pattern of Anticoagulants Based on CHA2DS2-VASc Score
Of the 2362 patients, 44.7% were on DOACs. Among the DOAC group, apixaban (51.0%) was prescribed most often followed by rivaroxaban (28.7%) and dabigatran (20.3%). Patients with CHA2DS2VASc score of >3 received prescription for warfarin more often than DOAC (P < .001). There was trend of prescribing warfarin more often with increasing CHA2DS2-VASc score in relation to DOACs (P for trend <.001; Figure 2A and B).

Prescription pattern of warfarin and DOAC based on the CHA2DS2-VASc score. A, Line diagram depicting the percentage of patients on either anticoagulant at a given CHA2DS2-VASc score. B, Bar chart depicting actual proportion of patients at a given CHA2DS2-VASc score. Warfarin was prescribed more often than DOACs in patients with high CHA2DS2-VASc scores. DOAC indicates direct oral anticoagulant.
Outcomes
Ischemic stroke
The incidence of new ischemic stroke was similar in both warfarin and DOAC groups (odds ratio [OR]: 0.77, 95% confidence interval [CI]: 0.57-1.05, P = .10; Figure 3). Prior history of stroke was the most important determinant for the occurrence of new ischemic stroke (OR: 7.28, 95% CI: 4.02-13.2, P < .001). Hypertension (OR: 2.09, 95% CI: 1.42-3.10, P < .001) and diabetes (OR: 1.46, 95% CI: 1.05-2.04, P = .02) were also significantly predictive of a new ischemic stroke in our patient population.

Outcomes of GI bleed, ischemic stroke, and intracranial hemorrhage in patients with AF in warfarin and DOAC groups. P value obtained after adjusting for patient variables. Risk of ICH is significantly lower in the DOAC group compared to warfarin. AF indicates atrial fibrillation; DOAC, direct oral anticoagulant; GI, gastrointestinal; ICH, intracranial hemorrhage.
Gastrointestinal bleed
Multivariate logistic regression analysis performed with GI bleed as the outcome showed similar odds in both warfarin and DOAC groups (OR: 0.91, 95% CI: 0.62-1.35, P = .66; Figure 3). Incidence of GI bleed was independently influenced by prior history of peptic ulcer disease (OR: 14.9, 95% CI: 6.7-33.0, P < .001) and antiplatelet drug use (OR: 2.3, 95% CI: 1.48-3.57, P < .001), as well as the presence of liver disease (OR: 3.37, 95% CI: 1.97-5.77, P < .001), hypertension (OR: 1.63, 95% CI: 1.02-2.62, P = .04), and renal disease (OR: 1.54, 95% CI: 1.02- 2.34, P = .04) in the study population. These factors were adjusted for in the logistic regression analysis.
Intracranial hemorrhage
There was a trend toward lower ICH in the DOAC group compared to warfarin after adjusting for other confounding variables (OR: 0.60, 95% CI: 0.36-1.01, P = .06).
Discussion
Our study specifically evaluated patients with a new diagnosis of nonvalvular AF who were initiated on chronic anticoagulation with either warfarin or DOACs. This real-world analysis suggests an increase in overall DOAC prescription rates over the study period (Supplementary Figure 1). In our analysis, we identified a trend of prescribing warfarin to patients with higher CHA2DS2-VASc score. Also, similar to the results of the large randomized controlled trials, incidence of ICH trended lower in the DOAC group. However, there was no difference between the groups in the incidence of GI bleed or ischemic stroke.
To the best of our knowledge, this is the first study in the United States that has assessed the prescription patterns of DOACs, well after the introduction of all 3 DOACs. Previous studies that evaluated the prescription pattern of DOAC and warfarin such as those using the data from Outcomes Registry for Better Informed Treatment of Atrial fibrillation (ORBIT-AF) registry, “Get with the Guidelines—Stroke” database, and the National Cardiovascular Data Registry (NCDR), Practice Innovation and Clinical Excellence (PINNACLE) registry were reported during a period when DOACs were just approved and the use was not as widespread. 7 -9
Steinberg et al described the pattern of dabigatran use in community practice between 2010 and 2011 using data from the ORBIT-AF registry. Dabigatran was prescribed only in 12% of the patients during the study period. 7 They also noted that patients who received dabigatran were younger (median age: 72 vs 75 years; P < .0001), more likely to be Caucasian (92% vs 89%; P = .005), and were less likely to have prior cardiovascular disease (4% vs 33%; P < .001). Our study also shows that patients who received a prescription for DOAC were younger and more likely to be Caucasian with relatively lower prevalence of hypertension, diabetes, and congestive heart failure compared to those prescribed warfarin. The trend toward warfarin prescription in older patients may in part be a cost issue, given that the Medicare population often do not receive support/co-pay cards easily (compared to patients younger than 65 years) and may thus have difficulty bearing the high cost of DOACs, thereby explaining the prescribing bias.
Camm et al in their prospective study on 39 670 patients from 35 countries, between 2010 and 2015, from the Global Anticoagulant Registry in the FIELD-Atrial Fibrillation noted that DOACs were more frequently prescribed than warfarin in the elderly patients. 10 However, in our study, the median age in the warfarin group was slightly higher than that in the DOAC group. Analysis of the study cohorts revealed that this was primarily due to the ease of use and perceived lower bleeding risk of DOACs. Cost may not have been a limiting factor in the study cohorts, which may account for the difference between their results and our study.
Patel et al analyzed the “Get with the Guidelines—Stroke” database on patients with AF who were admitted in the hospital after stroke/TIA between the years 2010 and 2012 and reported that only 11.1% of patients were discharged on a DOAC compared to 88.9% on warfarin. 8 The proportion of patients on DOAC had a relatively low CHA2DS2-VASc score. Likewise, Lauffenburger et al studied factors driving the anticoagulant selection in AF by analyzing the Medicare supplement claims database from 2010 to 2012 and found that patients with higher ischemic stroke risk (CHA2DS2-VASc ≥2) and high bleeding risk patients were less likely to receive DOACs. 11 Dabigatran and rivaroxaban were initiated in 29.9% and 7.9% of the patients in this cohort, respectively. Similar to both these study cohorts, 8,11 our study showed that there is a significant trend of using warfarin in patients with higher CHA2DS2-VASc score than the DOAC (P < .001). Of note, the use of DOAC has substantially increased to 46.3% in our study in the study period 2014 to 2017.
Data from NCDR PINNACLE registry from April 2008 to September 2014 showed a decline in warfarin use from 52.4% to 34.8% (P < .01) in patients with AF. Among the DOACs, rivaroxaban (12.3%) was used more commonly compared to dabigatran (7.2%) and apixaban (6.3%) at the end of the study period. In addition, cardiology practitioners were found to prescribe DOACs for patients with a lower risk of stroke, the reasons for which were unclear. The national patient registry cohort that included patients from years 2012 to 2015 also showed a trend of declining warfarin use from 59% to 24% in Danish population. 12 The Danish registry also showed an increase in apixaban use since its introduction and a decline in dabigatran use. Similar to these findings, apixaban was the most common DOAC prescribed (51.2%) in our study population. This may be due to better benefit–risk profile of apixaban and the wider scope of its use in patients with chronic kidney disease.
Meta-analyses of the randomized controlled trials comparing DOAC and warfarin showed that DOACs are similar or superior to warfarin in reducing stroke and ICH but associated with increased GI bleeding. 2 Real-world analyses examining outcomes of patients prescribed DOACs yielded better results. Propensity score-matched analysis of US claims database between 2013 and 2014 by Lip et al reported a decrease in major bleeding events with dabigatran (hazard ratio [HR]: 0.69; 95% CI, 0.50-0.96) and apixaban (HR: 0.53; 95% CI, 0.39-0.71) when compared to warfarin. However, the analysis did not separately assess ICH and GI bleed risk. 13 In a similar study, Yao et al evaluated the effectiveness and safety of dabigatran, rivaroxaban, and apixaban against warfarin in patients with AF using a large insurance database from 2010 to 2015. 14 They found a decreased risk of stroke with apixaban and a similar risk with dabigatran and rivaroxaban when compared to warfarin. For major bleeding, apixaban and dabigatran had a lower risk. Intracranial hemorrhage risk was found to be lower with all the DOACs when compared to warfarin. Another similar US claims database study showed lower risk of stroke (HR: 0.67, 95% CI, 0.59-0.76) and major bleeding (HR: 0.60, 95% CI, 0.54-0.65) with apixaban compared to warfarin. This study also showed that the benefits of using apixaban were consistent across the range of CHA2DS2-VASc scores. 15 Likewise, Chan et al found that rivaroxaban and dabigatran not only decreased the risk of ICH but also decreased the incidence of ischemic stroke when compared to warfarin. 16 This study was based on data from Asian patients and included patients who had received more than 1 anticoagulant therapy. Cha et al used the Korean National Health Insurance Service database to study the risk of ischemic stroke and ICH in Asian patients with high-risk AF (defined by them as CHA2DS2-VASc score ≥2) on DOAC or warfarin. 17 They described results similar to ours as they noted that DOACs are equally efficacious in preventing ischemic strokes and have lesser risk of ICH. This study also documented a significantly lower all-cause mortality rate with the use of dabigatran and apixaban.
Our results in a contemporary data set of patients with a higher proportion prescribed DOACs are comparable to the abovementioned large studies. There was a trend of lower ICH rates in the DOAC group. Stroke and GI bleeding were similar in adjusted analysis (Figure 3). Subanalysis of the effects of different types of DOAC on the outcomes, when compared to warfarin, did not yield statistically significant results in our study. This is likely due to the small sample size of each of the DOAC types. The event rates in our study may be higher than published landmark randomized clinical trials, however was not dramatically different. The apparent difference can be attributed to the higher mean CHA2DS2-VASc score in our study population, longer follow-up period in patients with higher CHA2DS2-VASc score, and the difference in the format in which the event rates were reported (such as 100 patient-year or %/year in landmark trials, unlike the total percentage of events as reported in this study).
Overall DOAC use is increasing in the United States. We believe that DOACs can be safely and effectively used in patients with high CHA2DS2-VASc score. These patients are likely to derive greater benefit than warfarin given their higher comorbidity index. However, the risk–benefit profile and comorbidities and patient preference should be taken into account prior to prescription. Further studies are also needed to explore the factors that determine favoring warfarin therapy in patients with AF.
Strengths and Limitations
The strength of our study includes analysis of a large, contemporary, real-world population of patients with AF, inclusion of patients on all 3 commonly used DOACs, and inclusion of major clinical end points. However, we acknowledge several limitations. First, baseline characteristics and outcomes were obtained from a clinical registry which, as with every retrospective study, is subject to misclassification. We do not have data on retention of patients within the system, and a proportion of the patients may have obtained their care from other facilities that may not have been captured by this database. We could not account for residual confounding imposed by variables such as socioeconomic class and medication adherence educational level in our study population. These factors play a significant role in determining clinical outcomes and could not be adjusted for. A large proportion of patients (1592 patients) were excluded from the analysis as we could not find prescription data on anticoagulation for this population. Our hypothesis is that these are patients who had new-onset AF in postoperative setting who were not anticoagulated, patients with sepsis or other severe systemic diseases who died prior to hospital discharge, or patients who were not a candidate for anticoagulation due to elevated bleeding risk. In addition, some of the patients may have been excluded as their anticoagulation may have been prescribed by other providers who are not in the University of Arkansas health system. To ensure accuracy, we decided to only include patients who actually received the prescription from our health system. The international normalized ratio (INR) control of the warfarin group was not well documented, and we did not have enough INR values to accurately determine the time in therapeutic range. From the available data, we have calculated a median time in therapeutic range of 36.5% (interquartile range: 23.3%-53.3%). This value was obtained using data from 722 patients in the warfarin group. However, we suspect the available laboratory data may not accurately depict the clinical picture. Again, we do not have data on DOAC compliance. These can be potential confounder in the analysis of the true effect—difference between the 2 groups. Our analysis of the different types of anticoagulants within the DOAC group did not yield statistically significant difference due to the relatively smaller sample size. Finally, we only included patients who were on a single anticoagulant (either a direct thrombin inhibitor or warfarin). We did not study the patients who were switched from one anticoagulant to the other during the study period. This was to keep the sample uniform and avoid inaccurate results due to interactions during the switching from one anticoagulant type to the other. Single tertiary care hospital-based database also limits its generalizability. However, unlike previous studies that obtained data from cardiology practices, our data are on all patients with AF, including patients from cardiology and noncardiology practices.
Conclusion
Direct oral anticoagulant prescription rates have increased since their introduction. Apixaban was the most commonly used DOAC in this study population, even though it was the last one to be approved for clinical use. Patients with high CHA2DS2-VASc score are prescribed DOACs less often than warfarin. Given the favorable risk–benefit profile of DOACs, they may be better suited for high-risk individuals. Further studies are needed to evaluate the reasons for favoring warfarin therapy in patients with high-risk AF and to identify patients who might benefit from warfarin therapy.
Supplemental Material
supplementary_figure_1 - Prescription Patterns and Outcomes of Patients With Atrial Fibrillation Treated With Direct Oral Anticoagulants and Warfarin: A Real-World Analysis
supplementary_figure_1 for Prescription Patterns and Outcomes of Patients With Atrial Fibrillation Treated With Direct Oral Anticoagulants and Warfarin: A Real-World Analysis by Ajoe John Kattoor, Naga Venkata Pothineni, Akshay Goel, Mahanazuddin Syed, Shorabuddin Syed, Hakan Paydak and Jawahar L. Mehta in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgments
Data for the study were provided by the Arkansas Clinical Data Repository (AR-CDR) maintained by the Department of Biomedical Informatics in the College of Medicine at the University of Arkansas for Medical Sciences (UAMS).
Author Contributions
Ajoe John Kattoor, MD, and Venkata Pothineni, MD, equally contributed to this work. Ajoe John Kattoor and Naga Venkata Pothineni contributed to contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, critically revised the manuscript, and agree to be accountable for all aspects of work ensuring itegrity and accuracy. Akshay Goel, Mahanazuddin Syed, and Shorabuddin Syed contributed to acquisition, analysis, and interpretation, critically revised manuscript, agrees to be accountable for all aspects of work ensuring itegrity and accuracy. Hakan Paydak and Jawahar L. Mehta contributed to contributed to conception and design, contributed to interpretation, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
